Responses of Nutrient Resorption to Human Disturbances in Phoebe bournei Forests
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Experimental Design
2.3. Field Sample Collection and Laboratory Analysis
2.4. Statistical Analysis
3. Results
3.1. Green Leaf, Senesced Leaf, and Soil Total Nutrient Contents of Phoebe Bournei Forest
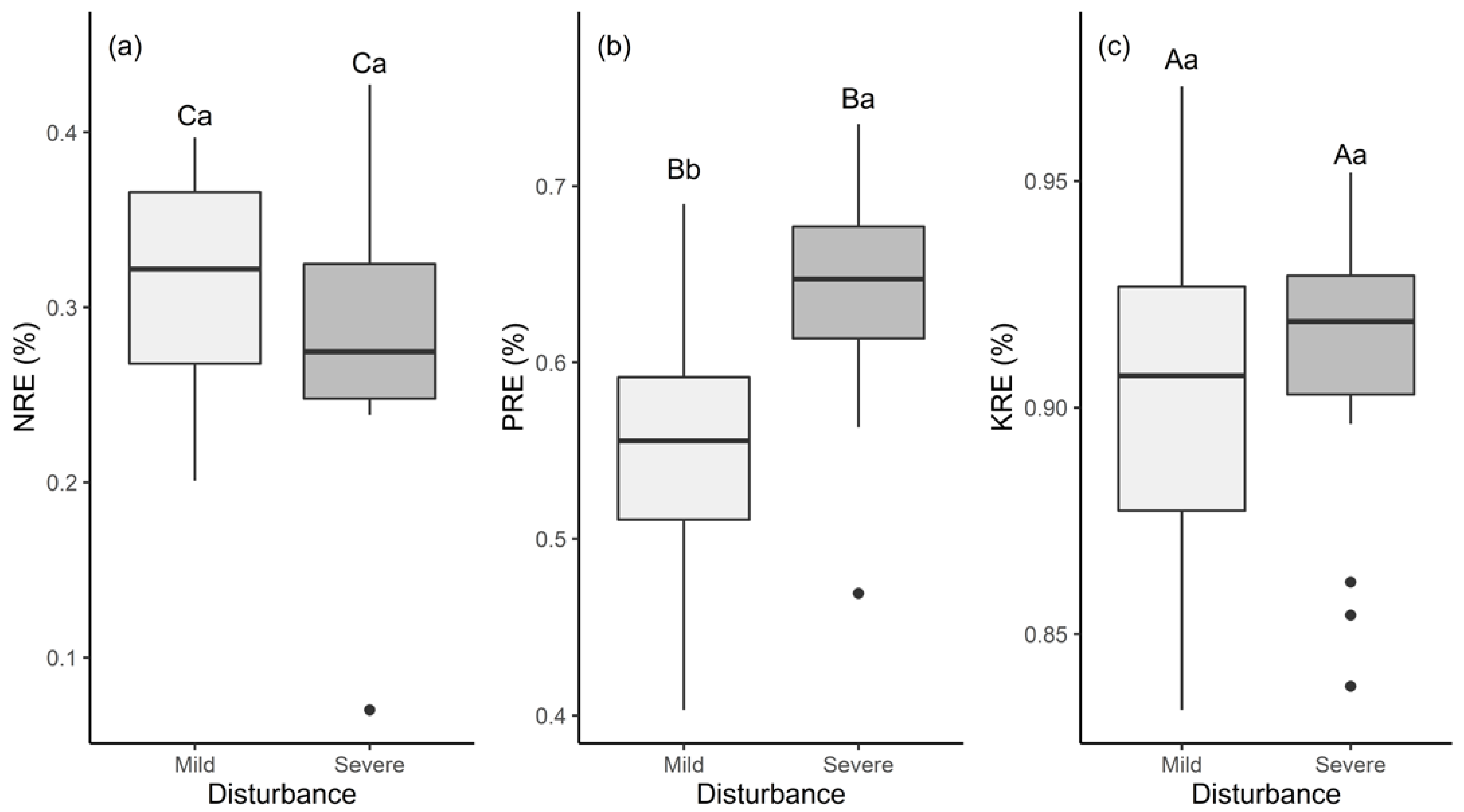
3.2. Leaf N, P, and K Resorption Efficiencies under Different Disturbances in the Phoebe Bournei Forest
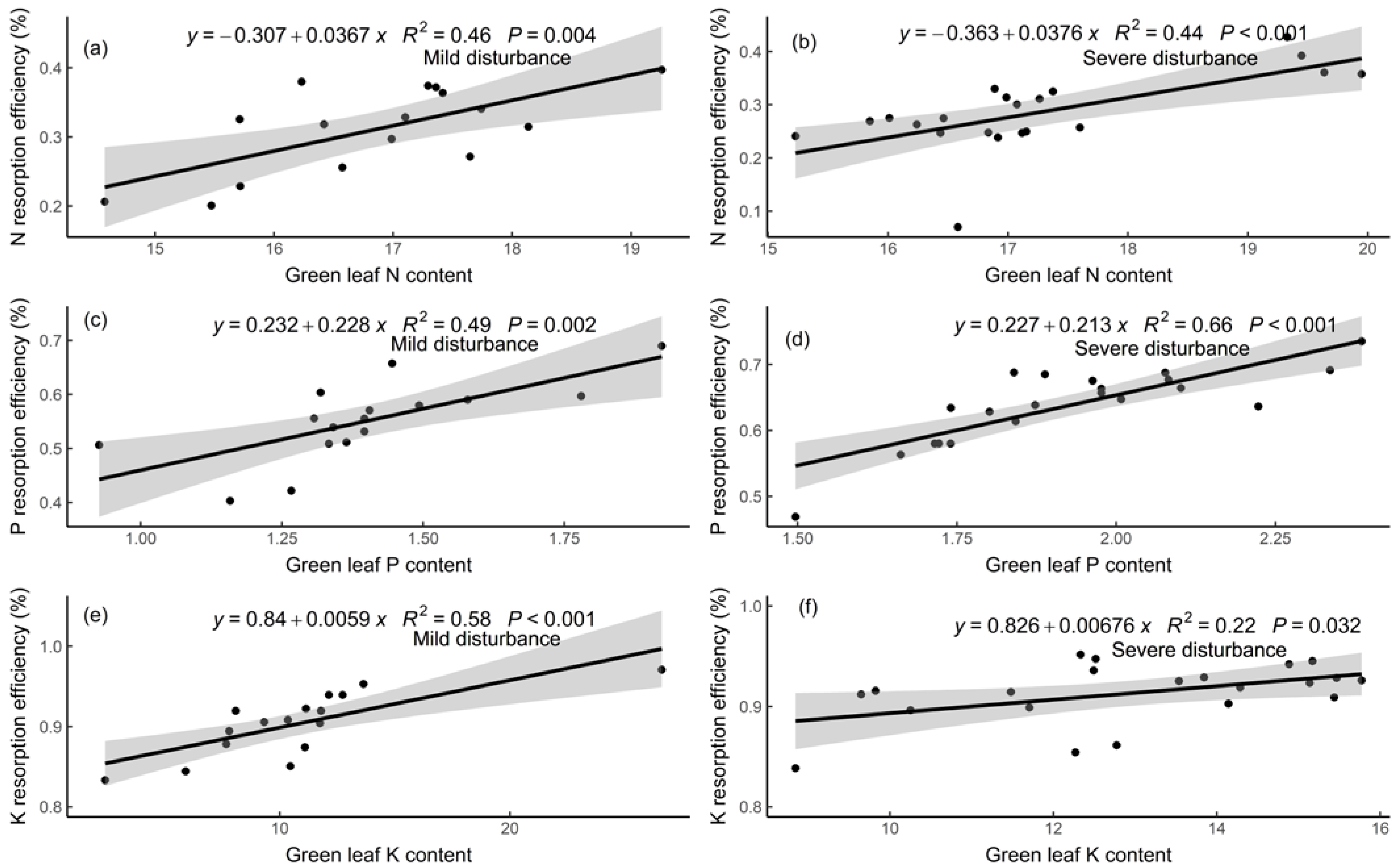
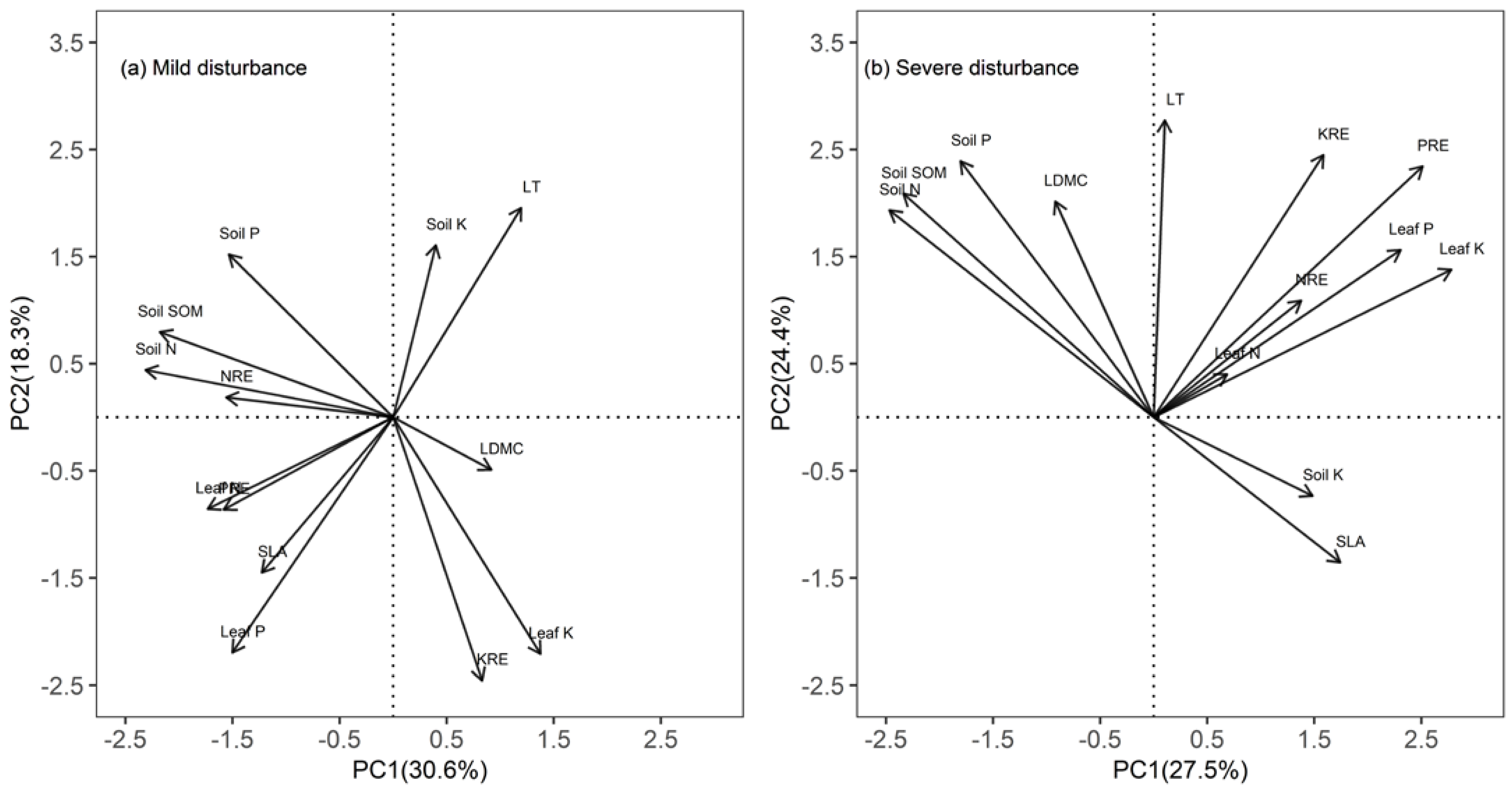
3.3. Relationships of Nutrient Resorption Efficiencies with Leaf Traits and Soil Nutrients
4. Discussion
4.1. Effects of Green Leaf Content on Soil Nutrients
4.2. Nutrient Resorption and Stoichiometric Characteristics under Different Disturbances
4.3. Relationships among Nutrient Resorption, Leaf Traits, and Soil Nutrients
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vergutz, L.; Manzoni, S.; Porporato, A.; Novais, R.F.; Jackson, R.B. Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol. Monogr. 2012, 82, 205–220. [Google Scholar] [CrossRef] [Green Version]
- Drenovsky, R.E.; Pietrasiak, N.; Short, T.H.; Silva, T. Global temporal patterns in plant nutrient resorption plasticity. Glob. Ecol. Biogeogr. 2019, 28, 728–743. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Reed, S.C.; Lü, X.; Xiao, K.; Wang, K.; Li, D. Global resorption efficiencies of trace elements in leaves of terrestrial plants. Funct. Ecol. 2021, 3, 1596–1602. [Google Scholar] [CrossRef]
- Zhang, T.; Li, F.Y.; Shi, C.; Li, Y.; Tang, S.; Baoyin, T. Enhancement of nutrient resorption efficiency increases plant production and helps maintain soil nutrients under summer grazing in a semi-arid steppe. Agric. Ecosys. Environ. 2020, 292, 106840. [Google Scholar] [CrossRef]
- Chen, H.; Reed, S.C.; Lu, X.; Xiao, K.; Wang, K.; Li, D. Coexistence of multiple leaf nutrient resorption strategies in a single ecosystem. Sci. Total Environ. 2021, 772, 144951. [Google Scholar] [CrossRef]
- Aerts, R. Nutrient resorption from senescing leaves of perennials: Are there general patterns? J. Ecol. 1996, 84, 597–608. [Google Scholar] [CrossRef]
- Killingbeck, K.T. Nutrients in senesced leaves: Keys to the search for potential resorption and resorption proficiency. Ecology 1996, 77, 1716–1727. [Google Scholar] [CrossRef]
- Urbina, I.; Grau, O.; Sardans, J.; Margalef, O.; Peguero, G.; Asensio, D.; LLusià, J.; Ogaya, R.; Gargallo-Garriga, A.; Van Langenhove, L.; et al. High foliar K and P resorption efficiencies in old-growth tropical forests growing on nutrient-poor soils. Ecol. Evol. 2021, 11, 8969–8982. [Google Scholar] [CrossRef]
- Brant, A.N.; Chen, H.Y.H. Patterns and mechanisms of nutrient resorption in plants. Crit. Rev. Plant Sci. 2015, 34, 471–486. [Google Scholar] [CrossRef]
- Huang, G.; Su, Y.; Mu, X.; Li, Y. Foliar nutrient resorption responses of three life-form plants to water and nitrogen additions in a temperate desert. Plant Soil 2018, 424, 479–489. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, J.; Wang, J.; Guo, Z.; Wang, G.G.; Zeng, D.; Wu, T. Tree stoichiometry and nutrient resorption along a chronosequence of Metasequoia glyptostroboides forests in coastal China. For. Ecol. Manag. 2018, 430, 445–450. [Google Scholar] [CrossRef]
- Chen, X.; Chen, H.Y.H. Foliar nutrient resorption dynamics of trembling aspen and white birch during secondary succession in the boreal forest of central Canada. For. Ecol. Manag. 2021, 505, 119876. [Google Scholar] [CrossRef]
- Ochieng, C.A.; Erftemeijer, P.L. Phenology, litterfall and nutrient resorption in Avicennia marina (Forssk.) Vierh in Gazi Bay, Kenya. Trees 2001, 16, 167–171. [Google Scholar] [CrossRef]
- Lü, X.T.; Hu, Y.Y.; Wolf, A.A.; Han, X.G. Species richness mediates within-species nutrient resorption: Implications for the biodiversity-productivity relationship. J. Ecol. 2019, 107, 2346–2352. [Google Scholar] [CrossRef]
- Tong, R.; Zhou, B.; Jiang, L.; Ge, X.; Cao, Y. Spatial patterns of leaf carbon, nitrogen, and phosphorus stoichiometry and nutrient resorption in Chinese fir across subtropical China. Catena 2021, 201, 105221–105229. [Google Scholar] [CrossRef]
- Akram, M.A.; Wang, X.; Hu, W.; Xiong, J.; Zhang, Y.; Deng, Y.; Ran, J.; Deng, J. Convergent variations in the leaf traits of desert plants. Plants 2020, 9, 990. [Google Scholar] [CrossRef]
- Wright, I.J.; Reich, P.B.; Westoby, M.; Ackerly, D.D.; Baruch, Z.; Bongers, F.; Cavender-Bares, J.; Chapin, T.; Cornelissen, J.H.; Diemer, M. The worldwide leaf economics spectrum. Nature 2004, 428, 821–827. [Google Scholar] [CrossRef]
- Pérez-Harguindeguy, N.; Díaz, S.; Garnier, E.; Lavorel, S.; Poorter, H.; Jaureguiberry, P.; Bret-Harte, M.S.; Cornwell, W.K.; Craine, J.M.; Gurvich, D.E.; et al. New handbook for standardised measurement of plant functional traits worldwide. Aust. J. Bot. 2013, 61, 167–234. [Google Scholar] [CrossRef]
- Wood, T.E.; Lawrence, D.; Wells, J.A. Inter-specific variation in foliar nutrients and resorption of nine canopy-tree species in a secondary neotropical rain forest. Biotropica 2011, 43, 544–551. [Google Scholar] [CrossRef]
- He, M.; Yan, Z.; Cui, X.; Gong, Y.; Li, K.; Han, W. Scaling the leaf nutrient resorption efficiency: Nitrogen vs phosphorus in global plants. Sci. Total Environ. 2020, 729, 138920. [Google Scholar] [CrossRef]
- Yan, T.; Zhu, J.; Yang, K. Leaf nitrogen and phosphorus resorption of woody species in response to climatic conditions and soil nutrients: A meta-analysis. J. For. Res. 2017, 29, 905–913. [Google Scholar] [CrossRef]
- Jiang, D.; Li, Q.; Geng, Q.; Zhang, M.; Xu, C.; Hu, G.; Shen, C.; Ruan, H.; Xu, X.; Ye, Q. Nutrient resorption and stoichiometric responses of poplar (Populus deltoids) plantations to N addition in a coastal region of eastern China. J. Plant Ecol. 2021, 14, 591–604. [Google Scholar] [CrossRef]
- Wu, Z.; Raven, P.H.; Hong, D. Flora of China; Sciencen Press: Beijing, China, 2008. [Google Scholar]
- Xiao, J.H.; Ding, X.; Li, L.; Ma, H.; Ci, X.Q.; Merwe, M.; Conran, J.G.; Li, J. Miocene diversification of a golden-thread nanmu tree species (Phoebe zhennan, Lauraceae) around the Sichuan Basin shaped by the East Asian monsoon. Ecol. Evol. 2020, 10, 10543–10557. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.F.; Koo, H.I.; Dell’Angelo, J.; D’Odorico, P.; Estes, L.; Kehoe, L.J.; Kharratzadeh, M.; Kuemmerle, T.; Machava, D.; Pais, A.d.J.R.; et al. Tropical forest loss enhanced by large-scale land acquisitions. Nat. Geosci. 2020, 13, 482–488. [Google Scholar] [CrossRef]
- Hasan, M.E.; Zhang, L.; Dewan, A.; Guo, H.; Mahmood, R. Spatiotemporal pattern of forest degradation and loss of ecosystem function associated with Rohingya influx: A geospatial approach. Land Degrad. Dev. 2020, 32, 3666–3683. [Google Scholar] [CrossRef]
- Qin, Y.; Xiao, X.; Wigneron, J.P.; Ciais, P.; Brandt, M.; Fan, L.; Li, X.; Crowell, S.; Wu, X.; Doughty, R.; et al. Carbon loss from forest degradation exceeds that from deforestation in the Brazilian Amazon. Nat. Clim. Chang. 2021, 11, 442–448. [Google Scholar] [CrossRef]
- Hao, J.; Wang, D.; Li, Y.; Yao, X.; Zhang, Y.; Zhan, M.; Qi, J. Effects of human disturbance on species diversity of Phoebe zhennan communitis in Jinfengshan Moutain in western Sichuan. Acta Ecol. Sini. 2014, 34, 6930–6942. [Google Scholar] [CrossRef]
- Zheng, J.; Liu, X.; Gao, R.; Yang, Z.; Yang, Y. Carbon storage and allocation in the Phoebe bournei plantations in Nanping, Fujian Province. J. Sub. Resour. Environ. 2009, 4, 59–65. [Google Scholar] [CrossRef]
- Parkinson, J.A.; Allen, S.E. A wet oxidation procedure suitable for the determination of nitrogen and mineral nutrients in biological material. Commun. Soil Sci. Plant Anal. 1975, 6, 1–11. [Google Scholar] [CrossRef]
- Van Heerwaarden, L.M.; Toet, S.; Aerts, R. Current measures of nutrient resorption efficiency lead to a substantial underestimation of real resorption efficiency: Facts and solutions. Oikos 2003, 101, 664–669. [Google Scholar] [CrossRef]
- Warton, D.I.; Duursma, R.A.; Falster, D.S.; Taskinen, S. Smatr 3-an R package for estimation and inference about allometric lines. Methods Ecol. Evol. 2012, 3, 257–259. [Google Scholar] [CrossRef]
- National Soil Census Office. Soil and Flora of China (Volumes 1 to 6); China Agricultural Press: Beijing, China, 1996.
- Sayer, E.J.; Banin, L.F. Tree nutrient status and nutrient cycling in tropical forest—Lessons from fertilization experiments. In Tropical Tree Physiology; Goldstein, G., Santiag, L., Eds.; Springer: Cham, Switzerland, 2016; Volume 6, pp. 275–297. [Google Scholar] [CrossRef]
- Mo, Q.; Li, Z.; Sayer, E.J.; Lambers, H.; Li, Y.; Zou, B.I.; Tang, J.; Heskel, M.; Ding, Y.; Wang, F. Foliar phosphorus fractions reveal how tropical plants maintain photosynthetic rates despite low soil phosphorus availability. Funct. Ecol. 2019, 33, 503–513. [Google Scholar] [CrossRef] [Green Version]
- van Breugel, M.; Craven, D.; Lai, H.R.; Baillon, M.; Turner, B.L.; Hall, J.S. Soil nutrients and dispersal limitation shape compositional variation in secondary tropical forests across multiple scales. J. Ecol. 2019, 107, 566–581. [Google Scholar] [CrossRef]
- Koerselman, W.; Meuleman, A.F. The vegetation N: P ratio: A new tool to detect the nature of nutrient limitation. J. Appl. Ecol. 1996, 33, 1441–1450. [Google Scholar] [CrossRef]
- Chapin, F.S.I.; Matson, P.A.; Mooney, H.A. Principles of Terrestrial Ecosystem Ecology; Springer: New York, NY, USA, 2002. [Google Scholar]
- He, X.; Augusto, L.; Goll, D.S.; Ringeval, B.; Wang, Y.; Helfenstein, J.; Huang, Y.; Yu, K.; Wang, Z.; Yang, Y.; et al. Global patterns and drivers of soil total phosphorus concentration. Earth Syst. Sci. Data 2021, 13, 5831–5846. [Google Scholar] [CrossRef]
- Sun, J.; Gao, P.; Li, C.; Wang, R.; Niu, X.; Wang, B. Ecological stoichiometry characteristics of the leaf–litter–soil continuum of Quercus acutissima Carr. and Pinus densiflora Sieb. in Northern China. Environ. Earth Sci. 2019, 78, 20. [Google Scholar] [CrossRef]
- Zhao, Y.; Liang, C.; Shao, S.; Chen, J.; Qin, H.; Xu, Q. Linkages of litter and soil C:N:P stoichiometry with soil microbial resource limitation and community structure in a subtropical broadleaf forest invaded by Moso bamboo. Plant Soil 2021, 465, 473–490. [Google Scholar] [CrossRef]
- Killingbeck, K.T. Nutrient Resorption. In Plant Cell Death Processes; Academic Press: Cambridge, MA, USA, 2004. [Google Scholar] [CrossRef]
- Kobe, R.K.; Lepczyk, C.A.; Iyer, M. Resorption efficiency decreases with increasing green leaf nutrients in a global data set. Ecology 2005, 86, 2780–2792. [Google Scholar] [CrossRef]
- Han, W.; Tang, L.; Chen, Y.; Fang, J. Relationship between the relative limitation and resorption efficiency of nitrogen vs phosphorus in woody plants. PLoS ONE 2013, 8, e83366. [Google Scholar] [CrossRef]
- Lü, X.T.; Reed, S.C.; Yu, Q.; Han, X.G. Nutrient resorption helps drive intra-specific coupling of foliar nitrogen and phosphorus under nutrient-enriched conditions. Plant Soil 2015, 398, 111–120. [Google Scholar] [CrossRef]
- Gerdol, R.; Iacumin, P.; Brancaleoni, L.; Wang, F. Differential effects of soil chemistry on the foliar resorption of nitrogen and phosphorus across altitudinal gradients. Funct. Ecol. 2019, 33, 1351–1361. [Google Scholar] [CrossRef]
- Akram, M.A.; Zhang, Y.; Wang, X.; Shrestha, N.; Malik, K.; Khan, I.; Ma, W.; Sun, Y.; Li, F.; Ran, J.; et al. Phylogenetic independence in the variations in leaf functional traits among different plant life forms in an arid environment. J. Plant Physiol. 2022, 272, 153671. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Luo, Y.; Yan, Z.; Chen, J.; Eziz, A.; Li, K.; Han, W. Resorptions of 10 mineral elements in leaves of desert shrubs and their contrasting responses to aridity. J. Plant Ecol. 2019, 12, 358–366. [Google Scholar] [CrossRef]
- Rodrigo, R.; Pettit, J.L.; Matula, R.; Kozák, D.; Bače, R.; Pavlin, J.; Janda, P.; Mikoláš, M.; Nagel, T.A.; Schurman, J.; et al. Historical mixed-severity disturbances shape current diameter distributions of primary temperate Norway spruce mountain forests in Europe. For. Ecol. Manag. 2022, 503, 119772. [Google Scholar] [CrossRef]
| Compartment | Disturbance Type | C (mg/g) | N (mg/g) | P (mg/g) | K (mg/g) |
|---|---|---|---|---|---|
| Green leaf | Mild | 541.60 ± 4.25 a | 16.85 ± 0.29 a | 1.40 ± 0.06 b | 10.81 ± 1.27 a |
| Severe | 549.17 ± 4.31 a | 17.26 ± 0.28 a | 1.93 ± 0.05 a | 12.94 ± 0.46 a | |
| Senesced leaf | Mild | 501.71 ± 7.01 a | 14.83 ± 0.24 b | 0.79 ± 0.02 b | 1.19 ± 0.08 a |
| Severe | 494.57 ± 7.53 a | 15.73 ± 0.26 a | 0.88 ± 0.02 a | 1.40 ± 0.09 a | |
| Soil (0–20 cm) | Mild | 23.73 ± 1.43 a | 1.45 ± 0.11 b | 0.89 ± 0.04 b | 28.35 ± 1.05 b |
| Severe | 24.55 ± 1.21 a | 1.78 ± 0.11 a | 1.06 ± 0.05 a | 42.08 ± 1.44 a | |
| Soil (20–40 cm) | Mild | 16.51 ± 1.06 a | 0.92 ± 0.08 b | 0.86 ± 0.04 b | 29.49 ± 1.11 b |
| Severe | 17.88 ± 1.28 a | 1.35 ± 0.11 a | 1.00 ± 0.05 a | 44.60 ± 1.80 a |
| Estimate | Standard Error | df | t | p | |
|---|---|---|---|---|---|
| Intercept | 0.93 | 0.11 | 35 | 8.63 | <0.001 |
| Green leaf N content | −0.04 | 0.01 | 35 | −5.88 | <0.001 |
| Intercept | 0.95 | 0.11 | 35 | 8.48 | <0.001 |
| Green leaf P content | −0.42 | 0.11 | 35 | −3.79 | <0.001 |
| Intercept | 0.98 | 0.03 | 35 | 28.52 | <0.001 |
| Green leaf K content | −0.06 | 0.02 | 35 | −2.62 | 0.01 |
| Property | Mild Disturbance | Severe Disturbance | ||
|---|---|---|---|---|
| Principal Components | Principal Components | |||
| 1 | 2 | 1 | 2 | |
| Green leaf N | −0.32 | −0.16 | 0.10 | 0.06 |
| Green leaf P | −0.28 | −0.41 | 0.34 | 0.23 |
| Green leaf K | 0.26 | −0.41 | 0.41 | 0.21 |
| NRE | −0.29 | 0.03 | 0.21 | 0.16 |
| PRE | −0.29 | −0.16 | 0.37 | 0.35 |
| KRE | 0.15 | −0.46 | 0.24 | 0.36 |
| LDMC | 0.17 | −0.09 | −0.14 | 0.30 |
| SLA | −0.23 | −0.27 | 0.26 | −0.20 |
| LT | 0.22 | 0.36 | 0.02 | 0.41 |
| Soil N | −0.43 | 0.08 | −0.37 | 0.29 |
| Soil P | −0.28 | 0.28 | −0.27 | 0.36 |
| Soil SOM | −0.40 | 0.15 | −0.35 | 0.31 |
| Soil K | 0.07 | 0.30 | 0.22 | −0.11 |
| Percent % | 30.6 | 18.3 | 27.5 | 24.4 |
| Cumulative percent % | 30.6 | 48.9 | 27.5 | 51.9 |
| Leaf N | Leaf P | Leaf K | NRE | PRE | KRE | LDMC | SLA | LT | Soil N | Soil P | Soil SOM | Soil K | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leaf N | −0.04 | 0.16 | 0.67 ** | 0.02 | 0.05 | −0.07 | 0.33 | −0.06 | 0.02 | 0.22 | 0.10 | 0.27 | |
| Leaf P | 0.38 | 0.51 * | 0.15 | 0.81 ** | 0.58 ** | 0.04 | 0.19 | 0.12 | −0.14 | −0.13 | −0.09 | 0.20 | |
| Leaf K | −0.23 | 0.14 | 0.44 * | 0.74 ** | 0.47 * | −0.14 | 0.35 | 0.38 | −0.30 | −0.07 | −0.27 | 0.27 | |
| NRE | 0.68 ** | 0.25 | −0.42 | 0.34 | 0.12 | −0.03 | 0.17 | 0.17 | −0.14 | 0.10 | −0.03 | −0.06 | |
| PRE | 0.14 | 0.70 ** | 0.04 | 0.32 | 0.72 ** | 0.08 | 0.08 | 0.45 * | −0.18 | −0.02 | −0.11 | 0.06 | |
| KRE | 0.06 | 0.27 | 0.68 ** | −0.26 | −0.03 | 0.31 | −0.03 | 0.34 | −0.03 | 0.11 | 0.04 | −0.02 | |
| LDMC | −0.31 | 0.16 | 0.42 | −0.06 | 0.40 | 0.10 | −0.69 ** | 0.44 * | 0.25 | 0.20 | 0.21 | −0.05 | |
| SLA | 0.48 | 0.40 | −0.13 | −0.01 | −0.14 | 0.06 | −0.63 ** | −0.42 | −0.25 | −0.19 | −0.23 | 0.23 | |
| LT | −0.47 | −0.31 | −0.02 | −0.29 | −0.15 | −0.14 | 0.40 | −0.58 * | 0.24 | 0.39 | 0.17 | −0.06 | |
| Soil N | 0.29 | 0.44 | −0.34 | 0.33 | 0.66 ** | −0.29 | −0.19 | 0.19 | −0.16 | 0.77 ** | 0.91 ** | −0.25 | |
| Soil P | 0.09 | 0.21 | −0.46 | −0.01 | 0.27 | −0.31 | −0.21 | 0.17 | 0.16 | 0.60 * | 0.80 ** | −0.21 | |
| Soil SOM | 0.38 | 0.25 | −0.34 | 0.32 | 0.57 * | −0.28 | −0.17 | 0.11 | −0.19 | 0.87 *** | 0.58 * | −0.41 | |
| Soil K | 0.13 | −0.37 | 0.02 | 0.14 | −0.26 | 0.02 | −0.13 | −0.34 | 0.31 | −0.14 | 0.11 | 0.09 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, D.; Peng, S.; Wang, J.; Hui, D. Responses of Nutrient Resorption to Human Disturbances in Phoebe bournei Forests. Forests 2022, 13, 905. https://doi.org/10.3390/f13060905
Zhu D, Peng S, Wang J, Hui D. Responses of Nutrient Resorption to Human Disturbances in Phoebe bournei Forests. Forests. 2022; 13(6):905. https://doi.org/10.3390/f13060905
Chicago/Turabian StyleZhu, Dehuang, Suhong Peng, Jinyan Wang, and Dafeng Hui. 2022. "Responses of Nutrient Resorption to Human Disturbances in Phoebe bournei Forests" Forests 13, no. 6: 905. https://doi.org/10.3390/f13060905






