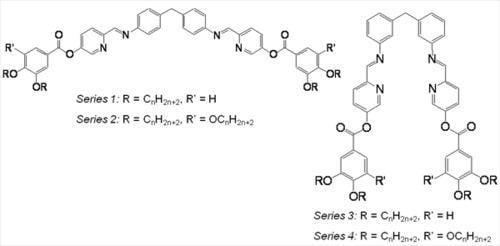
3.1. General
The starting materials which were commercially available were purchased from Aldrich and used as received. The solvents were purchased from Fisher Scientific (CH2Cl2, EtOAc, Et2O and MeOH) or Aldrich (anhydrous EtOH). Solvents were either used as received or dried; DCM was distilled from CaCl2 under a N2 atmosphere. Yields refer to chromatographically pure products. Thin-layer chromatography (TLC) was carried out on aluminum sheets coated with silica gel 60 (Merck 5554 mesh). Column chromatography was performed on silica gel 60 (Merck 230-400). Microanalyses were performed by the University of London microanalytical laboratories or by the University of Birmingham microanalytical services. Electron impact (EI) mass spectra were recorded at 70eV on a VG ProSpec mass spectrometer. Liquid secondary ion mass spectra (LSIMS) were recorded on a VG ZaBSpec mass spectrometer equipped with a cesium ion source and utilizing m-nitrobenzyl alcohol containing a trace of sodium acetate as the liquid matrix. 1H-NMR spectra were recorded on a Bruker AC 300 (300 MHz) spectrometer. 13C-NMR spectra were recorded on a Bruker AC 300 (75.5 MHz) spectrometer. The chemical shift values are expressed as δ values and the coupling constant values (J) are in Hertz (Hz). The following abbreviations are used for the signal multiplicities or characteristics: s, singlet; d, doublet; dd, doublet of doublets; t, triplet; m, muliplet; q, quartet; quint; quintet; br, broad. Transition temperatures were measured using a Mettler FP82 HT hot stage and central processor in conjunction with Leitz DMFRT Polarizing microscope as well as differential scanning calorimetry (DSC7 Perkin-Elmer).
Methyl 3,4-dihydroxybenzoate (8): A solution of 3,4-dihydroxybenzoic acid (6, 15.00 g, 97.30 mmol) and conc. H2SO4 (1.5 mL) in MeOH (200 mL) was stirred and heated under reflux overnight. The resultant yellow solution was allowed to cool to room temperature and the solvent removed in vacuo, affording a brown solid (14.60 g, 82%). mp: 126-129 °C; 1H-NMR (acetone-d6): δH 8.55 (br s, 1H, OH), 7.50 (d, J = 2.0 Hz, 1H, Ar-H), 7.44 (dd, 1H, Ar-H, J = 8.3, 2.0 Hz), 6.90 (d, 1H, Ar-H, J = 8.3 Hz,), 3.80 (s, 3H, OMe), 3.01 (br s, 1H, OH); 13C-NMR (acetone-d6): δC 167.0, 150.7, 145.5, 123.2, 122.7, 117.0, 115.7, 51.8; MS m/z (EIMS): 168 ([M]+, 54%), 137 ([M-OMe]+, 100%), 109 ([M-CO2Me]+, 20%); IR ν/cm-1 (nujol): 3468, 3263, 2927, 2854, 1690.
Methyl 3,4,5-trihydroxybenzoate (9): The same procedure as described for the preparation of 8 was followed, using 3,4,5-trihydroxybenzoic acid (7, 16.60 g, 97.40 mmol), conc. H2SO4 (0.5 mL) and MeOH (200 mL). This yielded a pale brown solid (14.40 g, 80%). mp: 120-124 °C; 1H-NMR (acetone-d6): δH 7.11 (s, 2H, Ar-H), 3.78 (s, 3H, OMe); 13C-NMR (acetone-d6): δC 167.9, 146.7, 139.5, 122.5, 110.5, 49.3; MS m/z (EIMS): 184 ([M]+, 55%), 153 ([M-OMe]+, 100%), 125 ([M-CO2Me]+, 17%); IR ν/cm-1 (nujol): 3468, 3263, 1690.
Methyl 3,4-dipentyloxybenzoate (10a): A suspension of K2CO3 (9.90 g, 71.63 mmol) in a solution of methyl 3,4-dihydroxybenzoate (8, 3.00 g, 17.84 mmol) and n-bromopentane (6.75 g, 44.69 mmol) in MeCN (100 mL) was stirred and heated under reflux overnight. The resultant brown mixture was allowed to cool to room temperature and concentrated in vacuo (30 mL). Water (250 mL) was added and the aqueous layer was extracted by washing with Et2O (3 x 100 mL). The combined organic layers were washed with brine solution (50 mL), dried (MgSO4), filtered and solvent removed in vacuo, yielding an orange oil as the crude product. The crude product was purified via silica gel column chromatography (gradient elution: 0 to 10 % EtOAc in hexane, increase polarity in increments of 5% per 150 mL of eluent used). The solvent was removed in vacuo to yield a colourless oil (4.84 g, 88%). 1H-NMR (CDCl3): δH 7.62 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.53 (d, , 1H, Ar-H, J = 2.2 Hz), 6.86 (d, 1H, Ar-H, J = 8.5 Hz), 4.05-4.02 (m, 4H, 2 x OCH2), 3.88 (s, 3H, OMe), 1.89-1.79 (m, 4H, 2 x CH2), 1.50-1.33 (m, 8H, 4 x CH2), 0.87 (t, 6H, 2 x CH3, J = 7.0Hz); 13C-NMR (CDCl3): δC 167.0, 153.1, 148.4, 123.5, 122.4, 114.1, 111.9, 69.2, 69.0, 51.9, 28.9, 28.2, 22.5, 14.1; MS m/z (EIMS): 308 ([M]+, 28 %), 168 ([M-(2 x C5H10)]+, 137 ([M-(2 x C5H10)-OMe]+, 24%); IR ν/cm-1 (nujol): 2926, 2856, 1718.
Using the same general procedure as described for 10a, the following compounds were prepared:
Methyl 3,4-diheptyloxybenzoate (10b): From K2CO3 (9.90 g, 71.63 mmol) methyl 3,4-dihydroxy-=benzoate (8, 3.00 g, 17.84 mmol) and n-bromoheptane (8.10 g, 45.23 mmol). This yielded a pale yellow oil (6.00 g, 92%). mp: 30-32 °C; 1H-NMR (CDCl3): δH 7.63 (dd, 1H, Ar-H, J = 8.5, 2.0 Hz), 7.53 (d, 1H, Ar-H, J = 2.0 Hz), 6.86 (d, 1H, Ar-H, J = 8.5 Hz), 4.05- 4.03 (m, 4H, 2 x OCH2), 3.88 (s, 3H, OMe), 1.85-1.80 (m, 4H, 2 x CH2), 1.31-1.25 (m, 16H, 8 x CH2), 0.88 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 167.2, 153.3, 148.6, 123.7, 122.5, 114.3, 112.0, 69.4, 69.1, 52.1, 32.0, 29.2, 26.1, 22.8, 14.2; MS m/z (EIMS): 365 ([M+H]+, 100%); IR ν/cm-1 (nujol): 2926, 2856, 1723.
Methyl 3,4-dinonyloxybenzoate (10c): From K2CO3 (9.90 g, 71.63 mmol), methyl 3,4-dihydroxy-benzoate (8, 3.00 g, 17.84 mmol) and n-bromononane (9.26 g, 44.70 mmol). This yielded a white solid (6.64 g, 88%). mp: 36-38 °C 1H-NMR (CDCl3): δH 7.62 (dd, 1H, Ar-H, J = 8.5, 2.0 Hz), 7.52 (d, 1H, Ar-H, J = 2.0 Hz), 6.85 (d, 1H, Ar-H, J = 8.5 Hz), 4.05-4.03 (m, 4H, 2 x OCH2), 3.87 (s, 3H, OMe), 1.88-1.76 (m, 4H, 2 x CH2), 1.46-1.25 (m, 24H, 12 x CH2), 0.87 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 167.0, 153.2, 148.5, 123.5, 122.4, 114.2, 111.9, 69.3, 69.0, 51.9, 31.9, 29.6, 29.4, 29.3, 29.1, 26.0, 22.7, 14.1; MS m/z (EIMS): 420 ([M]+, 46%), 154 ([M-(2 x C7H14)]+, 100%); IR ν/cm-1 (nujol): 2927, 2853, 1723.
Methyl 3,4-didodecanyloxybenzoate (10d): From K2CO3 (9.90 g, 71.63 mmol), methyl 3,4-dihydroxy-benzoate (8, 3.00 g, 17.84 mmol) and n-bromododecane (11.12 g, 44.62 mmol). This yielded a white solid (6.81 g, 76%). mp: 46-48 °C; 1H-NMR (CDCl3): δH 7.62 (dd, 1H, Ar-H, J = 8.5, 2.0 Hz), 7.52 (d, 1H, Ar-H, J = 2.0 Hz), 6.85 (d, 1H, Ar-H, J = 8.5 Hz), 4.05-4.03 (m, 4H, 2 x OCH2), 3.87 (s, 3H, OMe), 1.88-1.79 (m, 4H, 2 x CH2), 1.47-1.27 (m, 36H, 18 x CH2), 0.87 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 167.0, 157.3, 148.5, 123.5, 122.4, 114.2, 111.9, 69.3, 69.0, 51.9, 32.0, 29.7, 29.4, 29.2, 29.1, 26.0, 22.7, 14.2; MS m/z (EIMS): 505 ([M+H]+, 100%); IR ν/cm-1 (nujol): 2926, 2855, 1718.
Methyl 3,4,5-tripentyloxybenzoate (11a): Fromg methyl 3,4,5-trihydroxybenzoate (9, 3.83 g, 20.83 mmol) and n-bromopentane (11.01 g, 72.91 mmol), K2CO3 (11.50 g, 83.3 mmol) and MeCN (100 mL). This yielded a pale yellow oil (5.07 g, 61%). 1H-NMR (CDCl3): δH 7.24 (s, 2H, Ar-H), 4.01 (t, 6H, 3 x OCH2, J = 6.6 Hz), 3.88 (s, 3H, OMe), 1.86-1.69 (m, 6H, 3 x CH2), 1.48-1.32 (m, 12H, 6 x CH2), 0.92 (t, J = 7.0 Hz, 3H, CH3), 0.91 (t, 6H, 2 x CH3 J = 7.0 Hz); 13C-NMR (CDCl3): δC 165.4, 151.3, 140. 9, 123.0, 106.5, 71.9, 67.6, 50.5, 28.4, 27.4, 26.7, 21.0, 20.9, 12.5; MS m/z (EIMS): 394 ([M]+, 20%), 324 ([M-C5H10]+, 5%), 184 ([M-(3 x C5H10)]+, 100%); IR ν/cm-1 (nujol): 2925, 2854, 1718.
Methyl 3,4,5-triheptyloxybenzoate (11b): From K2CO3 (9.90 g, 71.63 mmol) methyl 3,4,5-trihydroxy-benzoate (9, 3.83 g, 20.82 mmol) and n-bromoheptane (13.04 g, 72.85 mmol). This yielded a yellow oil (5.97 g, 60%). 1H-NMR (CDCl3): δH 7.24 (s, 2H, Ar-H), 4.00 (t, 6H, 3 x OCH2, J = 6.6 Hz), 3.88 (s, 3H, OMe), 1.85-1.68 (m, 6H, 3 x CH2), 1.48-1.29 (m, 24H, 12 x CH2), 0.88 (t, 9H, 3 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 165.1, 151.0, 140.8, 122.9, 106.2, 71.6, 67.3, 50.2, 30.0, 28.5, 27.5, 27.3, 27.2, 24.2, 20.8, 12.2; MS m/z (EIMS): 478 ([M]+, 72 %), 380 ([M-C7H14]+, 25%), 184 ([M-(3 x C7H14)]+, 100%); IR ν/cm-1 (nujol): 2930, 2870, 1723.
Methyl 3,4,5-trinonyloxybenzoate (11c): From K2CO3 (9.90 g, 71.63 mmol) methyl 3,4,5-trihydroxy-benzoate (9, 3.83 g, 20.82 mmol) and n-bromononane (15.08 g, 72.85 mmol). This yielded a yellow oil (7.37 g, 63%). 1H-NMR (CDCl3): δH 7.24 (s, 2H, Ar-H), 4.00 (t, 6H, 3 x OCH2, J = 6.6 Hz), 3.88 (s, 3H, OMe), 1.85-1.68 (m, 6H, 3 x CH2), 1.48-1.27 (m, 36H, 18 x CH2), 0.88 (t, 9H, 3 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 165.8, 151.3, 140.9, 122.9, 106.8, 71.9, 67.7, 54.1, 33.9, 32.3, 31.6, 31.4, 31.3, 28.1, 24.7, 16.1; MS m/z (ESMS): 585 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2930, 2871, 1723.
Methyl 3,4,5-tridodecyloxybenzoate (11d): From K2CO3 (9.90 g, 71.63 mmol) methyl 3,4,5-trihydroxy-benzoate (9, 3.83 g, 20.82 mmol) and n-bromododecane (18.14 g, 72.85 mmol). This yielded a yellow oil (9.02 g, 63%). 1H-NMR (CDCl3): δH 7.24 (s, 2H, Ar-H), 4.00 (t, 6H, 3 x OCH2, J = 6.6 Hz), 3.88 (s, 3H, OMe), 1.85-1.68 (m, 6H, 3 x CH2), 1.48-1.29 (m, 54H, 27 x CH2), 0.88 (t, 9H, 3 x CH3, J = 6.99 Hz); 13C-NMR (CDCl3): δC 166.2, 152.0, 141.2, 123.1, 107.2, 72.6, 68.3, 51.0, 31.0, 29.4, 28.2, 28.4, 28.1, 25.0, 21.8, 13.2; MS m/z (ESMS): 688 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2930, 2870, 1723.
3,4-Dipentyloxybenzoic acid (12a): To a solution of methyl 3,4-dipentyloxybenzoate (10a, 4.84 g, 15.7 mmol) in MeOH (150 mL), an aqueous solution of sodium hydroxide (15 mL, 2.10 mol L-1) was added and the mixture was stirred and heated under reflux overnight. The resultant colourless solution was allowed to cool to room temperature and HCl (2 M) was added dropwise to acidify the solution, whereupon a white precipitate was formed, which was filtered off. The filtrate was concentrated in vacuo (~20 mL) and water (100 mL) was added. The aqueous layer was extracted with Et2OAc (3 x 100 mL). The combined organic layers were washed with brine solution (20 mL), dried (MgSO4) and filtered. The solvent was removed in vacuo, yielding a white solid (4.00 g, 87%). mp: 126-128 °C; 1H- NMR (CDCl3): δH 7.72 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.58 (d, 1H, Ar-H, J = 2.2 Hz), 6.89 (d, 1H, Ar-H, J = 8.5 Hz), 4.07-4.04 (m, 4H, 2 x OCH2), 1.88-1.82 (m, 4H, 2 x CH2), 1.49-1.38 (m, 8H, 4 x CH2), 0.87 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 171.7, 153.9, 148.5, 124.5, 121.4, 114.5, 111.8, 69.3, 69.0, 28.7, 28.2, 22.5, 14.1; MS m/z (EIMS): 294 ([M]+, 25%), 224 ([M-C5H10]+, 9%), 154 ([M-(2 x C5H10)]+; IR ν/cm-1 (nujol): 2926, 2856, 2646, 1684.
Following the same general procedure described for 12a, the following compounds were prepared:
3,4-Diheptyloxybenzoic acid (12b): From compound 11b (5.94 g, 16.30 mmol) and sodium hydroxide (15 mL, 2.18 mol L‑1). This yielded a white solid (4.70 g, 82%). mp: 120-123 °C; 1H-NMR (CDCl3): δH 7.70 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.56 (d, 1H, Ar-H, J = 2.2 Hz), 6.87 (d, 1H, Ar-H, J = 8.5 Hz), 4.05-4.02 (m, 4H, 2 x OCH2), 1.83-1.81 (m, 4H, 2 x CH2), 1.31-1.25 (m, 16H, 8 x CH2), 0.88 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 171.9, 153.7, 148.3, 124.3, 121.3, 114.3, 111.6, 69.1, 68.8, 31.0, 28.9, 25.8, 22.8, 13.9; MS m/z (EIMS): 351 ([M+H]+, 25%), 333 ([M-OH]+, 100%); IR ν/cm-1 (nujol): 2925, 2855, 2646, 1670s.
3,4-Dinonyloxybenzoic acid (12c): From compound 10c (6.42 g, 15.26 mmol) and sodium hydroxide (15 mL, 2.03 mol L‑1). This yielded a white solid (5.85 g, 94%). mp: 112-114°C; 1H-NMR (CDCl3): δH 7.71 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.52 (d, 1H, Ar-H, J = 2.2 Hz), 6.85 (d, 1H, Ar-H, J = 8.5 Hz), 4.07-4.03 (m, 4H, 2 x OCH2), 1.87–1.79 (m, 4H, 2 x CH2), 1.49–1.27 (m, 24H, 12 x CH2), 0.87 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 172.0, 154.1, 148.7, 124.7, 121.5, 114.6, 112.0, 69.4, 69.2, 32.1, 29.7, 29.6, 26.5, 26.2, 22.9, 14.3; MS m/z (EIMS): 406 ([M]+, 46%), 280 ([M-C7H14]+, 154 ([M-(2 x C7H14)]+, 100%); IR ν/cm-1 (nujol): 2926, 2854, 2644, 1670.
3,4-Didodecanyloxybenzoic acid (12d): From compound 10d (6.55 g, 12.98 mmol) and sodium hydroxide (15 mL, 1.87 mol L-1). This yielded a white solid (5.64 g, 89%). mp: 106-109 °C; 1H-NMR (CDCl3): δH 7.71 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.57 (d, 1H, Ar-H, J = 2.2 Hz), 6.88 (d, 1H, Ar-H, J = 8.5 Hz), 4.07-4.03 (m, 4H, 2 x OCH2), 1.87-1.79 (m, 4H, 2 X CH2), 1.52-1.26 (m, 36H, 18 x CH2), 0.87 (t, 6H, 2 x CH3 J = 6.6 Hz); 13C-NMR (CDCl3): δC 172.1, 154.3, 148.8, 124.9, 121.6, 114.7, 112.1, 69.6, 69.4, 32.3, 29.9, 29.8, 26.5, 26,4, 26.2, 25.6, 22.9, 14.5; MS m/z (ESMS): 513 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2926, 2854, 2644, 1670.
3,4,5-Tripentyloxybenzoic acid (13a): From compound 11a (4.62 g, 11.73 mmol) and sodium hydroxide (15 mL, 2.18 mol L-1). This yielded a white solid (4.46 g, 74%). mp: 48-50 °C; 1H-NMR (CDCl3): δH 7.12 (s, 2H, Ar-H), 3.89 (t, 2H, OCH2, J = 6.4 Hz), 3.79 (t, 4H, 2 x OCH2, J =5.9 Hz), 1.61-1.69 (m, 6H, 3 x CH2), 1.42-1.25 (m, 12H, 6 x CH2), 0.89 (t, 3H, CH3, J = 7.0 Hz), 0.85 (t, 6H, 2 x CH3, J = 6.8 Hz); 13C-NMR (CDCl3): δC; 173.4, 153.5, 142.7, 127.4, 108.9, 74.1, 69.7, 31.0, 30.1, 29.3, 29.2, 23.5, 14.9; MS m/z (EIMS): 380 ([M]+, 25%), 170 ([M-(3 x C5H11)]+, 100 %); IR ν/cm-1 (nujol): 2927, 2854, 2646, 1684.
3,4,5-Triheptyloxybenzoic acid (13b): From compound 11b (5.50 g, 11.51 mmol) and sodium hydroxide (15 mL, 2.18 mol L‑1). This yielded a white solid (4.02 g, 76%). mp: 47-49 °C; 1H-NMR (CDCl3): δH 7.31 (s, 2H, Ar-H), 4.03 (t, 2H, OCH2, J = 6.6 Hz), 4.01 (t, 4H, 2 x OCH2, J = 6.6 Hz), 1.86-1.70 (m, 6H, 3 x CH2), 1.52-1.29 (m, 24H, 12 x CH2), 0.88 (t, 9H, 3 x CH3, J = 6.6 Hz,); 13C-NMR (CDCl3): δC 173.8, 153.5, 142.9, 127.6, 109.0, 74.3, 69.8, 329, 32.8, 31.4, 30.4, 30.2, 27.2, 27.0, 23.6, 15.0; MS m/z (ESMS): 487 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2926, 2854, 2646, 1670.
3,4,5-Trinonyloxybenzoic acid (13c): From compound 11c (7.10 g, 12.63 mmol) and sodium hydroxide (15 mL, 1.68 mol dm-3). This yielded a white solid (5.40 g, 78%). mp: 47-49 °C; 1H-NMR (CDCl3): δH 7.30 (s, 2H, Ar-H), 4.03 (t, 2H, OCH2, J = 6.6 Hz), 4.01 (t, , 4H, 2 x OCH2, J = 6.6 Hz), 1.86-1.69 (m, 6H, 3 x CH2), 1.47-1.27 (m, 36H, 18 x CH2), 0.87 (t, 9H, 3 x CH3, J = 6.6 Hz,); 13C-NMR (CDCl3): δC 170.8, 150.8, 140.1, 121.6, 106.5, 71.5, 67.1, 29.8, 28.2, 27.6, 27.5, 27.3, 27.2, 24.0, 20,6, 12.0; MS m/z (ESMS): 547 ([M-H]-, 100%); IR ν/cm-1 (nujol): 2928, 2855, 2647, 1670.
3,4,5-Tridodecyloxybenzoic acid (13d): From compound 11d (8.80 g, 12.79 mmol) and sodium hydroxide (15 mL, 1.71 mol L-1). This yielded a white solid (6.71 g, 81%). mp: 47-49 °C; 1H-NMR (CDCl3): δH 7.30 (s, 2H, Ar-H), 4.03 (t, 2H, OCH2, J = 6.6 Hz), 4.01 (t, 4H, 2 x OCH2, J = 6.6 Hz), 1.86-1.69 (m, 6H, 3 x CH2), 1.47-1.25 (m, 54H, 27 x CH2), 0.87 (t, 9H, 3 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 171.3, 151.1, 141.1, 122.1, 106.8, 71.8, 67.4, 30.2, 28.6, 27.9, 27.6, 27.5, 24.3, 20.9, 12.3; MS m/z (ESMS): 697 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2926, 2854, 1674s.
5-Hydroxy-2-methylpyridine N-oxide (15): A solution of 5-hydroxy-2-methylpyridine (14, 15.0 g, 0.14 mol) and hydrogen peroxide 27.5 % (33 mL, 0.27 mol) in CH3CO2H (300 mL) was heated at 80 °C for 2.5 hrs. A colour change of solution was observed from orange to pale yellow. The solution was further stirred at room temperature for 24 hrs and concentrated in vacuo (50 mL). Acetone (400 mL) was added to force precipitation. The resulting pale yellow precipitate was collected by vacuum filtration (12.7 g, 74%). mp: 188.9-189.5 °C (decomposes); 1H-NMR (CD3OD): δH 7.93 (d, 1H, Py-H, J = 2.2 Hz), 7.32 (d, 1H, Py-H, J = 8.8 Hz), 7.03 (dd, 1H, Py-H, J = 8.8, 2.2 Hz), 2.41 (s, 3H, CH3); 13C-NMR (CD3OD): δC 158.7, 144.3, 136.0, 130.6, 122.1, 19.1; MS m/z (EIMS): 125 ([M]+, 50%), 109 ([M-O]+, 100%); IR ν/cm-1 (nujol): 2924, 2854, 1840, 1612.
2-Pyridylmethanol acetate (16): 5-Hydroxy-2-methylpyridine N-oxide (15, 17.5 g, 0.14 mol) was slowly added to (CH3CO)2O (170 mL) at 110 °C. The resultant dark brown reaction mixture was stirred for 2 hrs at 140 °C. EtOH (400 mL) was added and the solution was concentrated in vacuo to yield brown oil. CHCl3 (100 mL) was added and the solution was neutralised with saturated NaHCO3. The organic layers were collected through washings with saturated NaHCO3 (2 x 20 mL), dried (MgSO4) and filtered. The solvent was removed in vacuo to yield a brown oil, which was further dried under high vacuum for 2 hrs (26.1 g, 89%). 1H-NMR (CDCl3): δH 8.40 (d, 1H, Py-H, J = 2.6 Hz), 7.49 (d, 1H, Py-H, J = 8.5 Hz), 7.39 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 5.21 (s, 2H, CH2), 2.33 (s, 3H, CH3), 2.15 (s, 3H, CH3); 13C-NMR (CDCl3): δC 170.7, 168.9, 152.9, 146.7, 143.0, 130.0, 122.5, 66.3, 22.2, 21.0; MS m/z (FABMS): 210 ([MH]+, 100%); IR ν/cm-1 (nujol): 2923, 2854, 1747, 1582.
5-Hydroxy-2-hydroxymethylpyridine (17): A solution of 2-pyridylmethanol acetate (16) in conc. HCl (30 mL) was heated under reflux for 1.5 hrs. The reaction mixture was cooled to room temperature and concentrated in vacuo. CHCl3 (200 mL) was added and the solution neutralised carefully with saturated NaHCO3. The aqueous layers were collected through washings with saturated NaHCO3 and concentrated in vacuo. The brown solid was triturated with MeOH (500 mL) and filtrates collected through filtration. The solvent was removed in vacuo yielding a brown solid (8.3 g, 98%). 1H-NMR (CDCl3): δH 7.85 (d, 1H, Py-H, J = 2.6 Hz), 7.18 (d, 1H, Py-H, J = 8.5 Hz), 7.01 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 4.51 (s, 2H, CH2); 13C-NMR (CDCl3): δC 163.5, 147.8, 141.5, 128.0, 124.8, 66.7; MS m/z (FABMS): 125 ([M+H]+, 35%).
5-Hydroxypyridine 2-carbaldehyde (18): A suspension of 5-hydroxy-2-hydroxymethylpyridine (17, 2.50 g, 19.98 mmol) and activated MnO2 (1.74 g, 19.98 mmol) in iso-PrOH (100 mL) was heated under reflux for 3 hrs. The hot mixture was filtered through Celite® and washed thoroughly with hot MeOH (400 mL). The solution was allowed to cool to room temperature and concentrated in vacuo, yielding a brown solid. The solid was absorbed onto silica and purified via short silica gel column chromatography (gradient elution: 0 to 5% methanol in CH2Cl2, increase polarity in increments of 2 % per 150 mL of eluent used). The solvent was removed in vacuo to yield a brown solid (1.41 g, 57%). mp: 223-225 °C (decomposes); 1H-NMR (CD3OD): δH 9.60 (s, 1H, CHO), 7.90 (d, 1H, Py-H, J = 2.2 Hz), 7.74 (d, 1H, Py-H, J = 8.8 Hz), 6.84 (dd, 1H, Py-H, J = 8.8, 2.2 Hz), 13C-NMR (CD3OD): 193.0, 172.2, 146.5, 140.9, 130.2, 126.6; MS m/z (FABMS): 123 ([M]+, 50%), 109 ([M-CH2], 27%), 95 ([M-CO]+, 100%); IR ν/cm-1 (nujol): 2923, 2854, 1668, 1507.
3,4-Bisdipentyloxybenzoic acid 6-formylpyridin-3-ester (19a): To a solution of 5-hydroxypyridine-2-carbaldehyde (18, 0.50 g, 2.44 mmol) in dry CH2Cl2 (30 mL), cooled in an ice bath under a N2 atmosphere, compound 12a (0.72 g, 2.44 mmol), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC) (1.16 g, 60.55 mmol) and 4-dimethylaminopyridine (catalytical amount) were added. The solution was stirred at room temperature under a N2 atmosphere for 24 hrs. HCl (50 mL, 1 mol dm-3) was added, which yielded a white solid. MeOH (10 mL) was added to dissolve the precipitate. The organic layers were collected through washings with CH2Cl2 (3 x 30 mL), dried (MgSO4) and filtered. The solvent was removed in vacuo, to yield a brown solid. The solid was absorbed onto silica and purified via silica gel column chromatography (gradient elution: 0 to 10 % EtOAc in hexane, increase polarity in increments of 5% per 150 mL of eluent used). The solvent was removed in vacuo to yield a white solid (0.35 g, 36%); mp: 63-66 °C; 1H-NMR (CDCl3): δH 10.09 (s, 1H, CHO), 8.71 (d, 1H, Py-H, J = 8.5 Hz), 8.07 (d, 1H, Py-H, J = 2.6 Hz), 7.83 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.79 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 7.64 (d, 1H, Ar-H, J = 2.2 Hz), 6.94 (d, 1H, Ar-H, J = 8.5 Hz), 4.09 (t, 2H, OCH2, J = 6.6 Hz), 4.07 (t, 2H, OCH2, J = 6.6 Hz) 1.92-1.82 (m, 4H, 2 x CH2), 1.48-1.39 (m, 8H, 4 x CH2), 0.94 (t, 3H, CH3, J = 7.0 Hz), 0.93 (t, 3H, CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 192.1, 164.8, 155.4, 151.89, 150.8, 149.7, 145.0, 131.0, 125.7, 123.4, 120.9, 115.5, 112.8, 70.2, 70.0, 29.6, 29.5, 29.0, 23.3, 14.8; MS m/z (ESMS): 399 ([M]+, 20%), 277 ([M-OpyCO]+, 100%); IR ν/cm-1 (nujol): 2924, 2854, 1728.
Following the same general procedure described for 19a, the following compounds were prepared:
3,4-Bisdiheptyloxybenzoic acid 6-formylpyridin-3-ester (19b): From compound 18 (0.50 g, 2.44 mmol), compound 12b (0.85 g, 2.44 mmol) and EDC (1.16 g, 60.55 mmol) in dry CH2Cl2 (50 mL) to yield a white solid (0.40 g, 40%); mp: 66-68 °C; 1H-NMR (CDCl3): δH 10.08 (s, 1H, CHO), 8.70 (d, 1H, Py-H, J = 2.6 Hz), 8.07 (d, 1H, Py-H, J = 8.5 Hz), 7.82 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.79 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 7.64 (d, 1H, Ar-H, J = 2.2 Hz), 6.95 (d, 1H, Ar-H, J = 8.5 Hz), 4.09 (t, 2H, OCH2, J = 6.6 Hz), 4.06 (t, 2H, OCH2, J = 6.6 Hz), 1.89-1.83 (m, 4H, 2 x CH2), 1.49-1.30 (m, 16H, 8 x CH2), 0.89-0.84 (m, 6H, 2 x CH3); 13C-NMR (CDCl3): δC 192.2, 164.6, 152.7, 149.1, 148.1, 147.0, 142.3, 128.3, 123.0, 120.7, 118.2, 112.8, 110.1, 67.6, 67.3, 29.9, 27.3, 27.1, 24.1, 20.7, 12.2; MS m/z (FABMS): 478 ([M+Na]+, 25%); IR ν/cm-1 (nujol): 2925, 2854, 1728.
3,4-Bisdinonyloxybenzoic acid 6-formylpyridin-3-ester (19c): From compound 18 (0.50 g, 2.44 mmol), compound 12c (0.99 g, 2.44 mmol) and EDC (1.16 g, 60.55 mmol) in dry CH2Cl2 (50 mL). This yielded a white solid (0.45 g, 36%); mp: 68-70 °C; 1H-NMR (CDCl3): δH 10.08 (s, 1H, CHO), 8.71 (d, 1H, Py-H, J = 2.2 Hz), 8.07 (d, 1H, Py-H, J = 8.5 Hz), 7.83 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.79 (dd, 1H, Py-H, J = 8.5, 2.2 Hz), 7.64 (d, 1H, Ar-H, J = 2.2 Hz), 6.94 (d, 1H, Ar-H, J = 8.5 Hz), 4.09 (t, 2H, OCH2, J = 6.6 Hz), 4.06 (t, 2H, OCH2, J = 6.6 Hz), 1.89-1.82 (m, 4H, 2 x CH2), 1.49-1.28 (m, 24H, 12 x CH2), 0.89-0.85 (m, 6H, 2 x CH3); 13C-NMR (CDCl3): δC 192.1, 162.5, 153.7, 149.5, 148.5, 147.4, 142.7, 128.7, 123.4, 121.1, 118.6, 113.2, 110.5, 68.0, 67.7, 30.4, 28.1, 27.9, 27.8, 27.5, 24.5, 21.2, 12.6; MS m/z (ESMS): 534 ([M+Na]+, 85%); IR ν/cm-1 (nujol): 2930, 2855, 1730.
3,4-Bisdidodecyloxybenzoic acid 6-formylpyridin-3-ester (19d): From compound 18 (0.50 mg, 2.44 mmol), compound 12d (1.20 g, 2.44 mmol) and EDC (1.16 g, 60.55 mmol) in dry CH2Cl2 (50 mL). This yielded a white solid (0.46 g, 32%); mp: 70-71 °C: 1H-NMR (CDCl3): δH 10.09 (s, 1H, CHO), 8.71 (d, 1H, Py-H, J = 2.6 Hz), 8.71 (d, 1H, Py-H, J = 8.5 Hz), 7.83 (dd, 1H, Ar-H, J = 8.5, 2.2 Hz), 7.79 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 7.64 (d, 1H, Ar-H, J = 2.2 Hz), 6.95 (d, 1H, Ar-H, J = 8.5 Hz), 4.09 (t, 2H, OCH2, J = 6.6 Hz), 4.07 (t, 2H, OCH2, J = 6.6 Hz), 1.96-1.82 (m, 4H, 2 x CH2), 1.49-1.26 (m, 36H, 18 x CH2), 0.87 (t, 6H, 2 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 192.1, 164.0, 154.5, 150.9, 149.9, 148.8, 144.2, 130.2, 124.8, 122.5, 120.0, 114.5, 111.9, 69.4, 69.1, 31.9, 29.6, 29.4, 29.1, 26.0, 22.7, 14.1; MS m/z (ESMS): 618 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2929, 2856, 1731.
3,4,5-Trispentyloxybenzoic acid 6-formylpyridin-3-ester (20a): From compound 18 (0.50 g, 4.07 mmol), compound 13a (1.55 g, 4.07 mmol) and EDC (1.16 g, 6.06 mmol) in dry CH2Cl2 (50 mL). This yielded a brown oil (0.77 g, 40%); 1H-NMR (CDCl3): δH 10.09 (s, 1H, CHO), 8.70 (d, 1H, Py-H, J = 2.6 Hz), 8.08 (d, 1H, Py-H, J = 8.5 Hz), 7.77 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 7.39 (s, 2H, Ar-H), 4.07 (t, 2H, OCH2, J = 6.6 Hz), 4.04 (t, 4H, 2 x OCH2, J = 6.6 Hz) 1.89-1.72 (m, 6H, 3 x CH2), 1.50-1.26 (m, 12H, 6 x CH2), 0.93 (t, 9H, 3 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 192.2, 164.2, 153.3, 151.1, 150.3, 144.4, 144.0, 130.4, 122.8, 122.6, 109.0, 73.8, 69.6, 30.1, 29.1, 28.4, 22.6, 22.5, 14.1; MS m/z (EIMS): 485 ([M]+, 15%), 363 ([M-OPyCHO]+, 100%), 293 ([M-OPyCHO-C5H10]+, 75%), 153 ([M-OPyCHO-(3xC5H10)]+, 30%); IR ν/cm-1 (nujol): 2927, 2854, 1728.
3,4,5-Trisheptyloxybenzoic acid 6-formylpyridin-3-ester (20b): From compound 18 (0.50 g, 4.07 mmol), compound 13b (1.89 g, 4.07 mmol) and EDC (1.16 g, 6.06 mmol) in dry CH2Cl2 (50 mL). This yielded a brown oil (0.88 g, 39%); 1H-NMR (CDCl3): δH 10.09 (s, 1H, CHO), 8.70 (d, 1H, Py-H, J = 2.6 Hz), 8.07 (d, 1H, Py-H, J = 8.5 Hz), 7.77 (dd 1H, Py-H, J = 8.5, 2.6 Hz), 7.39 (s, 2H, Ar-H), 4.06 (t, 2H, OCH2, J = 6.6 Hz), 4.06 (t, 4H, 2 x OCH2, J = 6.6 Hz), 1.88-1.71 (m, 6H, 3 x CH2), 1.50-1.30 (m, 24H, 12 x CH2), 0.88 (t, 9H, 3 x CH3, J = 6.4 Hz); 13C-NMR (CDCl3): δC 192.4, 164.4, 153.5, 151.3, 150.5, 144.5, 144.2, 130.6, 122.9, 122.8, 109.2, 74.0, 69.7, 32.3, 32.2, 29.6, 29.4, 26.4, 23.0, 14.4; MS m/z (ESMS): 592 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2926, 2854, 1728.
3,4,5-Bistrinonylloxybenzoic acid 6-formylpyridin-3-ester (20c): From compound 18 (0.50 g, 4.07 mmol), compound 13c (2.23 g, 4.07 mmol) and EDC (1.16 g, 60.55 mmol) in dry CH2Cl2 (50 mL). This yielded a white solid (0.89 g, 34%); mp: 34-37 °C; 1H-NMR (CDCl3): δH 10.08 (s, 1H, CHO), 8.69 (d, 1H, Py-H, J = 2.6 Hz), 8.07 (d, 1H, Py-H, J = 8.5 Hz), 7.77 (dd, 1H, Py-H, J = 8.5, 2.6 Hz), 7.39 (s, 2H, Ar-H), 4.07 (t, 2H, OCH2, J = 6.4 Hz), 4.04 (t, 4H, 2 x OCH2, J = 6.6 Hz), 1.87-1.71 (m, 6H, 3 x CH3), 1.50-1.26 (m, 36H, 18 x CH2), 0.87 (t, 3H, CH3, J = 6.6 Hz), 0.86 (t, 6H, 2 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 194.7, 166.7, 155.8, 153.6, 152.8, 146.8, 146.5, 132.9, 125.3, 125.1, 111.5, 76.3, 72.0, 34.5, 33.0, 32.3, 32.2, 32.0, 28.7, 25.3, 16.7; MS m/z (ESMS): 676 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2927, 2855, 1730.
3,4,5-Trisdodecyloxybenzoic acid 6-formylpyridin-3-ester (20d): From compound 18 (0.50 g, 4.07 mmol), compound 13d (2.74 g, 2.44 mmol) and EDC (1.16 g, 60.55 mmol) in dry CH2Cl2 (50 mL). This yielded a white solid (0.89 g, 35%); mp: 41-43 °C: 1H-NMR (CDCl3): δH 10.08 (s, 1H, CHO), 8.69 (d, 1H, Py-H, J = 2.2 Hz), 8.07 (d, 1H, Py-H, J = 8.5 Hz), 7.77 (dd, 1H, Py-H, J = 8.5, 2.2 Hz), 7.39 (s, 2H, Ar-H), 4.06 (t, 2H, OCH2, J = 6.6 Hz), 4.04 (t, 4H, 2 x OCH2, J = 6.6 Hz), 1.85-1.71 (m, 6H, 3 x CH2), 1.49-1.25 (m, 54H, 27 x CH2), 0.86 (t, 9H, 3 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 191.3, 163.3, 152.4, 150.2, 149.4, 143.4, 143.1, 129.4, 121.8, 121.6, 108.1, 72.9, 68.6, 31.2, 29.6, 28.8, 28.6, 25.3, 21.9, 13.3; MS m/z (ESMS): 802 ([M+Na]+, 100%), 780 ([M+H]+, 20%); IR ν/cm-1 (nujol): 2927, 2856, 1731.
6-{N-[4-({4-[(E)-[(5-{[3, 4-bis (pentyloxy) phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino] phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(pentyloxy)benzoate (1a): To a solution of compound 19a (202 mg, 0.51 mmol) in anhydrous EtOH (4 mL) under N2 atmosphere, 4,4’-methylene dianiline (50 mg, 0.25 mmol) was added. The reaction mixture was stirred and heated at 50 °C for 24 hrs under a N2 atmosphere. The solid was collected through suction filtration and recrystallised in anhydrous PhMe. The yellow solid was collected through suction filtration (140 mg, 58%); mp: 158-159 °C; Calculated for C59H68N4O8: 73.72% C, 7.13% H, 5.83% N; found: 73.68% C, 7.10% H, 5.90% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.62 (d, 2H, Py-H, J = 2.2 Hz), 8.28 (d, 2H, Py-H, J = 8.5 Hz), 7.83 (dd, 2H, Ar-H, J = 8.5, 1.8 Hz), 7.71 (dd, 2H, Py-H, J = 8.5, 2.2 Hz), 7.65 (d, 2H, Ar-H, J = 1.8 Hz), 7.26 (s, 8H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.5 Hz), 4.09 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.05 (s, 2H, Ph2CH2), 1.90-1.81 (m, 8H, 4 x CH2), 1.55-1.34 (m, 16H, 8 x CH2), 0.94 (t, 6H, 2 x CH3, J = 7.0 Hz), 0.93 (t, 6H, 2 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC, 166.8, 161.5, 156.8, 154.4, 151.1, 151.4, 145.9, 142.3, 132.5, 132.3, 132.1, 127.2, 124.9, 123.9, 123.0, 117.1, 114.5, 71.9, 71.6, 43.5, 31.3, 31.2, 30.7, 24.5, 16.5; MS m/z (ESMS): 962 ([M+H]+, 100%); IR ν/cm-1 (nujol): 2927, 2854, 1729, 1598.
Following the same general procedure described for the preparation of 1a, the following compounds were prepared:
6-{N-[4-({4-[(E)-[(5-{[3,4-bis(heptyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]-phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(heptyloxy)benzoate (1b): From compound 19b (230 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (4 mL). This yielded a pale yellow solid (186 mg, 69%); mp: 136-137 °C; Calculated for C67H84N4O8: 74.97% C, 7.89% H, 5.22% N; found: 74.88% C, 7.70% H, 5.18% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.62 (d, 2H, Py-H, J = 2.6 Hz), 8.28 (d, 2H, Py-H, J = 8.5 Hz), 7.83 (dd, 2H, Ar-H, J = 8.5, 2.1 Hz), 7.71 (dd, 2H, Py-H, J = 8.5, 2.6 Hz), 7.65 (d, 2H, Ar-H, J = 2.1 Hz), 7.26 (s, 8H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.5 Hz), 4.09 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.04 (s, 2H, Ph2CH2), 1.91-1.80 (m, 8H, 4 x CH2), 1.55-1.34 (m, 32H, 16 x CH2), 0.89 (t, 6H, 2 x CH3, J = 6.99 Hz), 0.88 (t, 6H, 2 x CH3, J = 6.99 Hz); 13C-NMR (CDCl3): δC 165.5, 160.1, 155.5, 153.1, 150.1, 150.0, 144.5, 140.9, 131.2, 130.9, 130.7, 125.8, 123.5, 122.5, 121.6, 115.8, 113.1, 70.5, 70.3, 42.2, 32.9, 30.3, 30.2, 27.1, 23.7, 15.2; MS m/z (ESMS): 1096 ([M+Na]+, 100%); IR ν/cm-1 (nujol): 2926, 2855, 1729, 1598.
6-{N-[4-({4-[(E)-[(5-{[3,4-bis(nonyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(nonyloxy)benzoate (1c): From compound 19c (258 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (5 mL). This yielded a pale yellow solid (155 mg, 52%); mp: 129 °C; Calculated for C75H100N4O8: 75.98% C, 8.50% H, 4.73% N; found: 75.80% C, 8.40% H, 4.63% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.62 (d, 2H, Py-H, J = 2.6 Hz), 8.29 (d, 2H, Py-H, J = 8.8 Hz), 7.83 (dd, 2H, Ar-H, J = 8.5, 1.8 Hz), 7.71 (dd, 2H, Py-H, J = 8.8, 2.6 Hz), 7.65 (d, 2H, Ar-H, J = 1.8 Hz), 7.26 (s, 8H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.5 Hz), 4.09 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.05 (s, 2H, CH2), 1.89-1.80 (m, 8H, 4 x CH2), 1.49-1.29 (m, 48H, 24 x CH2), 0.88 (t, 6H, 2 x CH3, J = 7.0 Hz), 0.87 (t, 6H, 2 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 166.0, 160.7, 156.0, 155.2, 153.6, 150.6, 150.5, 145.0, 141.4, 131.7, 131.4, 126.4, 124.0, 123.1, 122.1, 116.3, 113.6, 71.1, 70.8, 42.8, 33.5, 31.2, 31.0, 30.9, 30.8, 30.7, 27.6, 24.3, 15.7; MS m/z (ESMS): 1207 ([M+Na]+); IR ν/cm-1 (nujol): 2926, 2854, 1727, 1598.
6-{N-[4-({4-[(E)-[(5-{[3,4-bis(dodecyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(dodecyloxy)benzoate (1d): From compound 19d (301 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (6 mL). This yielded a pale yellow solid (190 mg, 56%); mp: 126 °C; Calculated for C87H124N4O8: 77.18% C, 9.23% H, 4.14% N; found: 77.23% C, 9.31% H, 4.17% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.62 (d, 2H, Py-H, J = 2.6 Hz,), 8.29 (d, 2H, Py-H, J = 8.8 Hz), 7.83 (dd, 2H, Ar-H, J = 8.5, 1.8 Hz), 7.71 (dd, 2H, Py-H, J = 8.8, 2.6 Hz), 7.65 (d, 2H, Ar-H, J = 1.8 Hz), 7.26 (s, 8H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.5 Hz), 4.09 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.05 (s, 2H, Ph2CH2), 1.89-1.80 (m, 8H, 4 x CH2), 1.49-1.29 (m, 72H, 36 x CH2), 0.88 (t, 6H, 2 x CH3, J = 7.0 Hz), 0.87 (t, 6H, 2 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 165.5, 160.2, 155.6, 154.7, 153.1, 150.2, 150.1, 144.2, 141.0, 131.3, 131.1, 126.4, 123.6, 122.7, 121.7, 115.7, 110.0, 71.1, 70.8, 42.8, 33.5, 31.2, 31.0, 30.9, 30.8, 30.7, 27.6, 24.3, 15.7; MS m/z (ESMS): 1354 ([M]+, 100%); IR ν/cm-1 (nujol): 2927, 2854, 1727, 1597.
6-{N-[4-({4-[(E)-[(5-{[3,4,5-tris(pentyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(pentyloxy)benzoate (2a): From compound 20a (245 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (4 mL). This yielded a yellow solid (151 mg, 53%). mp: 67 °C; Calculated for C69H88N4O10: 73.12% C, 7.83% H, 4.94% N; found: 73.16% C, 7.87% H, 4.89% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.61 (d, 2H, Py-H, J = 2.6 Hz), 8.29 (d, 2H, Py-H, J = 8.8 Hz), 7.71 (dd, 2H, Py-H, J = 8.8, 2.6 Hz), 7.41 (s, 4H, Ar-H), 7.26 (s, 8H, Ar-H), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.05 (t, 8H, 4 x OCH2, J = 6.6 Hz), 4.05 (s, 2H, Ph2CH2), 1.89-1.72 (m, 12H, 6 x CH2), 1.53-1.31 (m, 24H, 12 x CH2), 0.93 (t, 18H, 6 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 164.7, 159.3, 153.4, 152.4, 149.3, 149.1, 143.9, 143.7, 140.2, 130.4, 130.2, 123.2, 122.7, 121.8, 109.1, 74.0, 69.7, 41.5, 31.3, 29.3, 28.6, 22.9, 22.8, 14.4; MS m/z (ESMS): 1133 ([M]+); IR ν/cm-1 (nujol): 2930, 2857, 1743, 1587.
6-{N-[4-({4-[(E)-[(5-{[3,4,5-tris(heptyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(heptyloxy)benzoate (2b): From compound 20b (287 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (4 mL). This yielded a yellow solid (197 mg, 60%); mp: 54-56 °C; Calculated for C81H112N4O10: 74.73% C, 8.67% H, 4.30% N; found: 74.78% C, 8.70% H, 4.32% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.61 (d, 2H, Py-H, J = 2.6 Hz), 8.29 (d, 2H, Py-H, J = 8.5 Hz), 7.70 (dd, 2H, Py-H, J = 8.5, 2.6 Hz), 7.40 (s, 4H, Ar-H), 7.26 (s, 8H, Ar-H), 4.07 (t, 4H, 2 x OCH2, J = 6.4 Hz), 4.05 (t, 8H, 4 x OCH2, J = 6.3 Hz), 4.05 (s, 2H, Ph2CH2), 1.88-1.71 (m, 12H, 6 x CH2), 1.50-1.27 (m, 48H, 12 x CH2), 0.88 (t, 6H, 2 x CH3, J = 6.6 Hz), 0.87 (t, 12H, 4 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 163.1, 157.7, 151.9, 150.1, 147.7, 147.6, 142.4, 142.2, 138.6, 128.8, 128.6, 121.6, 121.2, 120.2, 107.6, 72.4, 68.2, 39.9, 30.7, 30.6, 29.1, 28.1, 28.0, 27.8, 24.8, 21.4, 12.9; MS m/z (ESMS): 1302 ([M+H]+); IR ν/cm-1 (nujol): 2930, 2857, 1744s, 1590.
6-{N-[4-({4-[(E)-[(5-{[3,4,5-tris(nonyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino] phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(nonyloxy)benzoate (2c): From compound 20c (330 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (5 mL). This yielded a yellow solid (241 mg, 65%); mp: 60-62 °C; Calculated for C93H136N4O10: 75.98% C, 9.32% H, 3.81% N; found: 75.90% C, 9.27% H, 3.85% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.61 (d, 2H, Py-H, J = 2.6 Hz), 8.29 (d, 2H, Py-H, J = 8.5 Hz), 7.70 (dd, 2H, Py-H, J = 8.5, 2.6 Hz), 7.40 (s, 4H, Ar-H), 7.26 (s, 8H, Ar-H), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.05 (t, 8H, 4 x OCH2, J = 6.6 Hz), 4.05 (s, 2H, CH2), 1.88-1.71 (m, 12H, 6 x CH2), 1.50-1.21 (m, 72H, 36 x CH2), 0.88 (t, 6H, 2 x CH3, J = 7.0 Hz), 0.87 (t, 12H, 4 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 164.4, 159.0, 153.1, 152.1, 149.0, 148.8, 143.6, 143.4, 139.9, 130.1, 129.8, 122.8, 122.4, 121.4, 108.8, 73.7, 69.4, 41.4, 31.9, 30.4, 29.7, 29.6, 29.4, 29.3, 26.1, 22.7, 14.1; MS m/z (ESMS): 1471 ([M+H]+); IR ν/cm-1 (nujol): 2930, 2857, 1744, 1589.
6-{N-[4-({4-[(E)-[(5-{[3,4,5-tris(dodecyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino] phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(dodecyloxy)benzoate (2d): From compound 20d (398 mg, 0.51 mmol), 4,4’-methylenedianiline (50 mg, 0.25 mmol) and anhydrous EtOH (6 mL). This yielded a yellow solid (291 mg, 67%); mp 51 °C; Calculated for C111H172N4O10: 77.40% C, 10.06% H, 3.25% N; found: 77.33% C, 10.01% H, 3.27% N; 1H-NMR (CDCl3): δH 8.63 (s, 2H, HC=N), 8.61 (d, 2H, Py-H, J = 2.6 Hz), 8.29 (d, 2H, Py-H, J = 8.8 Hz), 7.70 (dd, 2H, Py-H, J = 8.8, 2.6 Hz), 7.40 (s, 4H, Ar-H), 7.27 (s, 8H, Ar-H), 4.06 (t, 4H, 2 x OCH2, J = 6.4 Hz), 4.04 (t, 8H, 4 x OCH2, J = 6.3 Hz), 4.04 (s, 2H, Ph2CH2), 1.89-1.71 (m, 12H, 6 x CH2), 1.48-1.25 (m, 108H, 54 x CH2), 0.87 (t, 18H, 9 x CH2, J = 6.6 Hz); 13C-NMR (CDCl3): δC 162.5, 157.1, 151.2, 150.2, 147.1, 146.9, 141.7, 141.5, 138.0, 128.2, 128.0, 120.9, 120.5, 119.6, 106.9, 71.8, 67.5, 30.1, 28.5, 27.8, 27.5, 24.2, 20.8, 12.3; MS m/z (ESMS): 1744 ([M+Na]+); IR ν/cm-1 (nujol): 2930, 2857, 1729, 1598.
6-{N-[3-({3-[(E)-[(5-{[3,4-bis(pentyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(pentyloxy)benzoate (3a): From compound 19a (60 mg, 0.16 mmol), 3,3’-methylenedianiline (10 mg, 0.08 mmol) and anhydrous EtOH (2 mL). This yielded a brown oil (60 mg, 85%). 1H-NMR (CDCl3): δH 8.61 (s, 2H, HC=N), 8.60 (d, 2H, Py-H, J = 2.3 Hz), 8.27 (d, 2H, Py-H, J = 8.7 Hz), 7.82 (dd, 2H, Ar-H, J = 8.5, 2.1 Hz), 7.71 (dd, 2H, Py-H, J = 8.7, 2.3 Hz), 7.65 (d, 2H, Ar-H, J = 2.1 Hz), 7.35 (t, 2H, Ar-H, J = 8.5 Hz), 7.15 (m, 6H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.5 Hz), 4.07 (m, 10H, Ph2CH2, OCH2), 1.88-1.68 (m, 8H, 4 x CH2), 1.52-1.31 (m, 16H, 8 x CH2), 0.93 (t, 12H, 4 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 160.0, 152.2, 151.5, 149.3, 143.8, 142.4, 130.6, 130.5, 128.8, 127.9, 125.1, 122.8, 122.4, 120.9, 119.2, 115.0, 112.3, 42.2, 29.2, 29.1, 28.6, 22.9, 14.4; MS m/z (+ve FAB): 961 ([M]+, 80%).
6-{N-[3-({3-[(E)-[(5-{[3,4-bis(heptyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(heptyloxy)benzoate (3b): From compound 19b (50 mg, 0.11 mmol), 3,3’-methylenedianiline (10 mg, 0.06 mmol) and anhydrous EtOH (2 mL). This yielded a brown oil (40 mg, 76%). 1H-NMR (CDCl3): δH 8.61 (s, 2H, HC=N), 8.61 (d, 2H, Py-H, J = 2.5 Hz), 8.27 (d, 2H, Py-H, J = 8.6 Hz), 7.83 (dd, J = 2H, Ar-H, 8.3, 2.0 Hz), 7.71 (dd, J = 2H, Py-H, 8.6, 2.3 Hz), 7.65 (d, 2H, Ar-H, J = 2.0 Hz), 7.35 (t, 2H, Ar-H, 7.5 Hz), 7.16-7.13 (m, 6H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.3 Hz), 4.09-4.05 (m, 10H, Ph2CH2, 4 x OCH2), 1.88-1.71 (m, 8H, 4 x CH2), 1.52-1.30 (m, 32H, 16 x CH2), 0.93 (m, 12H, 4 x CH3); 13C-NMR (CDCl3): δC 160.0, 143.8, 130.5, 129.8, 127.8, 125.1, 122.8, 122.4, 119.1, 115.0, 112.4, 32.2, 29.6, 29.4, 26.4, 26.3, 23.0, 14.5; MS m/z (+ve FAB): 1073 ([M]+); IR ν/cm-1 (nujol): 2911, 2851, 1729, 1598.
6-{N-[3-({3-[(E)-[(5-{[3,4-bis(dodecyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4-bis(dodecyloxy)benzoate (3c): From compound 19d (70 mg, 0.12 mmol), 3,3’-methylenedianiline (10 mg, 0.06 mmol) and anhydrous EtOH (2 mL). This yielded a white solid (70 mg, 83%); Calculated for C87H124N4O8·H2O: 76.2% C, 9.1% H, 4.1% N; found: 75.9% C, 9.0% H, 3.9% N; 1H-NMR (CDCl3): δH 8.60 (s, 2H, CHN), 8.61 (d, 2H, Py-H, J = 2.0 Hz), 8.27 (d, 2H, Py-H, J = 8.6 Hz), 7.83 (dd, J = 2H, Ar-H, 8.6, 2.0 Hz), 7.71 (d, 2H, Py-H, J = 8.3, 2.0 Hz), 7.65 (d, 2H, Ar-H, J = 2.0 Hz), 7.65 (s, 2H, Ar-H), 7.34 (t, 2H, Ar-H, J = 7.6 Hz), 7.16-7.13 (m, 6H, Ar-H), 6.94 (d, 2H, Ar-H, J = 8.6 Hz), 4.09 (m, 10H, Ph2CH2, 4 x OCH2), 1.88-1.71 (m, 8H, 4 x CH2), 1.48-1.27 (m, 72H, 36 x CH2), 0.93 (t, 12H, 4 x CH3, J = 7.0 Hz); MS m/z (+ve FAB): 1354 ([M]+); IR ν/cm-1 (nujol): 2933, 2840, 1734, 1685, 1593, 1516.
6-{N-[3-({3-[(E)-[(5-{[3,4,5-tris(pentyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(pentyloxy)benzoate (4a): From compound 20a (79 mg, 0.16 mmol), 3,3’-methylenedianiline (16 mg, 0.08 mmol) and anhydrous EtOH (2 mL). This yielded a brown oil (52 mg, 56%); Calculated for C69H88N4O10 73.12% C, 7.83% H, 4.94% N; found: 73.16% C, 7.87% H, 4.89% N; 1H-NMR (CDCl3): δH 8.60 (s, 2H, HC=N), 8.60 (d, 2H, Py-H, J = 2.6 Hz), 8.28 (d, 2H, Py-H, J = 8.6 Hz), 7.70 (dd, 2H, Py-H, J = 8.6, 2.6 Hz), 7.40 (s, 4H, Ar-H), 7.35 (t, 2H, Ar-H, J = 7.7 Hz), 7.16-7.13 (m, 6H, Ar-H), 4.07 (s, 2H, Ph2CH2), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.04 (t, 8H, 4 x OCH2, J = 6.6 Hz), 1.88-1.68 (m, 12H, 6 x CH2), 1.52-1.31 (m, 24H, 12 x CH2), 0.92 (t, 18H, 6 x CH3, J = 6.99 Hz); 13C-NMR (CDCl3): δC 165.1, 161.3, 154.9, 153.7, 152.9, 150.6, 145.3, 145.1, 143.8, 131.8, 131.2, 129.2, 124.2, 123.8, 123.7, 120.5, 110.5, 75.4, 71.1, 43.6, 31.8, 30.7, 30.0, 24.3, 24.2, 15.8; MS m/z (ESMS): 1133 ([M]+, 100%); IR ν/cm-1 (nujol): 2927, 2854, 1736, 1587.
6-{N-[3-({3-[(E)-[(5-{[3,4,5-tris(heptyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(heptyloxy)benzoate (4b): From compound 20b (287 mg, 0.51 mmol), 3,3’-methylene dianiline (50 mg, 0.25 mmol) and anhydrous EtOH (4 mL). This yielded a brown oil (195 mg, 59%); Calculated for C81H112N4O10 74.73% C, 8.67% H, 4.30% N; found: 74.78% C, 8.70% H, 4.32% N; 1H-NMR (CDCl3): δH 8.60 (s, 2H, HC=N), 8.60 (d, 2H, Py-H, J = 2.6 Hz), 8.28 (d, 2H, Py-H, J = 8.6 Hz), 7.70 (dd, 2H, Py-H, J = 8.6, 2.6 Hz), 7.40 (s, 4H, Ar-H), 7.35 (t, 2H, Ar-H, J = 7.7 Hz), 7.16-7.13 (m, 6H, Ar-H), 4.07 (s, 2H, Ph2CH2), 4.07 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.04 (t, 8H, 4 x OCH2, J = 6.6 Hz), 1.88-1.71 (m, 12H, 6 x CH2), 1.52-1.30 (m, 48H, 24 x CH2), 0.88 (t, 18H, 6 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 164.8, 159.9, 153.5, 152.4, 151.5, 149.2, 144.0, 143.8, 142.4, 130.5, 129.8, 127.9, 123.2, 122.8, 122.4, 119.1, 109.2, 74.0, 69.8, 42.2, 32.3, 32.2, 30.8, 29.7, 29.6, 29.4, 26.4, 23.0, 14.5; MS m/z (ESMS): 1324 ([M+Na]+), 1303; IR ν/cm-1 (nujol): 2927, 2854, 1739, 1586.
6-{N-[3-({3-[(E)-[(5-{[3,4,5-tris(nonyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(nonyloxy)benzoate (4c): From compound 20c (160 mg, 0.25 mmol), 3,3’-methylenedianiline (24 mg, 0.12 mmol) and anhydrous EtOH (4 mL). This yielded a white solid (117 mg, 65%); mp: 51 °C; Calculated for C93H136N4O10 75.98% C, 9.32% H, 3.81% N; found: 75.90% C, 9.37% H, 3.85% N; 1H-NMR (CDCl3): δH 8.60 (s, 2H, CHN), 8.60 (d, 2H, Py-H, J = 2.2 Hz), 8.28 (d, 2H, Py-H, J = 8.6 Hz), 7.69 (dd, 2H, Py-H, J = 8.6, 2.2 Hz), 7.40 (s, 4H, Ar-H), 7.35 (t, 2H, Ar-H, J = 7.7 Hz), 7.16-7.13 (m, 6H, Ar-H), 4.06 (s, 2H, Ph2CH2), 4.06 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.04 (t, 8H, 4 x OCH2, J = 6.6 Hz), 1.88-1.71 (m, 12H, 6 x CH2), 1.48-1.27 (m, 72H, 36 x CH2), 0.88 (t, 6H, 2 x CH3, J = 7.0 Hz), 0.87 (t, 12H, 4 x CH3, J = 7.0 Hz); 13C-NMR (CDCl3): δC 163.6, 158.7, 152.3, 151.2, 150.3, 148.0, 142.8, 142.6, 141.3, 129.3, 128.6, 126.7, 122.0, 121.6, 121.2, 118.0, 108.0, 72.8, 68.6, 41.0, 31.1, 29.6, 28.9, 28.8, 28.6, 28.5, 25.3, 21.9, 13.3; MS m/z (ESMS): 1471 ([M+H]+); IR ν/cm-1 (nujol): 2927, 2854, 1738, 1587.
6-{N-[3-({3-[(E)-[(5-{[3,4,5-tris(dodecyloxy)phenyl]carbonyloxy}pyridin-2-yl)methylidene]amino]- phenyl}methyl)phenyl]carboximidoyl}pyridin-3-yl 3,4,5-tris(dodecyloxy)benzoate (4d): From compound 20d (150 mg, 0.19 mmol), 3,3’-methylenedianiline (19 mg, 0.10 mmol) and anhydrous EtOH (4 mL). This yielded a white solid (110 mg, 67 %); mp: 55 °C; Calculated for C111H172N4O10 77.40% C, 10.06% H, 3.25% N; found: 77.33% C, 10.01% H, 3.27% N; 1H-NMR (CDCl3): δH 8.60 (s, 2H, CHN), 8.60 (d, 2H, Py-H, J = 2.6 Hz), 8.29 (d, 2H, Py-H, J = 8.5 Hz), 7.69 (dd, 2H, Py-H, J = 8.5, 2.6 Hz), 7.40 (s, 4H, Ar-H), 7.35 (t, 2H, Ar-H, J = 7.7 Hz), 7.16-7.14 (m, 6H, Ar-H), 4.06 (s, 2H, Ph2CH2), 4.06 (t, 4H, 2 x OCH2, J = 6.6 Hz), 4.07 (t, 8H, 4 x OCH2, J = 6.6 Hz), 1.87-1.71 (m, 12H, 6 x CH2), 1.48-1.25 (m, 108H, 54 x CH2), 0.87 (t, 18H, 6 x CH3, J = 6.6 Hz); 13C-NMR (CDCl3): δC 164.3, 159.4, 153.0, 151.9, 151.0, 148.7, 143.5, 143.3, 142.0, 130.0, 129.3, 127.4, 122.7, 122.3, 122.0, 118.7, 108.7, 73.6, 69.3, 41.7, 31.9, 30.3, 29.6, 29.3, 26.0, 22.6, 14.0; MS m/z (ESMS): 1744 ([M+Na)]+); IR ν/cm-1 (nujol): 2927, 2854, 1735, 1585.
[Cu2(3a)2][PF6]2: To a solution of compound 3a (25 mg, 0.026 mmol) in propan-2-ol (5 mL) under a nitrogen atmosphere, [Cu(MeCN)4][PF6] (10 mg, 0.026 mmol) was added to give a dark red solution, which was heated under reflux overnight and then cooled to room temperature. A dark red solid precipitated from the solution on standing and was collected by filtration and washed with diethyl ether (41 mg, 67 %); Calculated for Cu2C118H136N8O16P2F12·H2O: 59.7% C, 5.9% H, 4.7% N; found: 59.4% C, 5.9%, 5.0% N; 1H-NMR (CD2Cl2): δH 9.04 (br, 4H, HC=N), 8.46 (br, 4H, Py-H), 8.25 (br, 4H, Py-H), 8.08 (d, 4H, Ar-H, J = 8.0 Hz), 7.79 (d, 4H, Py-H, J = 8.6 Hz), 7.60 (s, 4H, Ar-H), 7.27 (t br, 4 H, Ar-H, J = 8.6 Hz), 7.00 (s br, 12H, Ar-H), 6.96 (d, 4H, Ar-H, J = 8.5 Hz), 4.09 (t, 8H, 4 x OCH2, J = 6.5 Hz), 4.04 (t, 8H, 4 x OCH2, J = 6.5 Hz), 3.69 (br s, 4H, Ph2CH2), 1.88-1.68 (m, 16H, 8 x CH2), 1.52-1.31 (m, 32H, 16 x CH2), 0.96 (q, 24H, 12 x CH3, J = 7.0 Hz); m/z (+ve FABMS): 2194 [(Cu2(3a)2)(PF6)]+; MS m/z (ESMS): 1024 [(Cu2(3a)2)]2+; IR ν/cm-1 (solid): 2953, 2926, 2867, 1727, 1594, 1556, 1521; UV/Vis (CH2Cl2) λmax (ε, L mol-1 cm-1): 495 (9,500), 312 (83,600) nm.
[Cu2(3b)2][PF6]2: The same procedure as described for the preparation of compound [Cu2(3a)2][PF6]2 was followed, using compound 3b (12 mg, 0.011 mmol) and [Cu(MeCN)4][PF6] (4 mg, 0.011 mmol). This yielded a dark red solid (20 mg, 70 %); 1H-NMR (CD2Cl2): δH 9.03 (br s, 4H, HC=N), 8.53 (br s, 4H, Py-H), 8.23 (d, 4H, Py-H, J = 8.3 Hz), 8.08 (d, J = 4H, Ar-H, 8.3 Hz), 7.77 (d, 4H, Py-H, J = 8.5 Hz), 7.59 (s, 4H, Ar-H), 7.25 (br t, 4H, J = 7.5 Hz), 7.00 (s br, 12H, Ar-H), 6.98 (br s, 4H, Ar-H), 4.08 (t, 8H, 4 x OCH2, J = 6.5 Hz), 4.02 (t, 8H, 4 x OCH2, J = 6.5 Hz), 3.66 (br s, 4H, Ph2CH2), 1.88-1.71 (m, 16H, 4 x CH2), 1.52-1.30 (m, 64H, 32 x CH2), 0.90 (m, 24H, 12 x CH3); m/z (+ve FAB MS): 2419 [Cu2(3b)2(PF6)]+, 1137 [Cu(3b)]+; MS m/z (ESI MS): [Cu2(3b)2]2+ ; IR ν/cm-1 (solid): 2922, 2852, 1727, 1595, 1556, 1513; UV/Vis (CH2Cl2) λmax (ε, L mol-1 cm-1): 480 (11,400), 294 (94,600) nm.
[Cu2(3c)2][PF6]2: The same procedure as described for the preparation of compound [Cu2(3a)2][PF6]2 was followed, using compound 3c (24 mg, 0.018 mmol) and [Cu(MeCN)4][PF6] (7 mg, 0.018 mmol). This yielded a dark red solid (56 mg, 65 %); The 1H-NMR spectrum of the red material was very broad and the sample did not ionise well in ESI-MS; IR ν/cm-1 (solid): 2919, 2853, 1732, 1662, 1600, 1508; UV/Vis (CH2Cl2) λmax (ε, L mol-1 cm-1): 482 (7 400), 298 (94 700) nm.