3.1. Microstructure Analysis
The SEM micrographs and X-ray diffraction (XRD) images of the Al-6Cu-xMn-yAg alloy are shown in
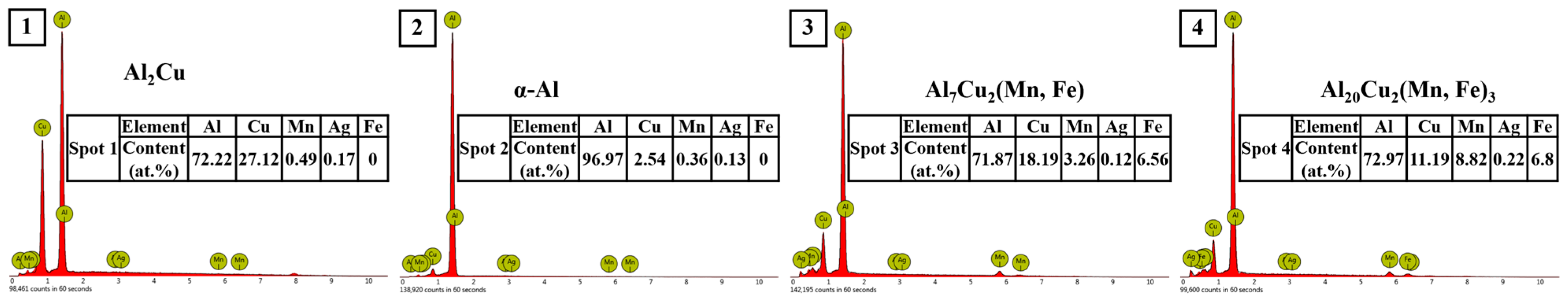
Figure 3. The chemical composition and energy dispersion spectrum (EDS) analyses of each phase of the alloy are shown in
Figure 4. As shown in
Figure 4, the as-cast microstructure of the alloy was mainly composed of α-Al, Al
2Cu, Al
20Cu
2Mn
3, and Al
7Cu
2Fe phases.
The dark gray α-Al (spot 4) was the matrix phase with a dendritic structure. We performed EDS analyses on the bright white reticular phase in the image (spots 1, 2, and 5), and the results showed that this phase was mainly composed of Al and Cu elements, and the atomic ratio of the Al and Cu elements was close to 2:1. Combined with the obvious Al2Cu diffraction peak found in the XRD diffraction pattern, we believe that it should be the Al2Cu phase. Some α-Al+Al2Cu eutectic network structures are distributed along α-Al grain boundaries or between dendrites. The sub-eutectic composition of Cu determines this microstructural characteristic. In the Al-Cu alloy of this experiment, the composition of Cu was 6 wt.%, which was much lower than the eutectic point composition (33.2 wt.%). Therefore, α-Al grains became the primary phases. With the continuous growth of the α-Al grains, aluminium atoms were constantly consumed, and Cu atoms were continuously enriched to the front of the solidification interface. When the concentration of Cu in the residual liquid reached 33.2 wt.%, a eutectic reaction occurred as follows: L→α-Al+Al2Cu; therefore, the eutectic network structure of both was distributed at the grain boundaries or interdendritic zone.
Further observation showed some light gray phases (spot 3), and some long strips (spot 6) were observed within part of the bright white phase. According to the EDS analysis, both phases contained large amounts of Al, Cu, Mn, and Fe elements. In the light gray phase (Spot 3), the content of Al was 74.9 at.%, the content of Cu was 18.2 at.%, and the total content of Mn and Fe was 6.67at.%. The peak of the Al
7Cu
2Fe phase appeared in the XRD diffraction pattern, and combined with the reference [
47], we believe that this phase was the Al
7Cu
2Fe phase. In other elongated phases (spot 6), the content of Al was 72.71 at.%, the content of Cu was 11.22 at.%, and the total content of Mn and Fe was 15.94 at.%. The peak of the Al
20Cu
2Mn
3 phase appeared in the XRD diffraction pattern, and combined with the reference [
48], we believe that this phase was the Al
20Cu
2Mn
3 phase. According to a previous study [
49], this was due to the phenomenon of mutual substitution between Fe and Mn atoms, resulting in the solid solution of the Fe and Mn elements into the original Al
7Cu
2Fe and Al
20Cu
2Mn
3 phases, respectively, forming Al
7Cu
2(Fe, Mn) and Al
20Cu
2(Mn, Fe)
3 phases. It seems that there was no significant change in the size and quantity of the α-Al grains and Al+Al
2Cu eutectic network structure as the Mn/Ag ratio increased.
3.2. Tensile Properties of as-Cast Al-Cu-xMn-yAg Alloys
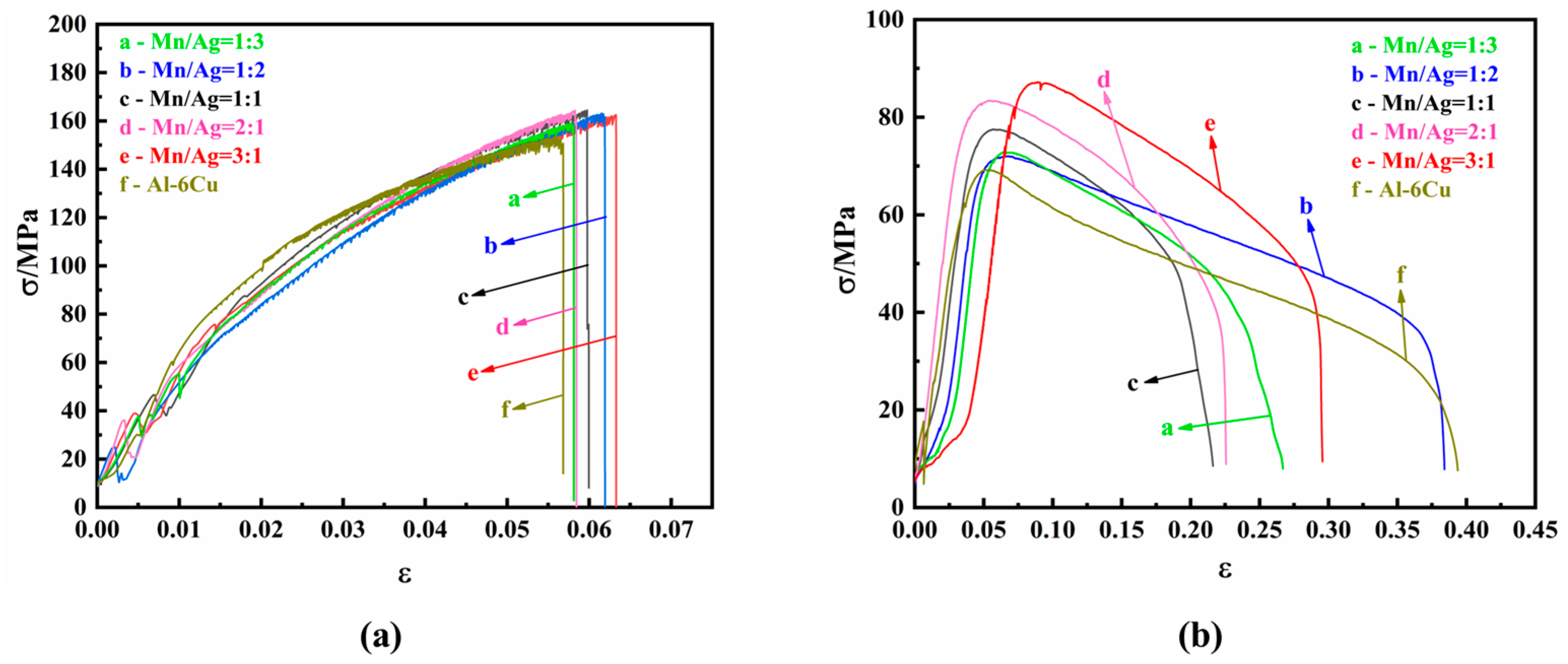
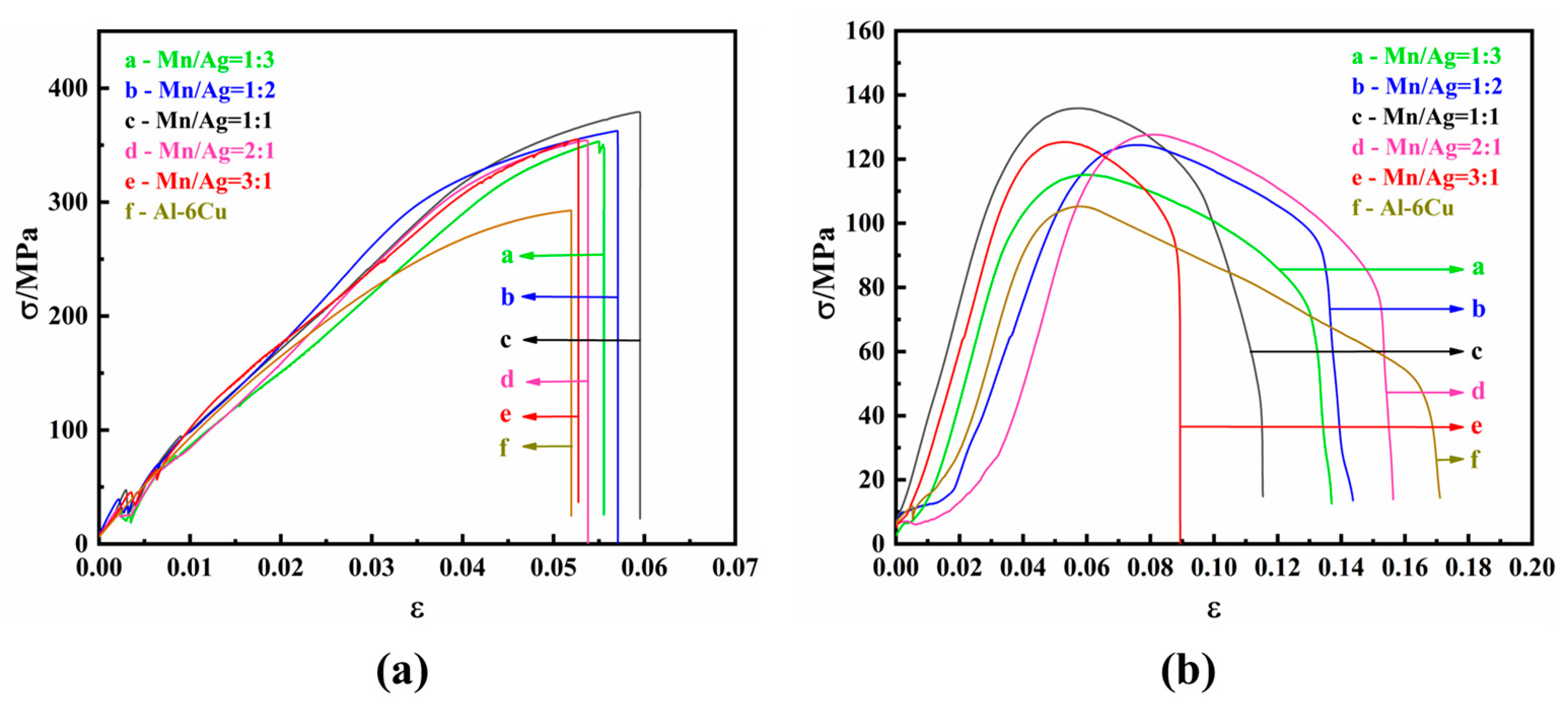
The mechanical properties of the as-cast alloys were studied by tensile tests at room temperature and a high temperature (350 °C). The tensile strain–stress curves are shown in
Figure 5, and the specific experimental data are shown in
Table 2 and
Table 3.
It can be seen from
Table 2 and
Figure 5a that the as-cast alloy had no noticeable difference in tensile properties at room temperature. When the Mn/Ag ratio was 2:1, the as-cast alloy had the best mechanical properties; its tensile strength was 164.34 MPa, and its elongation was 2.94%. The results showed that the Mn/Ag ratio change did not affect the tensile properties of the as-cast alloys at room temperature, which was consistent with the fact that the change in the Mg/Ag ratio had a relatively small impact on the microstructure of the as-cast alloy.
It can be seen from
Table 3 and
Figure 5b that the tensile properties of the as-cast alloy at a high temperature (350 °C) were significantly different from those at room temperature. With a decrease of the Mn/Ag ratio, the tensile strength of the cast alloy gradually decreased, and the elongation decreased first and then increased. When the Mn/Ag ratio was 3:1, the as-cast alloy had the best tensile strength, reaching 87.17 MPa, and the elongation after fracture was 25%. The above results show that Mn was dominant in strengthening the as-cast alloy at 350 °C.
According to the literature [
50], the bond strength is mainly controlled by the valence electrons of each atom. The more bonding electrons, the more muscular the bonding strength of the crystal structure, which is conducive to improving the high-temperature thermal strength; additionally, the atomic radii of Al, Cu, and Mn are 143.2 pm, 125 pm, and 139 pm [
51], respectively. Therefore, when Mn atoms replace some Cu atoms, specific geometric distortion will be caused, significantly affecting the redistribution and energetics of valence electrons [
52]. Mn belongs to the transitional family of elements and has an incomplete outer electron shell. After Mn atoms enter the lattice, some high-priced Mn atoms (+2~7) will replace low-priced Cu atoms (+1~2), causing local geometric distortion and losing more valence electrons. We infer that this will decrease local valence electron density and increase charge transfer, thus strengthening the interaction between Mn and the surrounding atoms. Thus, the structural stability of the Al
2Cu phase at high temperatures can be effectively improved. Although Ag atoms also appear in the Al
2Cu phase, like Mn atoms, the thermal stability of the Al
2Cu phase is limited due to the lower valence state of Ag atoms (+1). Therefore, this explains why the mechanical properties of the alloy increase with an increase in Mn content at 350 °C.
Figure 6 shows the room temperature and high temperature (350 °C) tensile fracture morphology of the as-cast Al-6Cu-0.4Mn-0.4Ag alloy. It can be seen from
Figure 6a that the fracture surface had some shallow dimples and some smooth cleavage planes. The analysis showed that the fracture mode of the alloy was mainly brittle fracture, which was consistent with the lower elongations of the alloys listed in
Table 2. The analysis showed that the cleavage planes and fragmented blocks were generated by the Al
2Cu phase. The Al
2Cu phase was a brittle intermetallic compound, and its coarse network structure led to stress concentrations that were apt to occur in this phase, ultimately resulting in cleavage planes and fragmentation, which accelerated the fracture of the alloy and could not effectively improve the tensile property of the alloy at room temperature. As can be seen from the high-temperature tensile fracture of the alloy in
Figure 6b, many dimples covered the surface of the tensile fracture. These thick and deep dimples indicated that the alloy underwent large plastic deformation before fracture, and dislocation slip and movement were apparent. The plasticity of the alloy was considerable because the matrix softens at high temperatures, reducing sensitivity to interface cracks, which could passivate the tip of the interface cracks, delay crack propagation, and enable Al
2Cu particles to play a precipitation-strengthening role resulting in no rapid fracture failure in the tensile process. At the bottom of the dimple were some white Al
2Cu phase deposits, which indicated that the Al
2Cu particles had a good strengthening effect.
3.3. Microstructure of the Heat-Treated Alloy
Figure 7 shows the microstructure and XRD patterns of the heat-treated alloys with different Mn/Ag ratios.
Figure 7a–d,e are the low-magnification images, and
Figure 7a
1–d
1,e
1 are the high-magnification images of the samples after metallographic corrosion.
Figure 8 shows the microstructure of the as-cast and T6 heat-treated Al-6Cu-0.4Mn-0.4Ag alloys. The EDS analysis results of the chemical composition of each phase are shown in
Figure 9. It can be seen from the microstructure of the SEM photos in
Figure 7 and the EDS components of each phase in
Figure 9 that the heat-treated alloy was mainly composed of α-Al, θ′-Al
2Cu, Al
20Cu
2(Mn, Fe)
3, and a small amount of Al
7Cu
2(Fe, Mn) phases.
During the heat treatment process, the alloys are heated to more than 500 °C, and most of the cast network θ-Al
2Cu will decompose and dissolve, and Cu solute atoms will dissolve into the Al matrix to form a single-phase solid solution of α-Al. However, there are still some coarse Al
2Cu phases in the aluminium matrix. We know that the limit solid solubility of Cu in aluminium is 5.65 wt.% [
53], and the designed alloy has a copper content of 6%, so a small amount of the θ-Al
2Cu phase will not dissolve into the matrix. According to Oswald’s ripening theory [
54], the energy of large particles is lower than that of small particles, and large particles are more stable than small ones, so small particles in the thermodynamic system will be swallowed by large particles [
55]. The residual θ-Al
2Cu in this experiment would also grow into coarse particles through the Oswald ripening mechanism during the solution–aging process. As shown in
Figure 7a–e, the remaining Al
2Cu particles were relatively large.
After aging treatment, fine dispersed θ′-Al
2Cu particles (100–400 nm) were precipitated from the alloy matrix, as shown in the high-magnification microstructure diagram (a
1–e
1) in
Figure 7. With the increase in Mn content in the Mn/Ag ratio, the quantity and size of θ′-Al
2Cu particles first increased and then decreased. When the Mn/Ag ratio was 1:1 (
Figure 7c), θ′-Al
2Cu particles had the most significant number and the smallest particle size, with an average particle size of 136 nm. This showed that the Mn/Ag ratio change specifically affected the quantity and size of the precipitated phase. Comparing the lengths of the red line segments in
Figure 8a,b showed that the α-Al dendrites were coarsened after heat treatment. The main reason was that intense thermal diffusion occurred during the solution treatment at more than 500 °C to make the grains merge and grow [
55]. These phenomena were consistent with some previous research [
56].
In the heat treatment process, Mn can promote the precipitation and refinement of the θ′-Al
2Cu phase, which is due to the low thermal diffusion coefficient of Mn, which will block the diffusion of Al and Cu, and it is easy to form a blockage. As the number of blocked atoms increases and the aggregated atomic clusters reach a specific critical size, nucleation begins. The increase in the amount of θ′-Al
2Cu phase precipitation represents a decrease in the size of each phase [
57]. At present, although there are no studies showing that the Ag element can refine the θ′-Al
2Cu phase by promoting the precipitation of the θ′-Al
2Cu phase, since the Ag element can segregate to the coherent interface of the θ′-Al
2Cu phase, forming an Ag-rich double layer, thereby reducing the interfacial energy and improving the thermal stability of the phase, at the same time, the Ag element will also segregate to approximately 40% of the semi-coherent lattice θ′/matrix interface, which can hinder the lateral growth of the θ′-Al
2Cu phase and cooperate with the Mn element to further refine the θ′-Al
2Cu phase [
40].
3.4. Tensile Properties of the Heat-Treated Alloys
The mechanical properties of the heat-treated alloys were studied by tensile tests at room temperature and a high temperature (350 °C). The tensile strain–stress curves are shown in
Figure 10, and the specific experimental data are shown in
Table 4 and
Table 5.
It can be seen that, at room temperature, with an increase of the Mn/Ag ratio, the tensile strength of the heat-treated alloy first increased and then decreased. When the Mn/Ag ratio was 1:1, the tensile mechanical properties of the heat-treated alloy were the best, reaching 379.14 MPa, 131.18% higher than that of the as-cast alloy, and the elongation after fracture was 2.87%. The above results showed that solution–aging heat treatment could significantly improve the mechanical properties of the alloy at room temperature.
It can be seen from
Table 4 and
Figure 10b that, at a high temperature (350 °C), with an increase of the Mn/Ag ratio, the tensile strength of the heat-treated alloy also first increased and then decreased, and the fracture elongation was the same. When the Mn/Ag ratio was 1:1, the tensile mechanical properties were the best, reaching 135.89 MPa, which was 75.30% higher than that of the as-cast alloy, and the elongation was 11.92%. The above results showed that heat treatment was essential in strengthening the alloy at high temperatures.
Table 6 shows some heat-resistant aluminium alloys’ tensile strength at 350 °C reported in recent years. It can be seen that the maximum high-temperature tensile strength of this study exceeded that of most heat-resistant aluminium alloys reported in recent years., indicating that it has potential high-temperature applications. The improvement of tensile properties at room temperature and high temperatures after heat treatment was mainly attributed to the dispersion strengthening of the fine dispersed θ′-Al
2Cu nanophase. The Orowan strengthening mechanism could explain this.
Orowan strengthening describes the strengthening effect resulting from the interaction between the reinforced particles and the dislocation motion in the matrix; that is, the second phase of dispersion in the matrix impedes the dislocation motion, thereby improving the material’s mechanical properties [
58]. The Orowan bypass stress Δ
σOrowan is expressed as follows [
59]:
where M is the average orientation factor (
MAl = 3.06), λ is the spacing between particles,
dp is the average particle size,
b is the Burgers vector,
VP is the volume fraction of strengthened particles,
Gm is the shear modulus of the matrix, and ν is the Poisson’s ratio. Orowan strengthening is usually initiated by particles smaller than 1 μm in size, and the θ′ phase in the heat-treated alloy in this study was between 0.1 and 0.4 μm in size. In this experiment, when the Mn/Ag ratio was 1:1, the θ′-Al
2Cu precipitates were the of an enormous amount and the smallest size, so the highest tensile mechanical properties were obtained, which accorded with the Orowan strengthening mechanism.
Table 6.
Comparison of tensile strength with other heat-resistant aluminum alloys at 350 °C.
Table 6.
Comparison of tensile strength with other heat-resistant aluminum alloys at 350 °C.
| Materials Composition (wt.%) | Temperature (°C) | σUTS (MPa) | Year | Ref. |
|---|
| Al-6Cu-0.4Mn-0.4Ag | 350 | 135.89 | 2024 | Present work |
| Al-5Cu-1.5Ni-0V | 350 | 92.06 | 2023 | [55] |
| Al-5Cu-1.5Ni-0.3V | 350 | 111.81 | 2023 | [55] |
| Al-6.5Cu-2Ni-0.5Zr-0.3Ti-0.25V | 350 | 127.5 | 2023 | [60] |
| Al-6Cu-2Ni-0.5V | 350 | 123.4 | 2023 | [57] |
| 9 wt.%Al3Zr/Al-6Cu-2Ni-0.5V | 350 | 136.9 | 2023 | [57] |
| Al-11.93Si-4.03Cu-0.97Mg-2.67Ni-0.52Fe | 350 | 103 | 2022 | [61] |
| rGO/Al | 350 | 128 | 2020 | [62] |
| (2% Al3Zr + 15.2% Al3Ni)/Al-1Mg-0.8Mn-0.8V | 350 | 82 | 2020 | [63] |
| Al-12.16Si-3.95Cu-1.01Mg-2.54Ni-0.47Fe | 350 | 100 | 2019 | [64] |
| Al-11.98Si-4.02Cu-1.02Mg-2.68Ni-0.62Fe | 350 | 106 | 2019 | [64] |
| Al-12Si-4Cu-2Ni-1Mg-AlNp | 350 | 106 | 2019 | [6] |
| Al-12.95Si-3.57Cu-0.72Mg-0.91Ni-0.53Fe-0.4Er | 350 | 117 | 2019 | [65] |
Figure 11 shows the room temperature and high temperature (350 °C) tensile fracture morphology of the heat-treated Al-6Cu-0.4Mn-0.4Ag alloy. It can be seen that the room temperature tensile fracture surface of the alloy was mainly composed of some white cleavage planes and some small dimples. Many massive Al
2Cu phases were distributed in the fracture, which was caused by the failure of the mid-layer sheet θ-Al
2Cu phase to dissolve entirely during the solid solution process. During the tensile process, these phases produced stress concentration and became the crack source, thus accelerating the fracture failure of the matrix alloy. Compared with the as-cast alloy, a large amount of the θ′-Al
2Cu phase was precipitated in the microstructure of the heat-treated alloy. The precipitation size of the θ′ phase was small, which made it difficult for the dislocation to the shear θ′ phase. Only when the external stress reached a certain degree could the θ′ phase be bypassed to deflect the α-Al phase along the surface of θ′-Al
2Cu, which could effectively inhibit crack growth and significantly affect the mechanical properties of the matrix alloy. It can be seen from the high-temperature tensile fracture of the heat-treated alloys in
Figure 11b that the high-temperature fracture of the T6 alloy was mainly composed of deep and wide dimples, and the high-temperature tensile fracture mode of the alloy belonged to ductile fracture. After heat treatment, more θ′-Al
2Cu nanophases were precipitated (
Figure 11b
1). It can be seen that many fine θ′-Al
2Cu phases were precipitated on the inner wall of the dimple. However, no small dimple was generated, indicating that the interface of the θ′-Al
2Cu nanophase and matrix was firmly bonded. It could play a good role in preventing the slip of matrix dislocation. θ′-Al
2Cu particles prevented the dislocation movement of the matrix, resulting in a decrease in plasticity and an increase in strength.
High-temperature mechanical properties are influenced by both the size and number of strengthening phase particles, as well as their strength and thermal stability. If the particles soften or coarsen at high temperatures, mechanical properties will significantly suffer. Recent studies have shown that adding Mn can promote the precipitation of the θ′ phase and refine the size of the θ′ during the artificial aging process. It can improve the high-temperature coarsening resistance of the θ′ phase, improving its thermal stability. The main reason is that Mn atoms segregate at the interface of θ′/α-Al, reducing the interface energy of both and thus improving the thermal stability of the θ′ phase. When Ag is added to an Al-Cu alloy, the Ag atom can form a diatomic layer at the θ′/α-Al coherent interface during the aging process, and the interface energy of the two phases can also be reduced, which promotes an increase in θ′. In this experiment, Mn and Ag were added to an Al-Cu alloy at the same time, and it was found that when Mn/Ag = 1:1, that is, the atomic ratio was about 2:1, the θ′-Al
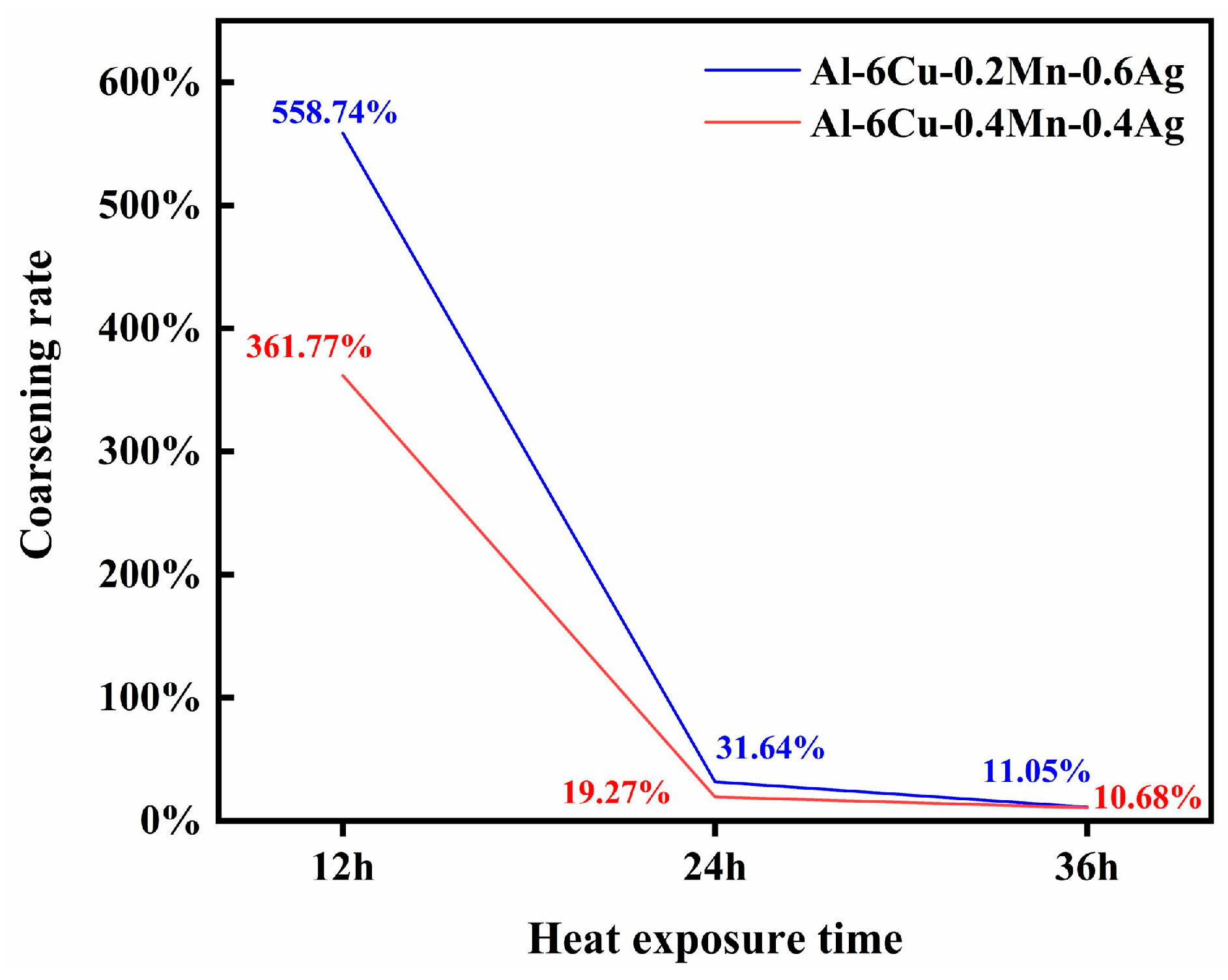
2Cu precipitation quantity was the largest and the size was the smallest. Therefore, we conclude that the joint segregation of Mn and Ag at the θ′/α-Al interface had a synergistic effect. The precipitation quantity and size of the θ′ phase could be minimized. Since the best high-temperature mechanical properties were also found in the Mn/Ag = 1:1 alloy, we believe that the thermal stability of the θ′ phase also plays a role. To this end, we conducted thermal exposure experiments on alloys with Mn/Ag ratios of 1:3 and 1:1 at different times to verify their anti-coarsening abilities. The result is shown in
Figure 12.
It can be seen that the θ′ phase size of both alloys increased with the extension of thermal exposure time, and the θ′-Al
2Cu phase gradually increased and grew in the longitudinal direction [
25]. Overall, the θ′ phase size of the Mn/Ag = 1:1 alloy was smaller at different stages. As shown in
Figure 13, its coarsening rate was also lower than that of the Mn/Ag = 1:3 alloy, so it can be confirmed that the Mn/Ag ratio had a significant effect on the anti-coarsening ability of the θ′ phase, that is, the effect of both on the thermal stability of the θ′ phase was also synergistic. This could be attributed to the segregation of Ag and Mn atoms at the Al/θ′-Al
2Cu interface. Specifically, phase field simulations have suggested the synergistic roles of thermodynamic and kinetics effects in retarding the coarsening of θ′-Al
2Cu precipitates [
66,
67,
68]. In the thermodynamics aspect, the θ′-Al
2Cu particles possess a plate-like morphology in the Al matrix with a coherent (001)
Al||(001)
θ′ interface along the broad faces and a semi-coherent (001)
Al||(001)
θ′ interface at the periphery of the plate. An atomically sharp Al/θ′-Al
2Cu semi-coherent interface (with low interfacial energy) is favorable at 350 °C [
52]. Mn and Ag show a strong segregation tendency at the Al/θ′-Al
2Cu interface, which provides powerful chemical bonds. Solute segregation can compensate for a lattice mismatch at the semi-coherent interface between Al and θ′-Al
2Cu; the chemical interaction of solutes at the interface helps reduce the interface energy [
14]. In the kinetics aspect, the thermal diffusion coefficient of the Mn atom is much lower than that of other atoms, which means that if it were introduced into the alloys, it would diffuse very slowly at high temperatures and could hinder the diffusion of other atoms, which is what we would expect, because it serves as a “coarsening barrier” and helps to hinder the suppression of the freely available vacancies in the Al matrix for Cu jumping (which leads to the coarsening of θ′-Al
2Cu) [
66,
69]. According to some reports [
25], it can be supposed that in the early stage of thermal exposure, Ag atoms with a relatively high thermal diffusion coefficient may first reach the Al/θ′-Al
2Cu interface and control the structure coarsening of θ′-Al
2Cu within a reasonable range. Subsequently, the Mn atoms with a lower thermal diffusion coefficient reach the interface to prevent the formation of a Cu surface on the interface and prevent the interface from becoming thicker and coarser.