Emerging Biomaterials for Maxillary Sinus Augmentation: From In Vitro Insights to In Vivo Clinical Translation
Abstract
1. Introduction
2. In Vitro Studies
3. Animal Models
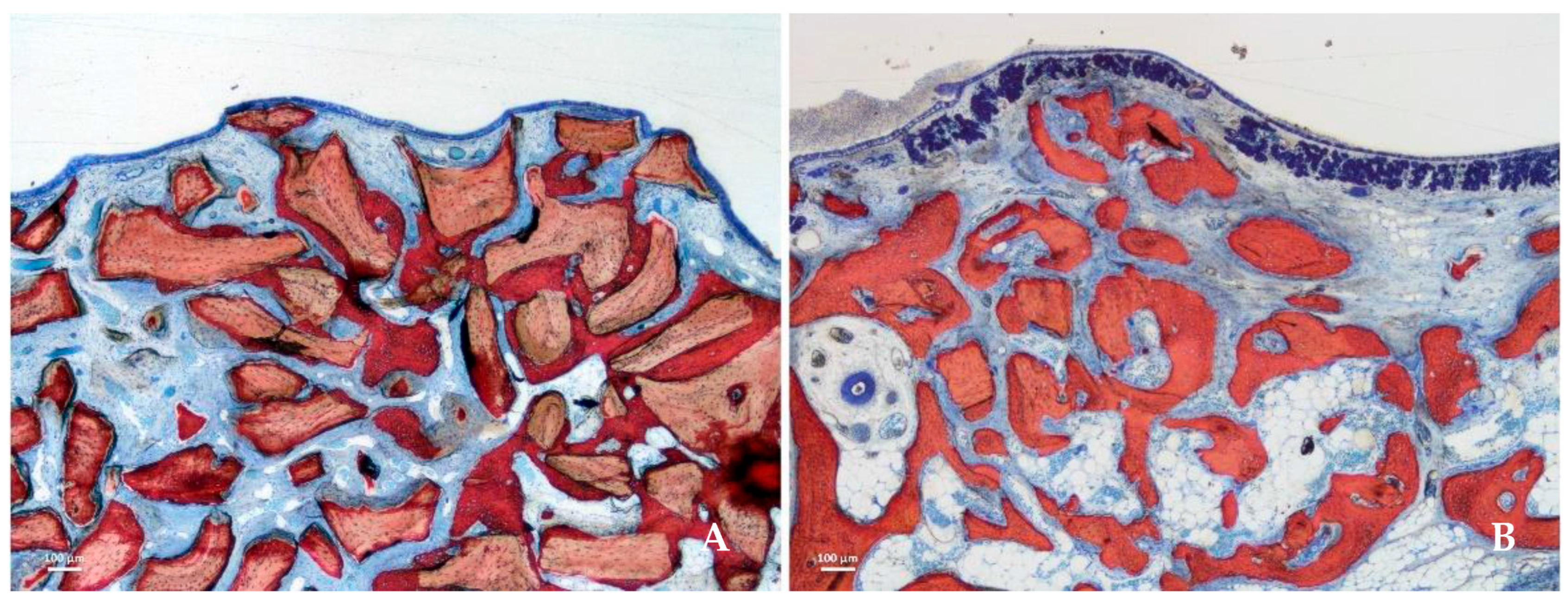
3.1. Deproteinized Bovine Bone Materials
3.2. Porcine-Derived Xenografts
3.3. Synthetic Polymers and Composite Materials
3.4. Growth-Factor-Enhanced Biomaterials
4. Clinical Cases
5. Discussion
6. Future Perspectives and Directions
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DBBM | deproteinized bovine bone mineral |
| β-TCP | β-tricalcium phosphate |
| HA/ | hydroxyapatite/ |
| β-TCP | β-tricalcium phosphate |
| PLGA | poly(lactic-co-glycolic acid) |
| BMP-2 | bone morphogenetic protein-2 |
| OCP | octacalcium phosphate |
| PRF | platelet-rich fibrin |
| BCP | biphasic calcium phosphate |
| LCM | lithography-based ceramic manufacturing |
| Micro-CT | X-ray micro computed tomography |
| FTIR | Fourier-transform infrared |
References
- Jang, H.-Y.; Kim, H.-C.; Lee, S.-C.; Lee, J.-Y. Choice of Graft Material in Relation to Maxillary Sinus Width in Internal Sinus Floor Augmentation. J. Oral Maxillofac. Surg. 2010, 68, 1859–1868. [Google Scholar] [CrossRef]
- Murphy, W.L.; Simmons, C.A.; Kaigler, D.; Mooney, D.J. Bone Regeneration via a Mineral Substrate and Induced Angiogenesis. J. Dent. Res. 2004, 83, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Hiatt, W.H.; Schallhorn, R.G.; Aaronian, A.J. The Induction of New Bone and Cementum Formation: IV. Microscopic Examination of the Periodontium Following Human Bone and Marrow Allograft, Autograft and Nongraft Periodontal Regenerative Procedures. J. Periodontol. 1978, 49, 495–512. [Google Scholar] [CrossRef] [PubMed]
- Younger, E.M.; Chapman, M.W. Morbidity at Bone Graft Donor Sites. J. Orthop. Trauma 1989, 3, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Ahlmann, E.; Patzakis, M.; Roidis, N.; Shepherd, L.; Holtom, P. Comparison of Anterior and Posterior Iliac Crest Bone Grafts in Terms of Harvest-Site Morbidity and Functional Outcomes. J. Bone Jt. Surg. Am. 2002, 84, 716–720. [Google Scholar] [CrossRef]
- Suda, A.J.; Schamberger, C.T.; Viergutz, T. Donor Site Complications Following Anterior Iliac Crest Bone Graft for Treatment of Distal Radius Fractures. Arch. Orthop. Trauma Surg. 2019, 139, 423–428. [Google Scholar] [CrossRef]
- Shin, S.Y.; Hwang, Y.J.; Kim, J.H.; Seol, Y.J. Long-Term Results of New Deproteinized Bovine Bone Material in a Maxillary Sinus Graft Procedure. J. Periodontal Implant Sci. 2014, 44, 259–264. [Google Scholar] [CrossRef]
- Caballé-Serrano, J.; Fujioka-Kobayashi, M.; Bosshardt, D.D.; Gruber, R.; Buser, D.; Miron, R.J. Pre-Coating Deproteinized Bovine Bone Mineral (DBBM) with Bone-Conditioned Medium (BCM) Improves Osteoblast Migration, Adhesion, and Differentiation In Vitro. Clin. Oral Investig. 2016, 20, 2507–2513. [Google Scholar] [CrossRef]
- Yu, X.; Tang, X.; Gohil, S.V.; Laurencin, C.T. Biomaterials for Bone Regenerative Engineering. Adv. Healthc. Mater. 2015, 4, 1268–1285. [Google Scholar] [CrossRef]
- Xiao, P.; Chen, C.; Shen, X.; Xu, A.; Ahmed, M.; Lu, H.; He, F. Bone Volume and Height Changes for Lateral Window Sinus Floor Elevation Using Two Types of Deproteinized Bovine Bone Mineral: A Retrospective Cohort Study of 1–4 Years. Clin. Oral Implant. Res. 2024, 35, 1493–1505. [Google Scholar] [CrossRef]
- Stacchi, C.; Berton, F.; Fiorillo, L.; Nicolin, V.; Lombardi, T.; Cicci, M.; Lenarda, R. Di Applied Sciences Fresh Frozen Allogeneic Bone Block in Maxillary Sinus Floor Elevation: Histomorphometric Analysis of a Bone Specimen Retrieved 15 Years after Grafting Procedure. Appl. Sci. 2019, 9, 1119. [Google Scholar] [CrossRef]
- Brett, E.; Flacco, J.; Blackshear, C.; Longaker, M.T.; Wan, D.C. Biomimetics of Bone Implants: The Regenerative Road. BioResearch Open Access 2017, 6, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hatt, L.P.; Armiento, A.R.; Mys, K.; Thompson, K.; Hildebrand, M.; Nehrbass, D.; Müller, W.E.G.; Zeiter, S.; Eglin, D.; Stoddart, M.J. Standard In Vitro Evaluations of Engineered Bone Substitutes Are Not Sufficient to Predict In Vivo Preclinical Model Outcomes. Acta Biomater. 2023, 156, 177–189. [Google Scholar] [CrossRef]
- Robinson, N.B.; Krieger, K.; Khan, F.M.; Huffman, W.; Chang, M.; Naik, A.; Yongle, R.; Hameed, I.; Krieger, K.; Girardi, L.N.; et al. The Current State of Animal Models in Research: A Review. Int. J. Surg. 2019, 72, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Al-Moraissi, E.A.; Alkhutari, A.S.; Abotaleb, B.; Altairi, N.H.; Del Fabbro, M. Do Osteoconductive Bone Substitutes Result in Similar Bone Regeneration for Maxillary Sinus Augmentation When Compared to Osteogenic and Osteoinductive Bone Grafts? A Systematic Review and Frequentist Network Meta-Analysis. Int. J. Oral Maxillofac. Surg. 2020, 49, 107–120. [Google Scholar] [CrossRef]
- Choy, C.S.; Lee, W.F.; Lin, P.Y.; Wu, Y.F.; Huang, H.M.; Teng, N.C.; Pan, Y.H.; Salamanca, E.; Chang, W.J. Surface Modified β-Tricalcium Phosphate Enhanced Stem Cell Osteogenic Differentiation In Vitro and Bone Regeneration In Vivo. Sci. Rep. 2021, 11, 9234. [Google Scholar] [CrossRef]
- Fricain, J.C.; Aid, R.; Lanouar, S.; Maurel, D.B.; Le Nihouannen, D.; Delmond, S.; Letourneur, D.; Amedee Vilamitjana, J.; Catros, S. In-Vitro and in-Vivo Design and Validation of an Injectable Polysaccharide-Hydroxyapatite Composite Material for Sinus Floor Augmentation. Dent. Mater. 2018, 34, 1024–1035. [Google Scholar] [CrossRef]
- da Silva Brum, I.; de Carvalho, J.J.; da Silva Pires, J.L.; de Carvalho, M.A.A.; dos Santos, L.B.F.; Elias, C.N. Nanosized Hydroxyapatite and β-Tricalcium Phosphate Composite: Physico-Chemical, Cytotoxicity, Morphological Properties and In Vivo Trial. Sci. Rep. 2019, 9, 19725. [Google Scholar] [CrossRef]
- Salamanca, E.; Pan, Y.H.; Sun, Y.S.; Hsueh, H.W.; Dorj, O.; Yao, W.L.; Lin, J.C.Y.; Teng, N.C.; Watanabe, I.; Abe, S.; et al. Magnesium Modified β-Tricalcium Phosphate Induces Cell Osteogenic Differentiation In Vitro and Bone Regeneration In Vivo. Int. J. Mol. Sci. 2022, 23, 1717. [Google Scholar] [CrossRef]
- Saito, K.; Inagaki, Y.; Uchihara, Y.; Okamoto, M.; Nishimura, Y.; Kawai, A.; Sugino, T.; Okamura, K.; Ogawa, M.; Kido, A.; et al. MgO-Enhanced β-TCP Promotes Osteogenesis in Both In Vitro and In Vivo Rat Models. Sci. Rep. 2024, 14, 19725. [Google Scholar] [CrossRef]
- Miki, M.; Botticelli, D.; Silva, E.R.; Xavier, S.P.; Baba, S. Incidence of Sinus Mucosa Perforations During Healing After Sinus Elevation Using Deproteinized Bovine Bone Mineral as Grafting Material: A Histologic Evaluation in a Rabbit Model. Int. J. Oral Maxillofac. Implants 2021, 36, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Yamada, R.; Xavier, S.P.; Nakajima, Y.; Silva, E.R.; Botticelli, D.; Teranishi, Y.; Baba, S. Impact of Collagenated and Non-Collagenated Deproteinized Bovine Bone Mineral on Schneiderian Membrane Integrity in Rabbits. Dent. J. 2025, 13, 19. [Google Scholar] [CrossRef]
- Maniwa, N.; Xavier, S.P.; Scombatti de Souza, S.L.; Silva, E.R.; Botticelli, D.; Morinaga, K.; Baba, S. Sequential Bone Repair in Rabbit Sinus Lifts Using Bio-Oss and Hyaluronic Acid–Polynucleotide Gel (Regenfast). J. Funct. Biomater. 2024, 15, 361. [Google Scholar] [CrossRef] [PubMed]
- Coyac, B.R.; Wu, M.; Bahat, D.J.; Wolf, B.J.; Helms, J.A. Biology of Sinus Floor Augmentation with an Autograft versus a Bone Graft Substitute in a Preclinical In Vivo Experimental Model. Clin. Oral Implants Res. 2021, 32, 916–927. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Z.; Esposito, M.; Xavier, S.P.; Silva, E.R.; Botticelli, D.; Buti, J.; Baba, S. On The Use of a Sticky Bone Substitute in the Presence of a Ruptured Schneider Membrane During Sinus Lift Procedures—An Experimental Within-Rabbit Study. Int. J. Oral Maxillofac. Implant. 2024, 4, 417–426. [Google Scholar] [CrossRef]
- Miyauchi, Y.; Izutani, T.; Teranishi, Y.; Iida, T.; Nakajima, Y.; Xavier, S.P.; Baba, S. Healing Patterns of Non-Collagenated Bovine and Collagenated Porcine Xenografts Used for Sinus Floor Elevation: A Histological Study in Rabbits. J. Funct. Biomater. 2022, 13, 276. [Google Scholar] [CrossRef]
- Lee, D.; Tae, K.; Yang, K.; Seol, J.; Moo, Y.; Jungwon, L. Comparison of Osteogenesis and Osseointegration Following Implant Placement with Simultaneous Maxillary Sinus Augmentation Using Particulate and Collagenated Block Types of Deproteinized Porcine Bone Mineral: A Radiographic and Histomorphometric Analysis. Clin. Oral Investig. 2023, 27, 5865–5874. [Google Scholar] [CrossRef]
- Mastrangelo, F.; Quaresima, R.; Sebastianelli, I.; Dedola, A.; Kuperman, S.; Azzi, L.; Mortellaro, C.; Muttini, A.; Mijiritsky, E. Poly D,L-Lactide-Co-Glycolic Acid Grafting Material in Sinus Lift. J. Craniofac. Surg. 2019, 30, 1076–1077. [Google Scholar] [CrossRef]
- Farjaminejad, S.; Farjaminejad, R.; Hasani, M.; Garcia-Godoy, F.; Abdouss, M.; Marya, A.; Harsoputranto, A.; Jamilian, A. Advances and Challenges in Polymer-Based Scaffolds for Bone Tissue Engineering: A Path Towards Personalized Regenerative Medicine. Polymers 2024, 16, 3303. [Google Scholar] [CrossRef]
- Cazzeli, F.; Akemi, M.; Cristina, C.; Claudia, A.; Rennó, M.; Andrade, L.D.; Ferreira, J.; Junior, S.; Salles, M.; Pinto, P. Distinct Healing Pattern of Maxillary Sinus Augmentation Using the Vitroceramic Biosilicate®: Study in Rabbits. Mater. Sci. Eng. C 2019, 99, 726–734. [Google Scholar] [CrossRef]
- Dou, Z.; Tang, H.; Chen, K.; Li, D.; Ying, Q.; Mu, Z.; An, C.; Shao, F.; Zhang, Y.; Zhang, Y.; et al. Highly Elastic and Self-Healing Nanostructured Gelatin/Clay Colloidal Gels with Osteogenic Capacity for Minimally Invasive and Customized Bone Regeneration. Biofabrication 2023, 15, 25001. [Google Scholar] [CrossRef]
- Sirak, S.V.; Giesenhagen, B.; Kozhel, I.V.; Schau, I.; Shchetinin, E.V.; Sletov, A.A.; Vukovic, M.A.; Grimm, W.-D. Osteogenic Potential of Porous Titanium. An Experimental Study in Sheep. J. Natl. Med. Assoc. 2018, 111, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Bystedt, H.; Rasmusson, L. Porous Titanium Granules Used as Osteoconductive Material for Sinus Floor Augmentation: A Clinical Pilot Study. Clin. Implant Dent. Relat. Res. 2009, 11, 101–105. [Google Scholar] [CrossRef]
- Susin, C.; Lee, J.; Fiorini, T.; De Freitas, R.M.; Chiu, H.C.; Prasad, H.S.; Buxton, A.N.; Wikesjö, U.M.E. Sinus Augmentation Using RhBMP-2/ACS in a Mini-Pig Model: Influence of an Adjunctive Ceramic Bone Biomaterial. J. Clin. Periodontol. 2018, 45, 1005–1013. [Google Scholar] [CrossRef] [PubMed]
- Ying, Y.; Li, B.; Liu, C.; Xiong, Z.; Bai, W.; Ma, P. Shape-Memory ECM-Mimicking Heparin-Modified Nanofibrous Gelatin Scaffold for Enhanced Bone Regeneration in Sinus Augmentation. ACS Biomater. Sci. Eng. 2022, 8, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Jang, T.S.; Kim, S.Y.; Lee, W.P. Octacalcium Phosphate Bone Substitute (Bontree®): From Basic Research to Clinical Case Study. Appl. Sci. 2021, 11, 7921. [Google Scholar] [CrossRef]
- Scarano, A.; Lorusso, F.; Staiti, G.; Sinjari, B.; Tampieri, A.; Mortellaro, C. Sinus Augmentation with Biomimetic Nanostructured Matrix: Tomographic, Radiological, Histological and Histomorphometrical Results after 6 Months in Humans. Front. Physiol. 2017, 8, 565. [Google Scholar] [CrossRef]
- Maglione, M.; Michelon, F.; Bevilacqua, L.; Bertolotto, M.; Rothweiler, R. Comparison of Two Graft Procedures in a Bilateral Sinus Lift by MRI and Histomorphometric Analysis: A Case Report. J. Oral Med. Oral Surg. 2024, 30, 21. [Google Scholar] [CrossRef]
- Shreehari, A.K.; Sahoo, N.K.; Kumar, D.; Malhi, R.S. Efficacy of Platelet-Rich Fibrin in Maxillary Sinus Augmentation and Simultaneous Implant Placement in Deficient Maxillary Ridges: A Pilot Comparative Study. Med. J. Armed Forces India 2024, 80, 420–427. [Google Scholar] [CrossRef]
- Trimmel, B.; Gyulai-Gaál, S.; Kivovics, M.; Jákob, N.P.; Hegedűs, C.; Szabó, B.T.; Dobó-Nagy, C.; Szabó, G. Evaluation of the Histomorphometric and Micromorphometric Performance of a Serum Albumin-Coated Bone Allograft Combined with A-PRF for Early and Conventional Healing Protocols after Maxillary Sinus Augmentation: A Randomized Clinical Trial. Materials 2021, 14, 1810. [Google Scholar] [CrossRef]
- La Monaca, G.; Iezzi, G.; Cristalli, M.P.; Pranno, N.; Sfasciotti, G.L.; Vozza, I. Comparative Histological and Histomorphometric Results of Six Biomaterials Used in Two-Stage Maxillary Sinus Augmentation Model after 6-Month Healing. Biomed Res. Int. 2018, 2018, 9430989. [Google Scholar] [CrossRef]
- Pereira, R.d.S.; de Carvalho, M.V.N.B.; Hochuli-Vieira, E.; Statkievicz, C.; Pereira Santos, D.L.; Augusto Neto, R.T.; Pinto, C.d.F.S.; Bennardo, F.; Mourão, C.F. Histomorphometric and Micro-CT Evaluation of Cerabone and Bio-Oss in Maxillary Sinus Lifting: A Randomized Clinical Trial. Medicina 2024, 60, 1834. [Google Scholar] [CrossRef]
- Souza, R.B.; Alves, H.D.L.; Oliveira, L.S.; Pires, F.R.; Vidigal, G.M.; Fischer, R.G. Histomorphometric and Microtomographic Analysis of Maxillary Sinus Augmentation Surgeries with and without Collagen Membrane on the Lateral Window: A Randomized Clinical Trial. Clin. Implant Dent. Relat. Res. 2024, 26, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Furlani, M.; Notarstefano, V.; Riberti, N.; D’Amico, E.; Pierfelice, T.V.; Mangano, C.; Giorgini, E.; Iezzi, G.; Giuliani, A. Healing Kinetics of Sinus Lift Augmentation Using Biphasic Calcium Phosphate Granules: A Case Series in Humans. Bioengineering 2025, 12, 848. [Google Scholar] [CrossRef]
- Mangano, C.; Riberti, N.; Orilisi, G.; Tecco, S.; Furlani, M.; Giommi, C.; Mengucci, P.; Giorgini, E.; Giuliani, A. Morphometric, Biomechanical and Macromolecular Performances of β-TCP Macro/Micro-Porous Lattice Scaffolds Fabricated via Lithography-Based Ceramic Manufacturing for Jawbone Engineering. J. Funct. Biomater. 2025, 16, 237. [Google Scholar] [CrossRef]
- Giuliani, A.; Iezzi, G.; Mazzoni, S.; Piattelli, A.; Perrotti, V.; Barone, A. Regenerative Properties of Collagenated Porcine Bone Grafts in Human Maxilla: Demonstrative Study of the Kinetics by Synchrotron Radiation Microtomography and Light Microscopy. Clin. Oral Investig. 2018, 22, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.H.; Shi, H.; Denes, C.E.; Cole, A.J.; Wang, Y.; Cheng, Y.Y.; Hesselson, D.; Roelofs, S.H.; Neely, G.G.; Jang, J.-H.; et al. Advanced Pathophysiology Mimicking Lung Models for Accelerated Drug Discovery. Biomater. Res. 2023, 27, 35. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (US) Committee on Methods of Producing Monoclonal Antibodies. Summary of Advantages and Disadvantages of In Vitro and In Vivo Methods. In Monoclonal Antibody Production; National Academies Press (US): Washington, DC, USA, 1999.
- Guo, J.; Weng, J.; Rong, Q.; Zhang, X.; Zhu, S.; Huang, D.; Li, X.; Chen, S. Investigation of Multipotent Postnatal Stem Cells from Human Maxillary Sinus Membrane. Sci. Rep. 2015, 5, 11660. [Google Scholar] [CrossRef]
- Wei, B.; Wei, M.; Huang, H.; Fan, T.; Zhang, Z.; Song, X. Mesenchymal Stem Cell-Derived Exosomes: A Promising Therapeutic Strategy for Age-Related Diseases. Cell Prolif. 2025, 58, e13795. [Google Scholar] [CrossRef]
- Zanette, R.d.S.S.; Fayer, L.; Vasconcellos, R.; de Oliveira, L.F.C.; Maranduba, C.M.d.C.; de Alvarenga, É.L.F.C.; Martins, M.A.; Brandão, H.d.M.; Munk, M. Cytocompatible and Osteoinductive Cotton Cellulose Nanofiber/Chitosan Nanobiocomposite Scaffold for Bone Tissue Engineering. Biomed. Mater. 2023, 18, 055016. [Google Scholar] [CrossRef]
- Shchukarev, A.; Ransjö, M.; Mladenovi, Ž. To Build or Not to Build: The Interface of Bone Graft Substitute Materials in Biological Media from the View Point of the Cells. In Biomaterials Science and Engineering; Pignatello, R., Ed.; Books on Demand: New York, NY, USA, 2011. [Google Scholar]
- Yu, S.; Bd, Y.T.; Bd, Y.W.; Bd, M.F.; BMed, S.L.; BMed, G.T.; BMed, Z.Y.; Miron, R.J.; Zhang, Y.; Yang, Z.; et al. Early Tissue and Healing Responses after Maxillary Sinus Augmentation Using Horizontal Platelet Rich Fibrin Bone Blocks. BMC Oral Health 2023, 23, 589. [Google Scholar] [CrossRef]
- Nakajima, Y.; Botticelli, D.; De Rossi, E.F.; Ferreira Balan, V.; Pires Godoy, E.; Ricardo Silva, E.; Xavier, S.P. Schneiderian Membrane Collateral Damage Caused by Collagenated and Non-Collagenated Xenografts: A Histological Study in Rabbits. Dent. J. 2023, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Valentini, P.; Stacchi, C. Prevention and Management of Intra-Operative Complications in Maxillary Sinus Augmentation: A Review. Clin. Implant Dent. Relat. Res. 2025, 27, e13397. [Google Scholar] [CrossRef]
- Schiavo-Di Flaviano, V.; Egido-Moreno, S.; González-Navarro, B.; Velasco-Ortega, E.; López-López, J.; Monsalve-Guil, L. Influence of Schneiderian Membrane Perforation on Implant Survival Rate: Systematic Review and Meta-Analysis. J. Clin. Med. 2024, 13, 3751. [Google Scholar] [CrossRef] [PubMed]
- Bunyatratchata, O.; Nimlamool, W. The Impact of Centrifugation Devices and Collection Tubes on Fibrin Characteristics and Growth Factor Release Under High- and Low-Speed Protocols. Dent. J. 2025, 13, 476. [Google Scholar] [CrossRef]
- Barakat, M.; Khalil, A.; Albash, Z. Efficacy of Platelet-Rich Albumin and Fibrin Gel in Two-Stage Lateral Sinus Lift: A Prospective Study. Ann. Med. Surg. 2025, 87, 5409–5416. [Google Scholar] [CrossRef]
- Padmanaban, A.M.; Ganesan, K. A Co-Culture System for Studying Cellular Interactions in Vascular Disease. Bioengineering 2024, 11, 1090. [Google Scholar] [CrossRef] [PubMed]
- Duan, Q.; Shao, H.; Luo, N.; Wang, F.; Cheng, L.; Ying, J.; Zhao, D. 3D-Printed Artificial Bone Scaffolds: The Design of Materials, the Incorporation of Bioactive Substances, and the Integration of Vascularized Tissue Flaps. Front. Bioeng. Biotechnol. 2025, 13, 1614727. [Google Scholar] [CrossRef]
- Inchingolo, A.M.; Marinelli, G.; Trilli, I.; Vecchio, G.D.; Noia, A.D.; Inchingolo, F.; Fabbro, M.D.; Palermo, A.; Inchingolo, A.D.; Dipalma, G. A Histological and Clinical Evaluation of Long-Term Outcomes of Bovine Bone-Derived Xenografts in Oral Surgery: A Systematic Review. J. Funct. Biomater. 2025, 16, 321. [Google Scholar] [CrossRef]
- Qi, J.; Wu, H.; Liu, G. Novel Strategies for Spatiotemporal and Controlled BMP-2 Delivery in Bone Tissue Engineering. Cell Transplant. 2024, 33, 9636897241276732. [Google Scholar] [CrossRef]
- Park, S.-Y.; Kim, K.-H.; Kim, S.; Lee, Y.-M.; Seol, Y.-J. BMP-2 Gene Delivery-Based Bone Regeneration in Dentistry. Pharmaceutics 2019, 11, 393. [Google Scholar] [CrossRef] [PubMed]



| Group | Biomaterial | Animal Model & Timing | Evaluation Method | Key Findings | Study Ref. |
|---|---|---|---|---|---|
| DBBM | DBBM (small vs. large granules) |
Rabbit
2, 4, 8 weeks | Histology; histomorphometric analysis of sinus membrane thickness and perforations; light microscopy. | Progressive Schneiderian membrane thinning and perforations in both particle sizes; effect intrinsic to DBBM. | [21] |
| Collagenated DBBM vs. DBBM |
Rabbit
2 and 12 weeks | Histology; histomorphometry of membrane integrity and new bone formation at early and late time points. | Early membrane protection with collagen, but by 12 weeks, both materials show similar thinning and perforation; similar bone formation. | [22] | |
| DBBM + hyaluronic acid + polynucleotides |
Rabbit
2 and 10 weeks | Histology; histomorphometric quantification of new bone, residual graft, and membrane alterations. | No improvement in bone formation or membrane condition; adjunct gel does not enhance DBBM performance. | [23] | |
| Autograft vs. DBBM substitute |
Mouse
Days 0, 9, 18, 120 | Histology; alkaline phosphatase (ALP) activity assays; histomorphometry at multiple time points; long-term tissue evaluation. | Autograft shows fast early osteogenesis; DBBM slower initially but supports later bone formation; DBBM remains slowly resorbing long-term. | [24] | |
| Porcine xenografts | Sticky porcine graft vs. particulate porcine |
Rabbit
8–10 weeks | Micro-CT for volume stability; histology; histomorphometry of new bone and graft displacement. | Sticky graft reduces particle extrusion; both show ~50% volume loss; similar bone regeneration. | [25] |
| Collagenated porcine xenograft vs. DBBM |
Rabbit
2–8 weeks | Histology; histomorphometry of bone formation and graft resorption; sinus wall bone contact analysis. | Faster resorption and greater shrinkage with porcine collagen graft; increased bone along sinus walls compared to DBBM | [26] | |
| Particulate porcine vs. collagenated porcine block |
Beagle dog
8 weeks | Histology; histomorphometry of new bone, residual graft, and osseointegration at 8 weeks. | Block graft shortens insertion time; new bone, graft remnants, and osseointegration similar between groups. | [27] | |
| Synthetic composites | PLGA scaffold |
Sheep
16–28 weeks | Micro-CT; histological analysis; histomorphometric evaluation of bone remodeling and scaffold integration. | Good biocompatibility, progressive bone remodeling and integration; promising long-term maturation pattern | [28] |
| Biosilicate® vitroceramic |
Rabbit
15, 45, 90 days | Histology; inflammatory cell scoring; histomorphometric analysis of bone and granulation tissue over time. | Adequate bone formation but persistent inflammation and granulation tissue; biocompatibility concerns for sinus use. | [30] | |
| Gelatin/nanoclay colloidal gel (injectable, self-healing) | Rat/Mouse | Histology; histomorphometry; immunohistochemical markers of osteogenesis; in vivo bone ingrowth assessment. | Supports cell adhesion, osteogenic differentiation, bone ingrowth; injectable and conformable; favorable resorption; promising for minimally invasive grafting. | [31] | |
| Porous titanium granules |
Sheep
30, 60, 90 days | Micro-CT; histology; histomorphometric evaluation of osteoconduction and bone–material contact. | Strong osteoconduction and high bone formation, but remains permanently in place; non-resorbable long-term. | [32] | |
| Bioactive/growth-factor enhanced | rhBMP-2 + absorbable collagen sponge; vs. addition of biphasic ceramic |
Mini-pig
Several weeks | Micro-CT; histology; histomorphometry of newly formed bone; comparative quantitative analysis. | BMP-2 drives strong osteoinduction regardless of scaffold type; ceramic addition provides no additional benefit. | [34] |
| Heparin-modified gelatin nanofiber gel + BMP-2 |
Rat
8 weeks | Micro-CT; histology; quantitative bone volume assessment; osteogenesis scoring. | Highest osteogenic response among compared materials; potent early bone formation with controlled BMP-2 release | [35] |
| Biomaterial | Project Aim and Evaluation Methods | Main Findings | Study Ref. |
|---|---|---|---|
| Octacalcium phosphate (OCP)–based synthetic graft: Bontree® |
Develop a fully synthetic, resorbable, bioactive alternative to xenografts.
Histological analysis; histomorphometry; immunohistochemical markers of osteogenesis (ALP activity, RANKL expression); clinical implant follow-up. | Enhanced osteoblastic activity, reduced osteoclastogenesis; successful human graft integration; promising biologically active synthetic substitute. | [36] |
| Mg-hydroxyapatite/collagen scaffold (MgHA–collagen) |
Create a bioinspired hybrid scaffold mimicking natural bone and remodeling faster than xenografts.
Cone Beam CT; histology; scanning electron microscopy; histomorphometric quantification at 6 months. | At 6 months, graft largely replaced by new bone; high cellularity and vascularization; no residual scaffold; stable regenerated volume. | [37] |
| PRF + DBBM or autograft |
Develop imaging-based (non-radiation) monitoring and test PRF-induced angiogenesis.
Contrast-enhanced magnetic resonance imaging (MRI); long-term radiological follow-up; qualitative and quantitative assessment of graft vascularization and remodeling. | PRF accelerates vascularization and bone maturation; MRI effective for long-term graft assessment; PRF side shows better remodeling at 10 years. | [38] |
| PRF vs. Allograft |
To evaluate whether PRF alone can serve as a grafting material for direct sinus lift, reducing the need for bone allografts.
The residual bone height (RBH), implant mobility, and soft tissue characteristics between and within the groups were compared and analyzed. | Both groups gained bone height, but PRF alone produced significantly greater RBH increase at 6–9 months; demonstrated predictable and stable sinus augmentation without use of graft materials. | [39] |
| A-PRF + serum albumin–coated allograft (SACBA) |
Assess biological enhancement to accelerate healing.
Histomorphometry; micro-CT–based trabecular microarchitecture analysis comparing 3- and 6-month healing periods. | Similar bone quality and volume at 3 and 6 months; suggests healing time can be reduced using A-PRF + SACBA combination. | [40] |
|
Six biomaterials:
MCBA–Mineralized Cancellous Bone Allograft; FDBA–Freeze-Dried Bone Allograft; ABB (Bio-Oss®)–Anorganic Bovine Bone; EB–Equine Bone; BCP–Biphasic Calcium Phosphate; Bioapatite–collagen composite |
Compare performance of allografts, xenografts, and synthetics under identical conditions.
Histological analysis of bone biopsies; (new bone, residual graft, connective tissue fractions). | All biocompatible; FDBA showed highest new bone (32%); ABB lowest (~16%); modern allografts may outperform xenografts in early formation. | [41] |
| Bio-Oss vs. Cerabone (bovine xenografts) |
Bio-Oss vs. Cerabone (bovine xenografts)
Micro-CT analysis was performed to assess bone volume fraction, trabecular thickness, number, and separation. | Both effective, but Cerabone produced significantly more new bone and higher bone volume fraction at 6 months than Bio-Oss, indicating superior regenerative performance. | [42] |
| Bio-Oss ± collagen membrane over lateral window |
Test whether membrane on osteotomy window improves bone yield.
Histomorphometric analysis of biopsies; micro-CT analysis of regenerated bone volume and architecture. | No difference in bone quantity or quality; membrane coverage of lateral window may be unnecessary. | [43] |
| Biphasic calcium phosphate (BCP) graft (30% HA/70% β-TCP) |
Track temporal regeneration.
Histology; Raman spectroscopy; macromolecular composition analysis; temporal assessment of mineral and collagen maturation. | Progressive remodeling at 6–12 months; β-TCP component resorbs steadily; increasing collagen and mineral organization; BCP acts as a dynamic scaffold. | [44] |
| β-TCP macro/micro-porous 3D-printed scaffold (Lithography-based Ceramic Manufacturing (LCM)) |
To create a fully synthetic, architecturally optimized scaffold for sinus augmentation and jawbone regeneration.
Postoperative evaluation: µCT, synchrotron imaging, histology, and Fourier Transform Infrared Imaging. | Highly porous, mechanically stable lattice supporting active new bone formation and controlled scaffold resorption in human biopsy; confirmed good osteoconductivity and biocompatibility. | [45] |
| Collagenated porcine bone (mp3®) |
Evaluate materials with preserved natural collagen for better clot stability and cell migration.
Synchrotron radiation X-ray microtomography; histological evaluation; 3D quantitative trabecular analysis over time. | Gradual graft resorption; excellent trabecular organization; improved early integration and cellular infiltration. | [46] |
| Observation | Interpretative Meaning | |
|---|---|---|
| Most frequently used polymer | Collagen (animal and human studies) | Collagen is the dominant polymer due to its biocompatibility, biological signaling capacity, and regulatory acceptance. |
| Roles of collagen | Structural matrix, biological carrier, barrier membrane | Collagen serves multifunctional roles: scaffold integration, growth-factor delivery, clot stabilization, and soft-tissue protection. |
| Synthetic polymers | PLGA used exclusively in animal models | Synthetic polymers are still in an exploratory or preclinical phase, likely due to regulatory and degradation predictability concerns. |
| Injectable polymeric systems | Gelatin, fibrin (PRF), polymeric gels | Injectable polymers improve handling, reduce invasiveness, and better adapt to sinus anatomy, favoring early healing. |
| Polymer–ceramic composites | Collagen–HA, gelatin–β-TCP, polymer–xenograft hybrids | Combining polymers with ceramics balances mechanical stability with biological activity and controlled resorption. |
| Purely ceramic vs. polymer-containing grafts | Ceramics dominate routine clinical use; polymers more common in experimental studies | Polymer-based systems are being tested to overcome the biological passivity of ceramics, but clinical adoption remains cautious. |
| Biological function of polymers | Angiogenesis support, cell recruitment, controlled degradation | Polymers act as biological modulators rather than load-bearing elements in sinus augmentation. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Riberti, N.; Furlani, M.; Giuliani, A. Emerging Biomaterials for Maxillary Sinus Augmentation: From In Vitro Insights to In Vivo Clinical Translation. Materials 2026, 19, 737. https://doi.org/10.3390/ma19040737
Riberti N, Furlani M, Giuliani A. Emerging Biomaterials for Maxillary Sinus Augmentation: From In Vitro Insights to In Vivo Clinical Translation. Materials. 2026; 19(4):737. https://doi.org/10.3390/ma19040737
Chicago/Turabian StyleRiberti, Nicole, Michele Furlani, and Alessandra Giuliani. 2026. "Emerging Biomaterials for Maxillary Sinus Augmentation: From In Vitro Insights to In Vivo Clinical Translation" Materials 19, no. 4: 737. https://doi.org/10.3390/ma19040737
APA StyleRiberti, N., Furlani, M., & Giuliani, A. (2026). Emerging Biomaterials for Maxillary Sinus Augmentation: From In Vitro Insights to In Vivo Clinical Translation. Materials, 19(4), 737. https://doi.org/10.3390/ma19040737









