Resistance of Concrete with Various Types of Coarse Aggregate to Coupled Effects of Thermal Shocks and Chemicals
Abstract
:1. Introduction
2. Materials and Method
2.1. Materials
2.2. Concrete Mixing and Specimen Preparation
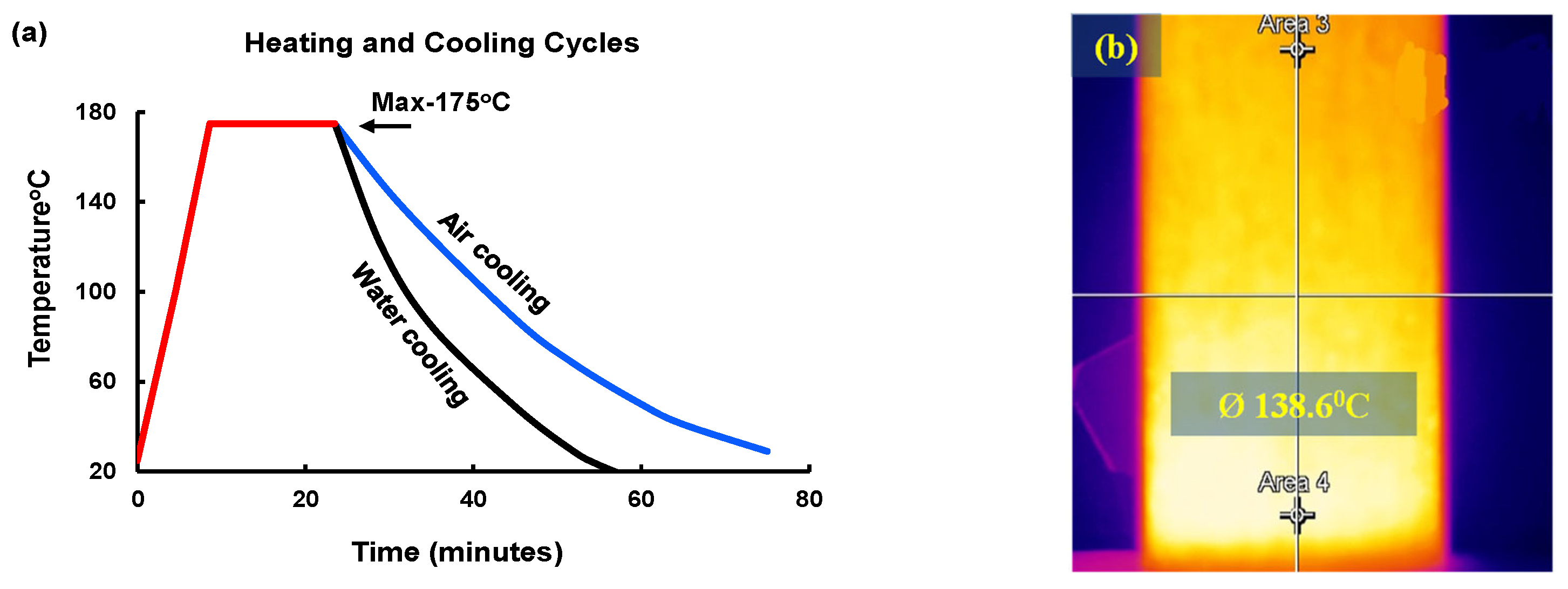
2.3. Thermal and Chemical (HC Fluids) Exposures
2.4. Tests
2.4.1. Residual Mechanical Properties Test
2.4.2. Stress–Strain Test
2.4.3. Determination of Thermal Properties
2.4.4. Fourier Transform Infrared Spectroscopy (FTIR) Analysis
2.4.5. Thermogravimetric (TG) and Differential Scanning Calorimetry (DSC) Analysis
2.4.6. Microstructural Investigation
3. Result and Discussion
3.1. Residual Mechanical Properties
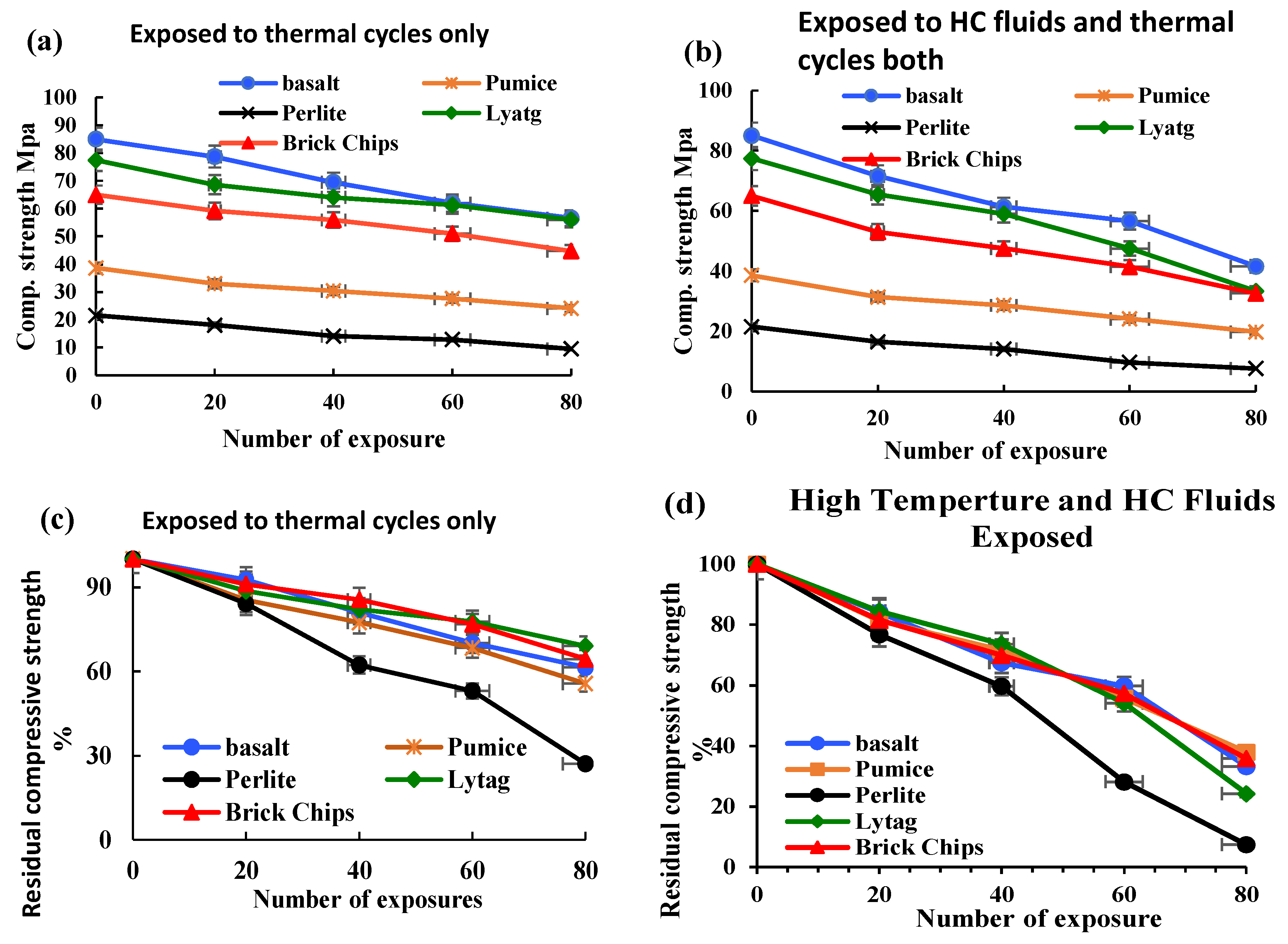
3.1.1. Compressive Strength
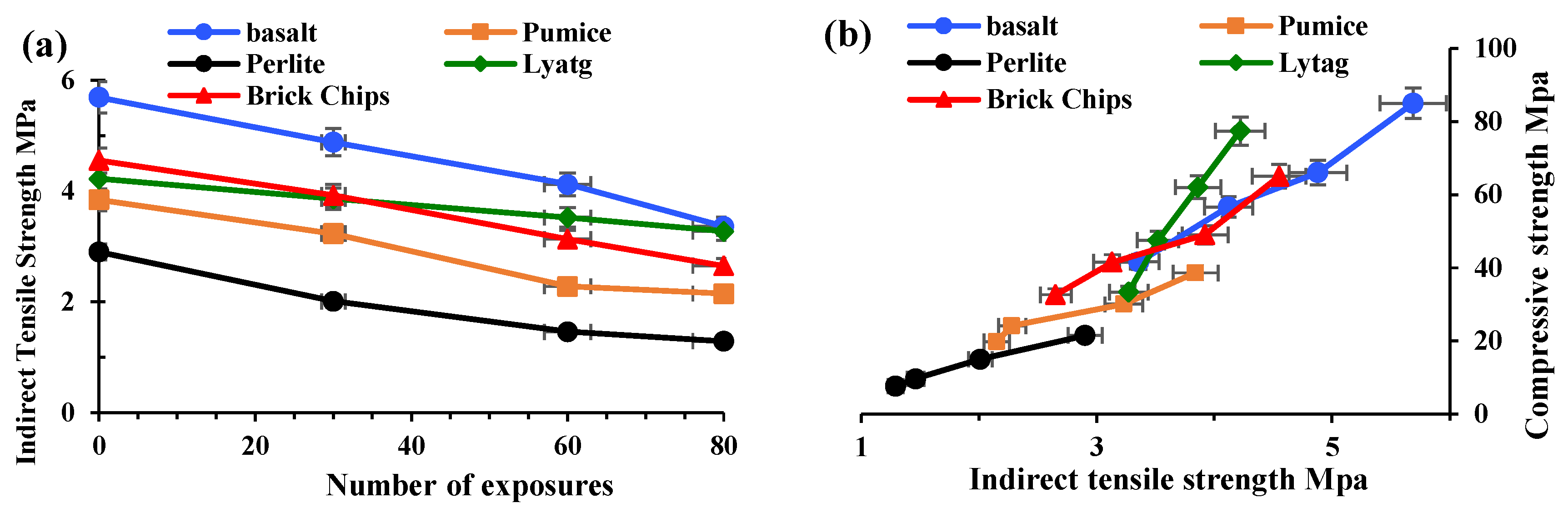
3.1.2. Indirect Tensile Strength
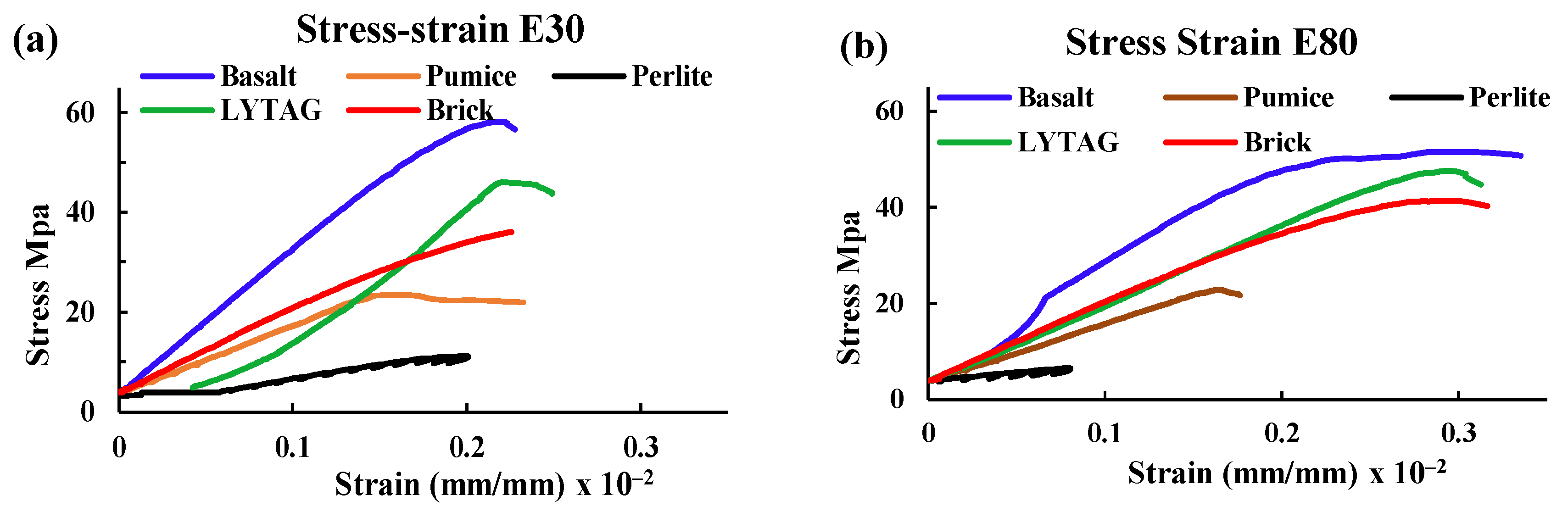
3.1.3. Stress–Strain
3.1.4. Concrete Spalling
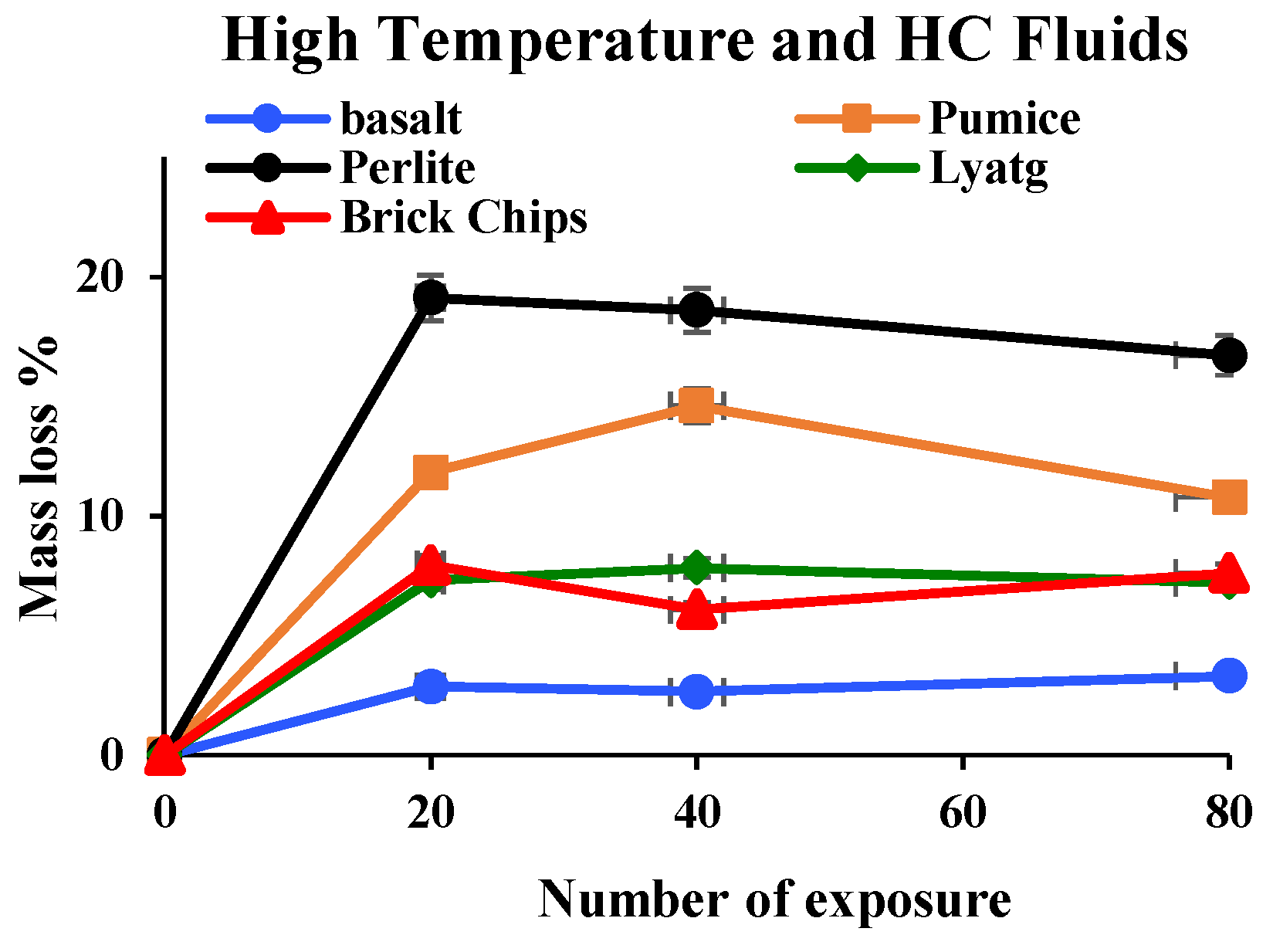
3.1.5. Mass Loss
3.2. Thermo-Mechanical Properties
3.2.1. Thermal Conductivity
3.2.2. Specific Heat
3.3. Thermochemical Properties
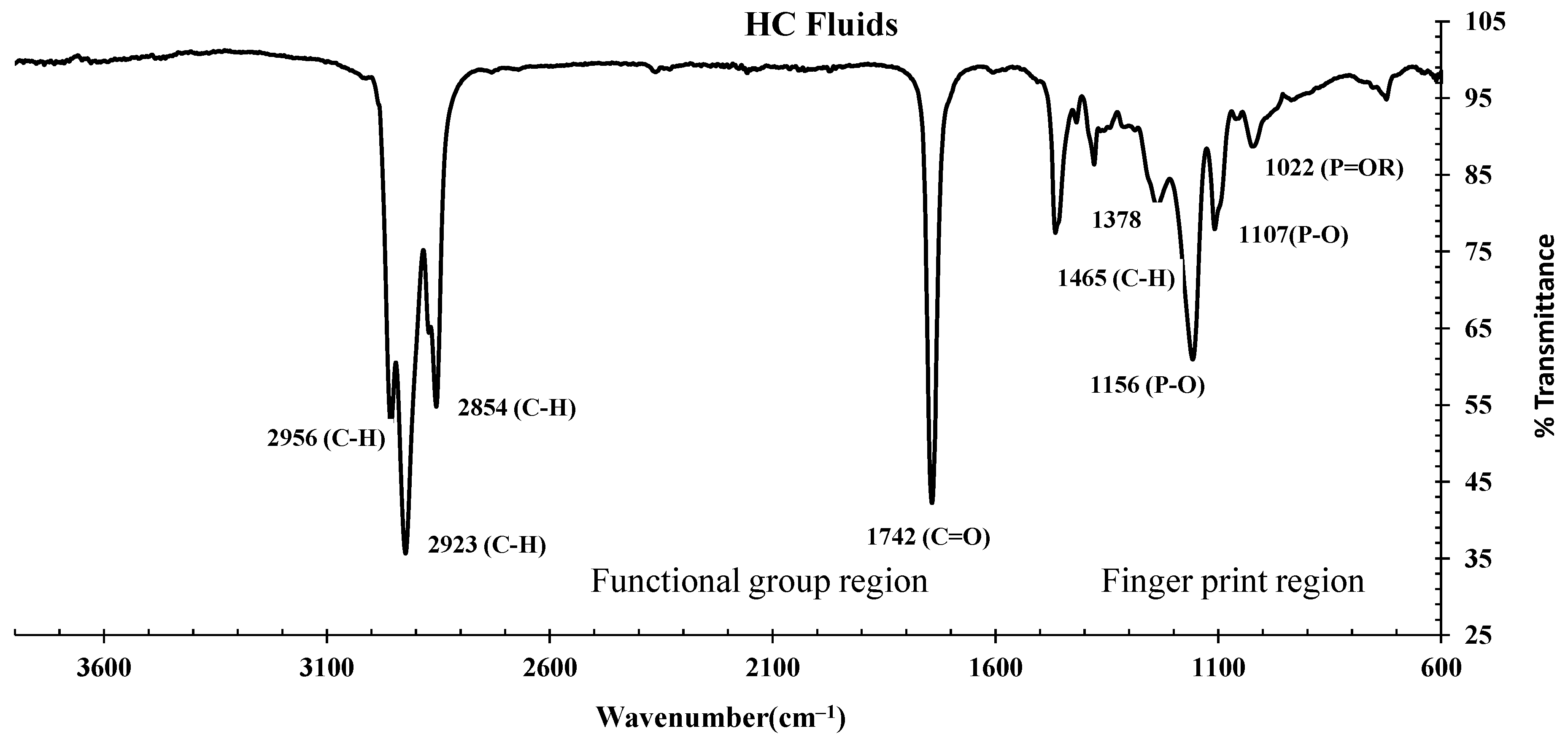
3.3.1. FTIR Spectrums of HC Fluids
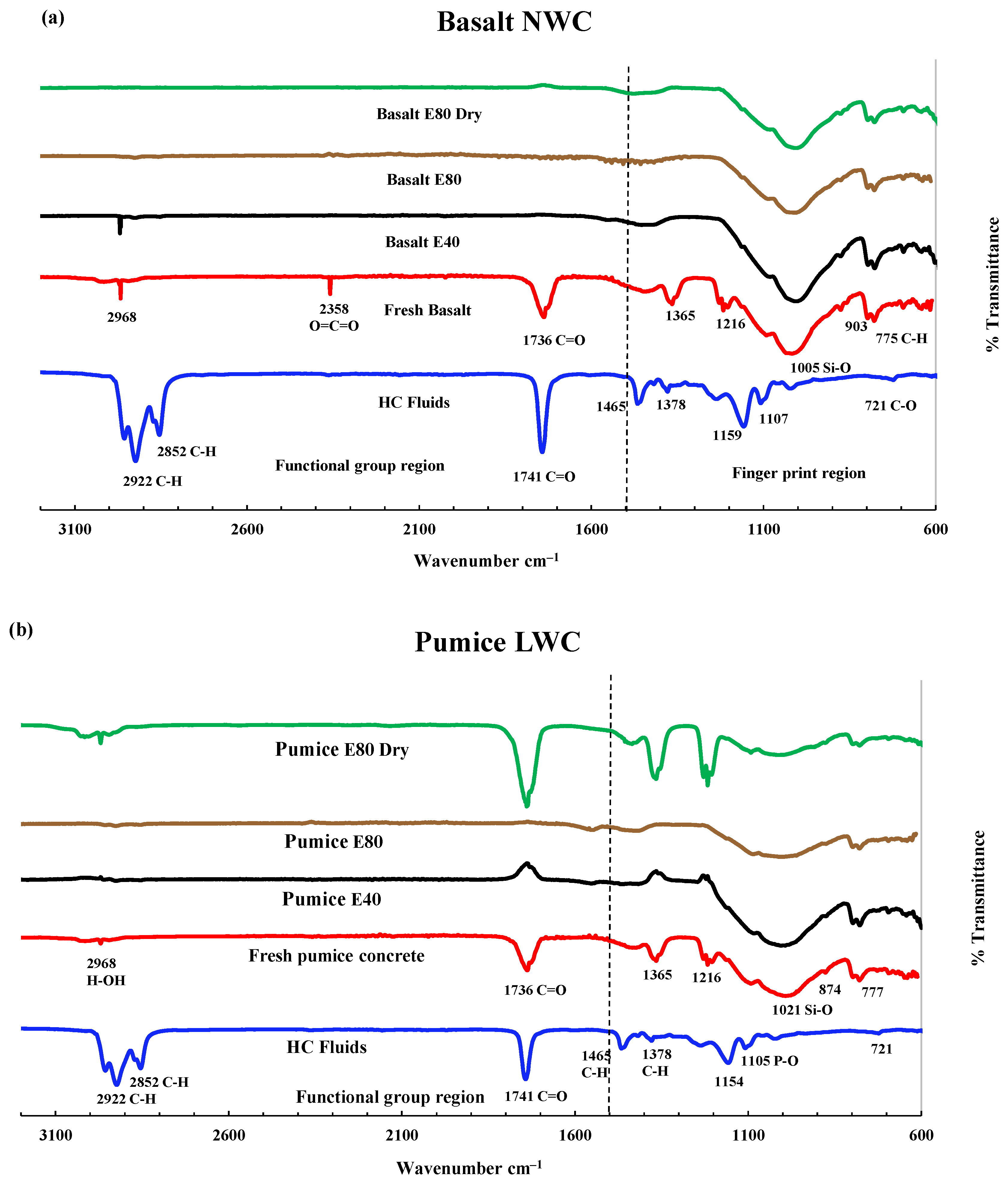
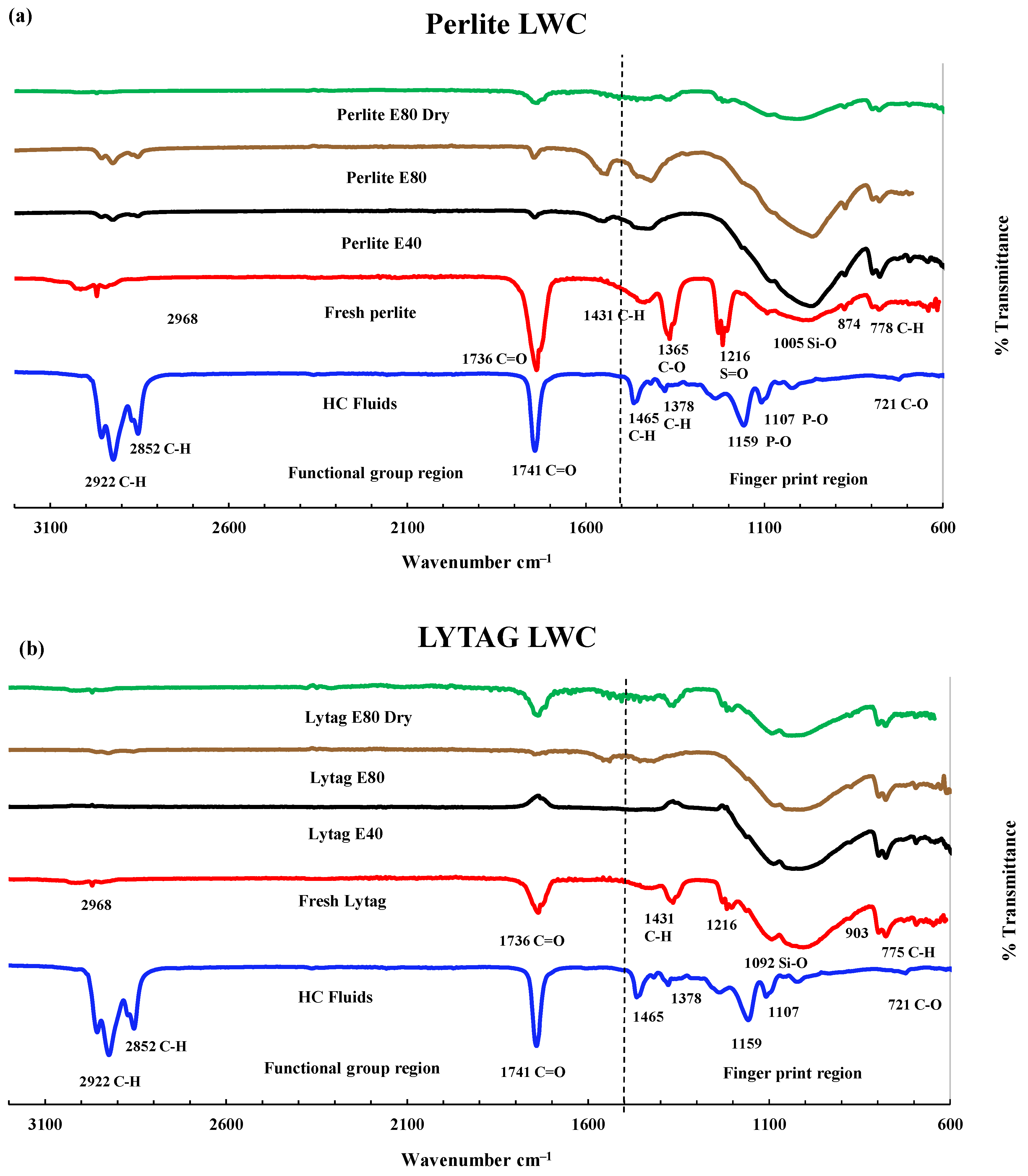
3.3.2. FTIR Analysis of LWAC Samples
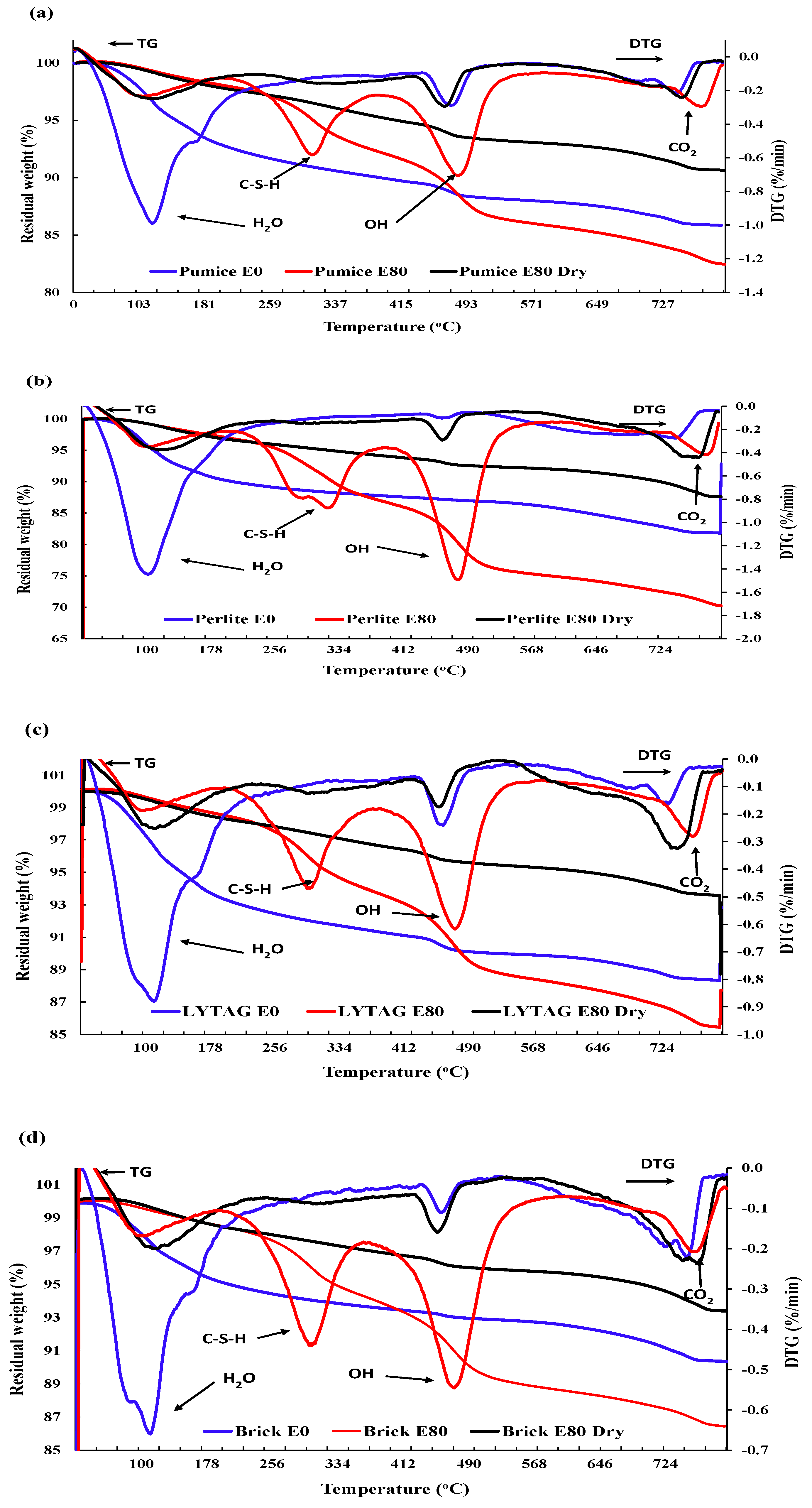
3.3.3. Thermogravimetric Analysis
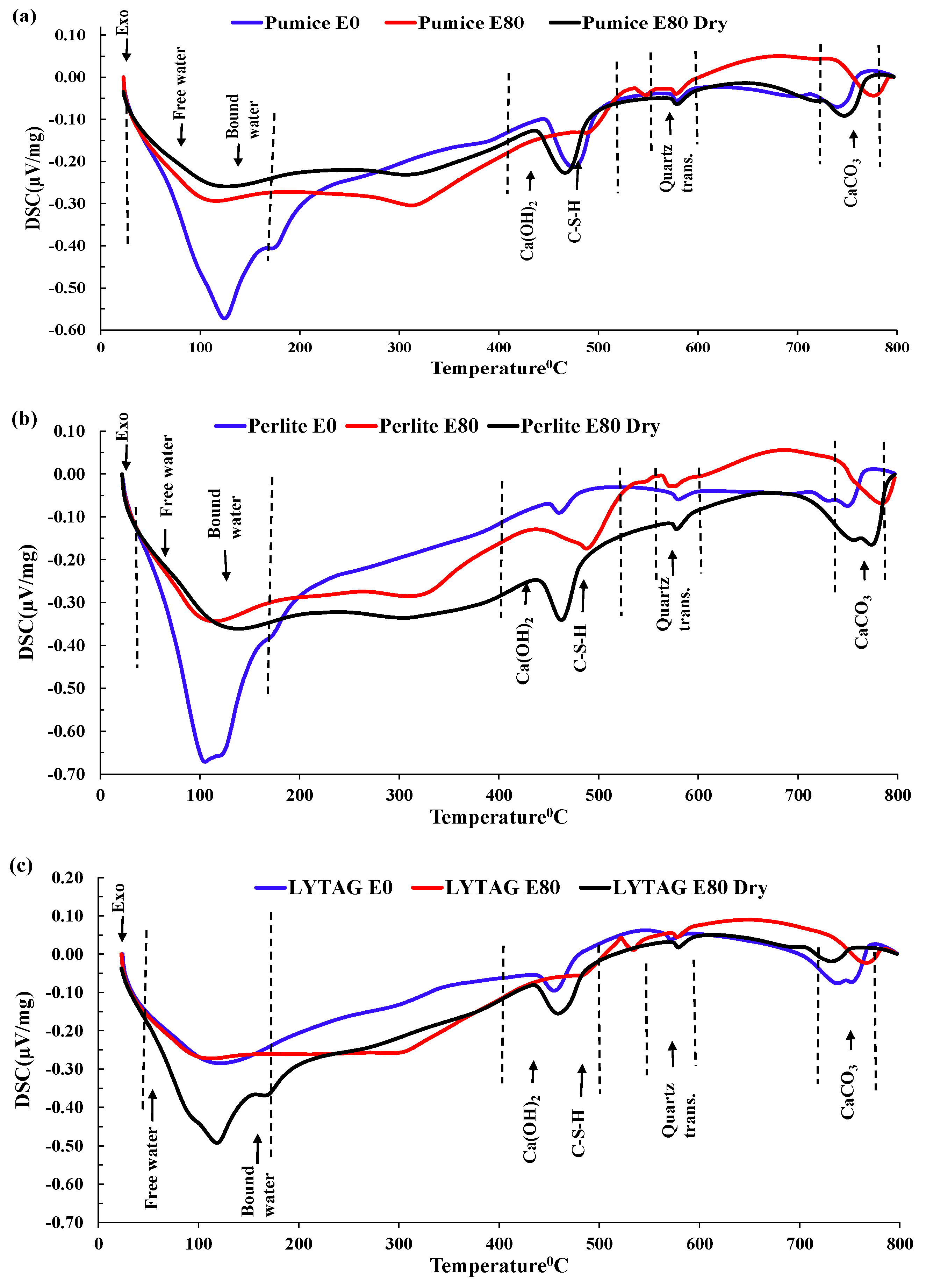
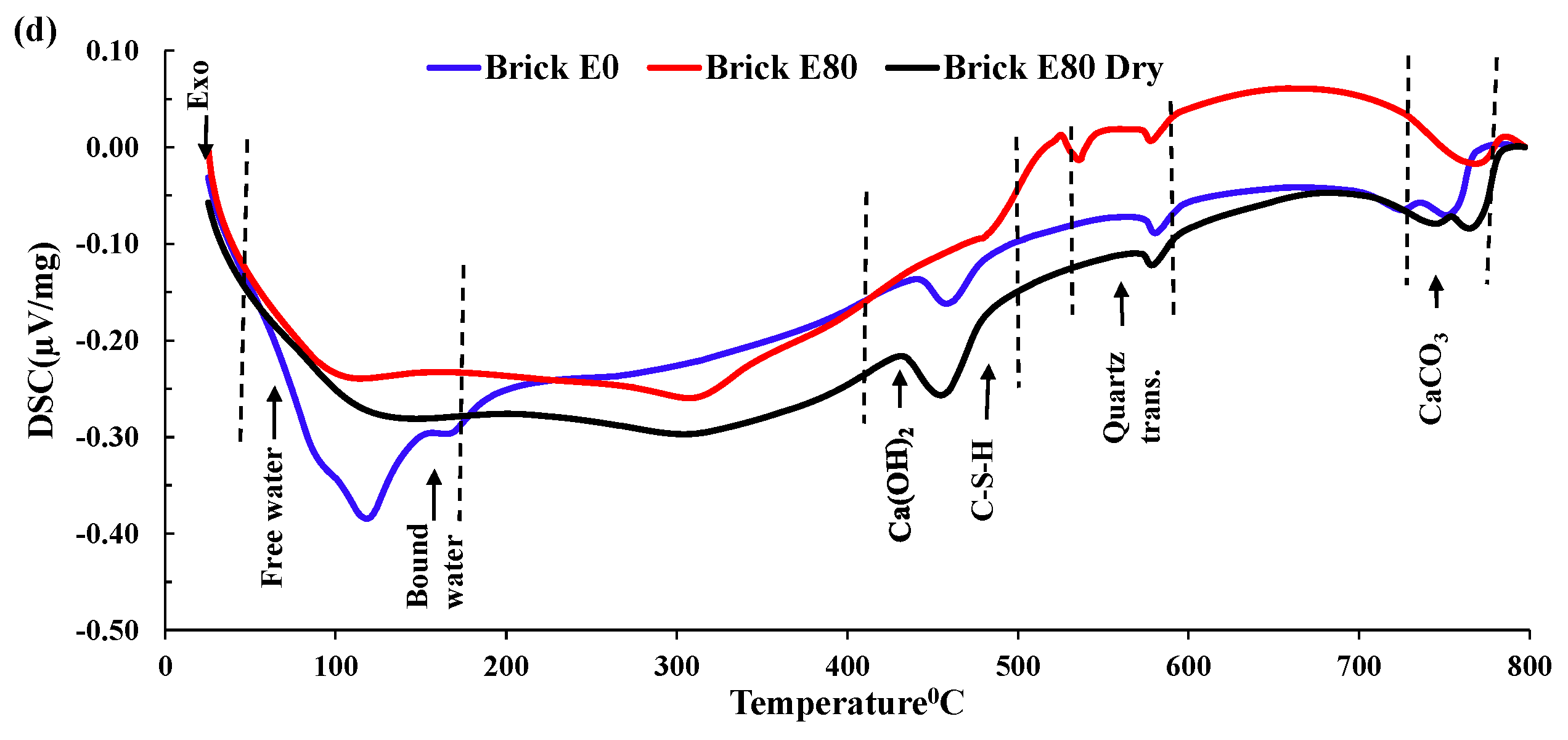
3.3.4. DSC Analysis
3.4. Morphology and Microcracks—Voids
3.4.1. Morphology of Aggregates
3.4.2. ITZ, Microcracks and Voids
4. Conclusions and Recommendation
- LWAC retained a moderate amount of residual mechanical strength after being exposed to repeated high temperatures and HC fluids. After 80 cycles of exposure to the coupled effects of HC fluids and high temperatures, both control and LWAC suffered a significant strength loss. Lytag and brick LWAC showed higher compressive strengths compared to other LWAC types and retained a residual compressive strength like the control specimen.
- The thermophysical properties of concrete are directly linked with the type and strength of the aggregate in concrete. LWAC showed much better thermal performances than NWA concrete under the repeated actions of HC fluids and high temperatures. Among the LWAC specimens used, perlite and pumice concrete specimens showed better thermal performances than lytag and brick aggregate concrete samples due to their porous microstructures.
- Mass loss was prominent for pumice and perlite aggregate concrete due to their higher percentages of porosity. TG and DSC tests showed that the decomposition of cement causes the evaporation of bound water, increasing the mass loss substantially.
- Concrete samples exposed to the coupled effects of HC fluids and high temperatures suffered from spalling damage. Basalt concrete (control) was detected to develop significant spalling. However, pumice, perlite and lytag LWAC experienced relatively lower spalling than did the control specimens. However, crushed brick concrete showed no spalling damage under the same exposure conditions.
- SEM scans revealed that the low porosity in basalt aggregate caused cracking in the cement paste and aggregates at elevated temperatures as the vapour pressure was not able to be released immediately. Therefore, basalt concrete experienced higher heat-induced microcracks compared to other LWAC tested.
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Khaiat, H.; Haque, M. Effect of initial curing on early strength and physical properties of a lightweight concrete. Cem. Concr. Res. 1998, 28, 859–866. [Google Scholar] [CrossRef]
- Hassanpour, M.; Shafigh, P.; Mahmud, H.B. Lightweight aggregate concrete fiber reinforcement—A review. Constr. Build. Mater. 2012, 37, 452–461. [Google Scholar] [CrossRef]
- Hossain, K.M.; Lachemi, M. Mixture design, strength, durability, and fire resistance of lightweight pumice concrete. ACI Mater. J. 2007, 104, 449. [Google Scholar]
- Yew, M.; Yew, M.; Beh, J.; Saw, L.; Lee, Y.; Lim, J. Fire resistance of lightweight foam concrete by incorporating lightweight bio-based aggregate. IOP Conf. Ser. Earth Environ. Sci. 2021, 920, 12009. [Google Scholar] [CrossRef]
- Hachemi, S.; Ounis, A. Performance of concrete containing crushed brick aggregate exposed to different fire temperatures. Eur. J. Environ. Civ. Eng. 2015, 19, 805–824. [Google Scholar] [CrossRef]
- Shideler, J.J. Lightweight-aggregate concrete for structural use. J. Proc. 1957, 54, 299–328. [Google Scholar]
- Lo, T.Y.; Tang, W.; Cui, H. The effects of aggregate properties on lightweight concrete. Build. Environ. 2007, 42, 3025–3029. [Google Scholar] [CrossRef]
- Demirdag, S.; Gunduz, L. Strength properties of volcanic slag aggregate lightweight concrete for high performance masonry units. Constr. Build. Mater. 2008, 22, 135–142. [Google Scholar] [CrossRef]
- Kockal, N.U.; Ozturan, T. Durability of lightweight concretes with lightweight fly ash aggregates. Constr. Build. Mater. 2011, 25, 1430–1438. [Google Scholar] [CrossRef]
- Hossain, K.M.A. Properties of volcanic pumice based cement and lightweight concrete. Cem. Concr. Res. 2004, 34, 283–291. [Google Scholar] [CrossRef]
- Go, C.-G.; Tang, J.-R.; Chi, J.-H.; Chen, C.-T.; Huang, Y.-L. Fire-resistance property of reinforced lightweight aggregate concrete wall. Constr. Build. Mater. 2012, 30, 725–733. [Google Scholar] [CrossRef]
- Demirboğa, R.; Örüng, İ.; Gül, R. Effects of expanded perlite aggregate and mineral admixtures on the compressive strength of low-density concretes. Cem. Concr. Res. 2001, 31, 1627–1632. [Google Scholar] [CrossRef]
- Poon, C.-S.; Azhar, S.; Anson, M.; Wong, Y.-L. Performance of metakaolin concrete at elevated temperatures. Cem. Concr. Compos. 2003, 25, 83–89. [Google Scholar] [CrossRef]
- Schneider, U. Concrete at high temperatures—A general review. Fire Saf. J. 1988, 13, 55–68. [Google Scholar] [CrossRef]
- Hager, I.; Tracz, T.; Śliwiński, J.; Krzemień, K. The influence of aggregate type on the physical and mechanical properties of high-performance concrete subjected to high temperature. Fire Mater. 2016, 40, 668–682. [Google Scholar] [CrossRef]
- Yehia, S.; AlHamaydeh, M.; Farrag, S. High-strength lightweight SCC matrix with partial normal-weight coarse-aggregate replacement: Strength and durability evaluations. J. Mater. Civ. Eng. 2014, 26, 04014086. [Google Scholar] [CrossRef]
- Shill, S.K.; Al-Deen, S.; Ashraf, M. Concrete durability issues due to temperature effects and aviation oil spillage at military airbase–A comprehensive review. Constr. Build. Mater. 2018, 160, 240–251. [Google Scholar] [CrossRef]
- Shill, S.K.; Al-Deen, S.; Ashraf, M.; Hossain, M.M. Residual properties of conventional concrete repetitively exposed to high thermal shocks and hydrocarbon fluids. Constr. Build. Mater. 2020, 252, 119072. [Google Scholar] [CrossRef]
- Shill, S.K.; Al-Deen, S.; Ashraf, M.; Elahi, M.A.; Subhani, M.; Hutchison, W. A comparative study on the performance of cementitious composites resilient to airfield conditions. Constr. Build. Mater. 2021, 282, 122709. [Google Scholar] [CrossRef]
- McVay, M.; Rish, J., III; Sakezles, C.; Mohseen, S.; Beatty, C. Cements resistant to synthetic oil, hydraulic fluid, and elevated temperature environments. Mater. J. 1995, 92, 155–163. [Google Scholar]
- Bingöl, A.F.; Gül, R. Compressive strength of lightweight aggregate concrete exposed to high temperatures. Ind. J. Eng. Mater. Sci. 2004, 11, 68–72. [Google Scholar]
- AS 3972-2010; General Purpose and Blended Cements. Australian Standard: Sydney, Australia, 2010.
- AS 1012.9:2014; Methods of Testing Concrete, Compressive Strength Tests—Concrete, Mortar and Grout Specimens. Australian Standard: Sydney, Australia, 2014.
- ASTM C518-21; Standard Test Method for Steady-State Thermal Transmission Properties by Means of the Heat Flow Meter Apparatus. ASTM International: West Conshohocken, PA, USA, 2017.
- McVay, M.C.; Smithson, L.D.; Manzione, C. Chemical damage to airfield concrete aprons from heat and oils. Mater. J. 1993, 90, 253–258. [Google Scholar]
- Shannag, M. Characteristics of lightweight concrete containing mineral admixtures. Constr. Build. Mater. 2011, 25, 658–662. [Google Scholar] [CrossRef]
- Hossain, M.M.; Al-Deen, S.; Hassan, M.K.; Shill, S.K.; Kader, M.A.; Hutchison, W. Mechanical and thermal properties of hybrid fibre-reinforced concrete exposed to recurrent high temperature and aviation oil. Materials 2021, 14, 2725. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.Y.N.; Peng, G.-F.; Anson, M. Fire behavior of high-performance concrete made with silica fume at various moisture contents. Mater. J. 1999, 96, 405–409. [Google Scholar]
- Dwaikat, M.B.; Kodur, V. Hydrothermal model for predicting fire-induced spalling in concrete structural systems. Fire Saf. J. 2009, 44, 425–434. [Google Scholar] [CrossRef]
- Shill, S.K.; Al-Deen, S.; Ashraf, M.; Rashed, M.; Hutchison, W. Consequences of aircraft operating conditions at military airbases: Degradation of ordinary mortar and resistance mechanism of acrylic and silica fume modified cement mortar. Road Mater. Pavement Des. 2022, 23, 98–111. [Google Scholar] [CrossRef]
- Kalifa, P.; Chene, G.; Galle, C. High-temperature behaviour of HPC with polypropylene fibres: From spalling to microstructure. Cem. Concr. Res. 2001, 31, 1487–1499. [Google Scholar] [CrossRef]
- Tandiroglu, A. Temperature-dependent thermal conductivity of high strength lightweight raw perlite aggregate concrete. Int. J. Thermophys. 2010, 31, 1195–1211. [Google Scholar] [CrossRef]
- Oktay, H.; Yumrutaş, R.; Akpolat, A. Mechanical and thermophysical properties of lightweight aggregate concretes. Constr. Build. Mater. 2015, 96, 217–225. [Google Scholar] [CrossRef]
- Kodur, V.; Sultan, M. Effect of temperature on thermal properties of high-strength concrete. J. Mater. Civ. Eng. 2003, 15, 101–107. [Google Scholar] [CrossRef]
- Demirboğa, R.; Gül, R. The effects of expanded perlite aggregate, silica fume and fly ash on the thermal conductivity of lightweight concrete. Cem. Concr. Res. 2003, 33, 723–727. [Google Scholar] [CrossRef]
- Daasch, L.; Smith, D. Infrared spectra of phosphorus compounds. Anal. Chem. 1951, 23, 853–868. [Google Scholar] [CrossRef]
- Arai, Y.; Sparks, D.L. ATR–FTIR spectroscopic investigation on phosphate adsorption mechanisms at the ferrihydrite–water interface. J. Colloid Interface Sci. 2001, 241, 317–326. [Google Scholar] [CrossRef]
- Mendes, A.; Gates, W.P.; Sanjayan, J.G.; Collins, F. NMR, XRD, IR and synchrotron NEXAFS spectroscopic studies of OPC and OPC/slag cement paste hydrates. Mater. Struct. 2011, 44, 1773–1791. [Google Scholar] [CrossRef]
- Sanati, M.; Andersson, A. DRIFT study of the oxidation and the ammoxidation of toluene over a TiO2 (B)-supported vanadia catalyst. J. Mol. Catal. 1993, 81, 51–62. [Google Scholar] [CrossRef]
- Varas, M.; De Buergo, M.A.; Fort, R. Natural cement as the precursor of Portland cement: Methodology for its identification. Cem. Concr. Res. 2005, 35, 2055–2065. [Google Scholar] [CrossRef]
- Fares, H.; Remond, S.; Noumowé, A.; Cousture, A. Microstructure et propriétés physico-chimiques de bétons autoplaçants chauffés de 20 à 600 °C. Eur. J. Environ. Civ. Eng. 2011, 15, 869–888. [Google Scholar]
- Alarcon-Ruiz, L.; Platret, G.; Massieu, E.; Ehrlacher, A. The use of thermal analysis in assessing the effect of temperature on a cement paste. Cem. Concr. Res. 2005, 35, 609–613. [Google Scholar] [CrossRef]
- Way, R.; Wille, K. Effect of heat-induced chemical degradation on the residual mechanical properties of ultrahigh-performance fiber-reinforced concrete. J. Mater. Civ. Eng. 2016, 28, 04015164. [Google Scholar] [CrossRef]
- Khoury, G. Compressive strength of concrete at high temperatures: A reassessment. Mag. Concr. Res. 1992, 44, 291–309. [Google Scholar] [CrossRef]
- Ye, G.; Liu, X.; De Schutter, G.; Taerwe, L.; Vandevelde, P. Phase distribution and microstructural changes of self-compacting cement paste at elevated temperature. Cem. Concr. Res. 2007, 37, 978–987. [Google Scholar] [CrossRef]
- Wu, T.; Yang, X.; Wei, H.; Liu, X. Mechanical properties and microstructure of lightweight aggregate concrete with and without fibers. Constr. Build. Mater. 2019, 199, 526–539. [Google Scholar] [CrossRef]









| Chemical Analysis (% by Mass) | Pumice | Perlite | Lytag | Brick | Basalt | Portland Cement | GPC * |
|---|---|---|---|---|---|---|---|
| Calcium oxide (CaO) | 3.37 | 2.14 | 3.44 | 8.095 | 4.64 | 64.5 | 4.29 |
| Silica (SiO2) | 71.41 | 75.64 | 41.35 | 56.67 | 58.24 | 20.20 | 60.06 |
| Alumina (Al2O3) | 10.46 | 10.91 | 20.89 | 11.76 | 15.72 | 4.8 | 17.52 |
| Iron oxide (Fe2O3) | 6.70 | 2.81 | 23.54 | 15.03 | 9.79 | 3.1 | 10.68 |
| Sulphur trioxide (SO3) | 0.86 | 0.81 | 1.09 | 0.44 | 0.668 | 2.70 | 0.901 |
| Magnesia (MnO) | 0.21 | 0.22 | 0.152 | 0.30 | 0.137 | 1.20 | 0.228 |
| Titanium oxide (TiO2) | 0.65 | - | 1.17 | 1.07 | 1.09 | 0.52 | 2.15 |
| Potassium Oxide (K2O) | 6.00 | 7.40 | 5.59 | 5.68 | 9.13 | 0.67 | 3.56 |
| Specific Gravity | 1.5 | 0.30 | 2.10 | 2.66 | - | - | |
| Moisture % | 45 | 35 | 15 | 12.5 | 4.3 | - | - |
| Designation of Mixture | Cement (kg/m3) | Sand (kg/m3) | Coarse Aggregate (kg/m3) | W/C Ratio | Superplasticizer (kg/m3) | Dry Unit Weight of Concrete (kg/m3) |
|---|---|---|---|---|---|---|
| Control | 462 | 792 | 1012 | 0.45 | 0 | 2323 |
| Pumice | 462 | 714 | 368 | 0.45 | 1.0 | 1897 |
| Perlite | 320 | 714 | 407 | 0.37 | 5.0 | 1578 |
| Lytag | 280 | 800 | 835 | 0.45 | 4.8 | 1909 |
| Crushed Brick | 462 | 750 | 1035 | 0.45 | 3.0 | 2094 |
| Types/Loss | Thermal Conductivity % | Specific Heat % | ||
|---|---|---|---|---|
| Heat Exposed | HC and Heat Exposed | Heat Exposed | HC and Heat Exposed | |
| Basalt | 63.91 | 61.94 | 35.04 | 31.13 |
| Brick | 54.68 | 37.09 | 16.34 | 10.26 |
| Lytag | 40.12 | 38.97 | 7.44 | 9.94 |
| Pumice | 39.86 | 33.57 | 15.85 | 15.94 |
| Perlite | 37.96 | 46.87 | 16.07 | 20.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hossain, M.M.; Al-Deen, S.; Shill, S.K.; Hassan, M.K. Resistance of Concrete with Various Types of Coarse Aggregate to Coupled Effects of Thermal Shocks and Chemicals. Materials 2024, 17, 791. https://doi.org/10.3390/ma17040791
Hossain MM, Al-Deen S, Shill SK, Hassan MK. Resistance of Concrete with Various Types of Coarse Aggregate to Coupled Effects of Thermal Shocks and Chemicals. Materials. 2024; 17(4):791. https://doi.org/10.3390/ma17040791
Chicago/Turabian StyleHossain, Muhammad Monowar, Safat Al-Deen, Sukanta Kumer Shill, and Md Kamrul Hassan. 2024. "Resistance of Concrete with Various Types of Coarse Aggregate to Coupled Effects of Thermal Shocks and Chemicals" Materials 17, no. 4: 791. https://doi.org/10.3390/ma17040791






