Preliminary Evidence for Domestication Effects on the Genetic Diversity of Guazuma crinita in the Peruvian Amazon
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. DNA Extraction
2.3. AFLP Amplification
2.4. Data Analysis
3. Results
3.1. AFLP Fingerprint
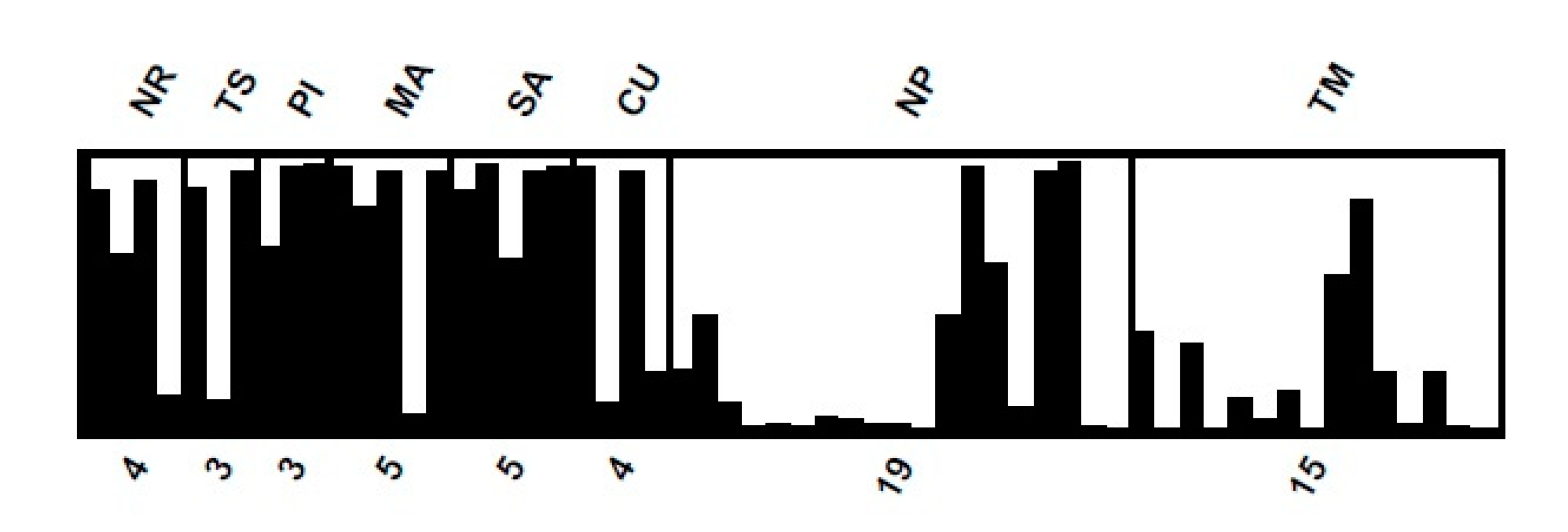
3.2. Genetic Diversity and Population Structure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Neill, G.; Dawson, I.; Sotelo-Montes, C.; Guarino, L.; Guariguata, M.; Current, D.; Weber, J.C. Strategies for genetic conservation of trees in the Peruvian Amazon. Biodivers Conserv. 2001, 10, 837–850. [Google Scholar] [CrossRef]
- Schnabel, A.; Hamrick, J.L. Organization of genetic diversity within and among populations of Gleditsia triacanthos (Leguminosae). Am. J. Bot. 1990, 77, 1060–1069. [Google Scholar] [CrossRef]
- Mertz, O.; Wadley, R.L.; Nielsen, U.; Bruun, B.T.; Colfer, C.J.P.; de Neergaard, A.; Jepsen, M.R.; Martinussen, T.; Zhao, Q.; Noweg, G.T.; et al. A fresh look at shifting cultivation: Fallow length and uncertain indicator of productivity. Agric. Syst. 2008, 96, 75–84. [Google Scholar] [CrossRef]
- Weber, J.C.; Sotelo-Montes, C.; Vidaurre, H.; Dawson, I.K.; Simons, A.J. Participatory domestication of agroforestry trees: An example from the Peruvian Amazon. Dev. Pract. 2001, 11, 425–433. [Google Scholar] [CrossRef]
- Lengkeek, A.G.; Muchugi-Mwangi, A.; Agufa, C.A.C.; Ahenda, J.O.; Dawson, I.K. Comparing genetic diversity in agroforestry systems with natural forest: A case study of the important timber tree Vitex fischeri in central Kenya. Agrofor. Syst. 2006, 67, 293–300. [Google Scholar] [CrossRef]
- Hollingsworth, P.; Dawson, I.; Goodall-Copestake, W.; Richardson, J.; Weber, J.C.; Sotelo-Montes, C.; Pennington, T. Do farmers reduce genetic diversity when they domesticate tropical trees? A case study from Amazonia. Mol. Ecol. 2005, 14, 497–501. [Google Scholar] [CrossRef]
- Sotelo-Montes, C.; Weber, J.C. Priorización de especies arbóreas para sistemas agroforestales en la selva baja del Perú. Agrofor. Am. 1997, 4, 12–17. [Google Scholar]
- Encarnación, F. Nomenclatura de las Especies Forestales Comunes en el Perú; Ministerio de Agricultura, Instituto Nacional Forestal y de Fauna: Lima, Peru, 1983; p. 149.
- Labarta, R.A.; Weber, J.C. Valorización económica de bienes tangibles de cinco especies arbóreas agroforestales en la Cuenca Amazónica Peruana. Rev. For. Centroam. 1998, 23, 12–21. [Google Scholar]
- Putzel, L.; Cronkleton, P.; Larson, A.; Pinedo-Vasquez, M.; Salazar, O.; Sears, R. Peruvian Smallholder Production and Marketing of Bolaina (Guazuma Crinita), a Fast-Growing Amazonian Timber Species: Call for a Pro-Livelihoods Policy Environment. Available online: http://www.cifor.org/library/4257 (accessed on 15 April 2017).
- Sotelo-Montes, C.; Vidaurre, H.; Weber, J.C.; Simons, A.J.; Dawson, I. Producción de semillas a partir de la domesticación participativa de árboles agroforestales en la amazonia peruana. In Proceedings of the Memorias del Segundo Symposio sobre Avances en la Producción de Semillas Forestales en América Latina. Proyecto de Semillas Forestales (PROSEFOR), Centro de Agricultura Tropical y de Enseñanza (CATIE) y International Union of Forest Research Organizations (IUFRO): Santo Domingo, República Dominica, 18–22 October 1999; Salazar, R., Ed.; CATIE: Turrialba, Costa Rica; pp. 65–72.
- IIAP (Instituto de Investigación de la Amazonía Peruana, PE). Evaluación Económica de Parcelas de Regeneración Natural y Plantaciones de Bolaina Blanca, Guazuma Crinita, en el Departamento de Ucayali; Instituto de Investigación de la Amazonía Peruana: Loreto, Peru, 2009; p. 54. [Google Scholar]
- Toledo, E.; Rincon, C. Utilización Industrial de Nuevas Especies Forestales en el Perú; Cámara Nacional Forestal, Instituto Nacional de Recursos Naturales, Organización Internacional de las Maderas Tropicales: Lima, Peru, 1996. [Google Scholar]
- Rochon, C.; Margolis, H.A.; Weber, J.C. Genetic variation in growth of Guazuma crinita (Mart.) trees at an early age in the Peruvian Amazon. Forest Ecol. Manag. 2007, 243, 291–298. [Google Scholar] [CrossRef]
- Hamrick, J.L.; Godt, M.J.; Sherman Broyles, S.L. Factors influencing levels of genetic diversity in woody plant species. New Forest 1992, 6, 95–124. [Google Scholar] [CrossRef]
- Ouborg, N.J.; Piquot, Y.; Van Groenendael, M. Population genetics, molecular markers and the study of dispersal in plants. J. Ecol. 1990, 87, 551–568. [Google Scholar] [CrossRef]
- Tuisima-Coral, L.; Hlásná-Čepková, P.; Lojka, B.; Weber, J.C.; Filomeno-Alves-Milo, S. Genetic diversity in Guazuma crinita from eleven provenances in the Peruvian Amazon revealed by ISSR markers. Bosque 2016, 37, 63–70. [Google Scholar] [CrossRef]
- Vodouhe, R.; Dansi, A. The “Bringing into Cultivation” Phase of the Plant Domestication Process and its Contributions to In Situ Conservation of Genetic Resources in Benin. Sci. World J. 2012, 176939. [Google Scholar] [CrossRef]
- Hernández, R.C.A.; Kafuri, L.A.; Isaza, R.A.; Arias, M.L. Analysis of genetic variation in clones of rubber (Hevea brasiliensis) from Asian, South and Central American origin using RAPDs markers. Rev. Col. Biotecnol. 2006, 8, 29–34. [Google Scholar]
- Thangjam, R. Inter-Simple Sequence Repeat (ISSR) Marker Analysis in Parkia timoriana (DC.) Merr. Populations from Northeast India. Appl. Biochem. Biotech. 2014, 172, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Nazareno, A.G.; Bemmels, J.B.; Dick, C.W.; Lohmann, L.I.G. Minimum sample sizes for population genomics: An empirical study from an Amazonian plant species. Mol. Ecol. Resour. 2017, 17, 1136–1147. [Google Scholar] [CrossRef] [PubMed]
- Rollo, A.; Ribeiro, M.M.; Costa, R.L.; Santos, C.; Clavo, P.Z.M.; Mandák, B.; Kalousová, M.; Vebrová, H.; Chuquilin, E.; Torres, S.G.; et al. Genetic Structure and Pod Morphology of Inga edulis Cultivated vs. Wild Populations from the Peruvian Amazon. Forests 2020, 11, 655. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Russell, J.R.; Fuller, J.D.; Macaulay, M.; Hatz, B.G.; Jahoor, A.; Powell, W.; Waugh, R. Direct comparison of levels of genetic variation among barley accessions detected by RFLPs, AFLPs, SSRs and RAPDs. Theor. Appl. Genet. 1997, 95, 714–722. [Google Scholar] [CrossRef]
- Vos, P.; Hogers, R.; Bleeker, M.; Reijans, M.; Lee van de, T.; Hornes, M.; Frijters, A.; Pot, J.; Peleman, J.; Kuiper, M.; et al. AFLP: A new technique for DNA fingerprinting. Nucleic Acids Res. 1995, 23, 4407–4414. [Google Scholar] [CrossRef]
- Mikulášková, E.; Fér, T.; Kučabová, V. The effect of different DNA isolation protocols and AFLP fingerprinting optimizations on error rate estimates in the bryophyte Campylopus introflexus. Lindbergia 2012, 35, 7–17. [Google Scholar]
- Yeh, F.C.; Yang, R.C.; Boyle, T.B.; Ye, Z.H.; Mao, J.X. POPGENE ver. 1.32 the User-Friendly Shareware for Population Genetic Analysis. Molecular Biology and Biotechnology Center, University of Alberta: Edmonton, AB, Canada, 1997. Available online: http://www.ualberta.ca/∼fyeh/ (accessed on 10 March 2020).
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research-an update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef] [PubMed]
- Perrier, X.; Jacquemoud-Collet, J.P. Darwin software. Available online: http://darwin.cirad.fr/darwin (accessed on 12 March 2020).
- Pritchard, J.; Stephens, M. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [PubMed]
- Pritchard, J.K.; Wen, W. Documentation for structure 2.0b. Available online: http://pritch.bsd.uchicago.edu (accessed on 1 April 2020).
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software structure: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef]
- Earl, D.A.; von Holdt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Rosenberg, N.A. DISTRUCT: A program for the graphical display of population structure. Mol. Ecol. Notes 2004, 4, 137–138. [Google Scholar] [CrossRef]
- Bonin, A.; Bellemain, E.; Bronken-Eidesen, P.; Pompanon, F.; Brochmann, C.; Taberlet, P. How to track and assess genotyping errors in population genetic studies. Mol. Ecol. 2004, 13, 3261–3273. [Google Scholar] [CrossRef]
- Weber, J.C.; Sotelo-Montes, C. Geographic variation in tree growth and wood density of Guazuma crinita Mart. in the Peruvian Amazon. New Forest 2008, 36, 29–52. [Google Scholar] [CrossRef]
- Weber, J.C.; Montes, C.S.; Cornelius, J.; Ugarte, J. Genetic variation in tree growth, stem form and mortality of Guazuma crinita in slower- and faster-growing plantations in the Peruvian Amazon. Silvae Genet. 2011, 30, 70–78. [Google Scholar] [CrossRef]
- White, T.L.; Adams, W.T.; Neale, D.B. Within Population Variation, Genetic Diversity, Mating Systems and Stand Structures; CABI Publishing, Ed.; Forest Genetics: Cambridge, UK, 2007; pp. 149–283. [Google Scholar]
- Hamrick, J.L.; Godt, M.J.W. Allozyme diversity in plant species. In Plant Population Genetics, Breeding and Genetic Resources; Brown, H.D., Clegg, M.T., Kahler, A.L., Weir, B.S., Eds.; Sinauer Associates: Sunderland, MA, USA, 1989; pp. 43–64. [Google Scholar]
- Reynel, C.; Pennington, R.; Pennington, T.; Flores, D.; Daza, C.A. Árboles útiles de la Amazonía Peruana, Manual de Identificación Ecológica y Propagación de las Especies; Universidad Nacional Agraria La Molina: Lima, Peru, 2003; p. 509. [Google Scholar]
- Abo-elwafa, A.K.; Shimada, M.T. Intra and inter specific variations in Lens revealed by RAPD markers. Theor. Appl. Genet. 1995, 90, 335–340. [Google Scholar] [CrossRef]
- Lu, Y.; Chen, C.; Wang, R.; Egan, A.; Fu, C. The effects of domestication on genetic diversity in Chimonanthus praecox: Evidence from cpDNA and AFLP data. J. Syst. Evol. 2015, 53, 239–251. [Google Scholar] [CrossRef]
- Simons, A.J.; MacQueen, D.J.; Stewart, J.L. Strategic concepts in the breeding of non-industrial trees. In Tropical Trees: The Potential for Domestication and the Rebuilding of Forest Resources, Proceedings of a conference organized by the Edinburgh Centre for Tropical Forests, Heriot-Watt University, Edinburgh, UK, 23-28 August 1992; Leakey, R.R.B., Newton, A.C., Eds.; HMSO: Edinburgh, UK, 1994; pp. 91–102. [Google Scholar]
- Aravanopoulos, F.A.P. Do Silviculture and Forest Management Affect the Genetic Diversity and Structure of Long-Impacted Forest Tree Populations? Forests 2018, 9, 355. [Google Scholar] [CrossRef]
- Ruņģis, D.; Luguza, S.; Bāders, E.; Šķipars, V.; Jansons, Ā. Comparison of Genetic Diversity in Naturally Regenerated Norway Spruce Stands and Seed Orchard Progeny Trials. Forests 2019, 10, 926. [Google Scholar] [CrossRef]
- Wang, B.; Mao, J.F.; Zhao, W.; Wang, X.R. Impact of geography and climate on the genetic differentiation of the subtropical pine Pinus yunnanensis. PLoS ONE 2013, 8, e67345. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.W.; Lewis, B.J.; Zhou, W.M.; Mao, C.R.; Wang, Y.; Zhou, L.; Yu, D.P.; Dai, L.M.; Qi, L. Genetic Diversity and Population Structure of Natural Pinus koraiensis Populations. Forests 2020, 11, 39. [Google Scholar] [CrossRef]
- Russell, J.R.; Weber, J.C.; Booth, A.; Powell, W.; Sotelo-Montes, C.; Dawson, I.K. Genetic variation of Calycophyllum spruceanum in the Peruvian Amazon basin, revealed by AFLP analysis. Mol. Ecol. 1999, 8, 199–204. [Google Scholar] [CrossRef]
- Nassar, J.M.; García-Rivas, A.E.; González, J.A. Patrones de diversidad genética en especies arbóreas de bosques fragmentados en Venezuela. Interciencia 2011, 36, 914–922. [Google Scholar]
- Pär, K.I.; Dahlberg, H. The effects of clonal forestry on genetic diversity in wild and domesticated stands of forest trees. Scand. J. For. Res. 2019, 34, 370–379. [Google Scholar] [CrossRef]
- Finkeldey, R.; Hattemer, H.H. Tropical Forest Genetics; Springer: Berlin/Heidelberg, Germany, 2007; p. 315. [Google Scholar] [CrossRef]
- Lemes, M.R.; Gribel, R.; Procor, J.; Grattapaglia, D. Population genetic structure of mahogany (Swietenia macrophylla King, Meliaceae) across the Brazilian Amazon, based on microsatellite at the loci: Implication for conservation. Mol. Ecol. 2003, 12, 2875–2883. [Google Scholar] [CrossRef]
- Kelly, B.A.; Hardy, O.J.; Bouver, J.M. Temporal and spatial genetic structure in Vitellaria paradoxa (shea tree) in an agroforestry system in southern Mali. Mol. Ecol. 2004, 13, 1231–1240. [Google Scholar] [CrossRef]
- Nelson, M.F.; Anderson, N.O. How many marker loci are necessary? Analysis of dominant marker data sets using two popular population genetic algorithms. Ecol. Evol. 2013, 3, 3455–3470. [Google Scholar] [CrossRef] [PubMed]
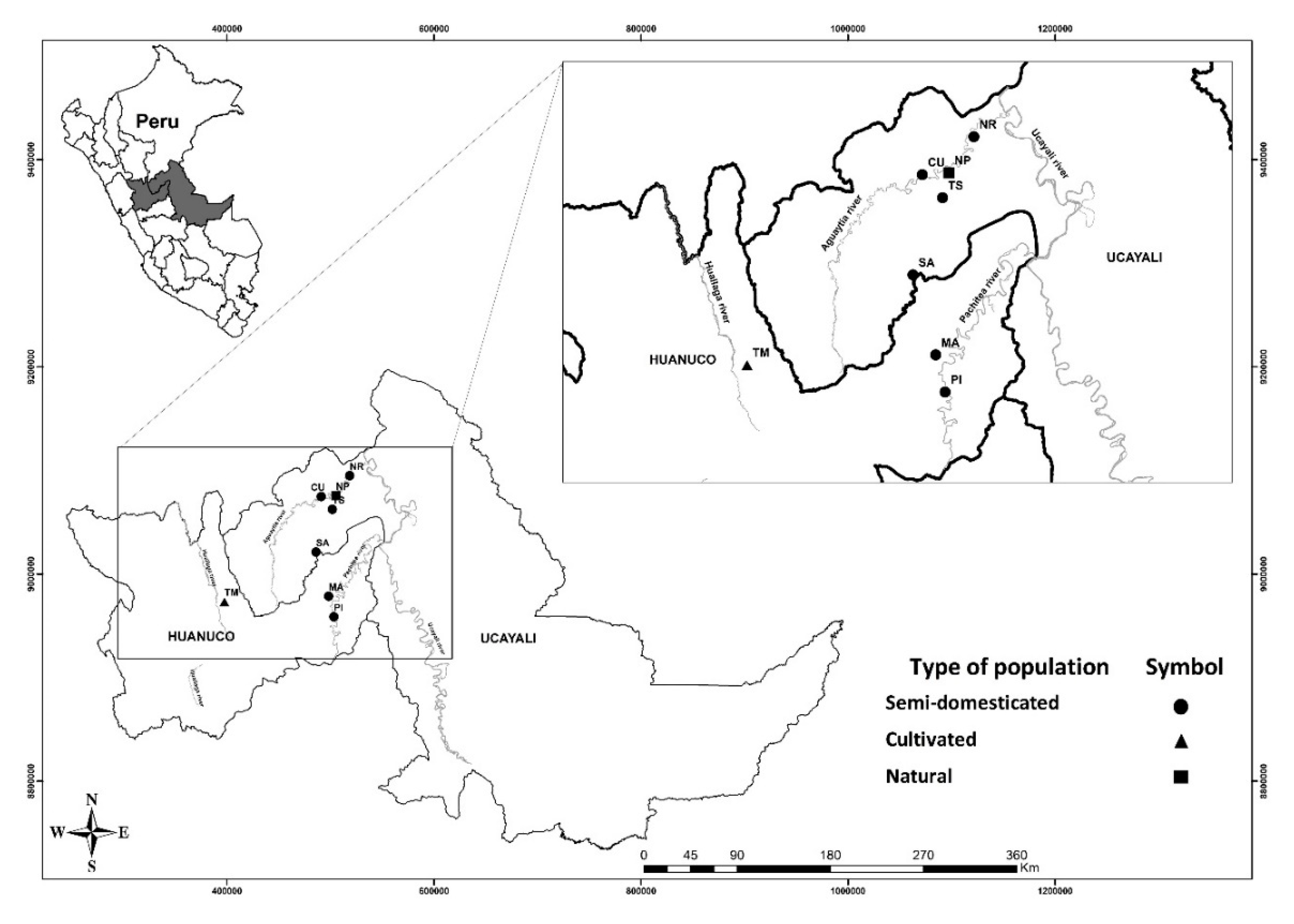
| Provenance | Region | No. of Samples (Code) | Population Type |
|---|---|---|---|
| Nuevo Piura (NP) | Ucayali | 19 (NP 1, 2, 3, ... 19) | Natural regeneration |
| Tingo Maria (TM) | Huanuco | 15 (TM 1, 2, 3, ... 15) | Cultivated 1 |
| Nueva Requena (NR) | Ucayali | 5 (NR 7, 8, 11, 12, 13) | Semi-domesticated 2 |
| Tahuayo Stream (TS) | Ucayali | 3 (TS 1, 2,9) | Semi-domesticated |
| San Alejandro (SA) | Ucayali | 5 (SA 1, 10, 11, 14, 15) | Semi-domesticated |
| Curimana (CU) | Ucayali | 4 (CU 3, 4, 6, 7) | Semi-domesticated |
| Puerto Inca (PI) | Huanuco | 3 (PI 1, 3, 13) | Semi-domesticated |
| Macuya (MA) | Huanuco | 5 (MA11, 33, 36, 42, 46) | Semi-domesticated |
| No. | Primer Combination | No. of Fragments | Fragment Range Size (pb) | Polymorphic Bands (%) |
|---|---|---|---|---|
| 1 | EcoRI-ACG/MseI- CTT | 35 | 58–231 | 20.6 |
| 2 | EcoRI-ACG/MseI- CTG | 23 | 60–215 | 13.5 |
| 3 | EcoRI-ACG/MseI- CTA | 19 | 69–198 | 11.1 |
| 4 | EcoRI-ACG/MseI- CAT | 27 | 52–176 | 15.9 |
| 5 | EcoRI-ACG/MseI- CAG | 10 | 71–140 | 5.9 |
| 6 | EcoRI-ACG/MseI- CAC | 29 | 64–336 | 17.0 |
| 7 | EcoRI-ACG/MseI- CAA | 28 | 52–228 | 16.4 |
| Total | 171 |
| Samples | N° of Samples | N° of PF | PPF (%) | Nei’s Gene Diversity (He) | Shannon Information Index (I) |
|---|---|---|---|---|---|
| Types of population | |||||
| Natural regeneration | 19 | 124 | 72.5 | 0.10 | 0.19 |
| Cultivated | 15 | 84 | 49.1 | 0.09 | 0.15 |
| Semi-domesticated | 24 | 93 | 54.4 | 0.09 | 0.16 |
| Provenances | |||||
| Nuevo Piura (NP) | 19 | 124 | 72.5 | 0.10 | 0.19 |
| Tingo Maria (CP) | 15 | 84 | 49.1 | 0.09 | 0.15 |
| Nueva Requena (SDP) | 4 | 35 | 20.5 | 0.06 | 0.10 |
| Tahuayo Stream (SDP) | 3 | 41 | 23.9 | 0.09 | 0.13 |
| San Alejandro (SDP) | 5 | 43 | 25.2 | 0.07 | 0.12 |
| Curimana (SDP) | 4 | 30 | 17.5 | 0.06 | 0.09 |
| Puerto Inca (SDP) | 3 | 31 | 18.1 | 0.07 | 0.10 |
| Macuya (SDP) | 5 | 32 | 18.7 | 0.07 | 0.10 |
| Species level | 58 | 170 | 99.4 | 0.11 | 0.20 |
| Provenances | NR | TS | SA | CR | NP | TM | PI | MA |
|---|---|---|---|---|---|---|---|---|
| NR | **** | 0.960 | 0.960 | 0.972 | 0.967 | 0.962 | 0.959 | 0.972 |
| TS | 0.040 | **** | 0.939 | 0.948 | 0.950 | 0.951 | 0.943 | 0.951 |
| SA | 0.041 | 0.063 | **** | 0.978 | 0.947 | 0.952 | 0.972 | 0.971 |
| CR | 0.028 | 0.054 | 0.023 | **** | 0.971 | 0.975 | 0.971 | 0.979 |
| NP | 0.033 | 0.051 | 0.054 | 0.029 | **** | 0.989 | 0.939 | 0.963 |
| TM | 0.038 | 0.050 | 0.049 | 0.026 | 0.011 | **** | 0.942 | 0.968 |
| PI | 0.042 | 0.059 | 0.029 | 0.030 | 0.063 | 0.059 | **** | 0.972 |
| MA | 0.029 | 0.050 | 0.030 | 0.022 | 0.037 | 0.032 | 0.029 | **** |
| Source of Variance | Degree of Freedom (df) | Sum of Square (SS) | Variance Component | Variance (%) | p Value a |
|---|---|---|---|---|---|
| Among population type | 2 | 99.43 | 1.60 | 12 | ˂0.001 |
| Among provenances | 5 | 69.95 | 0.58 | 4 | ˂0.001 |
| Within provenances | 50 | 583.56 | 11.67 | 84 | ˂0.001 |
| Total | 57 | 752.95 | 13.85 | 100 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuisima-Coral, L.L.; Hlásná Čepková, P.; Weber, J.C.; Lojka, B. Preliminary Evidence for Domestication Effects on the Genetic Diversity of Guazuma crinita in the Peruvian Amazon. Forests 2020, 11, 795. https://doi.org/10.3390/f11080795
Tuisima-Coral LL, Hlásná Čepková P, Weber JC, Lojka B. Preliminary Evidence for Domestication Effects on the Genetic Diversity of Guazuma crinita in the Peruvian Amazon. Forests. 2020; 11(8):795. https://doi.org/10.3390/f11080795
Chicago/Turabian StyleTuisima-Coral, Lady Laura, Petra Hlásná Čepková, John C. Weber, and Bohdan Lojka. 2020. "Preliminary Evidence for Domestication Effects on the Genetic Diversity of Guazuma crinita in the Peruvian Amazon" Forests 11, no. 8: 795. https://doi.org/10.3390/f11080795
APA StyleTuisima-Coral, L. L., Hlásná Čepková, P., Weber, J. C., & Lojka, B. (2020). Preliminary Evidence for Domestication Effects on the Genetic Diversity of Guazuma crinita in the Peruvian Amazon. Forests, 11(8), 795. https://doi.org/10.3390/f11080795






