4.1. Genetic Diversity
Prior to this study, little information was found in public databases for genetic studies of Mexican populations of
P. menziesii using molecular markers. Most of the reported studies focused mainly on morphological [
47] and phenological characteristics [
48], mating patterns [
15], and seed viability and dispersion [
12,
14]. Cruz-Nicolás et al. [
49] analyzed Mexican populations from the northern and central regions of Mexico. Using isozyme markers, Cruz-Nicolás et al. determined levels of genetic diversity of
Ht = 0.077 (
He = 0.051 for the central region). Although this was the first analysis of the levels of genetic diversity in Mexican populations, a direct comparison between isozymes and microsatellites would be inappropriate without taking into account their differing capacity to detect genetic variation [
27]. However, their results were the first to report low levels of genetic diversity in these populations and the presence of genetic differentiation (
FST = 0.298).
Previously, Amarasinghe and Carlson [
25] and Slavov et al. [
26] developed microsatellite markers and estimated genetic diversity parameters in
P. menziesii populations from Canada and the United States, respectively. From these primers, 12 SSR markers were used in the present study to determine the genetic diversity parameters in populations of
P. menziesii from the central region of Mexico. Polymorphism levels of the selected panel were high (
Table 4), with a mean value of 77.86% across the 12 populations of this study. The mean number of alleles (
Na = 6.08) and the heterozygosity (
Ht = 0.417) detected in this study were lower than those reported in other studies in
P. menziesii using microsatellites (
Na = 8,
He =0.673 [
25],
Na = 23,
He = 0.855 [
26] and
Na = 24.03,
He = 0.936 [
27]).
The mean number of alleles observed and expected heterozygosity parameters were compared with other studies that used a similar set of microsatellite markers. Molecular studies on pollination dynamics of
P. menziesii orchards [
50,
51] at the locus level (PmOSU_3B9, PmOSU_2D4, PmOSU_2G12, and PmOSU_3G9) displayed higher values (
Na = 20.2,
Ho = 0.651 [
50],
Na = 23,
Ho = 0.712,
He = 0.916 [
51]) than those detected in the Mexican populations with the same markers (
Na = 6.3,
Ho = 0.116,
He = 0.421). These results are in agreement with previous studies suggesting that Mexican populations from the central region display lower levels of genetic diversity than populations in other regions of North America [
25,
26,
27,
50,
51]. Populations of
P. menziesii in the central region of Mexico are fragmented, and present a discontinuous distribution in the form of small patches or stands that are isolated from one another [
1,
19]. Paleoclimatic studies suggest that
P. menziesii in Mexico emerged during the era of glaciation. During this period, the
P. menziesii populations of northern Mexico migrated southward, where they found refuge in the mountain systems. In the interglacial events, the populations expanded northward and to the high parts of the mountains, where today’s populations formed with a certain level of isolation. These climate changes could have caused bottlenecks that reduced genetic variation and led to interpopulation differentiation, while the populations distributed in the United States and Canada were secondary points of convergence in the interglacial periods, giving rise to greater genetic diversity [
19,
52].
In the case of small populations, reproductive isolation can lead to genetic drift [
53], increasing genetic differentiation among populations, homozygosity, and inbreeding [
54]. Low values of genetic diversity have also been reported in small populations of other species with restricted distribution in Mexico, such as
Picea mexicana Martínez (
Ht = 0.125) [
55], and in different species of
Abies that have isolated distribution, such as
A. flinckii Rushforth (
He = 0.064–0.158),
A. guatemalensis Rehder (
He = 0.044–0.094),
A. hickeli Flous & Gaussen (
He = 0.073–0.135) and
A. religiosa (
He = 0.008–0.235) [
52].
Generally, levels of genetic diversity are closely related to the degree of disturbance of the populations [
56,
57]. Values of genetic diversity obtained for populations of
P. menziesii for the central region are lower than those reported for populations in Canada and the United States. However, for a better estimate of the genetic diversity of the species in the country, it would be necessary to analyze populations from the northern region, since both regions have been found to be genetically different [
47,
48,
49].
4.2. Genetic Structure
At the locus level, we detected deficiencies of heterozygotes, as indicated by the values of Wright fixation indexes
Fis = 0.234 and
Fit = 0.452, in all the analyzed populations (
Table 3). High levels of homozygosity are characteristic of genetic drift acting on small populations as differentiation continues [
53]. According to Wright [
43], the genetic differentiation among populations is high when
Fst > 0.25. The results of the present study indicate that genetic variation exists among the populations (
Fst = 0.285). This result is counter to other studies, wherein the authors report no genetic differentiation [
27,
58]. The value of the coefficient of genetic differentiation,
θ = 0.27, analogous to the coefficient
Fst, was high. Populations with high levels of heterozygosity detected with microsatellite loci can have very low levels of differentiation [
59], as in the case of studies reported for other regions in North America, while in our study, the populations analyzed had a deficiency of heterozygotes and high values of differentiation (
Table 3).
Populations of
P. menziesii from the central region of Mexico have been characterized as having a high production of empty seeds, while the viable seeds are smaller in size with respect to populations from the northern region [
60]. Moreover, when evaluating germination, higher rates of germination and survival are observed in seeds from the northern region [
12]. These characteristic effects of inbreeding (morphological and genetic) observed in the central populations suggest that the process of genetic drift is associated with a high rate of selfing (deficiency of homozygotes,
Table 3).
Gene flow in the analyzed populations was limited, as indicated by the number of migrants (
Nm) per generation inferred from the data of the 12 loci analyzed (
Nm = 0.647,
Table 3). In populations of the state of Washington (USA) and British Columbia (Canada), a
Nm= 5.00 has been reported for this species [
58]. For the Pinaceae family, to which
P.
menziesii belongs, values of up to 17.2 migrants of the genus
Pinus [
61] have been reported. This limited gene flow is in agreement with the high levels of endogamy and genetic structure identified in these populations. It is possible that the reduced gene flow in the analyzed population is a consequence of population fragmentation and size, and of local genetic differentiation [
8].
The higher percentage of variation determined with the AMOVA was within the populations (73.05%). At a lower proportion (26.95%) was the variation detected is between populations. This could be due to the biology of the species. In general, long-living populations, panmictic, pollinated by wind, and of late succession have higher levels of variation within populations [
62,
63]. Additionally, metapopulation structure also has an impact in genetic diversity values. Metapopulation of species is caused by land fragmentation, creating spatially separated populations, which in some cases can interact. This fragmentation could favor genetic drift, hampering gene flow. As a result, there is a reduction of genetic diversity, especially in cross-pollinated species [
64].
The coefficient of genetic structure for the species, derived from the AMOVA, was
Φst = 0.278 (
p < 0.05), which is similar to the characteristic for long-living perennial plants
Φst = 0.25 [
62]. This value suggests the existence of genetic structure, as a consequence of restricted gene flow and evolution, regardless of the populations within the identified genetic groups.
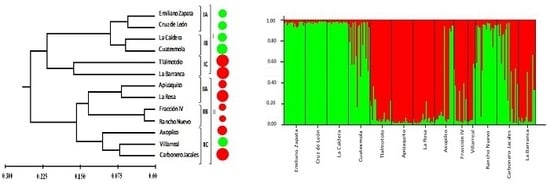
The PCoA, dendrogram, and Bayesian analysis of assignation of individuals (STRUCTURE) were consistent in identifying two genetic groups. However, the results regarding the genetic relatedness of the populations were inconsistent with the expectation that populations geographically closer would be more similar than those that are more distant (
Figure 1 and
Figure 3). These results are in agreement with the performed Mantel tests, where we found a positive correlation between geographic distance and genetic distance (
r = 0.240,
p = 0.102), although this correlation was not significant—a possible reason might be the small sampling range. These results suggest that, although geographic distances do not seem to play a significant role in genetic differentiation, there may exist barriers due to characteristics of the landscape that impede or reduce gene flow among the 12 populations studied.
Knowledge of genetic diversity values and population genetic differentiation is essential to identifying current threats and elucidating mechanisms to protect endangered species [
65,
66]. The results of this study show that the central populations of
P. menziessi analyzed still harbor significant levels of genetic diversity, despite the fragmentation status, illegal exploitation, overgrazing, inappropriate cone collecting, pest attack, and climate change [
67]. Given these precedents, it is imperative in situ conservation strategies be set up for natural populations with high genetic diversity and private alleles, particularly for populations Carbonero Jacales (
He = 0.383,
NP = 5), La Barranca (
He = 0.371,
NP = 4), Tlalmotolo (
He = 0.365,
NP = 1), La Rosa (
He = 0.364,
NP = 3), and Cuatemoxla (
He = 0.320,
NP = 2). In situ conservation must be complemented with ex situ conservation strategies. There are ongoing efforts by national institutions working on the recollection of cones and seeds from different populations of
P. menziessi in the country. Collection of vegetative samples should also be considered for in vitro conservation, particularly from those isolated populations and/or those with ancient trees. Populations from La Barranca, Carbonero Jacales, and Apizaquito are isolated from the main cluster of populations (
Figure 1), and are at high risk of genetic erosion due to their isolated status. The PCoA distribution also shows these populations are further away from the main clusters (
Figure 2). The results of the AMOVA indicate that genetic variation exists mainly within populations. Therefore, conservation efforts should focus on many individuals within populations as possible, to maintain this variation.