Long Exposure to Salt Stress in Jatropha curcas Leads to Stronger Damage to the Chloroplast Ultrastructure and Its Functionality Than the Stomatal Function
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Environmental Conditions
2.2. Optical Anatomy
2.3. Scanning Electron Microscopy
2.4. Transmission Electron Microscopy
2.5. Experimental Design and Statistical Analysis
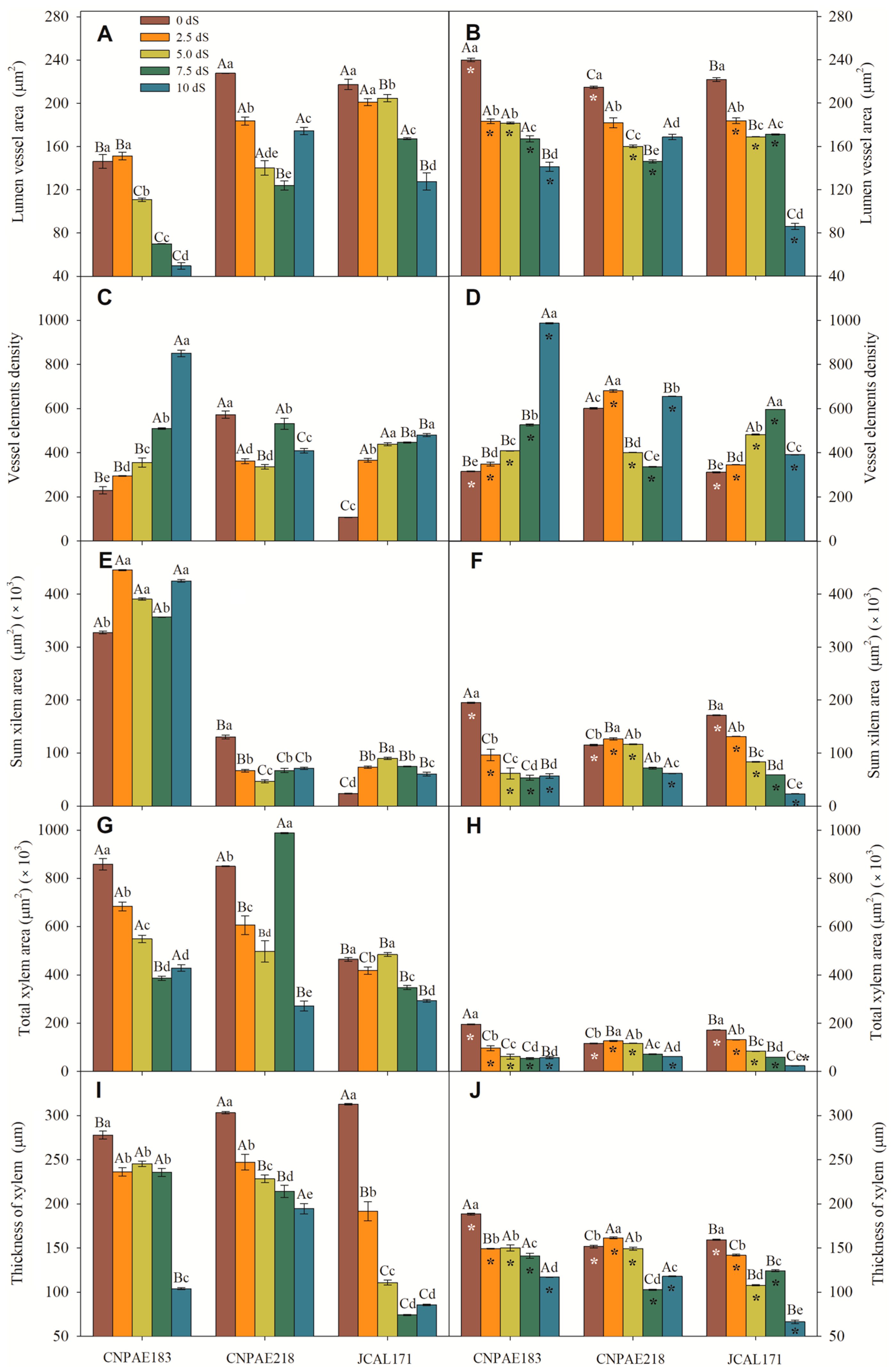
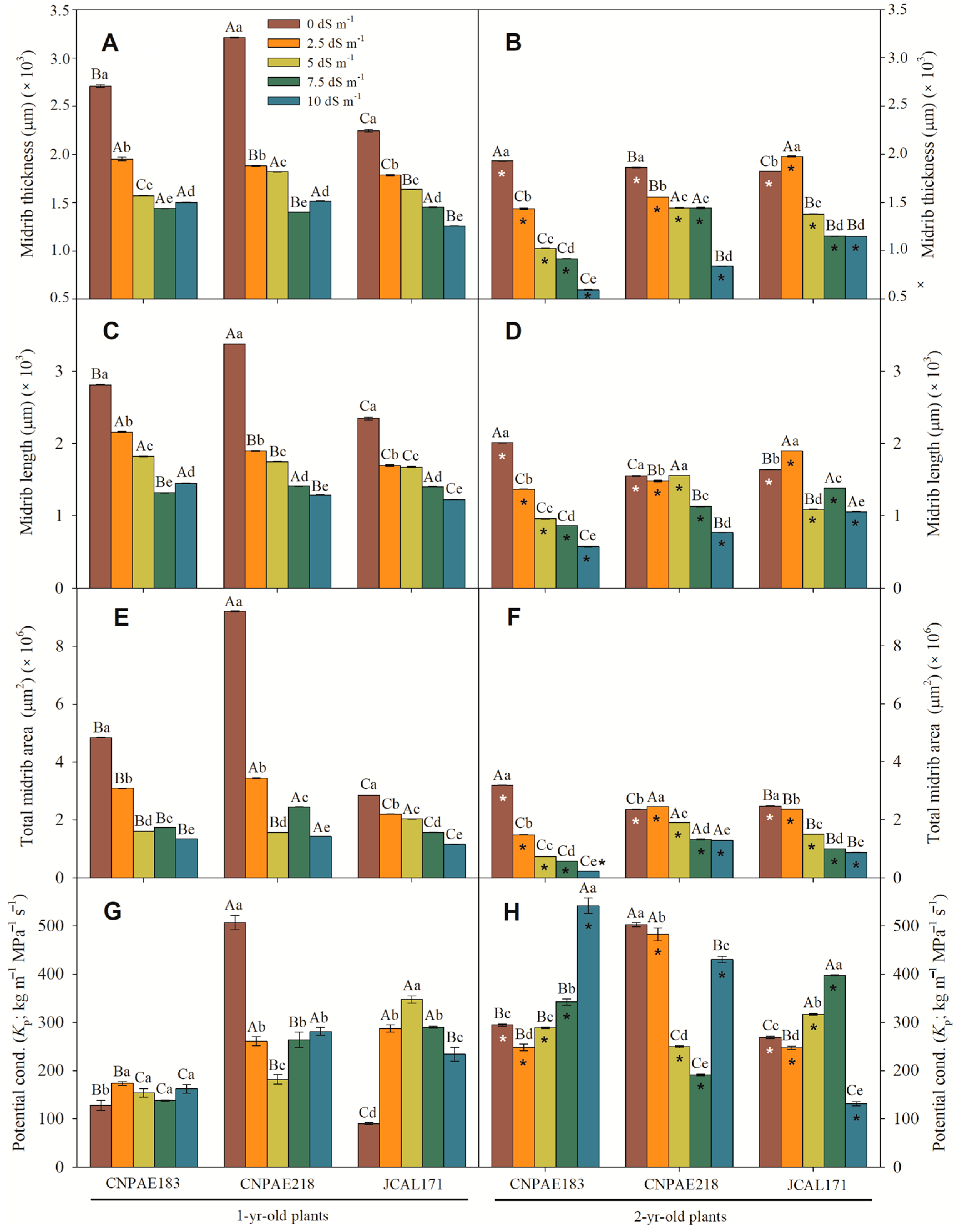
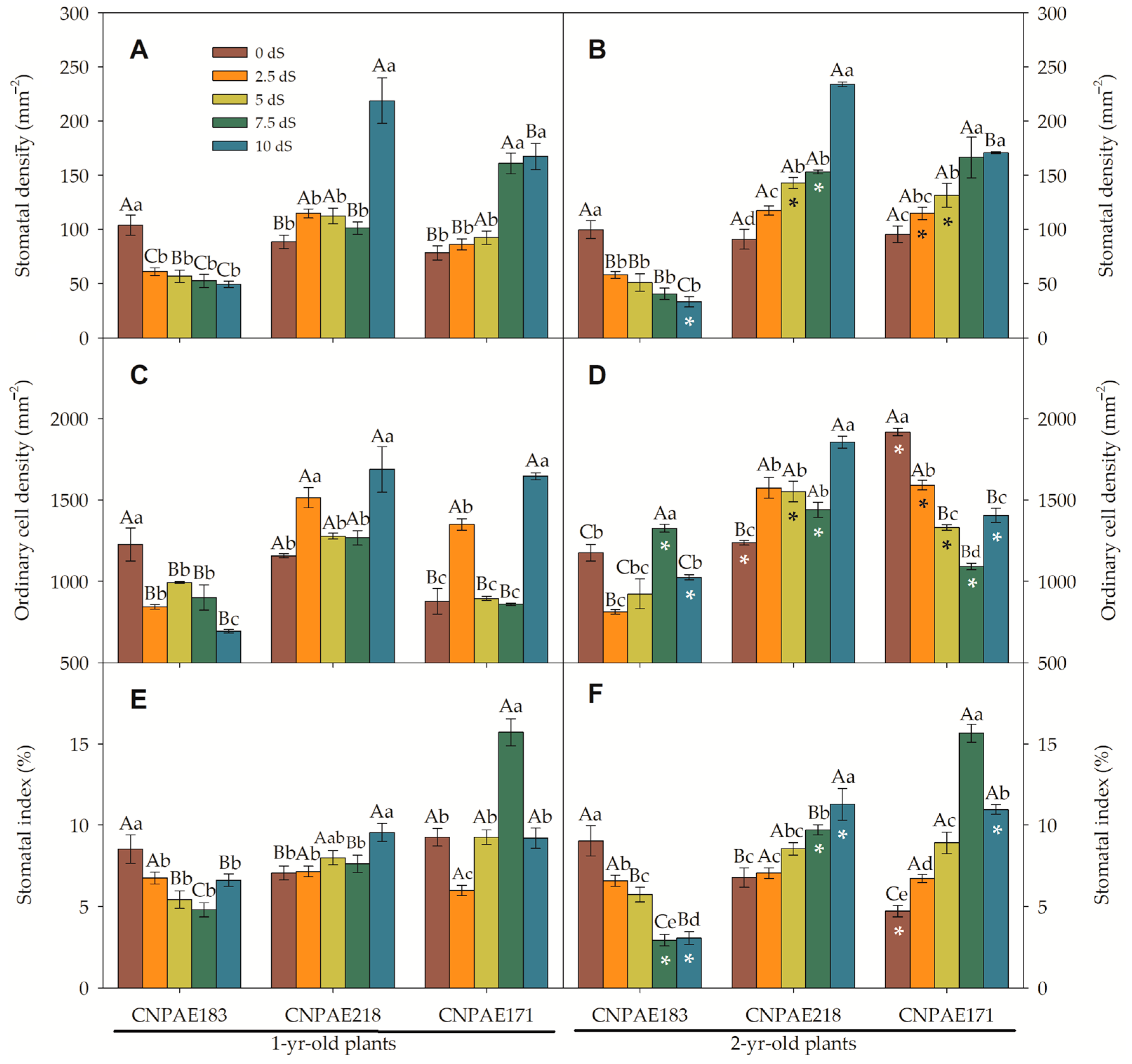
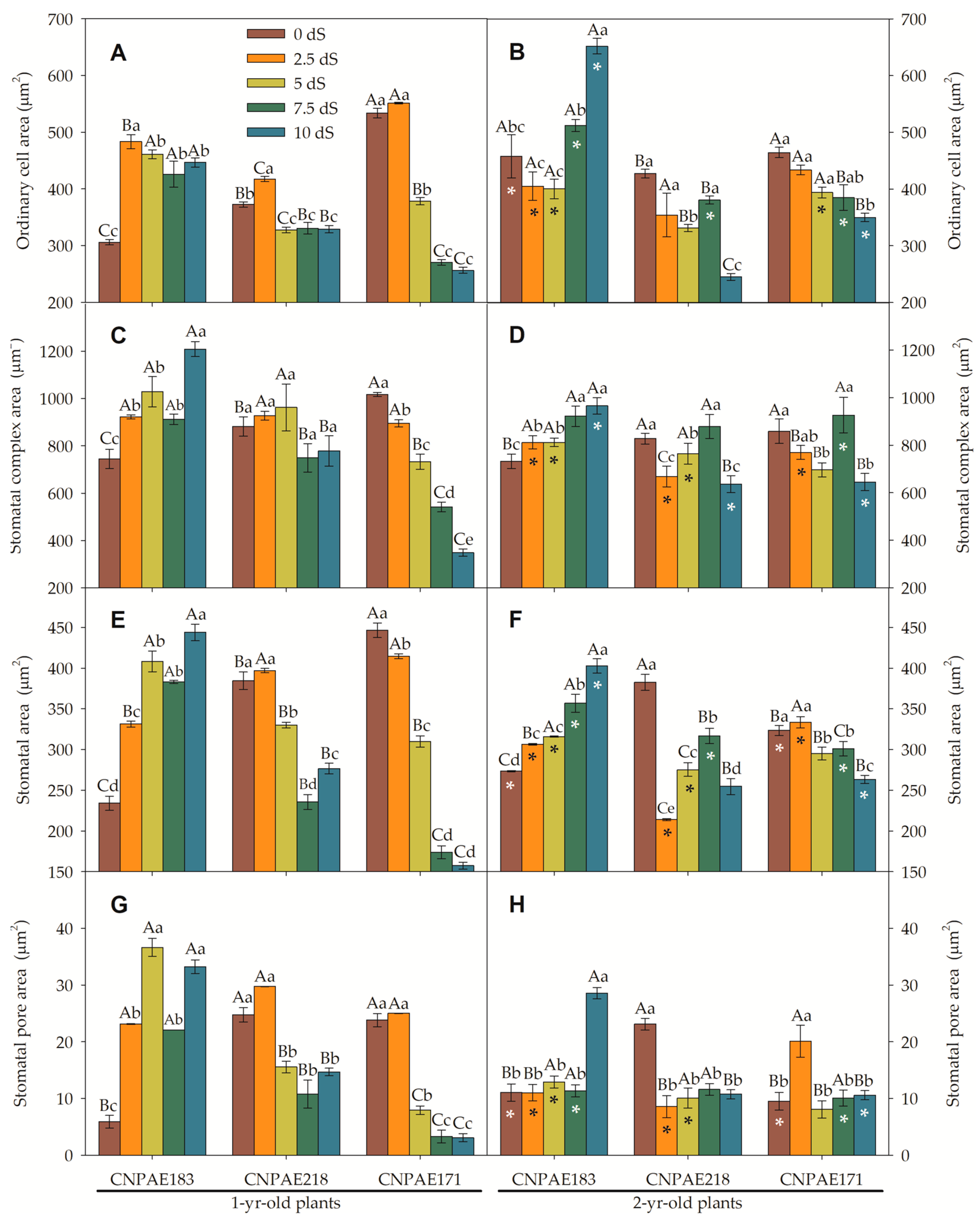
3. Results
3.1. Cross Section of Leaves of J. curcas Plants under Salt Stress
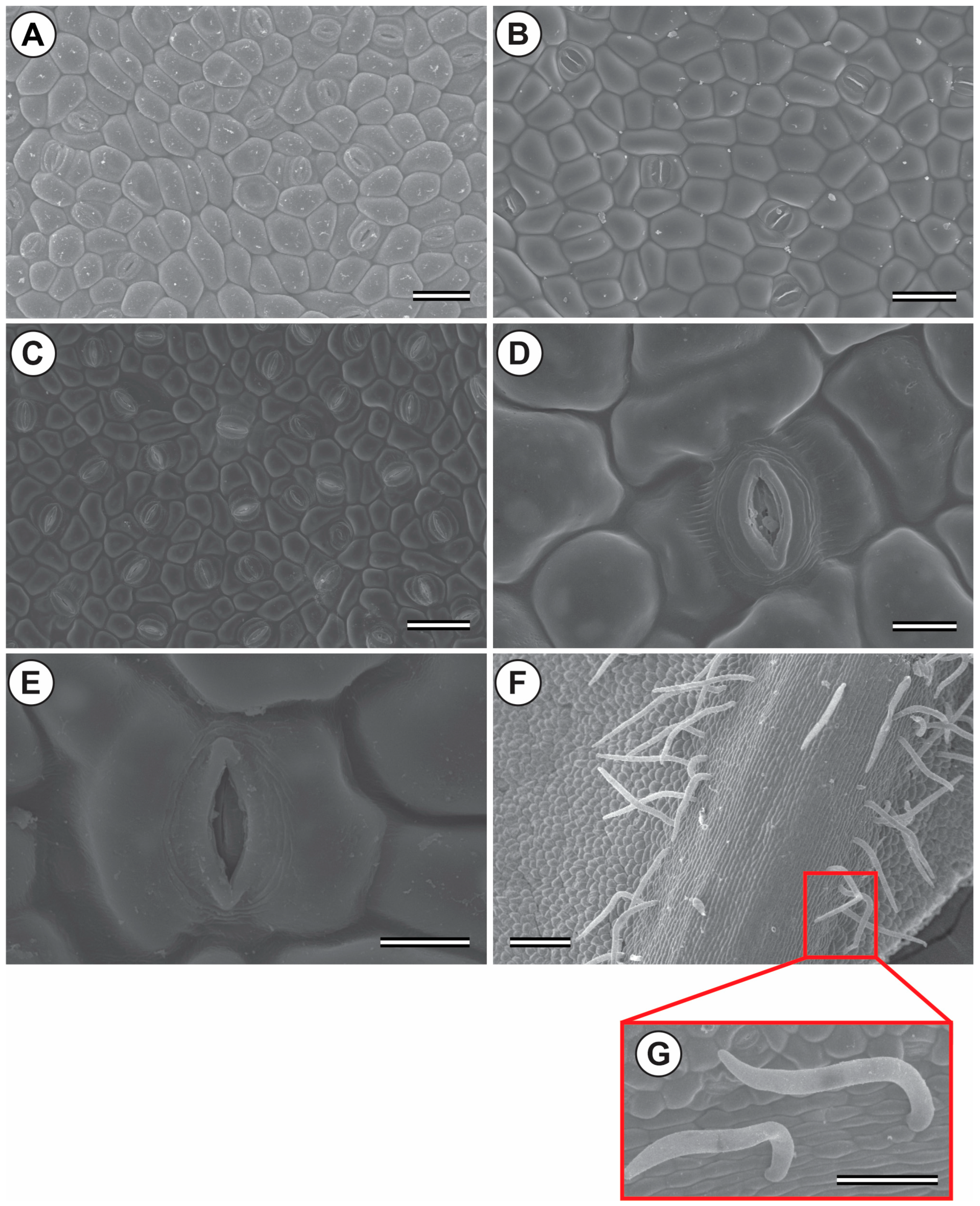
3.2. Scanning Electron Microscopy of the Epidermis (SEM)
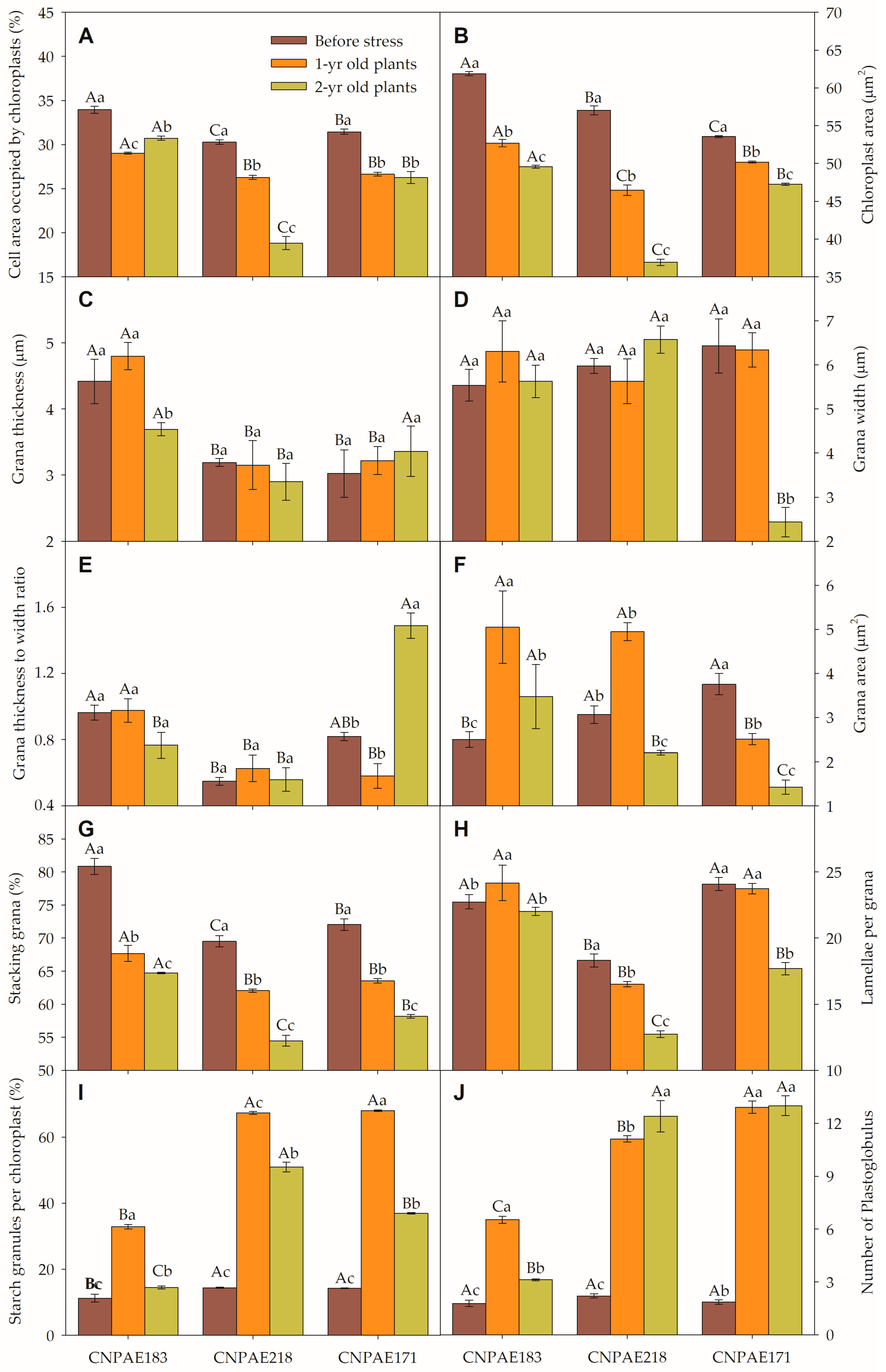
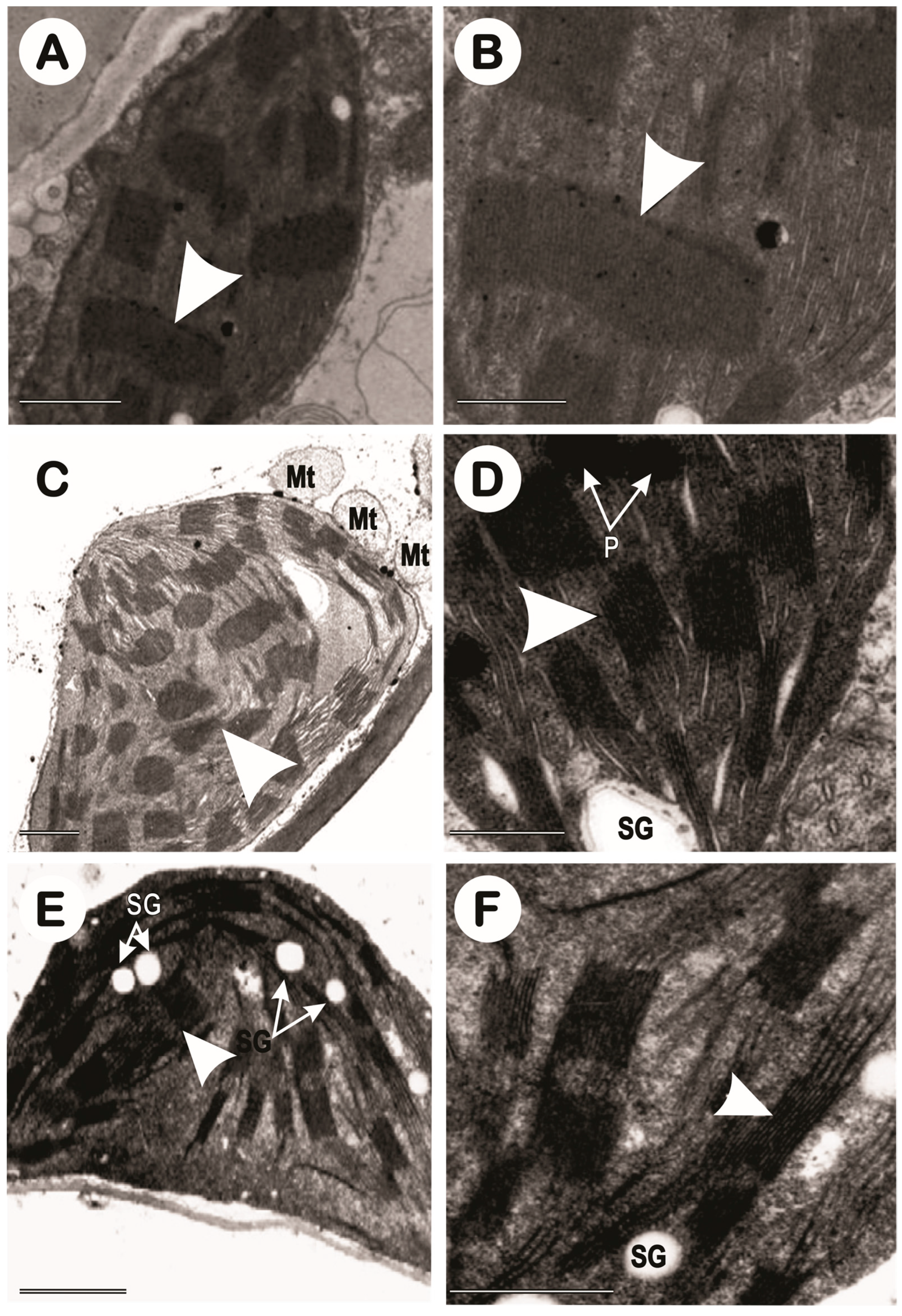
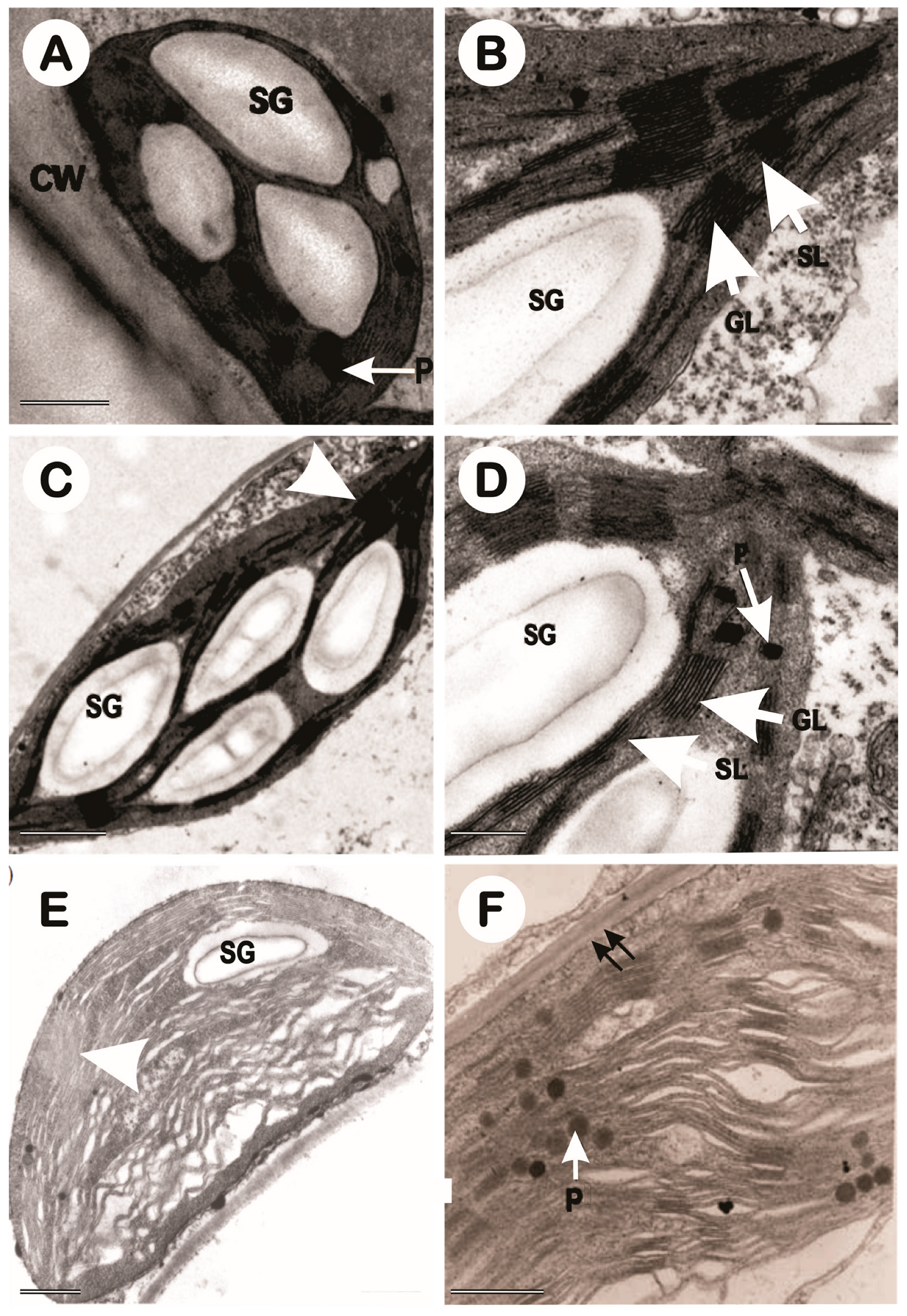
3.3. Transmission Electron Microscopy of Chloroplasts (TEM)
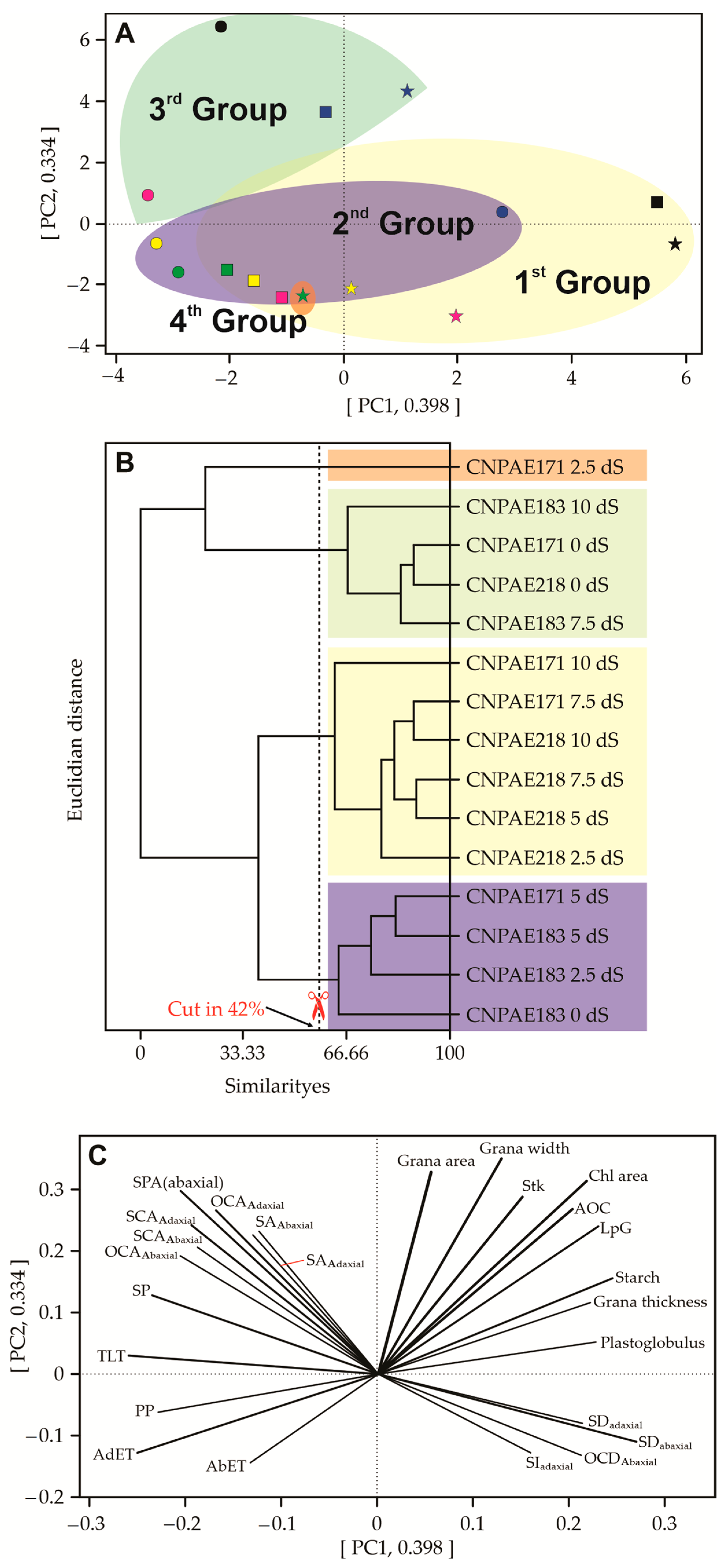
3.4. Principal Component Analysis (PCA)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Achten, W.M.J.; Maes, W.H.; Reubens, B.; Mathijs, E.; Singh, V.P.; Verchot, L.; Muys, B. Biomass production and allocation in Jatropha curcas L. seedlings under different levels of drought stress. Biomass Bioenergy 2010, 34, 667–676. [Google Scholar] [CrossRef]
- Díaz-López, L.; Gimeno, V.; Simón, I.; Martínez, V.; Rodrígues-Ortega, W.M.; García-Sánchez, F. Jatropha curcas seedlings show a water conservation strategy under drought conditions based on decreasing leaf growth and stomatal conductance. Agric. Water Manag. 2012, 105, 48–56. [Google Scholar] [CrossRef]
- Pompelli, M.F.; Barata-Luís, R.M.; Vitorino, H.S.; Gonçalves, E.R.; Rolim, E.V.; Santos, M.G.; Almeida-Cortez, J.S.; Endres, L. Photosynthesis, photoprotection and antioxidant activity of purging nut under drought deficit and recovery. Biomass Bioenergy 2010, 34, 1207–1215. [Google Scholar] [CrossRef]
- Silva, L.D.; Gomes, F.P.; Oliveira, P.S.; Almeida, F.R.; Pirovani, C.P.; Laviola, B.G.; Amaral, J.F.T. Plasticity of photosynthetic metabolism in Jatropha curcas genotypes under water stress. Gen. Mol. Res. 2019, 18, gmr18228. [Google Scholar] [CrossRef]
- Cabrales-Rodríguez, R.; Betancur-Hurtado, C.A.; Rodríguez-Páez, L.A. Cultivo del Piñón (Jatropha curcas L.); Manejo Nutricional y Usos en Córdoba, Colombia; Universidad de Córdoba: Montería, Colombia, 2019; p. 88. [Google Scholar]
- Bezerra-Neto, E.; Coelho, J.B.M.; Jarma-Orozco, A.; Rodríguez-Páez, L.A.; Pompelli, M.F. Modulation of photosynthesis under salinity and the role of mineral nutrients in Jatropha curcas L. J. Agron. Crop Sci. 2021, 208, 314–334. [Google Scholar] [CrossRef]
- Cabral, G.A.L.; Binneck, E.; Souza, M.C.P.; Silva, M.D.; Ferreira Neto, J.R.C.; Pompelli, M.F.; Endres, L.; Kido, E.A. First expressed TFome of physic nut (Jatropha curcas L.) after salt stimulus. Plant Mol. Biol. Rep. 2020, 38, 189–208. [Google Scholar] [CrossRef]
- Campos, M.L.O.; Hsie, B.S.; Granja, J.A.A.; Correia, R.M.; Silva, S.R.S.; Almeida-Cortez, J.S.; Pompelli, M.F. Photosynthesis and antioxidant activity mechanisms in Jatropha curcas L. under salt stress. Braz. J. Plant Physiol. 2012, 24, 55–67. [Google Scholar] [CrossRef]
- Dorta-Santos, M.A.; Barriola, I.; Wassner, D.F.; Ploschuk, E.L. Photosynthesis, fluorescence and mesophyll conductance responses to increasing salinity levels in Jatropha curcas at early vegetative stages. J. Agron. Crop Sci. 2020, 206, 52–63. [Google Scholar] [CrossRef]
- Gadelha, C.G.; Miranda, R.S.; Alencar, N.L.M.; Costa, J.H.; Prisco, J.T.; Gomes-Filho, E. Exogenous nitric oxide improves salt tolerance during establishment of Jatropha curcas seedlings by ameliorating oxidative damage and toxic ion accumulation. J. Plant Physiol. 2017, 212, 69–79. [Google Scholar] [CrossRef]
- Reubens, B.; Achten, W.M.J.; Maes, W.H.; Danjon, F.; Aerts, R.; Poesen, J.; Muys, B. More than biofuel? Jatropha curcas root system symmetry and potential for soil erosion control. J. Arid Environ. 2011, 75, 201–205. [Google Scholar] [CrossRef]
- Fairless, D. The little shrub that could—Maybe. Nature 2007, 449, 652–655. [Google Scholar] [CrossRef] [PubMed]
- Arockiasamy, S.; Kumpatla, J.; Hadole, S.; Yepuri, V.; Patil, M.; Shrivastava, V.; Rao, C.; Kancharla, N.; Jalali, S.; Varshney, A.; et al. Breeding and biotechnological efforts in Jatropha curcas L. for sustainable yields. Oil Crop Sci. 2021, 6, 180–191. [Google Scholar] [CrossRef]
- Pompelli, M.F.; Ferreira, P.P.B.; Chaves, A.R.M.; Figueiredo, R.C.Q.Q.; Martins, A.O.; Jarma-Orozco, A.; Batista-Silva, W.; Endres, L.; Araújo, W.L. Physiological, metabolic, and stomatal adjustments in response to salt stress in Jatropha curcas. Plant Physiol. Biochem. 2021, 168, 116–127. [Google Scholar] [CrossRef]
- Silva-Santos, L.; Corte-Real, N.; Dias-Pereira, J.; Figueiredo, R.C.B.Q.; Endres, L.; Pompelli, M.F. Salinity shock in Jatropha curcas leaves is more pronounced during recovery than during stress time. Braz. J. Dev. 2019, 5, 11359–11369. [Google Scholar] [CrossRef]
- Acosta-Motos, J.R.; Ortuño, M.F.; Bernal-Vicente, A.; Diaz-Vivancos, P.; Sanchez-Blanco, M.J.; Hernandez, J.A. Plant responses to salt stress: Adaptive mechanisms. Agronomy 2017, 7, 18. [Google Scholar] [CrossRef]
- Melo, G.M.; da Cunha, P.C.; Pereira, J.A.F.; Willadino, L.; Ulisses, C. Anatomical changes in the leaves and roots of Jatropha curcas L. cultivated under saline stress. Rev. Ciência Agric. 2011, 42, 670–674. [Google Scholar] [CrossRef]
- Ashraf, M.; Harris, P. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Arcoverde, G.B.; Rodrigues, B.M.; Pompelli, M.F.; Santos, M.G. Water relations and some aspects of leaf metabolism of Jatropha curcas young plants under two water deficit levels and recovery. Braz. J. Plant Physiol. 2011, 23, 123–130. [Google Scholar] [CrossRef]
- Corte-Real, N.; Miranda, P.V.V.C.; Endres, L.; Souza, E.R.; Pompelli, M.F. Tolerance to salinity in Jatropha curcas are genotype-dependent. Braz. J. Dev. 2019, 5, 22169–22199. [Google Scholar] [CrossRef]
- Gomes, M.F.C.; Binneck, E.; Ferreira-Neto, J.R.C.; Luz, G.A.; Benko-Iseppon, A.M.; Pompelli, M.F.E.L.; Kido, É.A. Transcriptome analysis uncovers the Jatropha curcas L. transportome after NaCl (150 mM) exposure. Physiol. Plant 2021, in press. [Google Scholar]
- Pompelli, M.F.; Jarma-Orozco, A.; Rodrígues-Páez, L.A. Salinity in Jatropha curcas: A review of physiological, biochemical, and molecular factors involved. Agriculture 2022, 12, 594. [Google Scholar] [CrossRef]
- Melcher, P.J.; Holbrook, N.M.; Burns, M.J.; Zwieniecki, M.A.; Cobb, A.R.; Brodribb, T.J.; Sack, L. Measurements of stem xylem hydraulic conductivity in the laboratory and field. Methods Ecol. Evol. 2012, 3, 685–694. [Google Scholar] [CrossRef]
- Oliveira, P.S.; Pereira, L.S.; Silva, D.C.; Souza Júnior, J.O.; Laviola, B.G.; Gomes, F.P. Hydraulic conductivity in stem of young plants of Jatropha curcas L. cultivated under irrigated or water deficit conditions. Ind. Crops Prod. 2018, 116, 15–23. [Google Scholar] [CrossRef]
- Fernández-Marín, B.; Gulías, J.; Figueroa, C.M.; Iñiguez, C.; Clemente-Moreno, M.J.; Nunes-Nesi, A.; Fernie, A.R.; Cavieres, L.A.; Bravo, L.A.; García-Plazaola, J.I.; et al. How do vascular plants perform photosynthesis in extremeenvironments? An integrative ecophysiological andbiochemical story. Plant J. 2020, 101, 979–1000. [Google Scholar] [CrossRef] [PubMed]
- Gauhl, E.; Björkman, O. Simultaneous measurements on the effect of oxygen concentration on water vapor and carbon dioxide exchange in leaves. Planta 1969, 88, 187–191. [Google Scholar] [CrossRef]
- Lin, J.; Li, J.P.; Yuan, F.; Yang, Z.; Wang, B.S.; Chen, M. Transcriptome profiling of genes involved in photosynthesis in Elaeagnus angustifolia L. under salt stress. Photosynthetica 2018, 56, 998–1009. [Google Scholar] [CrossRef]
- Sivakumar, P.; Sharmila, P.; Pardha-Saradhi, P. Proline alleviates salt-stress-induced enhancement in Ribulose-1,5-bisphosphate oxygenase activity. Biochem. Biophys. Res. Commun. 2000, 279, 512–515. [Google Scholar] [CrossRef]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2016, 90, 856–867. [Google Scholar] [CrossRef]
- Muchate, M.S.; Nikalje, G.C.; Rajurkar, N.S.; Suprasanna, P.; Nikam, T.D. Plant salt stress: Adaptive responses, tolerance mechanism and bioengineering for salt tolerance. Bot Rev. 2016, 82, 371–406. [Google Scholar] [CrossRef]
- El-Banna, M.F.; Abdelaal, K.A.A. Response of strawberry plants grown in the hydroponic system to pretreatment with H2O2 before exposure to salinity stress. J. Plant Prod. 2018, 9, 989–1001. [Google Scholar] [CrossRef]
- Bongi, G.; Loreto, F. Gas-exchange properties of saltstressed olive (Olea europea L.) leaves. Plant Physiol. 1989, 90, 1408–1416. [Google Scholar] [CrossRef] [PubMed]
- Terletskaya, N.; Kurmanbayeva, M. Change in leaf anatomical parameters of seedlings of different wheat species under conditions of drought and salt stress. Pak. J. Bot 2017, 49, 857–865. [Google Scholar]
- Toscano, S.; Ferrante, A.; Tribulato, A.; Romano, D. Leaf physiological and anatomical responses of Lantana and Ligustrum species under different water availability. Plant Physiol. Biochem. 2018, 127, 380–392. [Google Scholar] [CrossRef]
- Akcin, T.A.; Akcin, A.; Yalcin, E. Anatomical adaptations to salinity in Spergularia marina (Caryophyllaceae) from Turkey. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2014, 85, 625–634. [Google Scholar] [CrossRef]
- Trebst, A.; Tsujimoto, H.; Arnon, D. Separation of light and dark phases in the photosynthesis of isolated chloroplasts. Nature 1958, 182, 351–355. [Google Scholar] [CrossRef]
- Buchanan, B.B.; Wolosiuk, R.A. Photosynthesis: The carbon reactions. In Plant Physiology, 5th ed.; Taiz, L., Zeiger, E., Eds.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2010; pp. 199–242. [Google Scholar]
- Atabayeva, S.; Nurmahanova, A.; Minocha, S.; Ahmetova, A.; Kenzhebayeva, S.; Aidosova, S.; Nurzhanova, A.; Zhardamalieva, A.; Asrandina, S.; Alybayeva, R.; et al. The effect of salinity on growth and anatomical attributes of barley seedling (Hordeum vulgare L.). Afr. J. Biotechnol. 2013, 12, 2366–2377. [Google Scholar]
- Souza, M.C.P.; Silva, M.D.; Binneck, E.; Cabral, G.A.L.; Iseppon, A.M.B.; Pompelli, M.F.; Endres, L.; Kido, E.A. RNA-Seq transcriptome analysis of Jatropha curcas L. accessions after salt stimulus and unigene-derived microsatellite mining. Ind. Crops Prod. 2020, 147, 112168. [Google Scholar] [CrossRef]
- Mendes, K.R.; Batista-Silva, W.; Dias-Pereira, J.; Pereira, M.P.S.; Souza, E.V.; Serrão, J.E.; Granja, J.A.A.; Pereira, E.C.; Gallacher, D.J.; Mutti, P.R.; et al. Leaf plasticity across wet and dry seasons in Croton blanchetianus (Euphorbiaceae) at a tropical dry forest. Sci. Rep. 2022, 12, 954. [Google Scholar] [CrossRef]
- Sterck, F.J.; Zweifel, R.; Sass-Klaassen, U.; Chowdhury, Q. Persisting soil drought reduces leaf specific conductivity in Scots pine (Pinus sylvestris) and pubescent oak (Quercus pubescens). Tree Physiol. 2008, 28, 529–536. [Google Scholar] [CrossRef]
- Scholz, A.; Klepsch, M.; Karimi, Z.; Jansen, S. How to quantify conduits in wood? Front. Plant Sci. 2013, 4, 56. [Google Scholar] [CrossRef]
- Karnovsky, M.J. A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J. Cell Biol. 1965, 27, 137–138. [Google Scholar]
- Pompelli, M.F.; Martins, S.C.; Celin, E.F.; Ventrella, M.C.; DaMatta, F.M. What is the influence of ordinary epidermal cells and stomata on the leaf plasticity of coffee plants grown under full-sun and shady conditions? Braz. J. Biol. 2010, 70, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.K.; Huang, C.F. Stochastic flow network reliability with tolerable error rate. Qual. Technol. Quant. Manag. 2013, 10, 57–73. [Google Scholar] [CrossRef]
- Hsie, B.S.; Mendes, K.R.; Antunes, W.C.; Endres, L.; Campos, M.L.O.; Souza, F.C.; Santos, N.D.; Singh, B.; Arruda, E.C.P.; Pompelli, M.F. Jatropha curcas L. (Euphorbiaceae) modulates stomatal traits in response to leaf-to-air vapor pressure deficit. Biomass Bioenergy 2015, 81, 273–281. [Google Scholar] [CrossRef]
- Leigh, A.; Sevanto, S.; Close, J.D.; Nicotra, A.B. The influence of leaf size and shape on leaf thermal dynamics: Does theory hold up under natural conditions? Plant Cell Environ. 2016, 40, 237–248. [Google Scholar] [CrossRef]
- Pompelli, M.F.; Mendes, K.R.; Ramos, M.V.; Santos, J.N.B.; Youssef, D.T.A.; Pereira, J.D.; Endres, L.; Jarma-Orozco, A.; Solano-Gomes, R.; Jarma-Arroyo, B.; et al. Mesophyll thickness and sclerophylly among Calotropis procera morphotypes reveal water-saved adaptation to environments. J. Arid Land 2019, 11, 795–810. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Barrón, E.; Camarero, J.J.; Vilagrosa, A.; Gil-Pelegrín, E. Morphological and physiological divergences within Quercus ilex support the existence of different ecotypes depending on climatic dryness. Ann. Bot. 2014, 114, 301–313. [Google Scholar] [CrossRef]
- Gil-Pelegrín, E.; Saz, M.A.; Cuadrat, J.M.; Peguero-Pina, J.J.; Sancho-Knapik, D. Oaks Under Mediterranean-Type Climates: Functional Response to Summer Aridity. In Oaks Physiological Ecology. Exploring the Functional Diversity of Genus quercus L.; Gil-Pelegrín, E., Peguero-Pina, J.J., Sancho-Knapik, D., Eds.; Springer: London, UK, 2017; pp. 137–193. [Google Scholar]
- Taratima, W.; Samattha, C.; Maneerattanarungroj, P.; Trunjaruen, A. Physiological and anatomical response of rice (Oryza sativa L.) ‘Hom Mali Daeng’ at different salinity stress levels. Acta Agrobot. 2023, 76, 764. [Google Scholar] [CrossRef]
- Ola, H.; Elbar, A.; Farag, E.; Eisa, S.S.; Habib, S.A. Morpho-anatomical changes in salt stressed kallar grass (Leptochloa fusca L. Kunth). J. Agric. Biol. Sci. 2012, 8, 158–166. [Google Scholar]
- Barhoumi, Z.; Djebali, W.; Chaïbi, W.; Abdelly, C.; Smaoui, A. Salt impact on photosynthesis and leaf ultrastructure of Aeluropus littoralis. J. Plant Res. 2007, 120, 529–537. [Google Scholar] [CrossRef]
- Bastias, E.; Gonzales-Moro, M.B.; Gonzales-Murua, C. Zea mays L. amylacea from the Lluta Valley (Arica Chile) tolerates salinity stress when high levels of boron are available. Plant Soil. 2005, 267, 73–84. [Google Scholar] [CrossRef]
- Clemente-Moreno, M.J.; Gago, J.; Díaz-Vivancos, P.; Bernal, A.; Miedes, E.; Bresta, P.; Liakopoulos, G.; Fernie, A.R.; Hernández, J.A.; Flexas, J. The apoplastic antioxidant system and altered cell wall dynamics influence mesophyll conductance and the rate of photosynthesis. Plant J. 2019, 99, 1031–1046. [Google Scholar] [CrossRef] [PubMed]
- Taratima, W.; Ritmaha, T.; Jongrungklang, N.; Maneerattanarungroj, P.; Kunpratum, N. Effect of stress on the leaf anatomy of sugarcane cultivars with different drought tolerance (Saccharum officinarum, Poaceae). Rev. Biol. Trop. 2020, 68, 1159–1170. [Google Scholar] [CrossRef]
- Cavalcante, P.G.S.; Santos, C.M.; Filho, H.C.L.W.; Avelino, J.R.L.; Endres, L. Morpho-physiological adaptation of Jatropha curcas L. to salinity stress. Aust. J. Crop Sci. 2018, 12, 563–571. [Google Scholar] [CrossRef]
- Masrahi, Y.; Al-Namazi, A.; Alammari, B.; Alturki, T. Adaptations facilitate the invasion of Cylindropuntia rosea (DC.) Backeb. (Cactaceae) in the highlands of southwestern Saudi Arabia. Plant Signal. Behav. 2022, 17, 2144593. [Google Scholar] [CrossRef]
- Hunsche, M.; Bürling, K.; Saied, A.S.; Schmitz-Eiberger, M.; Sohail, M.; Gebauer, J.; Noga, G.; Buerkert, A. Effects of NaCl on surface properties, chlorophyll fluorescence and light remission, and cellular compounds of Grewia tenax (Forssk.) Fiori and Tamarindus indica L. leaves. Plant Growth Regul. 2010, 61, 253–263. [Google Scholar] [CrossRef]
- Tsuda, M.; Tyree, M.T. Plant hydraulic conductance measured by the high pressure flow meter in crop plants. J. Exp. Bot. 2000, 51, 823–828. [Google Scholar] [CrossRef]
- Poorter, L.; McDonald, I.; Alarcón, A.; Fichtler, E.; Licona, J.-C.; Peña-Claros, M.; Sterck, F.; Villegas, Z.; Sass-Klaassen, U. The importance of wood traits and hydraulic conductance for the performance and life history strategies of 42 rainforest tree species. New Phytol. 2010, 185, 481–492. [Google Scholar] [CrossRef]
- Dodd, I.C. Abscisic acid and stomatal closure: A hydraulic conductance conundrum? New Phytol. 2013, 197, 6–8. [Google Scholar] [CrossRef]
- Meinzer, F.; Grantz, D. Coordination of stomatal, hydraulic, and canopy boundary layer properties: Do stomata balance conductances by measuring transpiration? Physiol. Plant 1991, 83, 324–329. [Google Scholar] [CrossRef]
- North, G.B.; Brinton, E.K.; Browne, M.G.; Gillman, M.G.; Roddy, A.B.; Kho, T.L.; Wang, E.; Fung, V.A.; Brodersen, C.R. Hydraulic conductance, resistance, and resilience: How leaves of a tropical epiphyte respond to drought. Am. J. Bot. 2019, 106, 943–957. [Google Scholar] [CrossRef] [PubMed]
- Ladjal, M.; Huc, R.; Ducrey, M. Drought effects on hydraulic conductivity and xylem vulnerability to embolism in diverse species and provenances of Mediterranean cedars. Tree Physiol. 2005, 25, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Linton, M.J.; Sperry, J.S.; Williams, D.G. Limits to water transport in Juniperus osteosperma and Pinus edulis: Implications for drought tolerance and regulation of transpiration. Funct. Ecol. 1998, 12, 906–911. [Google Scholar] [CrossRef]
- Hacke, U.G.; Sperry, J.S.; Pittermann, J. Drought experience and cavitation resistance in six shrubs from the Great Basin, Utah. Basic Appl. Ecol. 2000, 1, 31–41. [Google Scholar] [CrossRef]
- Santana, T.A.; Oliveira, P.S.; Silva, L.D.; Laviola, B.G.F.; de Almeida, A.-A.; Gomes, F.P. Water use efficiency and consumption in different Brazilian genotypes of Jatropha curcas L. subjected to soil water deficit. Biomass Bioenergy 2015, 75, 119–125. [Google Scholar] [CrossRef]
- Franks, P.J.; Gibson, A.; Bachelard, E.P. Xylem permeability and embolism susceptibility in seedlings of Eucalyptus camaldulensis Dehnh. from two different climatic zones. Aust. J. Plant Physiol. 1995, 22, 15–21. [Google Scholar] [CrossRef]
- Tyree, M. Dynamic measurements of root hydraulic conductance using a high pressure flowmeter in the laboratory and field. J. Exp. Bot. 1995, 46, 83–94. [Google Scholar] [CrossRef]
- Charrier, G.; Torres-Ruiz, J.M.; Badel, E.; Burlett, R.; Choat, B.; Cochard, H.; Delmas, C.E.L.; Domec, J.C.; Jansen, S.; King, A.; et al. Evidence for hydraulic vulnerability segmentation and lack of xylem refilling under tension. Plant Physiol. 2016, 172, 1657–1668. [Google Scholar] [CrossRef]
- Xu, T.; Zhang, L.; Li, Z. Computational fluid dynamics model and flow resistance characteristics of Jatropha curcas L xylem vessel. Sci. Rep. 2020, 10, 14728. [Google Scholar] [CrossRef]
- Neves, E.L.; Funch, L.S.; Viana, B.F. Comportamento fenológico de três espécies de Jatropha (Euphorbiaceae) da Caatinga, semi-árido do Brasil. Braz. J. Bot. 2010, 33, 155–166. [Google Scholar] [CrossRef]
- Krishnamurthy, L.; Zaman-Allah, M.; Marimuthu, S.; Wani, S.P.; Kesava Rao, A.V.R. Root growth in Jatropha and its implications for drought adaptation. Biomass Bioenergy 2012, 39, 247–252. [Google Scholar] [CrossRef]
- Tavecchio, N.; Reinoso, H.; Ruffini Castiglione, M.; Spanò, C.; Pedranzani, H.E. Anatomical studies of two Jatropha species with importance for biodiesel production. J. Agric. Sci. 2016, 8, 84–94. [Google Scholar] [CrossRef]
- Lei, Z.Y.; Han, J.M.; Yi, X.P.; Zhang, W.F.; Zhang, Y.L. Coordinated variation between veins and stomata in cotton and its relationship with water-use efficiency under drought stress. Photosynthetica 2018, 56, 1326–1335. [Google Scholar] [CrossRef]
- Drake, P.L.; Froend, R.H.; Franks, P.J. Smaller, faster stomata: Scaling of stomatal size, rate of response, and stomatal conductance. J. Exp. Bot. 2013, 64, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Carins, M.R.; Jordan, G.J.; Brodribb, T.J. Differential leaf expansion can enable hydraulic acclimation to sun and shade. Plant Cell Environ. 2012, 35, 1407–1418. [Google Scholar] [CrossRef]
- Hameed, M.; Ashraf, M.; Naz, N.; Al-Qurainy, F. Anatomical adaptations of Cynodon dactylon (L.) Pers. from the salt range Pakistan to salinity stress. I. Root and stem anatomy. Pak. J. Bot. 2010, 42, 279–289. [Google Scholar]
- Franks, P.J.; Drake, P.L.; Beerling, D.J. Plasticity in maximum stomatal conductance constrained by negative correlation between stomatal size and density: An analysis using Eucalyptus globulus. Plant Cell Environ. 2009, 32, 1737–1748. [Google Scholar] [CrossRef]
- Rahman, A.A.N.S.; Rahman, M.; Shimanto, M.H.; Kibria, M.G.; Islam, M.A. Stomatal size and density trade-off varies with leaf phenology and species shade tolerance in a South Asian moist tropical forest. Funct. Plant Biol. 2022, 49, 307–318. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, G. Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J. Exp. Bot. 2008, 59, 3317–3325. [Google Scholar] [CrossRef]
- Zhang, S.B.; Guan, Z.J.; Sun, M.; Zhang, J.-J.; Cao, K.-F.; Hu, H. Evolutionary association of stomatal traits with leaf vein density in Paphiopedilum, Orchidaceae. PLoS ONE 2012, 7, e40080. [Google Scholar] [CrossRef]
- Dunford, S. Translocation in the Phloem. In Plant Physiology, 5th ed.; Taiz, L., Zeiger, E., Eds.; Sinauer Associates Inc.: Sunderland, MA, USA, 2010; pp. 271–303. [Google Scholar]
- DaMatta, F.M.; Cunha, R.L.; Antunes, W.C.; Martins, S.C.V.; Araujo, W.L.; Fernie, A.R.; Moraes, G.A.B.K. In field-grown coffee trees source-sink manipulation alters photosynthetic rates, independently of carbon metabolism, via alterations in stomatal function. New Phytol. 2008, 178, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Rao, I.M.; Merchant, A. Source-sink relationships in crop plants and their influence on yield development and nutritional quality. Front. Plant Sci. 2018, 9, 1889. [Google Scholar] [CrossRef] [PubMed]
- Zhai, L.; Yan, A.; Shao, K.; Wang, S.; Wang, Y.; Chen, Z.-H.; Xu, J. Large vascular bundle phloem area 4 enhances grain yield and quality in rice via source–sink–flow. Plant Physiol. 2023, 191, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Haibo, D.; Zhang, W.; Hua, B.; Zhu, Z.; Zhang, J.; Zhang, Z. Cucumber STACHYOSE SYNTHASE is regulated by its cis-antisense RNA asCsSTS to balance source–sink carbon partitioning. Plant Cell 2023, 35, 435–452. [Google Scholar] [CrossRef]
- Miehe, W.; Czempik, L.; Klebl, F.; Lohaus, G. Sugar concentrations and expression of SUTs suggest active phloem loading in tall trees of Fagus sylvatica and Quercus robur. Tree Physiol. 2023, 43, 805–816. [Google Scholar] [CrossRef]
- Chen, L.; Ganguly, D.R.; Shafik, S.H.; Danila, F.; Grof, C.P.L.; Sharwood, R.E.; Furbank, R.T. The role of SWEET4 proteins in the post-phloem sugar transport pathway of Setaria viridis sink tissues. J. Exp. Bot. 2023, 74, 2968–2986. [Google Scholar] [CrossRef]
- Taylor, A.O.; Craig, A.S. Plants under climatic stress: II. low temperature, high light effects on chloroplast ultrastructure. Plant Physiol. 1971, 47, 719–725. [Google Scholar] [CrossRef]
- Ackerson, R.C.; Hebert, E.R. Osmoregulation in cotton in response to water stress: I. alterations in photosynthesis, leaf conductance, translocation, and ultrastructure. Plant Physiol. 1981, 67, 484–488. [Google Scholar] [CrossRef]
- Capellades, M.; Lemeur, R.; Debergh, P. Effects of sucrose on starch accumulation and rate of photosynthesis in rosa cultured in vitro. Plant Cell Tissue Organ Cult. 1991, 25, 21–26. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, H.; Han, Y.; Cheng, Z.; Lv, Q.; Pompelli, M.F.; Pereira, J.D.; Araújo, W.L. Long Exposure to Salt Stress in Jatropha curcas Leads to Stronger Damage to the Chloroplast Ultrastructure and Its Functionality Than the Stomatal Function. Forests 2023, 14, 1868. https://doi.org/10.3390/f14091868
Cao H, Han Y, Cheng Z, Lv Q, Pompelli MF, Pereira JD, Araújo WL. Long Exposure to Salt Stress in Jatropha curcas Leads to Stronger Damage to the Chloroplast Ultrastructure and Its Functionality Than the Stomatal Function. Forests. 2023; 14(9):1868. https://doi.org/10.3390/f14091868
Chicago/Turabian StyleCao, Huijuan, Yongguang Han, Ziyi Cheng, Qian Lv, Marcelo F. Pompelli, Jaqueline Dias Pereira, and Wagner L. Araújo. 2023. "Long Exposure to Salt Stress in Jatropha curcas Leads to Stronger Damage to the Chloroplast Ultrastructure and Its Functionality Than the Stomatal Function" Forests 14, no. 9: 1868. https://doi.org/10.3390/f14091868






