High-Frequency Plasma Electrolytic Oxidation of an Al–Si Alloy: Influence of Al2O3 and SiO2 Additives on Coating Microstructure and Tribological Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate Preparation
2.2. Formation of PEO Coatings
| Electrolyte Composition | NaAlO2 (g/L) | KOH (g/L) | Al2O3 (g/L) | SiO2 (g/L) | |
|---|---|---|---|---|---|
| E1 | Base electrolyte | 24 | 1 | – | – |
| E2 | Base + SiO2 | 24 | 1 | – | 3.0 |
| E3 | Base + Al2O3 | 24 | 1 | 3.0 | – |
| E4 | Base + Al2O3 + SiO2 (1 g/L each) | 24 | 1 | 1.0 | 1.0 |
| E5 | Base + Al2O3 + SiO2 (2 g/L each) | 24 | 1 | 2.0 | 2.0 |
| E6 | Base + Al2O3 + SiO2 (3 g/L each) | 24 | 1 | 3.0 | 3.0 |
2.3. Coating Analysis
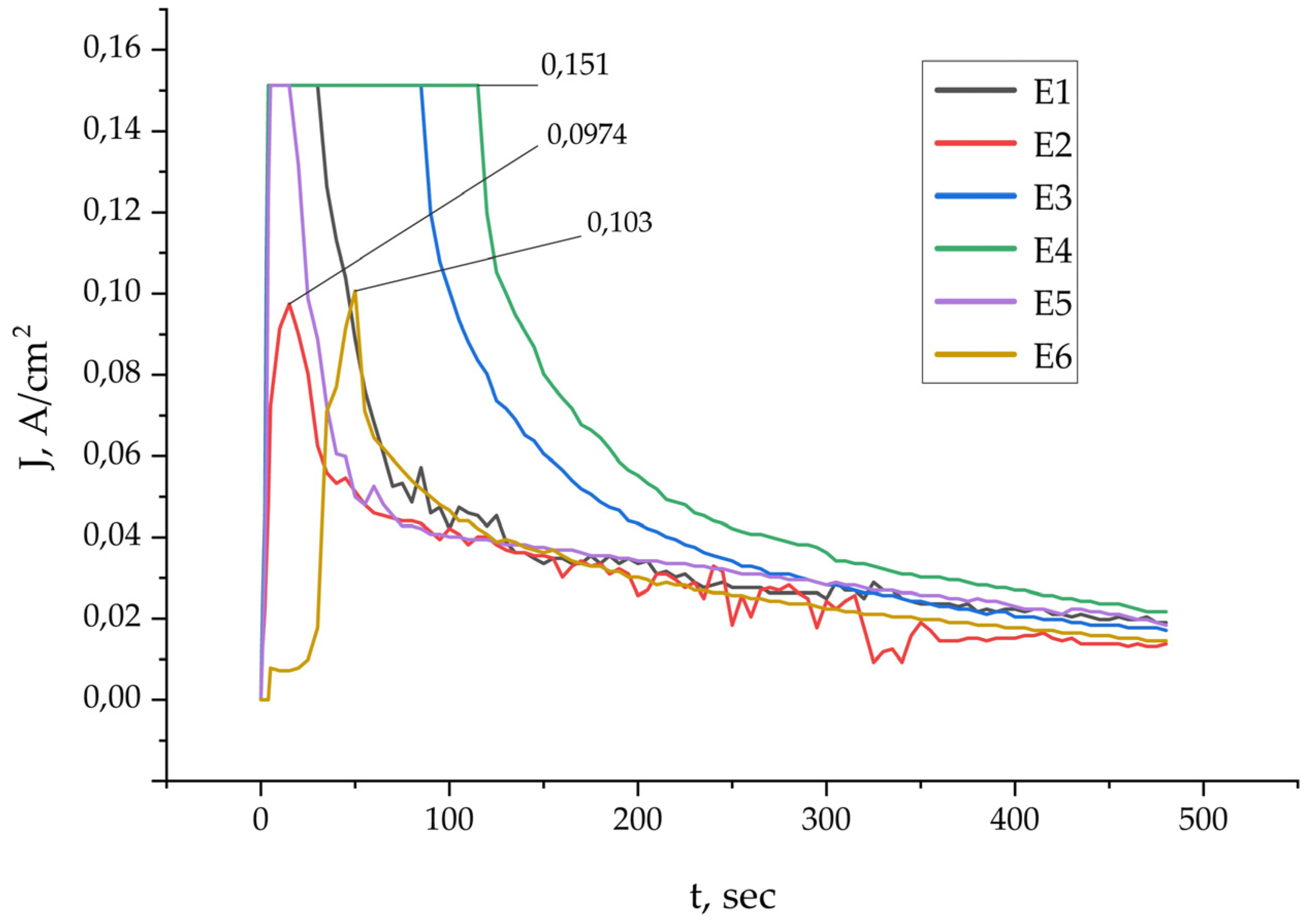
3. Results and Discussion
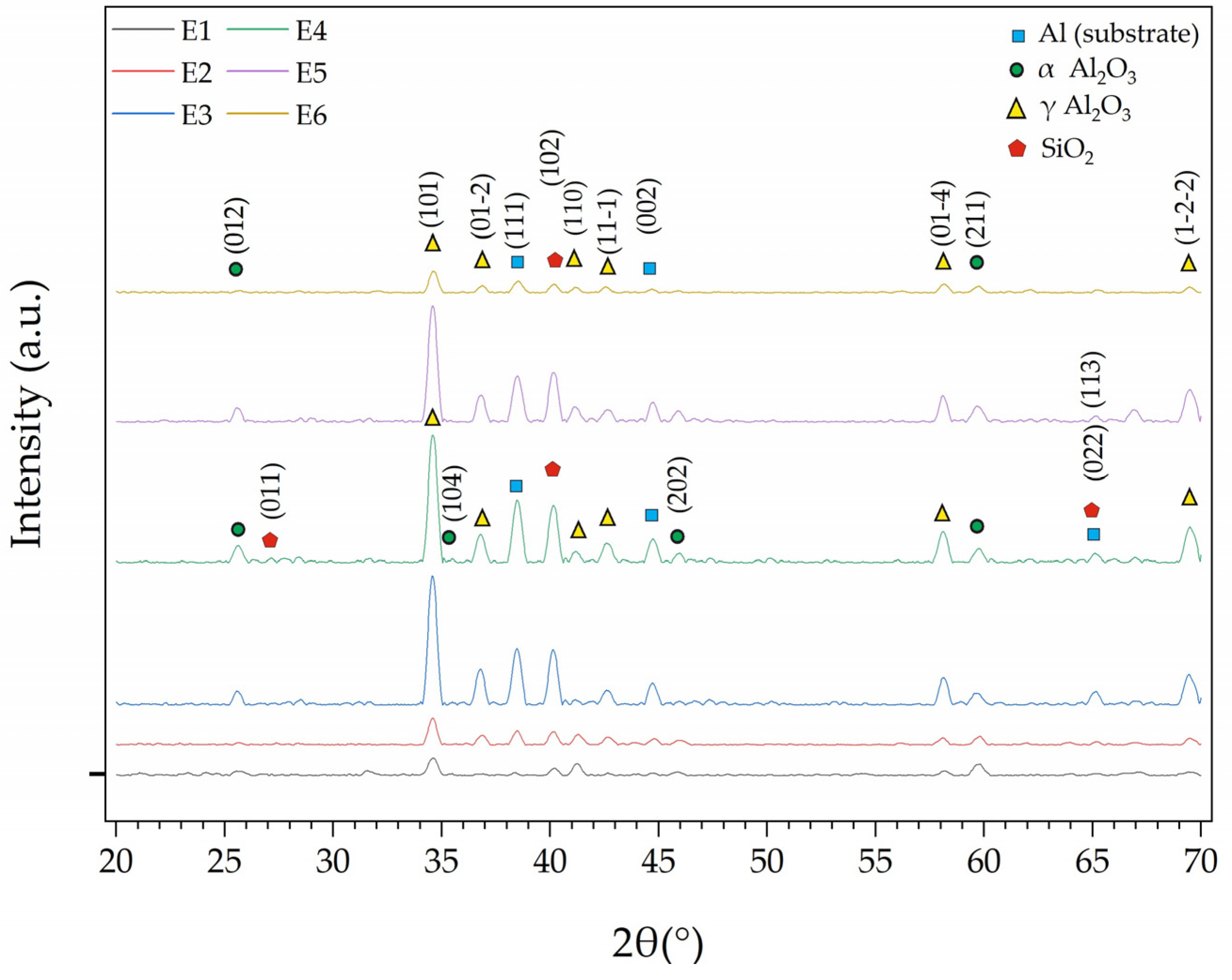
3.1. Analysis of PEO Coatings
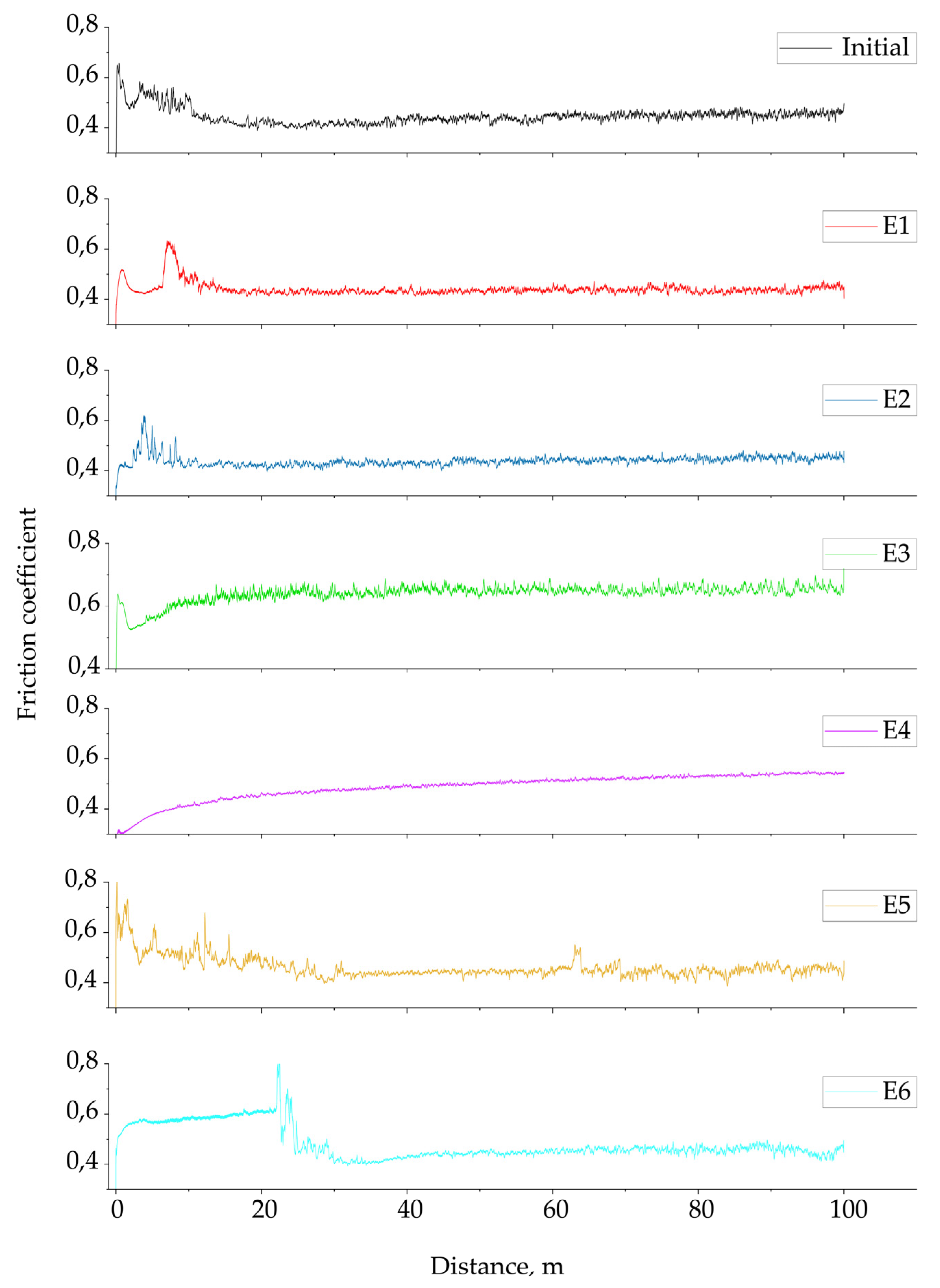
3.2. Tribological Tests
4. Conclusions
- Coatings E1 and E2 exhibited improved wear resistance compared to the initial sample (η_wear = +32% and +41%), and their microhardness increased to 230 HV(E1) and 173 HV (E2). This contributed to the reduction in the wear rate of these coatings. In particular, sample E2 showed a slightly improved surface morphology, with finer and more uniformly distributed pores.
- The E3 coating with added Al2O3 showed the most promising results: the wear rate decreased to 0.030 × 10−3 mm3/N·m, an improvement of approximately 96%. Although the microhardness increased only moderately (260 HV), the XRD results indicated that the α-Al2O3 phase was more pronounced in this coating. Therefore, the high wear resistance is explained mainly by the coating’s density and phase composition, not just by its hardness.
- No synergistic effect was observed in the E4–E6 systems with the combined Al2O3 + SiO2. The E4–E6 samples showed low wear resistance due to defects caused by particle agglomeration and unstable discharges. Although the coating produced in electrolyte E4 showed a smoother and more uniform surface appearance.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fernández-López, P.; Alves, S.A.; San-Jose, J.T.; Gutierrez-Berasategui, E.; Bayón, R. Plasma Electrolytic Oxidation (PEO) as a Promising Technology for the Development of High-Performance Coatings on Cast Al-Si Alloys: A Review. Coatings 2024, 14, 217. [Google Scholar] [CrossRef]
- Matykina, E.; Arrabal, R.; Mohedano, M.; Mingo, B.; Gonzalez, J.; Pardo, A.; Merino, M.C. Recent Advances in Energy Efficient PEO Processing of Aluminium Alloys. Trans. Nonferrous Met. Soc. China 2017, 27, 1439–1454. [Google Scholar] [CrossRef]
- Alves, S.A.; Fernández-Lopéz, P.; Lopéz-Ortega, A.; Fernández, X.; Quintana, I.; San-José, J.T.; Bayón, R. Enhanced Tribological Performance of Cylinder Liners Made of Cast Aluminum Alloy with High Silicon Content through Plasma Electrolytic Oxidation. Surf. Coat. Technol. 2022, 433, 128146. [Google Scholar] [CrossRef]
- Zhang, J.; Dai, W.; Wang, X.; Wang, Y.; Yue, H.; Li, Q.; Yang, X.; Guo, C.; Li, C. Micro-Arc Oxidation of Al Alloys: Mechanism, Microstructure, Surface Properties, and Fatigue Damage Behavior. J. Mater. Res. Technol. 2023, 23, 4307–4333. [Google Scholar] [CrossRef]
- Sikdar, S.; Menezes, P.V.; Maccione, R.; Jacob, T.; Menezes, P.L. Plasma Electrolytic Oxidation (PEO) Process—Processing, Properties, and Applications. Nanomaterials 2021, 11, 1375. [Google Scholar] [CrossRef]
- Yerokhin, A.L.; Nie, X.; Leyland, A.; Matthews, A.; Dowey, S.J. Plasma Electrolysis for Surface Engineering. Surf. Coat. Technol. 1999, 122, 73–93. [Google Scholar] [CrossRef]
- Rakhadilov, B.K.; Tabiyeva, Y.Y.; Uazyrkhanova, G.K.; Zhurerova, L.G.; Baizhan, D. Influence of Electrolytic-Plasma Surface Quenching on the Structure and Strength Properties of Ferritic-Pearlite Class Wheel Steel. Eurasian J. Phys. Funct. Mater. 2020, 4, 167–173. [Google Scholar] [CrossRef]
- Pesode, P.; Barve, S. Surface Modification of Titanium and Titanium Alloy by Plasma Electrolytic Oxidation Process for Biomedical Applications: A Review. Mater. Today Proc. 2021, 46, 594–602. [Google Scholar] [CrossRef]
- Sagidugumar, A.; Dogadkin, D.; Turlybekuly, A.; Kaliyev, D. Calcium Phosphate Coatings Deposited on 3D-Printed Ti–6Al–4V Alloy by Plasma Electrolytic Oxidation. Coatings 2024, 14, 696. [Google Scholar] [CrossRef]
- Parfenov, E.; Farrakhov, R.; Aubakirova, V.; Stotskiy, A.; Nagumothu, R.; Yerokhin, A. Frequency Response Evaluation as Diagnostic and Optimization Tool for Pulsed Unipolar Plasma Electrolytic Oxidation Process and Resultant Coatings on Zirconium. Materials 2023, 16, 7681. [Google Scholar] [CrossRef]
- Tu, W.; Zhu, Z.; Zhuang, X.; Cheng, Y.; Skeldon, P. Effect of Frequency on Black Coating Formation on AZ31 Magnesium Alloy by Plasma Electrolytic Oxidation in Aluminate-Tungstate Electrolyte. Surf. Coat. Technol. 2019, 372, 34–44. [Google Scholar] [CrossRef]
- Mann, R.; Hansal, W.; Hansal, S. Effects of Pulsed Current on Plasma Electrolytic Oxidation. Trans. Inst. Met. Finish. 2014, 92, 297–304. [Google Scholar] [CrossRef]
- Lu, X.; Mohedano, M.; Blawert, C.; Matykina, E.; Arrabal, R.; Kainer, K.U.; Zheludkevich, M.L. Plasma Electrolytic Oxidation Coatings with Particle Additions—A Review. Surf. Coat. Technol. 2016, 307, 1165–1182. [Google Scholar] [CrossRef]
- Gamba, M.; Cristoforetti, A.; Fedel, M.; Ceriani, F.; Ormellese, M.; Brenna, A. Plasma Electrolytic Oxidation (PEO) Coatings on Aluminum Alloy 2024: A Review of Mechanisms, Processes, and Corrosion Resistance Enhancement. Appl. Surf. Sci. Adv. 2025, 26, 100707. [Google Scholar] [CrossRef]
- Lu, C.; Feng, X.; Yang, J.; Jia, J.; Yi, G.; Xie, E.; Sun, Y. Influence of Surface Microstructure on Tribological Properties of PEO-PTFE Coating Formed on Aluminum Alloy. Surf. Coat. Technol. 2019, 364, 127–134. [Google Scholar] [CrossRef]
- Xie, H.; Cheng, Y.; Li, S.; Cao, J.; Cao, L. Wear and Corrosion Resistant Coatings on Surface of Cast A356 Aluminum Alloy by Plasma Electrolytic Oxidation in Moderately Concentrated Aluminate Electrolytes. Trans. Nonferrous Met. Soc. China 2017, 27, 336–351. [Google Scholar] [CrossRef]
- Ghafaripoor, M.; Raeissi, K.; Santamaria, M.; Hakimizad, A. The Corrosion and Tribocorrosion Resistance of PEO Composite Coatings Containing α-Al2O3 Particles on 7075 Al Alloy. Surf. Coat. Technol. 2018, 349, 470–479. [Google Scholar] [CrossRef]
- Nadimi, M.; Dehghanian, C.; Etemadmoghadam, A. Influence of SiO2 Nanoparticles Incorporating into Ceramic Coatings Generated by PEO on Aluminium Alloy: Morphology, Adhesion, Corrosion, and Wear Resistance. Mater. Today Commun. 2022, 31, 103587. [Google Scholar] [CrossRef]
- Fernández-López, P.; Alves, S.A.; López-Ortega, A.; San José-Lombera, J.T.; Bayón, R. High Performance Tribological Coatings on a Secondary Cast Al–Si Alloy Generated by Plasma Electrolytic Oxidation. Ceram. Int. 2021, 47, 31238–31250. [Google Scholar] [CrossRef]
- Huang, Z.-Q.; Wang, R.-Q.; Zhang, H.; Shen, X.-J.; Zhang, X.-Z.; He, Y.; Huang, C.; Shen, D.-J.; Li, D.-L. Effect of Nanosized Silicon Dioxide Additive on Plasma Electrolytic Oxidation Coatings Fabricated on Aluminium. Int. J. Electrochem. Sci. 2020, 15, 11191–11202. [Google Scholar] [CrossRef]
- Cheng, Y.; Mao, M.; Cao, J.; Peng, Z. Plasma Electrolytic Oxidation of an Al-Cu-Li Alloy in Alkaline Aluminate Electrolytes: A Competition between Growth and Dissolution for the Initial Ultra-Thin Films. Electrochim. Acta 2014, 138, 417–429. [Google Scholar] [CrossRef]
- Rogov, A.B.; Yerokhin, A.; Matthews, A. The Role of Cathodic Current in Plasma Electrolytic Oxidation of Aluminum: Phenomenological Concepts of the “Soft Sparking” Mode. Langmuir 2017, 33, 11059–11069. [Google Scholar] [CrossRef] [PubMed]
- Guglielmi, P.; Davoodi, F.; Palumbo, G.; Carbone, G. Tribological Behaviour of Microindented 100Cr6 Steel Surfaces in Dry Contact Conditions. Int. J. Adv. Manuf. Technol. 2024, 133, 2381–2400. [Google Scholar] [CrossRef]
- Zhu, Z.; Wang, Y.; Shi, D.; Wang, J.; Liu, Y.; Wu, C.; Su, X. Optimizing the Microstructure and Enhancing the Mechanical Prsoperties of Hypereutectic Al–Si Alloys by Mg Addition. J. Mater. Res. Technol. 2024, 32, 457–469. [Google Scholar] [CrossRef]
- Kaseem, M.; Fatimah, S.; Nashrah, N.; Ko, Y.G. Recent Progress in Surface Modification of Metals Coated by Plasma Electrolytic Oxidation: Principle, Structure, and Performance. Prog. Mater. Sci. 2021, 117, 100735. [Google Scholar] [CrossRef]
- Tsai, D.-S.; Chou, C.-C. Review of the Soft Sparking Issues in Plasma Electrolytic Oxidation. Metals 2018, 8, 105. [Google Scholar] [CrossRef]
- Dogadkin, D.; Sagidugumar, A.; Kaliyev, D.; Dmitriev, N.; Kozhakhmetov, Y. The Role of Signal Waveforms in Plasma Electrolytic Oxidation. Coatings 2025, 15, 36. [Google Scholar] [CrossRef]
- Krishtal, M.M.; Katsman, A.V.; Polunin, A. V Effects of Silica Nanoparticles Addition on Formation of Oxide Layers on AlSi Alloy by Plasma Electrolytic Oxidation: The Origin of Stishovite under Ambient Conditions. Surf. Coat. Technol. 2022, 441, 128556. [Google Scholar] [CrossRef]
- Dehnavi, V.; Liu, X.Y.; Luan, B.L.; Shoesmith, D.W.; Rohani, S. Phase Transformation in Plasma Electrolytic Oxidation Coatings on 6061 Aluminum Alloy. Surf Coat Technol 2014, 251, 106–114. [Google Scholar] [CrossRef]
- Algahtani, A.; Mahmoud, E.R.I. Erosion and Corrosion Resistance of Plasma Electrolytic Oxidized 6082 Aluminum Alloy Surface at Low and High Temperatures. J. Mater. Res. Technol. 2019, 8, 2699–2709. [Google Scholar] [CrossRef]
- Pezzato, L.; Kostelac, L.; Tonelli, L.; Elsayed, H.; Kajánek, D.; Bernardo, E.; Martini, C.; Dabalà, M.; Brunelli, K. Effect of Different Types of Glass Powders on the Corrosion and Wear Resistance of Peo Coatings Produced on 6061 Aluminum Alloy. Met. Mater. Int. 2025, 31, 636–653. [Google Scholar] [CrossRef]
- Florea, V.A.; Toderaș, M.; Danciu, C. The Influence of Roughness of Surfaces on Wear Mechanisms in Metal–Rock Interactions. Coatings 2025, 15, 150. [Google Scholar] [CrossRef]
- Michalak, M.; Sokołowski, P.; Szala, M.; Walczak, M.; Łatka, L.; Toma, F.-L.; Björklund, S. Wear Behavior Analysis of Al2O3 Coatings Manufactured by APS and HVOF Spraying Processes Using Powder and Suspension Feedstocks. Coatings 2021, 11, 879. [Google Scholar] [CrossRef]
- Seto, N.; Endo, K.; Sakamoto, N.; Hirose, S.; Akedo, J. Hard α-Al2O3 Film Coating on Industrial Roller Using Aerosol Deposition Method. J. Therm. Spray. Technol. 2014, 23, 1373–1381. [Google Scholar] [CrossRef]
- Yang, H.; Dong, Y.; Li, X.; Gao, Y.; He, W.; Liu, Y.; Mu, X.; Zhao, Y.; Fu, W.; Wang, X.; et al. Development of a Mechanically Robust Superhydrophobic Anti-Corrosion Coating Using Micro-HBN/Nano-Al2O3 with Multifunctional Properties. Ceram. Int. 2025, 51, 491–505. [Google Scholar] [CrossRef]

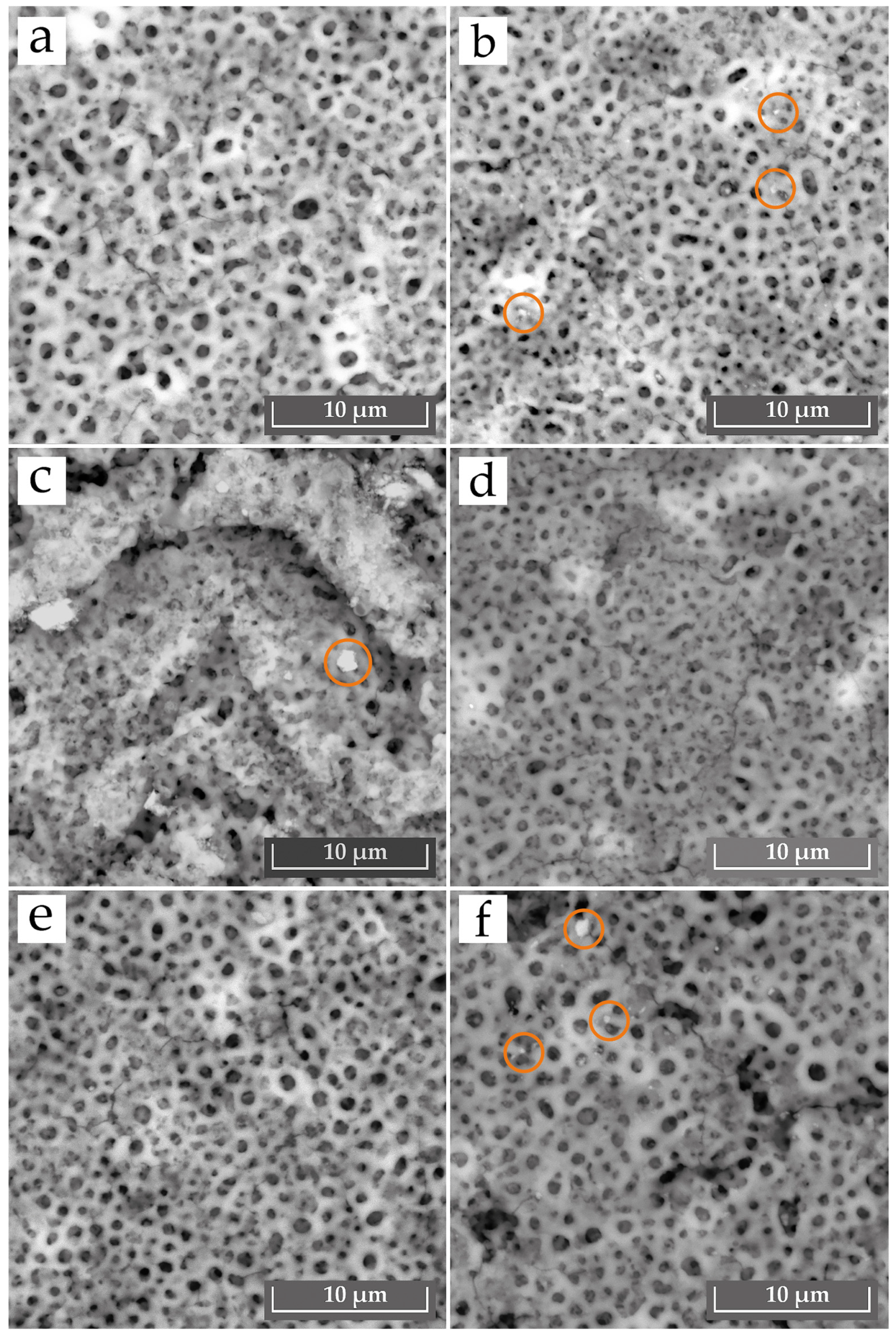



| Element (wt.%) | O | Al | Si | Cr | Mn | Fe | Cu | Zn |
|---|---|---|---|---|---|---|---|---|
| Spectrum 1 | 2.24 | 58.83 | 9.95 | 0.91 | 6.18 | 19.65 | 2.01 | 0.22 |
| Spectrum 2 | 2.25 | 93.96 | 2.07 | 0.00 | 0.00 | 0.13 | 0.77 | 0.83 |
| Spectrum 3 | 2.29 | 67.88 | 28.21 | 0.00 | 0.00 | 0.00 | 0.95 | 0.67 |
| Spectrum 4 | 3.51 | 70.68 | 6.07 | 0.52 | 4.19 | 12.45 | 2.10 | 0.47 |
| Spectrum 5 | 2.13 | 72.18 | 13.09 | 0.14 | 1.53 | 9.53 | 0.75 | 0.64 |
| Std. deviation | 0.58 | 12.96 | 10.02 | 0.40 | 2.73 | 8.41 | 0.68 | 0.23 |
| Sample | Spectrum | Al (wt.%) | O (wt.%) | Si (wt.%) | Fe (wt.%) | C (wt.%) |
|---|---|---|---|---|---|---|
| E1 | 1 | 56.59 ± 0.21 | 31.70 ± 0.16 | 6.50 ± 0.05 | - | 5.21 ± 0.32 |
| 2 | 60.83 ± 0.23 | 28.02 ± 0.15 | 5.66 ± 0.05 | - | 5.49 ± 0.32 | |
| 3 | 56.49 ± 0.22 | 33.29 ± 0.17 | 4.84 ± 0.05 | - | 5.38 ± 0.33 | |
| E2 | 1 | 40.75 ± 0.05 | 45.77 ± 0.14 | 6.34 ± 0.02 | 0.60 ± 0.03 | 6.54 ± 0.20 |
| 2 | 45.62 ± 0.15 | 45.96 ± 0.14 | 7.93 ± 0.05 | 0.49 ± 0.02 | - | |
| 3 | 51.73 ± 0.15 | 30.20 ± 0.17 | 11.38 ± 0.04 | 0.28 ± 0.04 | 6.31 ± 0.30 | |
| E3 | 1 | 12.95 ± 0.10 | 45.65 ± 0.11 | 5.26 ± 0.04 | 36.14 ± 0.21 | - |
| 2 | 12.20 ± 0.04 | 46.42 ± 0.14 | 5.32 ± 0.06 | 36.06 ± 0.17 | - | |
| 3 | 2.87 ± 0.14 | 36.91 ± 0.16 | 1.04 ± 0.04 | 59.18 ± 0.18 | - | |
| E4 | 1 | 49.88 ± 0.19 | 39.14 ± 0.17 | 5.52 ± 0.05 | - | 5.45 ± 0.31 |
| 2 | 83.17 ± 0.44 | 7.43 ± 0.12 | 2.75 ± 0.05 | - | 6.65 ± 0.48 | |
| 3 | 69.91 ± 0.33 | 13.51± 0.14 | 9.78 ± 0.07 | - | 6.81 ± 0.42 | |
| E5 | 1 | 50.62 ± 0.25 | 35.47 ± 0.21 | 7.01 ± 0.06 | - | 6.90 ± 0.40 |
| 2 | 71.73 ± 0.54 | 5.33 ± 0.15 | 13.84 ± 0.13 | - | 9.10 ± 0.66 | |
| 3 | 61.18 ± 0.30 | 20.40 ± 0.13 | 10.43 ± 0.07 | - | 8.00 ± 0.39 | |
| E6 | 1 | 47.19 ± 0.03 | 46.43 ± 0.13 | 5.56 ± 0.02 | 0.82 ± 0.17 | - |
| 2 | 60.05 ± 0.13 | 30.06 ± 0.17 | 3.76 ± 0.04 | 0.89 ± 0.04 | 5.24 ± 0.28 | |
| 3 | 40.46 ± 0.15 | 48.37 ± 0.18 | 5.89 ± 0.03 | 0.80 ± 0.07 | 4.48 ± 0.32 |
| Sample | Coefficient of Friction | η (Friction) % | Wear Rate (10−3 mm3(Nm)−1) | η (Wear) % | Surface Roughness (Ra) |
|---|---|---|---|---|---|
| Initial | 0.446 ± 0.034 | - | 0.730 ± 0.141 | - | - |
| E1 | 0.441 ± 0.027 | +1.1 | 0.495 ± 0.100 | +32.2 | 0.223 ± 0.016 |
| E2 | 0.439 ± 0.022 | +1.6 | 0.431 ± 0.248 | +41.0 | 0.193 ± 0.039 |
| E3 | 0.640 ± 0.030 | −43.5 | 0.030 ± 0.002 | +95.9 | 0.376 ± 0.038 |
| E4 | 0.487 ± 0.053 | −9.2 | 1.209 ± 0.777 | −65.8 | 0.153 ± 0.008 |
| E5 | 0.462 ± 0.045 | −3.6 | 1.923 ± 0.512 | −163.4 | 0.356 ± 0.027 |
| E6 | 0.482 ± 0.065 | −8.1 | 1.048 ± 0.396 | −43.6 | 0.163 ± 0.028 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uazyrkhanova, G.; Sagidugumar, A.; Kozhakhmetov, Y.; Moldabayeva, G.; Kaliyev, D.; Rudenko, S.; Kantay, N. High-Frequency Plasma Electrolytic Oxidation of an Al–Si Alloy: Influence of Al2O3 and SiO2 Additives on Coating Microstructure and Tribological Performance. Materials 2025, 18, 5334. https://doi.org/10.3390/ma18235334
Uazyrkhanova G, Sagidugumar A, Kozhakhmetov Y, Moldabayeva G, Kaliyev D, Rudenko S, Kantay N. High-Frequency Plasma Electrolytic Oxidation of an Al–Si Alloy: Influence of Al2O3 and SiO2 Additives on Coating Microstructure and Tribological Performance. Materials. 2025; 18(23):5334. https://doi.org/10.3390/ma18235334
Chicago/Turabian StyleUazyrkhanova, Gulzhaz, Amangeldi Sagidugumar, Yernat Kozhakhmetov, Gulzhaz Moldabayeva, Daniyar Kaliyev, Sergey Rudenko, and Nurgamit Kantay. 2025. "High-Frequency Plasma Electrolytic Oxidation of an Al–Si Alloy: Influence of Al2O3 and SiO2 Additives on Coating Microstructure and Tribological Performance" Materials 18, no. 23: 5334. https://doi.org/10.3390/ma18235334
APA StyleUazyrkhanova, G., Sagidugumar, A., Kozhakhmetov, Y., Moldabayeva, G., Kaliyev, D., Rudenko, S., & Kantay, N. (2025). High-Frequency Plasma Electrolytic Oxidation of an Al–Si Alloy: Influence of Al2O3 and SiO2 Additives on Coating Microstructure and Tribological Performance. Materials, 18(23), 5334. https://doi.org/10.3390/ma18235334






