Comparative Study of Acid Etching and SLA Surface Modification for Titanium Implants
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Surface Treatments
2.2. Roughness and Hydrophilicity Assessment
2.3. Bioactivity Assessment by Immersion in Simulated Body Fluid
2.4. In Vitro Study
2.5. Cell Culture
2.6. Cell Viability Assay
2.7. Cell Differentiation
2.8. Alkaline Phosphatase Activity
2.9. Mineralization Assay
2.10. Statistical Analysis
3. Results
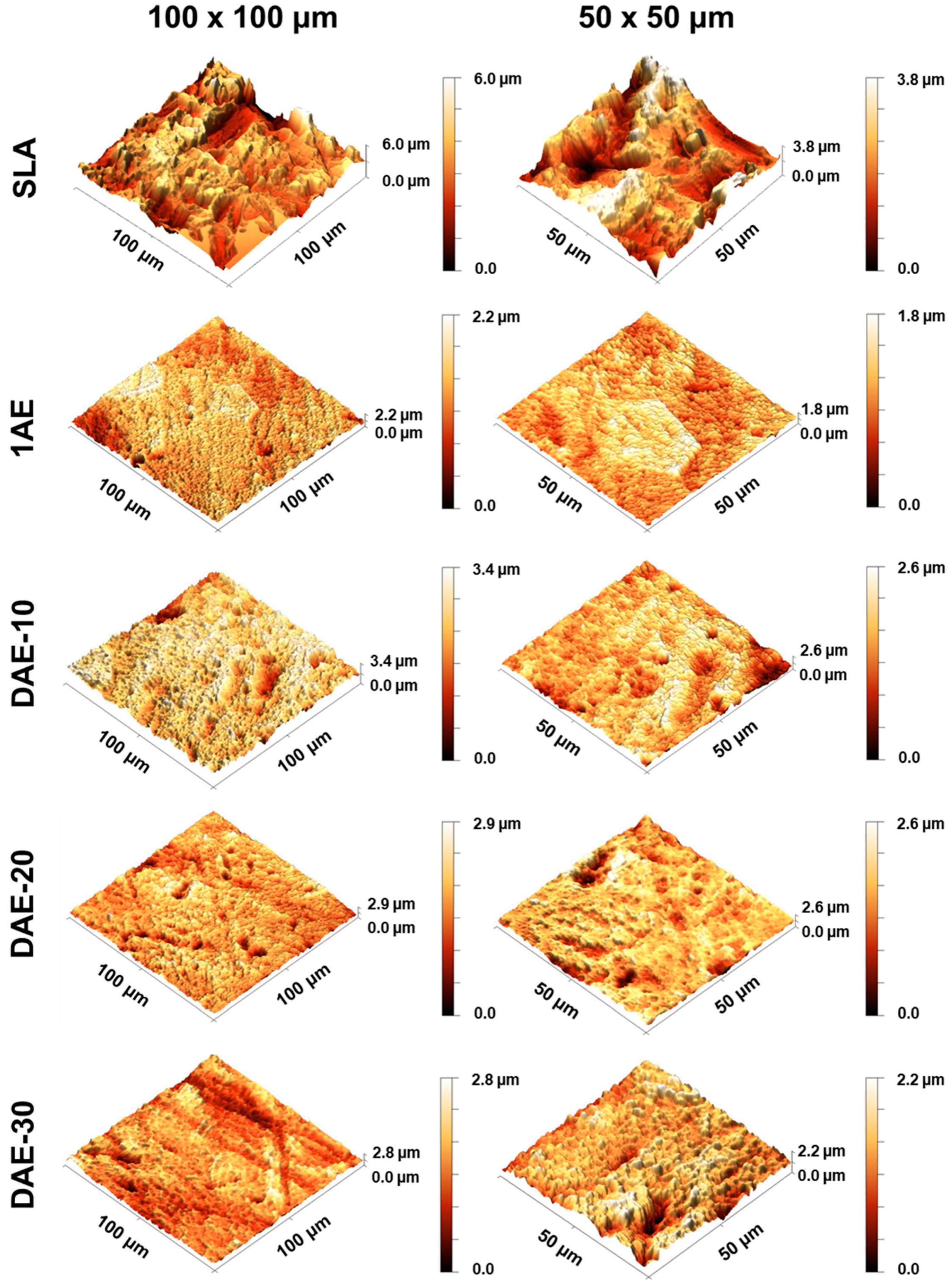
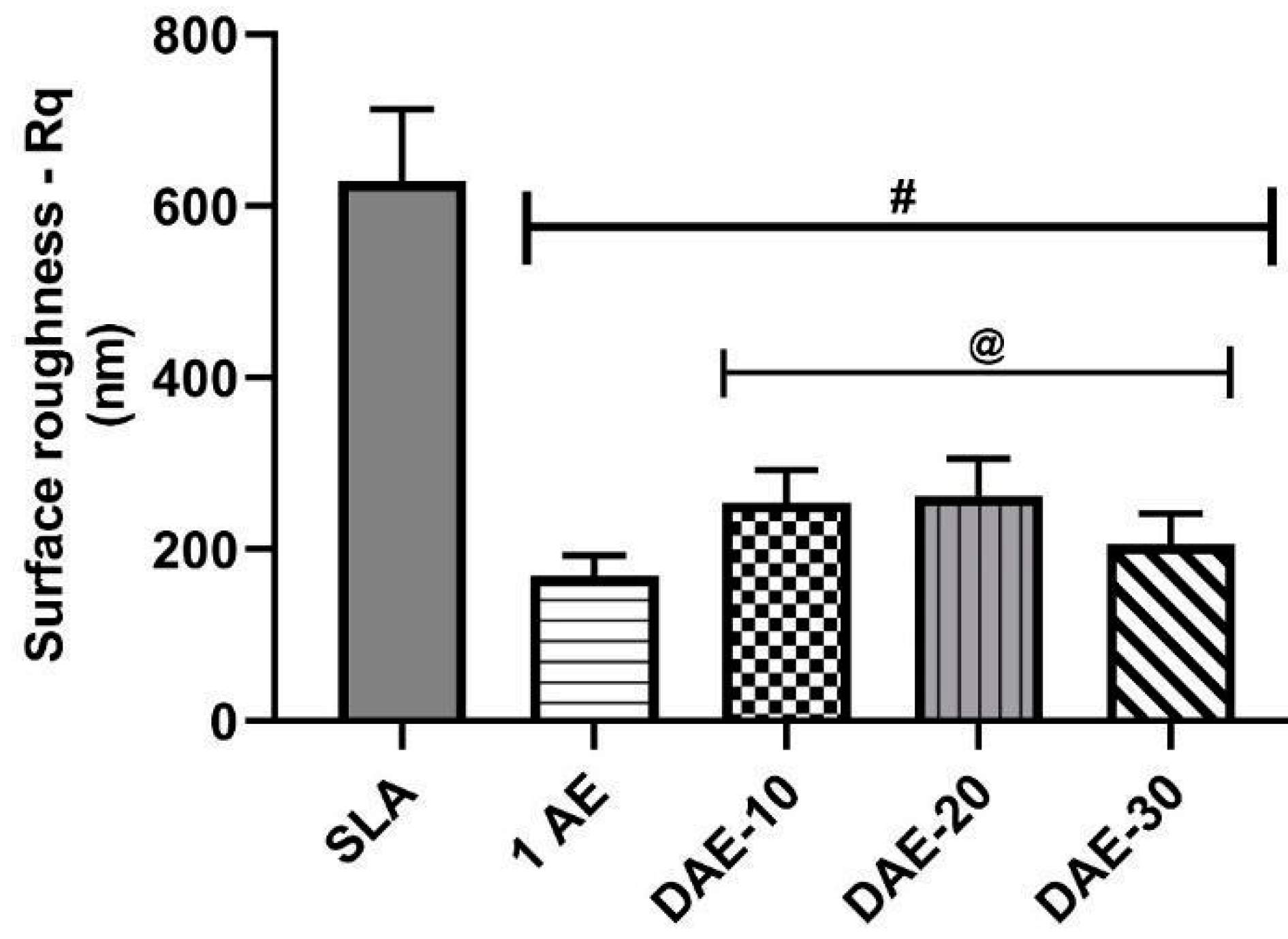
3.1. Roughness Assessment
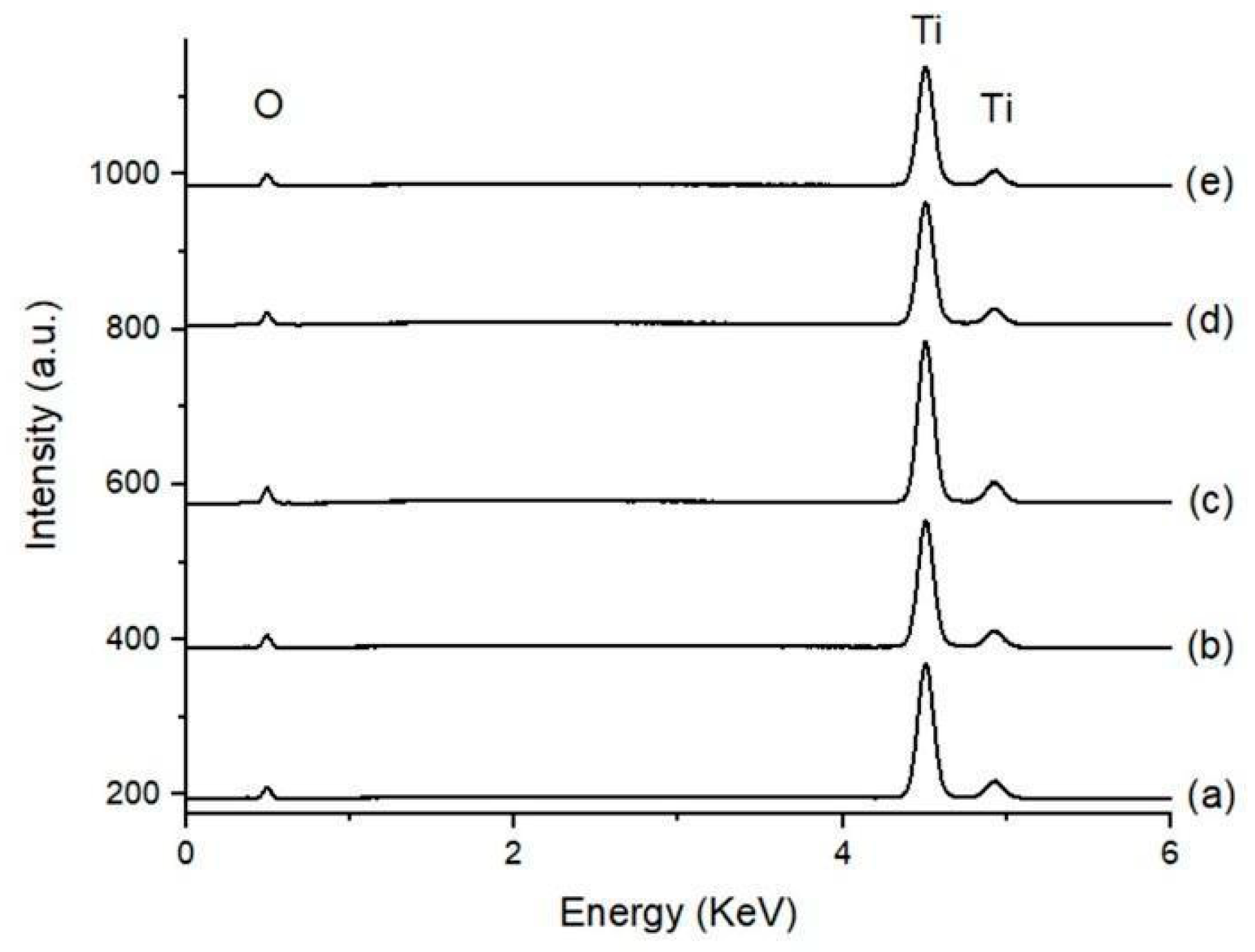
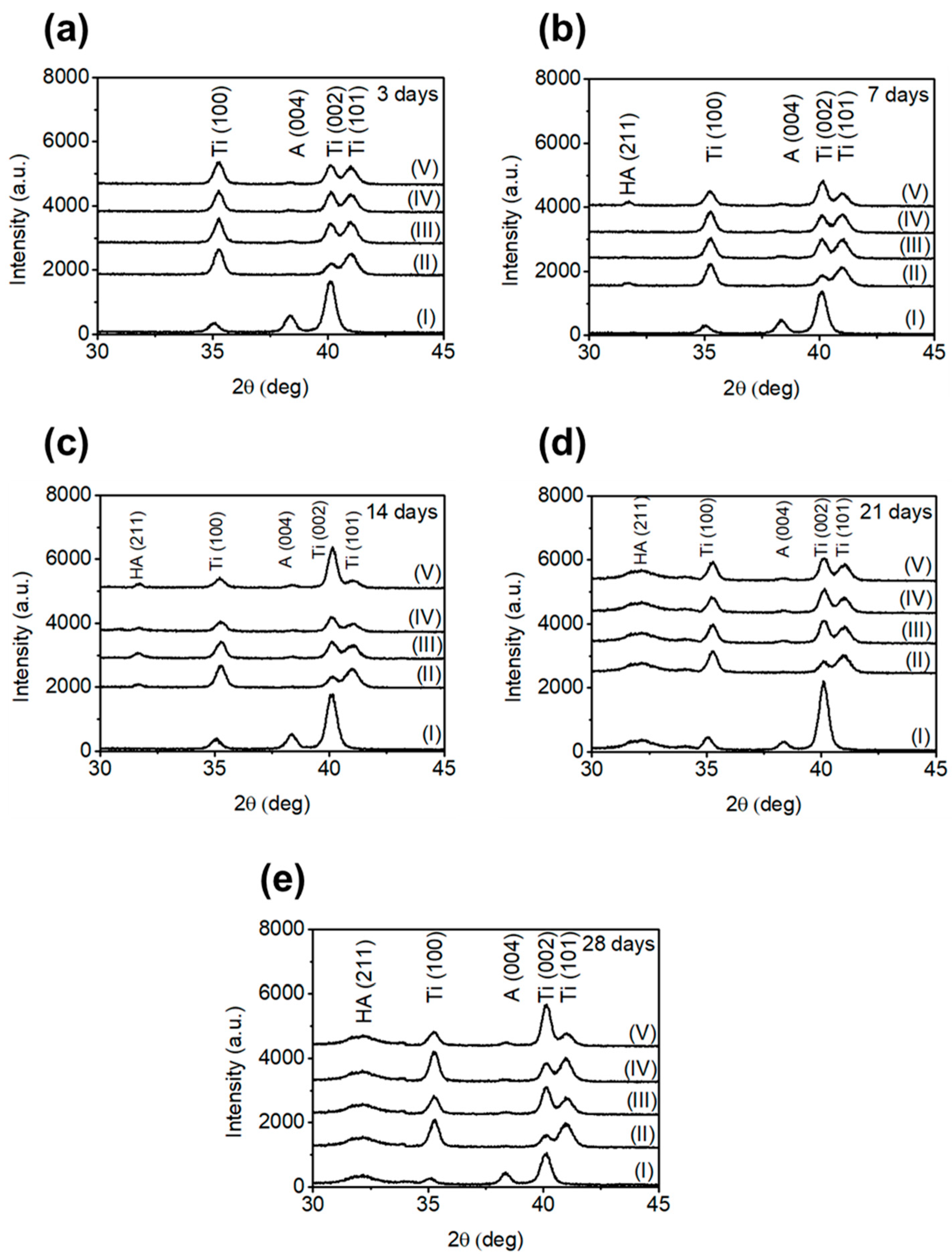
3.2. Surface Chemical Composition
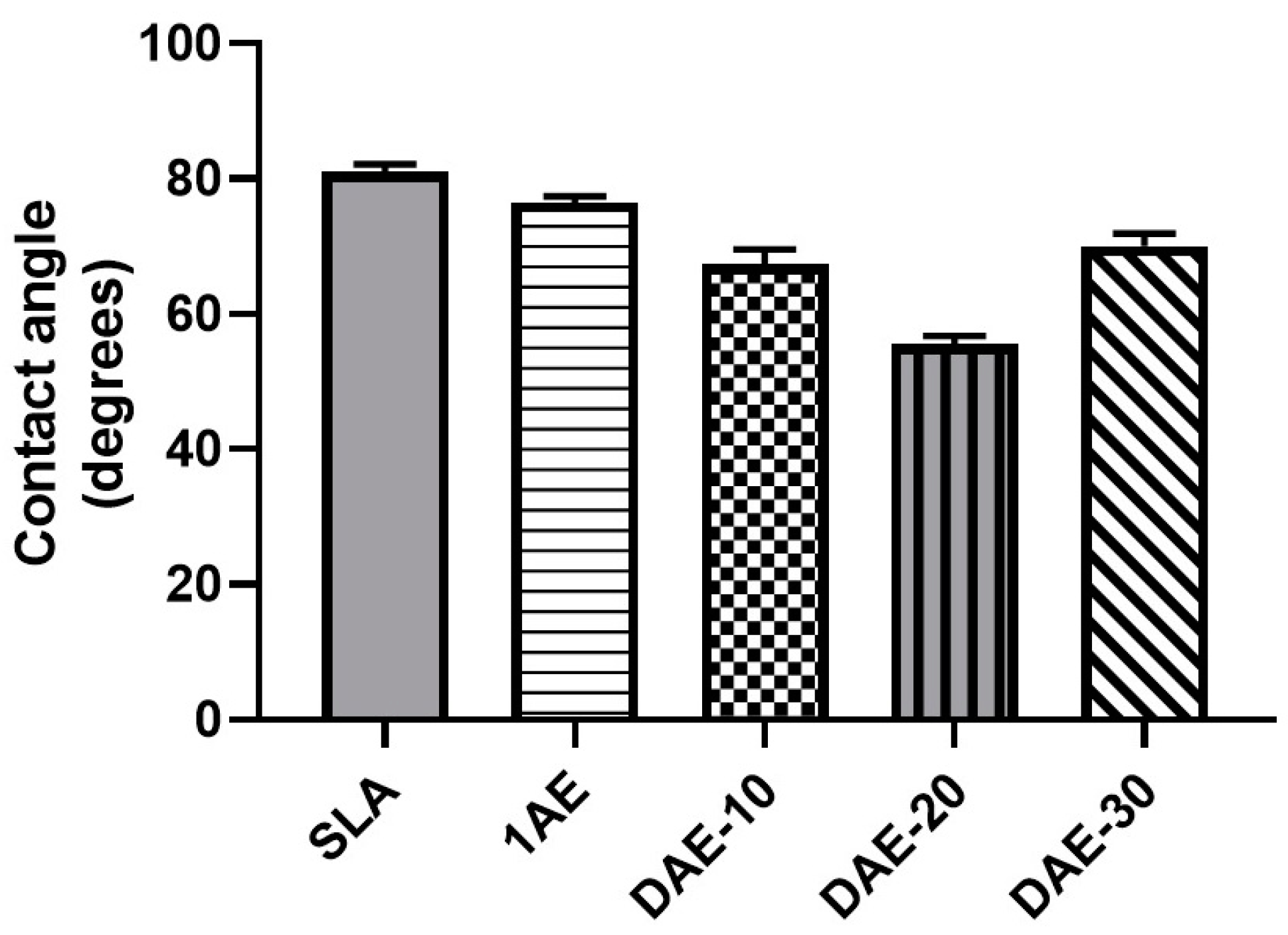
3.3. Surface Wettability
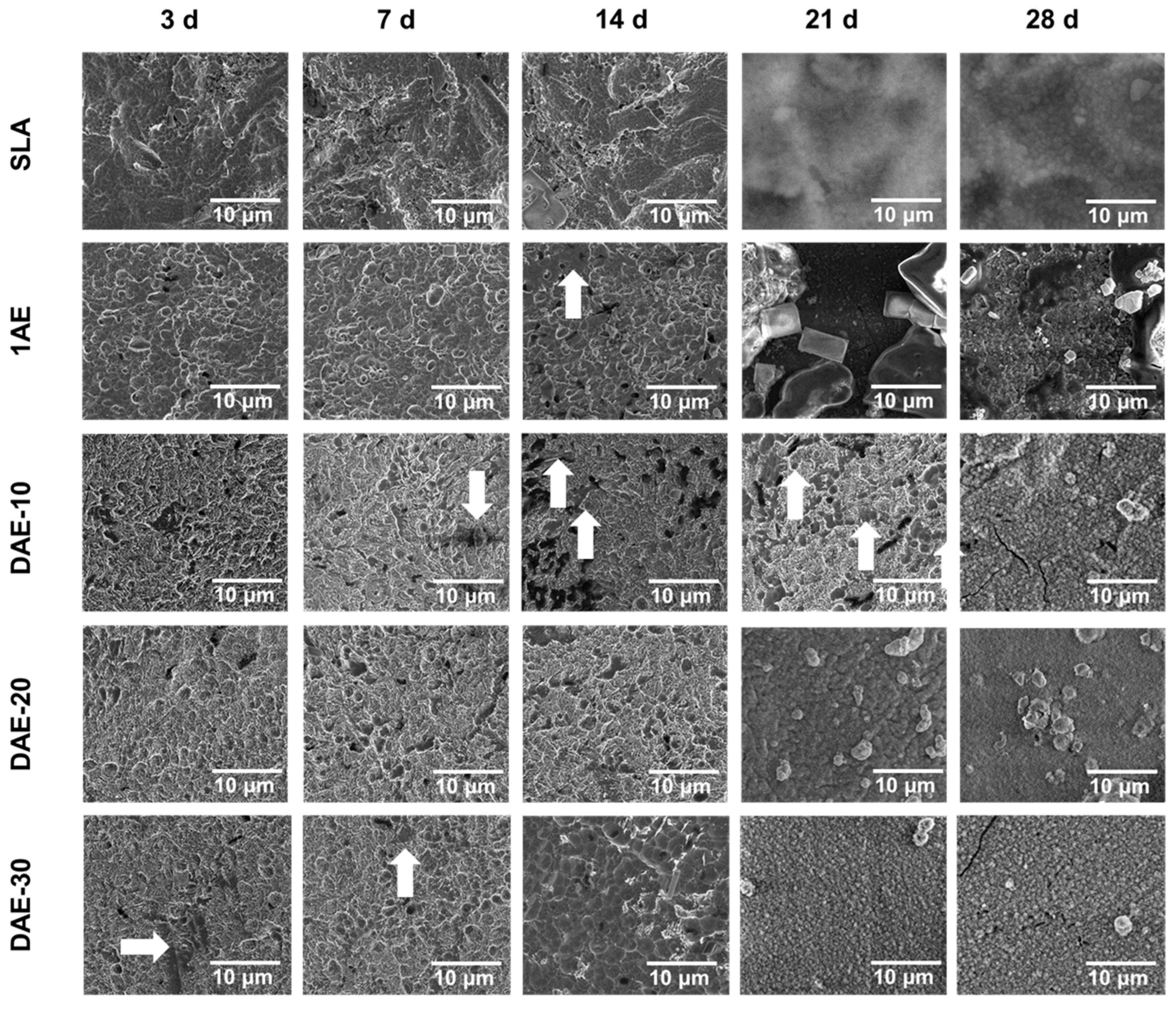
3.4. Bioactivity Assessment by Immersion in SBF
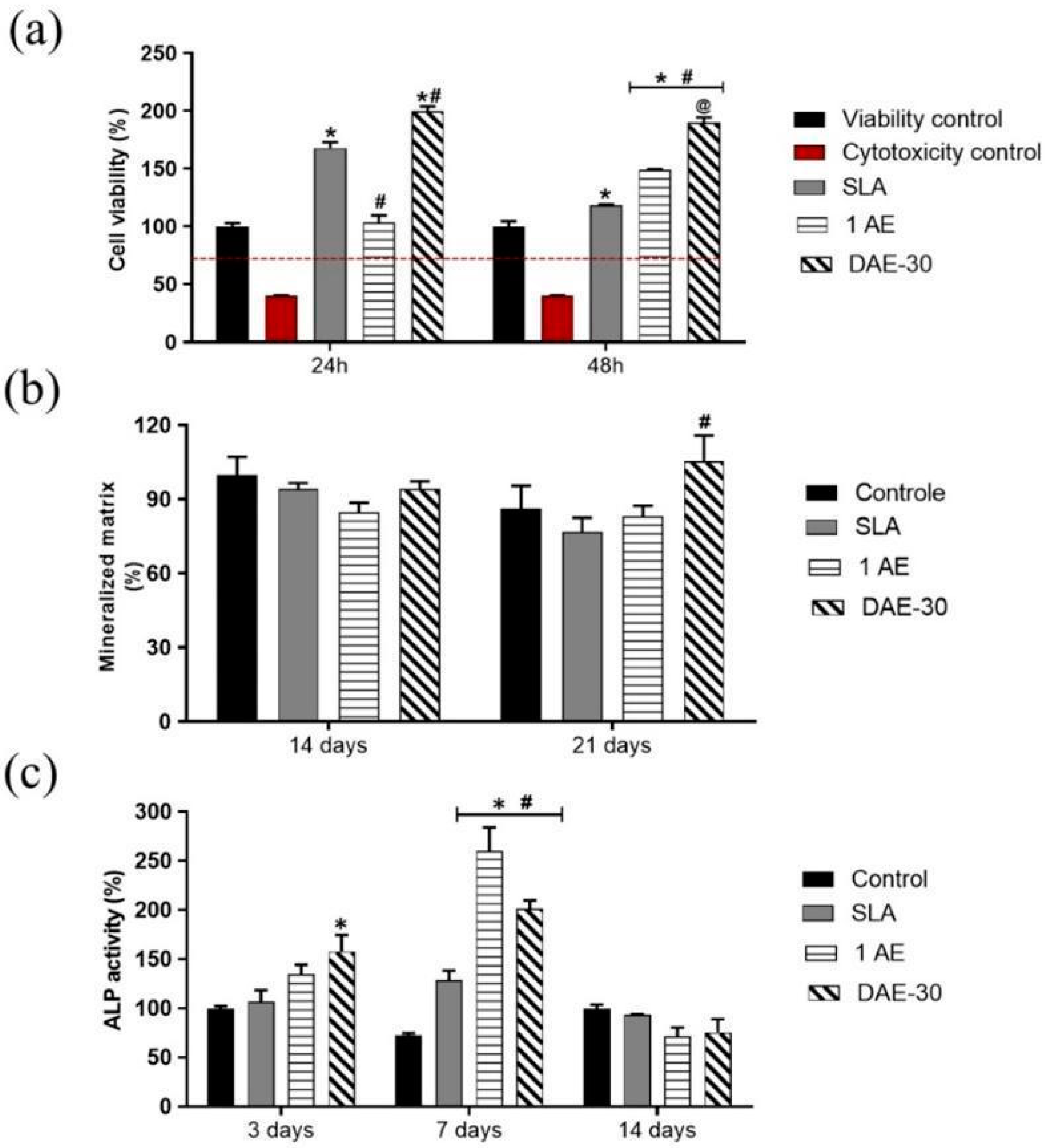
3.5. In Vitro Tests
4. Discussion
5. Limitations in Research and Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Davenport, R. Patterns of death, 1800–2020: Global rates and causes. In The Routledge History of Death Since 1800; Taylor & Francis: London, UK; New York, NY, USA, 2021; pp. 21–44. [Google Scholar]
- Alghamdi, H.S.; Jansen, J.A. The development and future of dental implants. Dent. Mater. J. 2020, 39, 167–172. [Google Scholar] [CrossRef] [PubMed]
- van Noort, R. Titanium: The implant material of today. J. Mater. Sci. 1987, 22, 3801–3811. [Google Scholar] [CrossRef]
- Singh, A.G.; Saini, P.; Pabla, B.S. In-vitro mineralization of bio-coated 3D printed metallic implant under simulated body conditions. Mater. Today Proc. 2022, 50, 1946–1952. [Google Scholar] [CrossRef]
- Eliades, T. Passive film growth on titanium alloys: Physicochemical and biologic considerations. Int. J. Oral Maxillofac. Implants 1997, 12, 621–627. [Google Scholar] [PubMed]
- Bocchetta, P.; Chen, L.Y.; Tardelli, J.D.C.; Reis, A.C.; Almeraya-Calderón, F.; Leo, P.L. Passive layers and corrosion resistance of biomedical Ti-6Al-4V and β-Ti alloys. Coatings 2021, 11, 487. [Google Scholar] [CrossRef]
- Mitsuo, N. Mechanical properties of biomedical titanium alloys. Mater. Sci. Eng. A 1998, 243, 231–236. [Google Scholar]
- Li, J.; Jansen, J.A.; Walboomers, X.F.; van den Beucken, J.J. Mechanical aspects of dental implants and osseointegration: A narrative review. J. Mech. Behav. Biomed. Mater. 2020, 103, 103574. [Google Scholar] [CrossRef] [PubMed]
- Brånemark, P.I.; Adell, R.; Breine, U.; Hansson, B.O.; Lindström, J.; Ohlsson, A. Intra-osseous anchorage of dental prostheses. I. Experimental studies. Scand. J. Plast. Reconstr. Surg. 1969, 3, 81–100. [Google Scholar] [CrossRef]
- Pandey, C.; Rokaya, D.; Bhattarai, B.P. Contemporary concepts in osseointegration of dental implants: A review, BioMed Res. Int. 2022, 2022, 6170452. [Google Scholar]
- de Souza, V.Z.; Manfro, R.; Joly, J.C.; Elias, C.N.; Peruzzo, D.C.; Napimoga, M.H.; Martinez, E.F. Viability and collagen secretion by fibroblasts on titanium surfaces with different acid-etching protocols. Int. J. Implant Dent. 2019, 5, 41. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, K.; Dong, K.; Cui, N.; Lu, T.; Han, Y. Enhanced osteogenic differentiation of osteoblasts on CaTiO3 nanotube film. Colloids Surf. B Biointerfaces 2020, 187, 110773. [Google Scholar] [CrossRef] [PubMed]
- Szmukler-Moncler, S.; Perrin, D.; Ahossi, V.; Magnin, G.; Bernard, J.P. Biological properties of acid etched titanium implants: Effect of sandblasting on bone anchorage. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 68, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.D.; Liu, T.T.; Wang, Q.Q.; Zhang, J.; Cao, M.S. Surface modification and functionalities for titanium dental implants. ACS Biomater. Sci. Eng. 2023, 9, 4442–4461. [Google Scholar] [CrossRef] [PubMed]
- López-Valverde, N.; Flores-Fraile, J.; Ramírez, J.M.; Sousa, B.M.; Herrero-Hernández, S.; López-Valverde, A. Bioactive surfaces vs. conventional surfaces in titanium dental implants: A comparative systematic review. J. Clin. Med. 2020, 9, 2047. [Google Scholar] [CrossRef]
- Velasco-Ortega, E.; Jimenez-Guerra, A.; Monsalve-Guil, L.; Ortiz-Garcia, I.; Nicolas-Silvente, A.I.; Segura-Egea, J.J.; Lopez-Lopez, J. Long-Term clinical outcomes of treatment with dental implants with acid etched surface. Materials 2020, 13, 1553. [Google Scholar] [CrossRef]
- El-Banna, A.; Bissa, M.W.; Khurshid, Z.; Zohaib, S.; Asiri, F.Y.; Zafar, M.S. Surface modification techniques of dental implants. In Dental Implants; Woodhead Publishing: Cambridge, UK, 2020; pp. 49–68. [Google Scholar]
- Nobles, K.P.; Janorkar, A.V.; Williamson, R.S. Surface modifications to enhance osseointegration–Resulting material properties and biological responses. J. Biomed. Mater. Res. B Appl. Biomater. 2021, 109, 1909–1923. [Google Scholar] [CrossRef]
- ASTM F67-13(2017); Standard Specification for Unalloyed Titanium, for Surgical Implant Applications (UNS R50250, UNS R50400, UNS R50550, UNS R50700). ASTM International: West Conshohocken, PA, USA, 2024.
- Nečas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Open Phys. 2012, 10, 181–188. [Google Scholar] [CrossRef]
- Kokubo, T.; Miyaji, F.; Kim, H.M.; Nakamura, T. Spontaneous formation of bonelike apatite layer on chemically treated titanium metals. J. Am. Ceram. Soc. 1996, 79, 1127–1129. [Google Scholar] [CrossRef]
- Yeo, I.S.L. Modifications of dental implant surfaces at the micro- and nano-level for enhanced osseointegration. Materials 2019, 13, 89. [Google Scholar] [CrossRef]
- Bharati, S.; Sinha, M.K.; Basu, D. Hydroxyapatite coating by biomimetic method on titanium alloy using concentrated SBF. Bull. Mater. Sci. 2005, 28, 617–621. [Google Scholar] [CrossRef]
- Bächle, M.; Kohal, R.J. A systematic review of the influence of different titanium surfaces on proliferation, differentiation and protein synthesis of osteoblast-like MG63 cells. Clin. Oral Implants Res. 2004, 15, 683–692. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Choi, S.H.; Ryu, J.J.; Koh, S.Y.; Park, J.H.; Lee, I.S. The biocompatibility of SLA-treated titanium implants. Biomed. Mater. 2008, 3, 025011. [Google Scholar] [CrossRef] [PubMed]
- Sezin, M.L.; Croharé, L.; Ibañez, J.C. Microscopic study of surface microtopographic characteristics of dental implants. Open Dent. J. 2016, 10, 139–147. [Google Scholar] [CrossRef]
- Hung, K.Y.; Lin, Y.C.; Feng, H.P. The effects of acid etching on the nanomorphological surface characteristics and activation energy of titanium medical materials. Materials 2017, 10, 1164. [Google Scholar] [CrossRef] [PubMed]
- Dhonde, M.; Dhonde, K.S.; Murty, V.V.S. Novel sol–gel synthesis of Al/N co-doped TiO2 nanoparticles and their structural, optical and photocatalytic properties. J. Mater. Sci. Mater. Electron. 2018, 29, 18465–18475. [Google Scholar] [CrossRef]
- Quéré, D. Wetting and roughness. Annu. Rev. Mater. Res. 2008, 38, 71–99. [Google Scholar] [CrossRef]
- Marchezini, E.; de Almeida, T.C.S.; Oliveira, F.P.; Albergaria, J.D.S.; Ghosh, S.; Tavares, M.A.B.; Leite, R.R.; Silva, G.A.B.; Martins, M.D. Comparative study of nanostructured TiO2 and SLA surface modifications for titanium implants: Surface morphology and in vitro evaluation. Mater. Res. 2022, 25, 1–10. [Google Scholar] [CrossRef]
- Weitzel, A.P.R.; de Mendonça, R.; Azzi, P.C.; Vieira, G.M.; de Almeida, T.C.; Rodrigues, C.F.; Rodrigues, E.M.; de Siqueira, J.G.; Nunes, E.H.; Martins, M.D. Comparative study of calcium phosphate deposition on nanotubular and sandblasted large grit acid-etched titanium substrates. Surf. Coat. Technol. 2023, 473, 130036. [Google Scholar] [CrossRef]
- Habibovic, P.; Barrère, F.; Van Blitterswijk, C.A.; de Groot, K.; Layrolle, P. Biomimetic hydroxyapatite coating on metal implants. J. Am. Ceram. Soc. 2002, 85, 517–522. [Google Scholar] [CrossRef]
- Song, W.H.; Jun, Y.K.; Han, Y.; Hong, S.H. Biomimetic apatite coatings on micro-arc oxidized titania. Biomaterials 2004, 25, 3341–3349. [Google Scholar] [CrossRef]
- Wen, C.; Guan, S.; Peng, L.; Ren, C.X.Z.H.; Wang, X.; Hu, Z. Characterization and degradation behavior of AZ31 alloy surface modified by bone-like hydroxyapatite for implant applications. Appl. Surf. Sci. 2009, 255, 6433–6438. [Google Scholar] [CrossRef]
- Maxian, S.H.; Zawadsky, J.P.; Dunn, M.G. In vitro evaluation of amorphous calcium phosphate and poorly crystallized hydroxyapatite coatings on titanium implants. J. Biomed. Mater. Res. 1993, 27, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Kim, H.M.; Kawashita, M. Novel bioactive materials with different mechanical properties. Biomaterials 2003, 24, 2161–2175. [Google Scholar] [CrossRef] [PubMed]
- Massaro, C.; Baker, M.A.; Cosentino, F.; Ramires, P.A.; Klose, S.; Milella, E. Surface and biological evaluation of hydroxyapatite-based coatings on titanium deposited by different techniques. J. Biomed. Mater. Res. 2001, 58, 651–657. [Google Scholar] [CrossRef]
- Forsgren, J.; Svahn, F.; Jarmar, T.; Engqvist, H. Formation and adhesion of biomimetic hydroxyapatite deposited on titanium substrates. Acta Biomater. 2007, 3, 980–984. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Cheng, T.C.; Fang, T.H.; Li, M.H. The fabrication and characteristics of hydroxyapatite film grown on titanium alloy Ti-6Al-4V by anodic treatment. J. Mater. Res. Technol. 2020, 9, 4817–4825. [Google Scholar] [CrossRef]
- Jaafar, A.; Hecker, C.; Árki, P.; Joseph, Y. Sol–gel derived hydroxyapatite coatings for titanium implants: A review. Bioengineering 2020, 7, 127. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Valenti, C.; Federici, M.I.; Coniglio, M.; Betti, P.; Pancrazi, G.P.; Tulli, O.; Masciotti, F.; Nanussi, A.; Pagano, S. Mechanical and biological properties of polymer materials for oral appliances produced with additive 3D printing and subtractive CAD-CAM techniques compared to conventional methods: A systematic review and meta-analysis. Clin. Oral Investig. 2024, 28, 396. [Google Scholar] [CrossRef] [PubMed]
- Almeida, T.; Valverde, T.; Martins, T.; Oliveira, F.d.P.; Cunha, P.d.S.; Tavares, M.A.B.; Rodrigues, E.M.; Albergaria, J.D.S.; Vieira, G.M.; Gomes, D.A.; et al. Enhanced osteogenic response by collagen type I coating on surface-modified titanium bone implants. Mater. Today Commun. 2024, 39, 108535. [Google Scholar] [CrossRef]
- Martin, J.Y.; Schwartz, Z.; Hummert, T.W.; Schraub, D.M.; Simpson, J.; Lankford, J., Jr.; Dean, D.D.; Cochran, D.L.; Boyan, B. Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells (MG63). J. Biomed. Mater. Res. 1995, 29, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Rupp, F.; Liang, L.; Geis-Gerstorfer, J.; Scheideler, L.; Hüttig, F. Surface characteristics of dental implants: A review. Dent. Mater. 2018, 34, 40–57. [Google Scholar] [CrossRef] [PubMed]
- Prins, H.J.; Braat, A.K.; Gawlitta, D.; Dhert, W.J.A.; Egan, D.A.; Tijssen-Slump, E.; Yuan, H.; Coffer, P.J.; Rozemuller, H.; Martens, A.C. In vitro induction of alkaline phosphatase levels predicts in vivo bone forming capacity of human bone marrow stromal cells. Stem Cell Res. 2014, 12, 428–440. [Google Scholar] [CrossRef] [PubMed]




| Group | Surface Treatment |
|---|---|
| SLA | Sandblasted + NaOH + HNO3 and HF |
| 1AE | H2SO4 + HCl at 60 °C for 60 min |
| DAE-10 | H2SO4 + HCl at 60 °C for 60 min + HNO3 and HF at 25 °C (10 s) |
| DAE-20 | H2SO4 + HCl at 60 °C for 60 min + HNO3 and HF a 25 °C (20 s) |
| DAE-30 | H2SO4 + HCl at 60 °C for 60 min + HNO3 and HF a 25 °C (30 s) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vieira, G.M.; Almeida, T.C.S.; Oliveira, F.P.; Azzi, P.C.; Rodrigues, C.F.; Souza, R.L.; Lacerda, S.M.S.N.; Lages, F.S.; Martins, M.D. Comparative Study of Acid Etching and SLA Surface Modification for Titanium Implants. Materials 2025, 18, 1632. https://doi.org/10.3390/ma18071632
Vieira GM, Almeida TCS, Oliveira FP, Azzi PC, Rodrigues CF, Souza RL, Lacerda SMSN, Lages FS, Martins MD. Comparative Study of Acid Etching and SLA Surface Modification for Titanium Implants. Materials. 2025; 18(7):1632. https://doi.org/10.3390/ma18071632
Chicago/Turabian StyleVieira, Gabriel M., Tatiane C. S. Almeida, Fernanda P. Oliveira, Patrícia C. Azzi, Caio F. Rodrigues, Rafael L. Souza, Samyra Maria S. N. Lacerda, Frederico S. Lages, and Maximiliano D. Martins. 2025. "Comparative Study of Acid Etching and SLA Surface Modification for Titanium Implants" Materials 18, no. 7: 1632. https://doi.org/10.3390/ma18071632
APA StyleVieira, G. M., Almeida, T. C. S., Oliveira, F. P., Azzi, P. C., Rodrigues, C. F., Souza, R. L., Lacerda, S. M. S. N., Lages, F. S., & Martins, M. D. (2025). Comparative Study of Acid Etching and SLA Surface Modification for Titanium Implants. Materials, 18(7), 1632. https://doi.org/10.3390/ma18071632






