1. Introduction
Pine wood nematode disease (PWD) is a devastating forest disease caused by pine wood nematode (PWN). The stem pests of pine trees are responsible for the short-distance natural transmission medium of the disease, and human economic and logistics activities are the main factors associated with the long-distance transmission and spread of the disease [
1]. Symptoms of the PWN disease were first discovered in Japan in 1905 [
2], but the PWN was identified as the causal agent behind the disease only in 1971. In the 1980s, the disease was introduced to China and South Korea and then to Portugal and Spain, gradually becoming one of the most dangerous tree diseases worldwide [
3,
4,
5,
6,
7]. Since the first discovery of PWD in China in 1982, the disease has developed rapidly in just a few decades. Its rapid spread in tropical and subtropical regions has caused serious damage. In recent years, it has gradually invaded the warm temperate zone; its range has crossed the 10 °C average annual temperature boundary and expanded to mid-temperate zones such as Liaoning Province and high-altitude areas such as the Qinling Mountains [
8,
9]. The reason why the population of PWN that has invaded can expand to high altitude and high-latitude low-temperature regions is associated with the long-distance transport of man-made logistics and economic activities, and the inheritance of low-temperature adaptation of PWN itself [
10]. In China, 60 million hectares of pine forest face the threat of a PWD epidemic [
11]. In 2019, PWD continued to spread, and the area south of the Yangtze River experienced a breakout. The epidemic developed from a single location into an area with an irregular circular shape and continued to spread rapidly from west to north [
12]. According to the 2020 PWN disease epidemic area announcement issued by the Chinese State Forestry and Grassland Administration (No. 4, 2020), the disease has spread to 666 county-level administrative regions in 18 provinces (regions, cities) in China.
The disease can harm more than 70 species, including 58 species of pinaceae trees [
13]. PWD is currently known to naturally infect 25 species of pinaceae trees in China, including
P. bungeana,
P. pinaster,
P. densiflora,
P. densiflora var.
umbraculifera,
P. thunbergii,
P. massoniana,
P. taeda,
P. elliottii,
P. thunbergii ×
P. massoniana,
P. luchuensis,
P. taiwanensis,
P. caribaea,
P. kesiya,
P. yunnan ensis,
P. tabuliformis,
P. armandii,
P. koraiensis,
P. virginiana,
P. palustris,
P. greggii,
P. strobus var.
chiapensis,
Abies holophylla,
Larix olgensis,
L. kaempferi, and
L. principis-rupprechtii [
14,
15,
16,
17,
18]. These pinaceae trees grow in large numbers from southern to northern China, and most of them are highly or moderately susceptible to PWN.
P. tabuliformis is one of the main afforestation tree species in China’s “Three-North” region [
19]. Currently, the total area of
P. tabuliformis in Liaoning Province is approximately 700 km
2, with a total stock volume of approximately 36 million m
3. In areas of
P. tabuliformis afforestation in Liaoning Province, soil and water conservation, landscaping, timber supplies and the underforest economy have played a substantial role in the development of the ecological environment of the forests [
20].
The adaptation of PWN pathogenicity to different pine species is one of the key factors affecting the spread of the disease. A large number of domestic and foreign studies have shown that there are differences in the pathogenicity of PWN from different geographic sources [
21,
22], and PWN isolates from different host sources exhibit different levels of virulence to different tree species. Siliang (2013) [
23] found through inoculation experiments on 18 species of PWN strains that the pathogenicity of different host source strains is different. Overall, the strains of
P. massoniana,
P. kesiya,
P. thunbergii, and
P. elliottii are highly pathogenic to
P. massoniana.
P. elliottii and
P. tabuliformis are moderately pathogenic, while
P. caribaea is weakly pathogenic. Xuelian (2007) [
24] studied the population morphology and pathogenic variation in PWN in China and showed that even strains from the same geographic origin are not completely consistent in their morphology and pathogenicity. Inoculation experiments on two-year-old
Pine. thunbergii showed that 90 PWN strains had extremely significant differences in pathogenicity; of these strains, AMA3 from Anhui had the strongest pathogenicity.
Due to the short outbreak of PWN in northern China, there are fewer studies on the pathogenicity of PWN in Liaoning Province. There is also a lack of relevant experimental data on the pathogenicity experiment of PWN artificially inoculated with P. tabuliformis. More research and experimentation are needed to determine whether PWN strains in northern and southern China exhibit differences in morphology and pathogenicity in different hosts and climate regions. This study intends to compare the morphology and pathogenicity of eight strains from southern China and eight strains from Liaoning Province in order to provide further evidence explaining the cause of the outbreak of PWNs in northern China.
2. Materials and Methods
2.1. Pine Seedlings for Testing
Two-year-old
P. thunbergia and
P. tabuliformis seedlings (purchased in a Dianzhuang flower and seedling shop and originating from Suqian city, Jiangsu Province, China) in pots were subjected to inoculation. The seedlings were kept in the greenhouse of Xiashu Forest Farm at Nanjing Forestry University, watered regularly and managed in the manner way. See
Table 1 for more details regarding the tested seedlings.
2.2. Pine Wood Nematodes for Testing
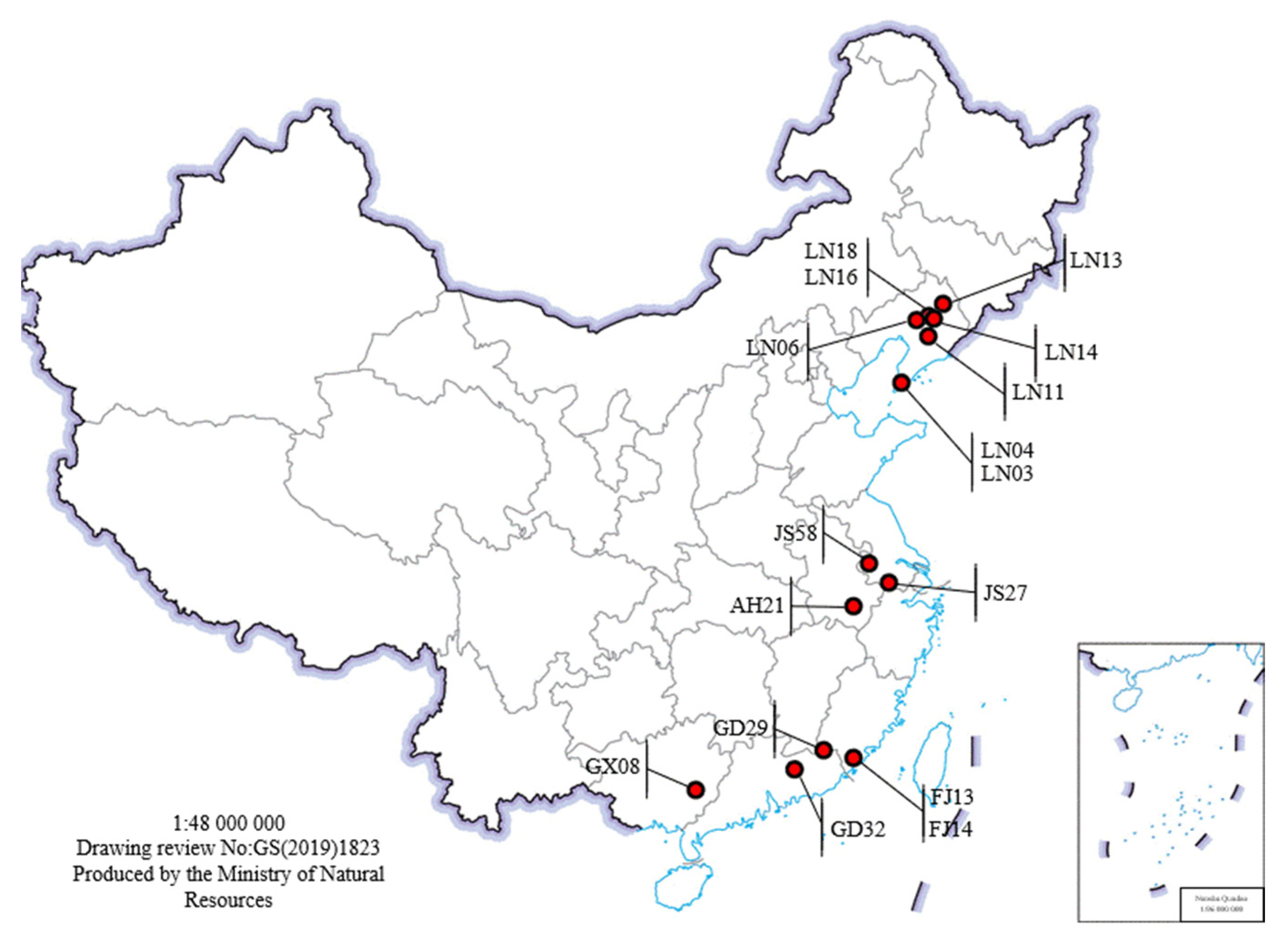
The 16 PWN strains used in this experiment were isolated and purified from infected wood collected by the Forest Pathology Laboratory of Nanjing Forestry University. See
Table 2 and
Figure 1 for details.
2.3. Activation and Cultivation of Nematodes
Botrytis cinerea is a fungus belonging to Sclerotiniaceae and Botryotinia, and it can be used as a source of nutrients for PWN. B. cinerea was inoculated onto potato dextrose agar (PDA) medium and placed in an incubator at 25 °C for 4–7 days. After the mycelium covered the culture dish, the suspensions of 16 strains isolated in the laboratory were removed from refrigeration at 4 °C and inoculated onto the B. cinerea at an ultraclean workbench. The culture dishes were sealed with sealing film and placed into a constant-temperature incubator at 25 °C. When the B. cinerea had been eaten by the nematodes, the nematodes were separated by the Baermann funnel method to obtain a fresh nematode suspension.
2.4. Morphological Measurement Indexes for the Nematodes (the De Man Formula)
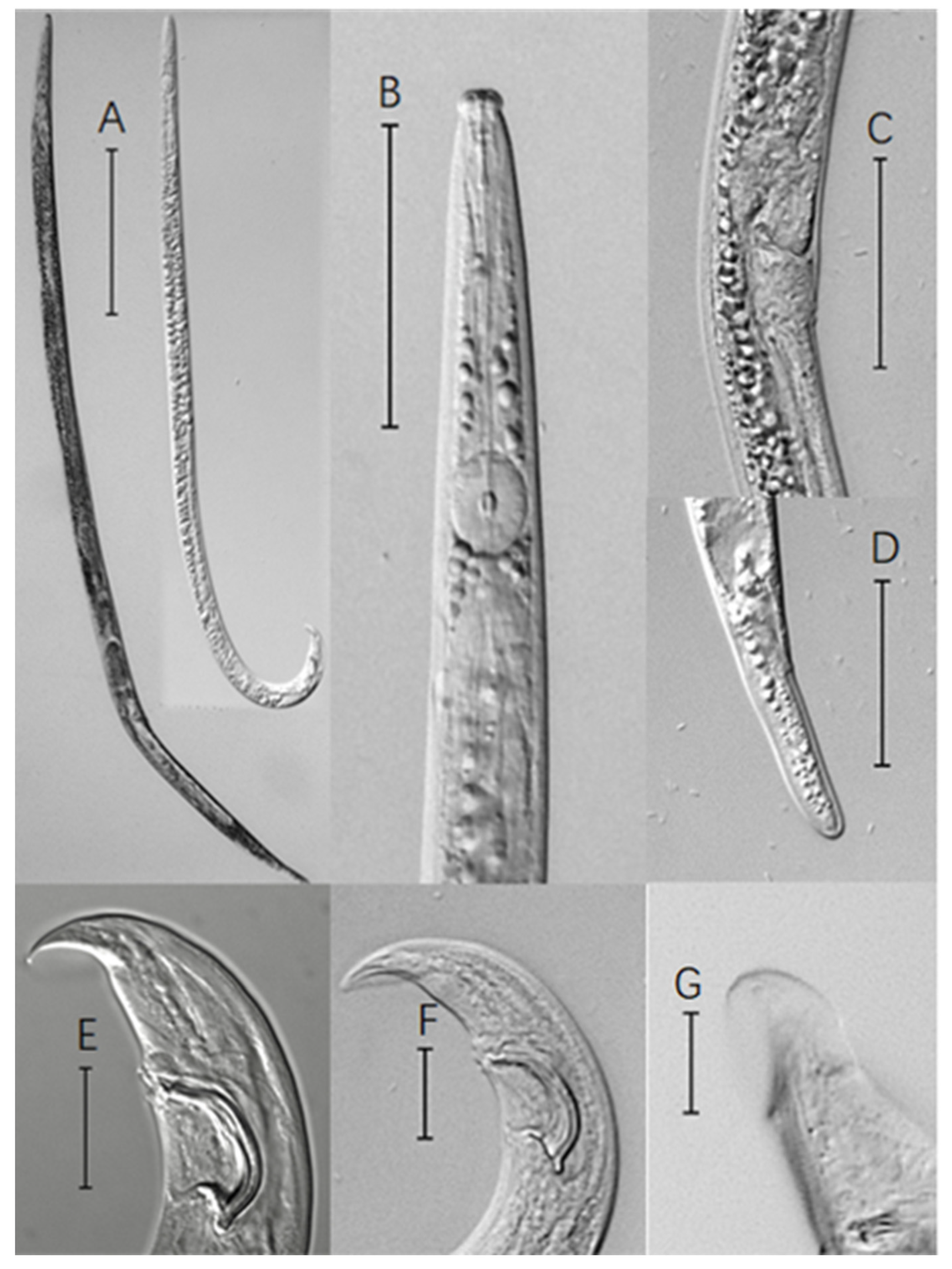
A total of 10 μL of the mixed nematode suspension was dropped onto a glass slide, with the bottom of the slide heated to kill the nematode. Then, a cover slip was placed gently over the slide, which was then viewed under the microscope for observation and measurement. Thirty nematodes of each strain were randomly selected for morphological measurement of the following indicators. N is the number of specimens measured, L is the body length (μm), LW is the maximum body width (μm), a is the body length/maximum body width ratio, b is the body length/tail length ratio, V is the distance from the vulva to the top of the head × 100/Body length, WMB is the width of the middle oesophageal bulb, Stylet is the length of the stylus (μm), Spi is the length of the spicule (μm), and Tail is the length of the tail (μm).
2.5. Determination of Pathogenicity
The propagative nematodes separated with the Baermann funnel method were collected in a 10 mL centrifuge tube and washed with sterile water 3 times to avoid the influence of nematode or fungal secretions on the inoculation result. Inoculation method: The artificial tree bark inoculation method was used for the pine trees. A sterile scalpel blade was used to cut a 0.3 cm deep incision into the tree bark. A small amount of sterile cotton was inserted into the incision. The incision with the cotton inside was wrapped with parafilm, leaving room for the spout of a funnel, and an appropriate amount of sterile water was added through the funnel. After checking for water leakage, the nematode solution (3000 nematodes per pine seedling) was added with a micro-injector. Each pine wood nematode strain was used to inoculate 5 P. thunbergia and 5 P. tabuliformis seedlings, and the control seedlings were injected with sterile water. The inoculations were performed on 22 June 2019. The inoculated pine seedlings were placed in a greenhouse at 30 °C with 18 h of light for cultivation. The presence of pine trees was observed daily, and the earliest time of the appearance of wilting symptoms of pine seedlings and the number of wilting pine seedlings caused by each strain were recorded.
2.6. Pathogenicity Indicators
According to the method of Fuyuan et al. (1998) [
25] and Qi et al. (2020) [
26], the disease incidence level of individual pine trees was recorded as follows: level 0, normal; level 1, fewer than half of the needles exhibited chlorosis, fewer than 1/4 of the needles exhibited yellowing; level 2, more than half of the needles exhibited chlorosis, 1/4~3/4 of the needles exhibited yellowing; level 3, more than 3/4 of the needles exhibited yellowing, fewer than half of the needles had turned red; and level 4 (number of disease symptom stage is 0–4), more than half of the needles had turned red, the plant is dying or has died.
2.7. Data Collection and Statistical Analysis
The nematodes were observed and counted under a Leica DM500 (Leica microsystems Gmbh, Wetzlar, Germany) microscope, and the nematodes were photographed and measured using the Carl Zeiss M2 imager (Carl Zeiss Microlmaging Gmbh, d-37081, Gottingen, Germany). The whole and part of the nematodes were photographed under 10×/0.30, 20×/0.50, and 40×/0.75 objective lenses (eyepiece is 10×) and corresponding scales were added to the photos, then we measured the length of the morphological index through the line segment to obtain the data. The morphological indexes included the body length and maximum width, the middle esophageal bulb width, the stylus length, the female distance from the vulva to the top of the head, the female tail length and the male spicule length. Each indicator was measured 30 times. SPSS Statistics 25.0 software was used to perform statistical analysis. Single-factor analysis of variance and Duncan multiple comparison analysis of variance were performed on the experimental results. Prism software was used for the mapping.
4. Discussion
According to previous studies, differences in pathogenicity among PWN strains may be related to the host [
23,
27], geographic location [
21], environment [
28,
29], associated bacterial species [
30], and strain fecundity [
31,
32]. This study showed that the same strain exhibits different levels of pathogenicity to different host pine trees. For instance, AH21 was more pathogenic to
P. thunbergii than JS58, but its pathogenicity to
P. tabuliformis was not as strong as that of JS58. This result suggested that PWN showed host specialization. The pathogenicity of strains isolated from different hosts to different pine also varied. Strains isolated from the same host showed similar pathogenicity. For instance, the strains LN18, LN16, and LN14 isolated from
L. gmelinii exhibited similar virulence and showed strong virulence against
P. thunbergii and
P. tabuliformis. Siliang (2013) [
23] pointed out that the strains with the same host source and inoculated target were more pathogenic. However, this correlation was not obvious in our study, and the pathogenicity of the strain still depended on the strain itself.
The pathogenicity of strains isolated from the same host source but different geographical populations may be different. The results of this experiment showed that the pathogenicity of the nematodes isolated from
P. massoniana was highly differentiated. This is because
P. massoniana itself has a large distribution area in southern China, and its disease resistance in different provenances varies [
25,
27]. Peigen et al. (1995) [
33] used pine wood nematode strains from China and Japan to inoculate
P. massoniana,
P. thunbergii,
Cedrus deodara,
P. elliottii and
P. taeda, respectively, and the study showed that there were certain differences in the pathogenicity of different hosts in different regions. In addition, the two strains from China and Japan, respectively, also differed in host specificity. The strains from China had strong pathogenicity to
P. thunbergii and basically no pathogenicity to
C. deodara, while strains from Japan had strong pathogenicity to
C. deodara and
P. elliottii. Generally, the PWN isolated from
P. thunbergii had weaker pathogenicity than that isolated from
P. massoniana. However, our results showed that the pathogenicity of strains isolated on
P. thunbergii is not always stronger than that of
P. massoniana. This illustrated that the geographical origin factor is generally, but not always, stronger than the host source factor. Nevertheless, the pathogenicity differences between strains with different geographical origins did not show absolute regularity. Zhiyu et al. (2002) [
28] measured the pathogenicity of pine wood nematode populations in China, Japan, and Canada to 3–4-year-old
P. thunbergii, and found that their pathogenicity was significantly differentiated. The study also showed there were both strong and weakly pathogenic groups in Nanjing. Thus, different strains of PWN had different pathogenicity levels, but the results had little correlation with geographical factors.
A comparison of the morphology and pathogenicity of two PWN strains in China by Ruocheng et al. (2019) [
34] showed that the female body length of the northern strain FCBX was longer than that of strain AMA3 in the south. The results of the inoculation experiment on
P. thunbergii showed that the strain FCBX is more pathogenic than the strain AMA3. However, it was not sufficient to only study these two individual strains, and
P. thunbergii was considered to be the most susceptible pine species for PWN. The inoculation experiment on
P. thunbergi cannot adequately explain the pathogenicity relationship between these two strains. Based on the experimental results, the female body length of GD32 was longer than all northern strains except LN04, and pathogenicity among the eight strains of Liaoning Province was similar and maintained at a high level, while the pathogenicity difference between the eight southern strains was greater. This pathogenic differentiation was more remarkable in the
P. tabuliformis inoculation experiment. There were not only highly pathogenic GD32 and JS27, but also moderately pathogenic GD29, JS58, GX08, FJ14, AH21 and weakly pathogenic FJ13 among the eight southern strains. Thus, there was no absolute northern strain whose pathogenicity is stronger than southern strains. Whether in the
P. thunbergii inoculation experiment or the
P. tabuliformis inoculation experiment, the southern strain GD32 was more pathogenic than all eight northern strains. From this, we can speculate that if the southern strain GD32 is introduced and established in the northern regions, it would quickly spread and cause damage in the north.