Liquid Chromatographic/Mass Spectrometric Study on the Role of Beech (Fagus sylvatica L.) Wood Polyphenols in Red Heartwood Formation
Abstract
1. Introduction
2. Materials and Methods
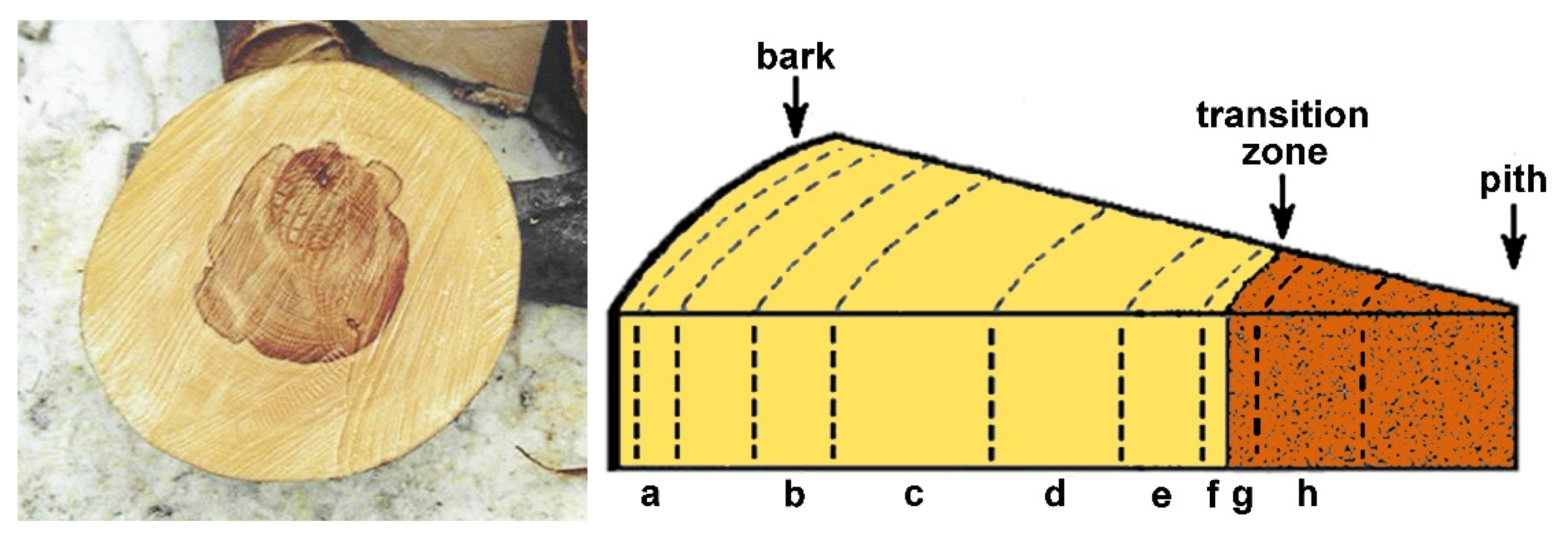
2.1. Sample Collection and Extraction
2.2. The HPLC-PDA-ESI-MS/MS Separation and Relative Quantitative Determination of Beech Wood Polyphenols
2.3. MALDI-TOF Analysis of the Extracts
2.4. Chemicals
2.5. Statistical Evaluation
3. Results and Discussion
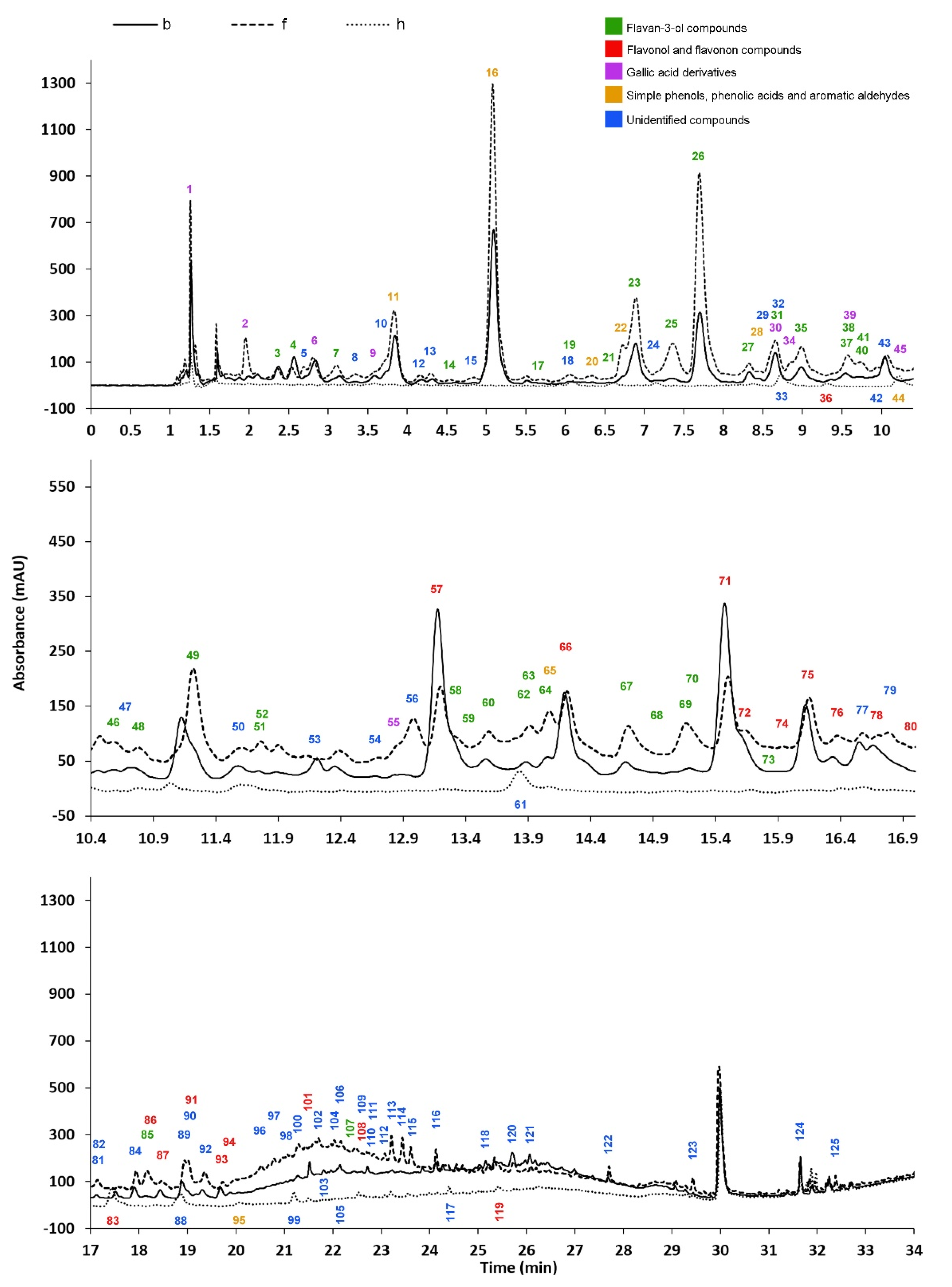
3.1. Separation and Identification of Polyphenols Using HPLC-PDA-ESI-MS/MS
3.2. Relative Quantitative Assessment of Polyphenols
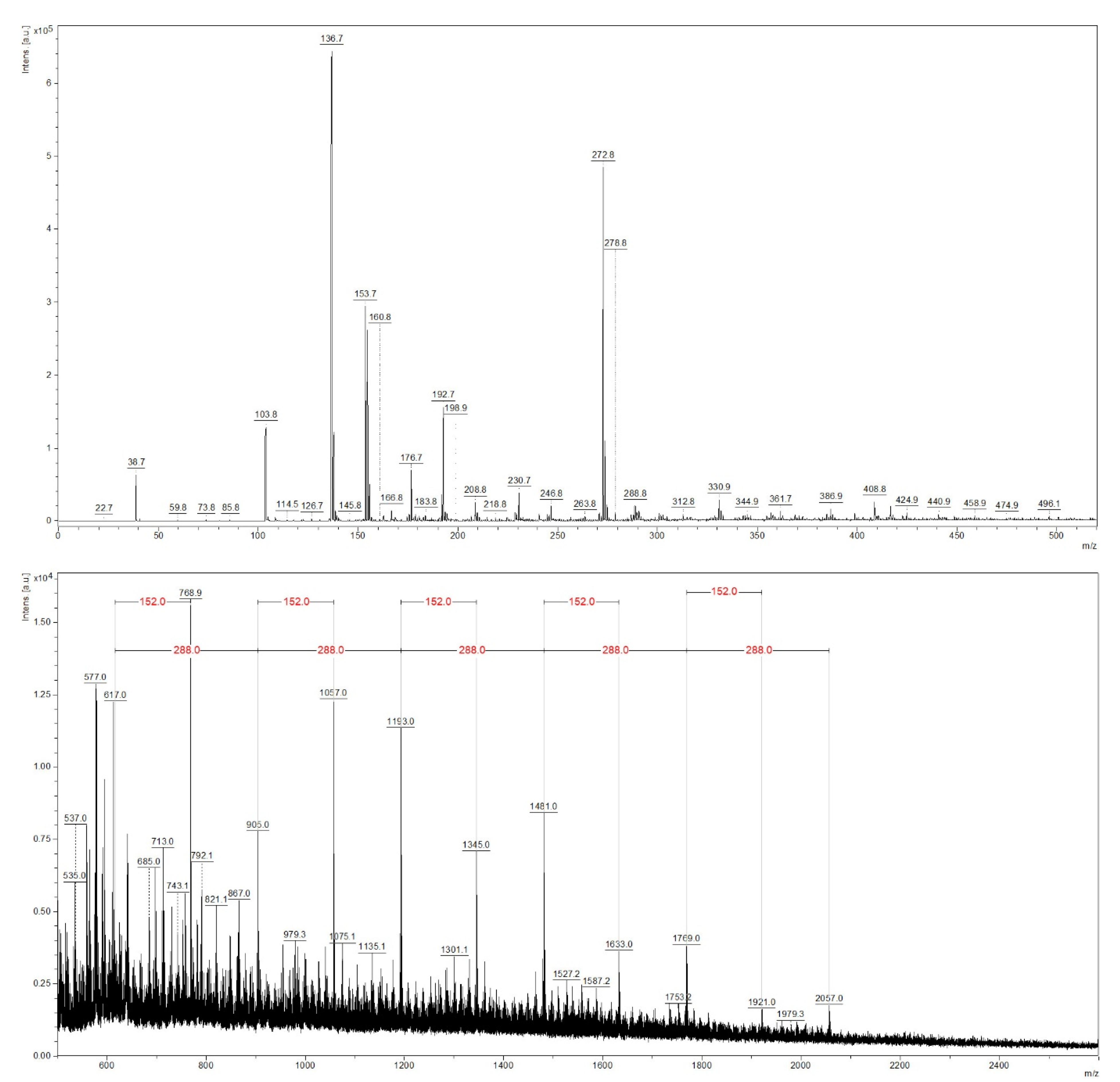
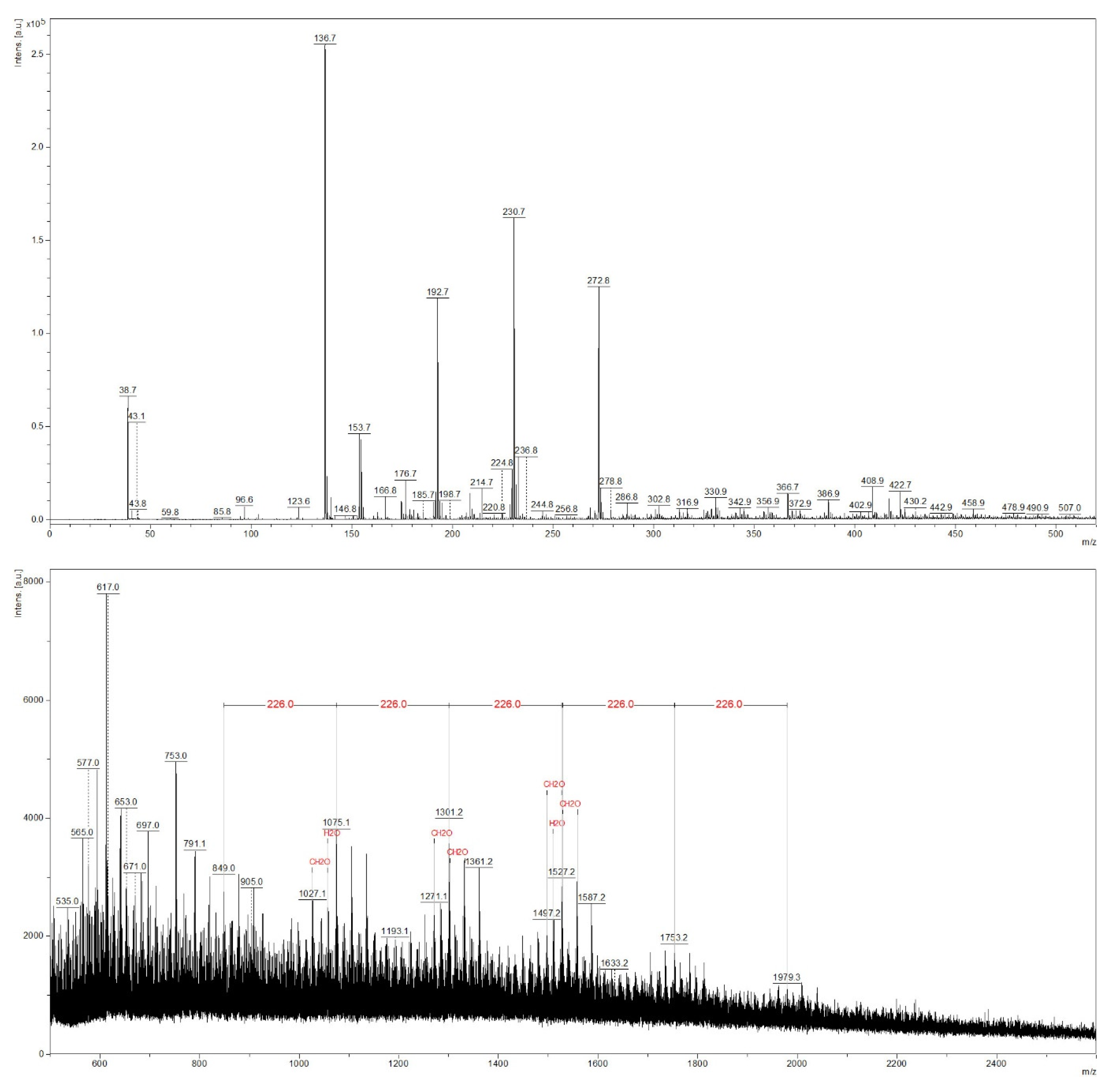
3.3. MALDI-TOF Analysis of Sapwood and Red Heartwood Extracts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Allen, C.D.; Breshears, D.D.; McDowell, N.G. On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 2015, 6, 1–55. [Google Scholar] [CrossRef]
- Molnár, S. Faanyagismeret; Mezőgazdasági Szaktudás Kiadó: Budapest, Hungary, 2004. [Google Scholar]
- Knoke, T. Predicting red heartwood formation in beech trees (Fagus sylvatica L.). Ecol. Model. 2003, 169, 295–312. [Google Scholar] [CrossRef]
- Nečesany, V. Forstliche Aspekte bei der Entstehung des Falschkerns bei der Rotbuche. Holz Zent. Bl 1969, 95, 563–564. [Google Scholar]
- Seeling, U. Kerntypen im Holz—Konsequenzen für die Verwertung am Beispiel der Buche (Fagus sylvatica L.). Schweiz Z Forstwes 1998, 149, 991–1004. [Google Scholar]
- Tarp, P.; Helles, F.; Holten-Andersen, P.; Bo Larsen, J.; Strange, N. Modelling near-natural silvicultural regimes for beech—An economic sensitivity analysis. For. Ecol. Manag. 2000, 130, 187–198. [Google Scholar] [CrossRef]
- Zell, J.; Hanewinkel, M.; Seeling, U. Financial optimisation of target diameter harvest of European beech (Fagus sylvatica) considering the risk of decrease of timber quality due to red heartwood. For. Policy Econ. 2004, 6, 579–593. [Google Scholar] [CrossRef]
- Hörnfeldt, R.; Drouin, M.; Woxblom, L. False heartwood in beech Fagus sylvatica, birch Betula pendula, B. papyrifera and ash Fraxinus excelsior—An overview. Ecol. Bull. 2010, 53, 61–75. [Google Scholar]
- Klement, I.; Vilkovska, T. Color characteristics of red false heartwood and mature wood of beech (Fagus sylvatica L.) determining by different chromacity coordinates. Sustainability 2019, 11, 690. [Google Scholar] [CrossRef]
- Sorz, J.; Hietz, P. Is oxygen involved in beech (Fagus sylvatica) red heartwood formation? Trees 2008, 22, 175–185. [Google Scholar] [CrossRef]
- Bonifazi, G.; Calienno, L.; Capobianco, G.; Lo Monaco, A.; Pelosi, C.; Picchio, R.; Serranti, S. Modeling color and chemical changes on normal and red heart beech wood by reflectance spectrophotometry, Fourier transform infrared spectroscopy and hyperspectral imaging. Polym. Degrad. Stab. 2015, 113, 10–21. [Google Scholar] [CrossRef]
- Seeling, U.; Becker, G.; Schwarz, C. Stand der Buchenrotkernforschung und Zerstörungsfreie Erfassung des Rotkerns bei Buche (Fagus sylvatica L.); Interner Abschlußbericht; Institut für Forstbenutzung und Forstliche Arbeitswissenschaft Universität Freiburg, University of Freiburg: Freiburg, Germany, 1999. [Google Scholar]
- Herrmann, E. Über die Kernbildung der Rotbuche. Z. F Forst-u Jagdwes. 1902, 34, 596–617. [Google Scholar]
- Mayer-Wegelin, H. Die Verkernung des Buchenholzes. In Silvae Orbis; CIS: Berlin, Germany, 1944; Volume 15, pp. 227–236. [Google Scholar]
- Sachsse, H. Kerntypen der Rotbuche. Forstarchiv 1991, 62, 238–242. [Google Scholar]
- Beimgraben, T. Stand der Rotkernforschung aus Holzanatomischer und Holztechnologischer Sicht, Materialien RVNA 1/03—Regionales Vermarktungsprojekt Rotkernige Buche; Regionalverband Neckar-Alb: Mössingen, Germany, 2003. [Google Scholar]
- Büren, S.V. Der Farbkern der Buche in der Schweiz Nördlich der Alpen—Untersuchung über die Verbreitung, die Erkennung am Stehendem Baum und die Ökonomischen Auswirkungen. Ph.D. Thesis, ETH Zürich, Zürich, Switzerland, 1997. [Google Scholar]
- Dietrichs, H.H. Studies of the chemistry and physiology of the transformation of sapwood into heartwood in Fagus sylvatica L. A contribution to the problem of heartwood formation. Mitt BundesforschAnst Forst Holzw 1964, 58, 141. [Google Scholar]
- Hofmann, T.; Albert, L.; Rétfalvi, T. Quantitative TLC analysis of (+)-catechin and (-)-epicatechin from Fagus sylvatica L. with and without red heartwood. J. Planar Chromatogr.-Mod. 2004, 17, 350–354. [Google Scholar] [CrossRef]
- Hofmann, T. A Kémiai Paraméterek Szerepe a Bükk (Fagus sylvatica L.) álgesztesedésében. Ph.D. Thesis, University of West Hungary, Sopron, Hungary, 2006. [Google Scholar]
- Hofmann, T.; Albert, L.; Rétfalvi, T.; Visi-Rajczi, E.; Brolly, G. TLC analysis of the in-vitro reaction of beech (Fagus sylvatica L.) wood enzyme extract with catechins. J. Planar Chromatogr.-Mod. 2008, 21, 83–88. [Google Scholar] [CrossRef]
- Visi-Rajczi, E.; Albert, L.; Hofmann, T.; Sárdi, É.; Koloszár, J.; Varga Sz Csepregi, I. Storage and accumulation of nonstructural carbohydrates in trunks of Fagus sylvatica L. in relation to discoloured Wood. In Proceedings of the International Conference Chemical Technology of Wood, Pulp and Paper, Bratislava, Slovak Republic, 17–18 September 2003; pp. 330–334. [Google Scholar]
- Dietrichs, H.H. The behaviour of carbohydrates during heartwood formation. Holzforschung 1964, 18, 14–24. [Google Scholar] [CrossRef]
- Magel, E.A.; Höll, W. Storage carbohydrates and adenine nucleotides in trunks of Fagus sylvatica in relation to discoloured wood. Holzforschung 1993, 47, 19–25. [Google Scholar] [CrossRef]
- Magel, E.; Jay-Allemand, C.; Ziegler, H. Formation of heartwood substances in the stemwood of Robinia pseudoacaia L. II: Distribution of nonstructural carbohydrates and wood extractives across the trunk. Trees 1994, 8, 165–171. [Google Scholar] [CrossRef]
- Magel, E.; Hübner, B. Distribution of phenylalanine ammonia lyase and chalcone synthase within trunks of Robinia pseudoacacia L. Bot. Acta 1997, 110, 314–322. [Google Scholar] [CrossRef]
- Yang, J.; Kamdem, D.P.; Keathley, D.E.; Han, K.-H. Seasonal changes in gene expression at the sapwood–heartwood transition zone of black locust (Robinia pseudoacacia) revealed by cDNA microarray analysis. Tree Physiol. 2004, 24, 461–474. [Google Scholar] [CrossRef]
- Beritognolo, I.; Magel, E.; Abdel-Latif, A.; Charpentier, J.P.; Jayallemand, C.; Breton, C. Are flavonoids de novo synthesised in Juglans nigra L. sapwood tissues being transformed into heartwood? Tree Physiol. 2002, 22, 291–300. [Google Scholar] [CrossRef]
- Burtin, P.; Jay-Allemand, C.; Charpentier, J.P.; Janin, G. Natural wood colouring process in Juglans sp. (J. nigra, J. regia and hybrid J. nigra x J. regia) depends on native phenolic compounds accumulated in the transition zone between sapwood and heartwood. Trees 1998, 12, 258–264. [Google Scholar] [CrossRef]
- Lim, K.-J.; Paasela, T.; Harju, A.; Venäläinen, M.; Paulin, L.; Auvinen, P.; Kärkkäinen, K.; Teeri, T.H. Developmental changes in Scots pine transcriptome during heartwood formation. Plant Physiol. 2016, 172, 1403–1417. [Google Scholar] [CrossRef] [PubMed]
- Hillis, W.E. Chemical aspects of heartwood formation. Wood Sci. Technol. 1968, 2, 241–259. [Google Scholar] [CrossRef]
- Vek, V.; Oven, P.; Poljanšek, I.; Ters, T. Contribution to understanding the occurrence of extractives in red heart of beech. BioResources 2015, 10, 970–985. [Google Scholar] [CrossRef][Green Version]
- Vek, V.; Oven, P.; Poljanšek, I. Review on lipophilic and hydrophilic extractives in tissues of common beech. Drv. Ind. 2016, 67, 85–96. [Google Scholar] [CrossRef]
- Mämmelä, P. Phenolics in selected European hardwood species by liquid chromatography–electrospray ionisation mass spectrometry. Analyst 2001, 126, 1535–1538. [Google Scholar] [CrossRef]
- Hofmann, T.; Nehebaj, E.; Albert, L. The high-performance liquid chromatography/multistage electrospray mass spectrometric investigation and extraction optimization of beech (Fagus sylvatica L.) bark polyphenols. J. Chromatogr. A 2015, 1393, 96–105. [Google Scholar] [CrossRef]
- Hofmann, T.; Nehebaj, E.; Albert, L. Antioxidant properties and detailed polyphenol profiling of European hornbeam (Carpinus betulus L.) leaves by multiple antioxidant capacity assays and high-performance liquid chromatography/multistage electrospray mass spectrometry. Ind. Crop. Prod. 2016, 87, 340–349. [Google Scholar] [CrossRef]
- Visi-Rajczi, E.; Hofmann, T.; Albert, L.; Mátyás, C. Tracing the acclimation of European beech (Fagus sylvatica L.) populations to climatic stress by analyzing the antioxidant system. iForest 2021, 14, 95–103. [Google Scholar] [CrossRef]
- Hillis, W.E.; Inoue, T. The formation of polyphenols in trees—III.: The effect of enzyme inhibitors. Phytochemistry 1966, 5, 491–499. [Google Scholar] [CrossRef]
- Barry, K.M.; Davies, N.W.; Mohamed, C.L. Identification of hydrolysable tannins in the reaction zone of Eucalyptus nitens wood by high performance liquid chromatography–electrospray ionisation mass spectrometry. Phytochem. Anal. 2001, 12, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Hashida, K.; Tabata, M.; Kuroda, K.; Otsuka, Y.; Kubo, S.; Makino, R.; Kubojima, Y.; Tonosaki, M.; Ohara, S. Phenolic extractives in the trunk of Toxicodendron vernicifluum: Chemical characteristics, contents and radial distribution. J. Wood Sci. 2014, 60, 160–168. [Google Scholar] [CrossRef]
- Ricci, A.; Parpinello, G.P.; Palma, A.S.; Teslić, N.; Brilli, C.; Pizzi, A.; Versari, A. Analytical profiling of food-grade extracts from grape (Vitis vinifera sp.) seeds and skins, green tea (Camellia sinensis) leaves and Limousin oak (Quercus robur) heartwood using MALDI-TOF-MS, ICP-MS and spectrophotometric methods. J. Food Compost. Anal. 2017, 59, 95–104. [Google Scholar] [CrossRef]
- Rockenbach, I.I.; Jungfer, E.; Ritter, C.; Santiago-Schübel, B.; Thiele, B.; Fett, R.; Galensa, R. Characterization of flavan-3-ols in seeds of grape pomace by CE, HPLC-DAD-MSn and LC-ESI-FTICR-MS. Food Res. Int. 2012, 48, 848–855. [Google Scholar] [CrossRef]
- Tran, J.C.; Doucette, A.A. Cyclic polyamide oligomers extracted from nylon 66 membrane filter disks as a source of contamination in liquid chromatography/mass spectrometry. Am. Soc. Mass. Spectrom. 2006, 17, 652–656. [Google Scholar] [CrossRef]
- Pérez-Jimnez, J.; Díaz-Rubio, M.; Saura-Calixto, F. Non-extractable polyphenols in plant foods: Nature, isolation and analysis. In Polyphenols in Plants, 2nd ed.; Watson, R.R., Ed.; Academic Press: London, UK, 2014; pp. 203–218. [Google Scholar]
| Flavan-3-ols | |||
|---|---|---|---|
| Peak | Compound Name | [M − H]− m/z | MS/MS m/z |
| 26 | (+)-Catechin | 289 | 245, 203, 125, 123, 109 |
| 49 | (–)-Epicatechin | 289 | 245, 203, 125, 123, 109 |
| 21, 23, 25, 38, 46, 52, 67 | Procyanidin B dimer | 577 | 451, 425, 407, 289, 245, 125 |
| 3, 4, 7, 27, 35, 40, 58, 60, 62, 73 | Procyanidin C trimer | 865 | 739, 695, 755, 407, 289, 125 |
| 31, 37, 63, 70 | Procyanidin D tetramer | 1153 | 865, 695, 577, 451, 407, 289, 245, 125 |
| 41, 48, 51, 68 | Procyanidin E pentamer | 1441 | [M − 2H]2−: 720; 865, 577, 451, 425, 289, 125 |
| 59, 64, 69 | Procyanidin dimer monogallate | 729 | 603, 559, 577, 451, 441, 425, 407, 289 |
| 85 | (epi)catechin monogallate | 441 | 289, 245, 205, 169, 125, 109 |
| 14, 17, 19 | (epi)catechin-O-hexoside | 451 | 331, 289, 271, 245, 203, 125, 109 |
| 107 | (epi)afzelechin-O-hexoside | 435 | 315, 273, 179, 167 |
| Flavonol and flavonon compounds | |||
| Peak | Compound name | [M − H]− m/z | MS/MS m/z |
| 91 | Isorhamnetin | 315 | 300, 271, 255, 227 135 |
| 101 | Isorhamnetin-O-pentoside | 447 | 315, 314, 300, 271, 243 |
| 74, 86, 108 | Isorhamnetin-O-hexoside | 477 | 315, 314, 300, 271 |
| 83 | Taxifolin | 303 | 285, 275, 217, 177, 151, 125 |
| 71, 75, 87, 93 | Taxifolin-O-pentoside | 435 | 303, 285, 257, 179, 151, 107 |
| 57, 66, 78 | Taxifolin-O-hexoside | 465 | 285, 303, 339, 257, 179, 151, 107 |
| 36 | Quercetin | 301 | 300, 271, 255 |
| 94 | Quercetin-O-hexoside | 463 | 301, 300, 271, 255, 215, 179, 151 |
| 119 | Naringenin | 271 | 177, 151, 119, 107, 93 |
| 72, 76, 80 | Naringenin-C-hexoside | 433 | 387, 343, 313, 283, 271, 193, 151, 119, 107, 93 |
| Gallic acid derivatives | |||
| Peak | Compound name | [M − H]− m/z | MS/MS m/z |
| 1, 2, 6, 9 | Monogalloyl glucose | 331 | 169, 125 |
| 34 | Unknown galloylglucose derivative | 511 | 467, 313, 197, 169, 125 |
| 30, 45, 55 | Digalloyl glucose | 483 | 439, 313, 169, 125 |
| 39 | Gallic acid + taxifolin derivative | 453 | 313, 285, 169, 125 |
| Simple phenols, phenolic acids and aromatic aldehydes | |||
| Peak | Compound name | [M − H]− m/z | MS/MS m/z |
| 11 | Vanillic acid-O-hexoside | 397 | 329, 235, 167, 152, 123, 108 |
| 22 | Dihydro-coumaric acid-O-hexoside | 327 | 179, 165, 147, 121 |
| 16, 20 | Syringic acid-O-hexoside | 359 | 239, 207, 197, 182, 167, 153, 138, 123, 95 |
| 28, 65 | Coniferin isomer * | 387 | 341, 207, 179, 164, 161, 146 |
| 44 | Syringic acid | 197 | 182, 167, 153, 138, 123, 121, 95 |
| 95 | Sinapaldehyde | 207 | 192, 177, 163 |
| Unidentified compounds | |||
| Peak | Compound name | [M − H]− m/z | MS/MS m/z |
| 5 | Unknown | 191 | 111, 103 |
| 8 | Unknown | 315 | 201, 165, 152, 108 |
| 10 | Unknown | 343 | 205, 163, 135, 93 |
| 12 | Unknown | 391 | 183 |
| 13 | Unknown | 345 | 183, 168, 139, 124 |
| 15 | Unknown | 863 | 341, 165 |
| 18 | Unknown | 241 | 211, 193, 178, 163, 153 |
| 24 | Unknown | 269 | 209, 138, 131 |
| 29 | Unknown | 439 | 331, 195, 180, 165, 150 |
| 32 | Unknown-O-hexoside * | 391 | 345, 183, 168, 153, 161, 85 |
| 33 | Unknown | 271 | 256, 241, 213, 185 |
| 42, 43 | Unknown | 463 | 417, 305, 235, 181, 166, 135, 121, 109 |
| 47 | Unknown | 431 | 305, 123, 108 |
| 50 | Unknown-O-hexoside * | 431 | 385, 223, 205, 161, 153, 123 |
| 53 | Unknown | 405 | 357, 161 |
| 54 | Unknown | 371 | 353, 249, 175, 121, 99 |
| 56 | Unknown | 461 | 415, 149 |
| 61 | Unknown | 187 | 119, 92 |
| 77 | Unknown | 575 | 538, 449, 289, 217, 203 |
| 79 | Unknown | 449 | 269, 259, 152 |
| 81 | Unknown-O-hexoside | 539 | 377, 359, 313, 209, 193, 180, 149 |
| 82 | Unknown | 497 | 481, 313, 297, 169, 153, 124 |
| 84 | Unknown | 441 | 397, 330, 217, 205, 187, 133, 109 |
| 88 | Unknown | 419 | 404, 373, 359, 344, 313, 281, 175, 124 |
| 89 | Unknown | 551 | 419, 401, 373, 359, 358 |
| 90 | Unknown | 403 | 223, 179, 161, 101 |
| 92 | Unknown | 551 | 419, 401, 373, 359, 358 |
| 96 | Unknown | 449 | 281, 167, 137, 93 |
| 97 | Unknown | 415 | 337, 283, 180, 161 |
| 98 | Unknown | 503 | 371, 293, 163, 125 |
| 99 | Unknown | 263 | 243, 203, 152 |
| 100 | Unknown | 553 | 421, 406, 391, 373, 195, 177 |
| 102 | Unknown | 455 | 289, 183, 168, 124 |
| 103 | Unknown | 417 | 402, 387, 371, 190, 181, 166, 151, 137 |
| 104 | Unknown | 535 | 337, 323, 197, 175, 160 |
| 105 | Unknown | 547 | 432, 163 |
| 106 | Unknown | 571 | 523, 493, 345, 327, 165, 150, 122 |
| 109 | Unknown | 403 | 388, 373, 181, 166, 151, 137 |
| 110 | Unknown | 571 | 523, 493, 345, 327, 165, 150, 122 |
| 111 | Unknown | 465 | 341, 281, 168, 153, 137, 93 |
| 112 | Unknown | 557 | 509, 361, 165, 150 |
| 113 | Unknown | 341 | 326, 311 |
| 114 | Unknown | 555 | 507, 326, 195, 165, 150 |
| 115 | Unknown | 617 | 569, 421, 391, 165, 150 |
| 116 | Unknown | 585 | 537, 359, 345, 195, 165, 150 |
| 117 | Unknown | 521 | 473, 337, 175, 160, 153, 135 |
| 118 | Unknown | 581 | 551, 533, 503, 367, 355, 325, 190, 165, 150, 122 |
| 120 | Unknown | 809 | 565, 535, 417, 195, 165, 150, 122 |
| 121 | Unknown | 579 | 355, 337, 321, 193, 178, 150 |
| 122 | Unknown | 537 | 522, 491, 249, 193, 175, 160, 134 |
| 123 | Unknown | 487 | 409 |
| 124 | Unknown | 471 | 427 |
| 125 | Unknown | 327 | 281, 265 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hofmann, T.; Guran, R.; Zitka, O.; Visi-Rajczi, E.; Albert, L. Liquid Chromatographic/Mass Spectrometric Study on the Role of Beech (Fagus sylvatica L.) Wood Polyphenols in Red Heartwood Formation. Forests 2022, 13, 10. https://doi.org/10.3390/f13010010
Hofmann T, Guran R, Zitka O, Visi-Rajczi E, Albert L. Liquid Chromatographic/Mass Spectrometric Study on the Role of Beech (Fagus sylvatica L.) Wood Polyphenols in Red Heartwood Formation. Forests. 2022; 13(1):10. https://doi.org/10.3390/f13010010
Chicago/Turabian StyleHofmann, Tamás, Roman Guran, Ondrej Zitka, Eszter Visi-Rajczi, and Levente Albert. 2022. "Liquid Chromatographic/Mass Spectrometric Study on the Role of Beech (Fagus sylvatica L.) Wood Polyphenols in Red Heartwood Formation" Forests 13, no. 1: 10. https://doi.org/10.3390/f13010010
APA StyleHofmann, T., Guran, R., Zitka, O., Visi-Rajczi, E., & Albert, L. (2022). Liquid Chromatographic/Mass Spectrometric Study on the Role of Beech (Fagus sylvatica L.) Wood Polyphenols in Red Heartwood Formation. Forests, 13(1), 10. https://doi.org/10.3390/f13010010









