Minocycline Inhibits Tick-Borne Encephalitis Virus and Protects Infected Cells via Multiple Pathways
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture and Virus Propagation
2.2. Preparation of Compounds
2.3. Cytotoxicity of Compounds
2.4. Measurement of Virus Titers
2.5. Indirect Immunofluorescence (IFA) Assay
2.6. Western Blots
2.7. Real-Time Reverse Transcription PCR (RT-qPCR)
2.8. Transcriptome Sequencing
2.9. Detection of the Inflammatory Factor IL-6
2.10. Statistical Analysis
3. Result
3.1. Evaluation of the Safety and Efficacy of Tetracycline Antibiotics in Vero Cells
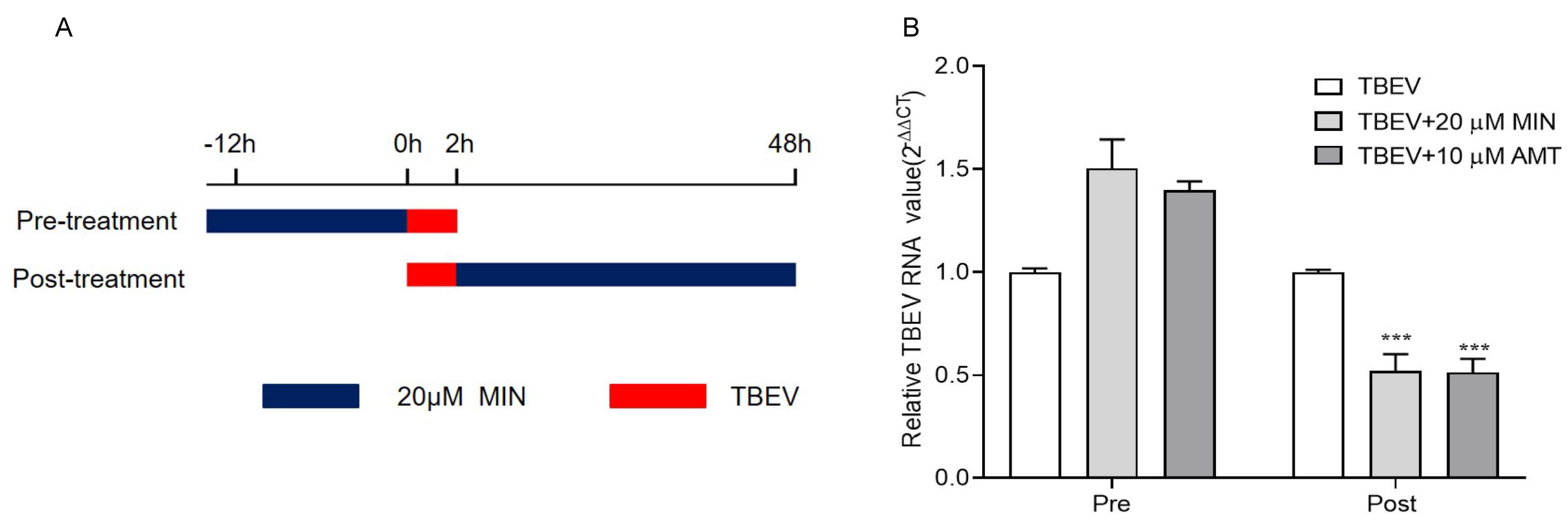
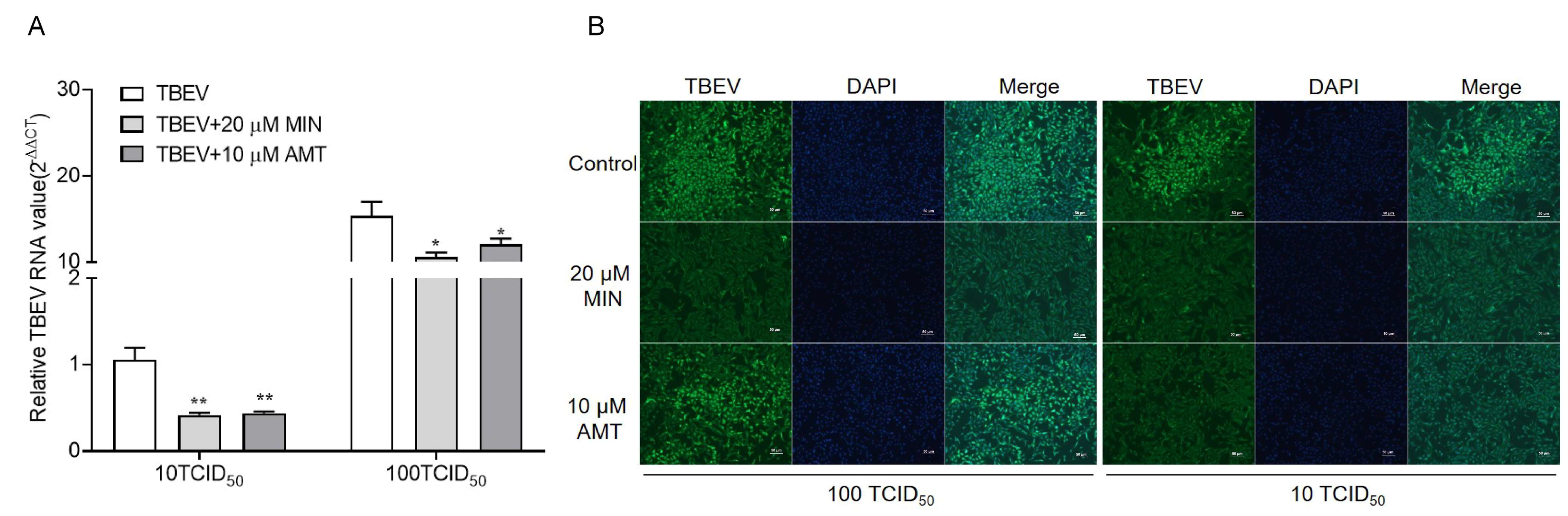
3.2. MIN Inhibits TBEV Replication in Vero Cells
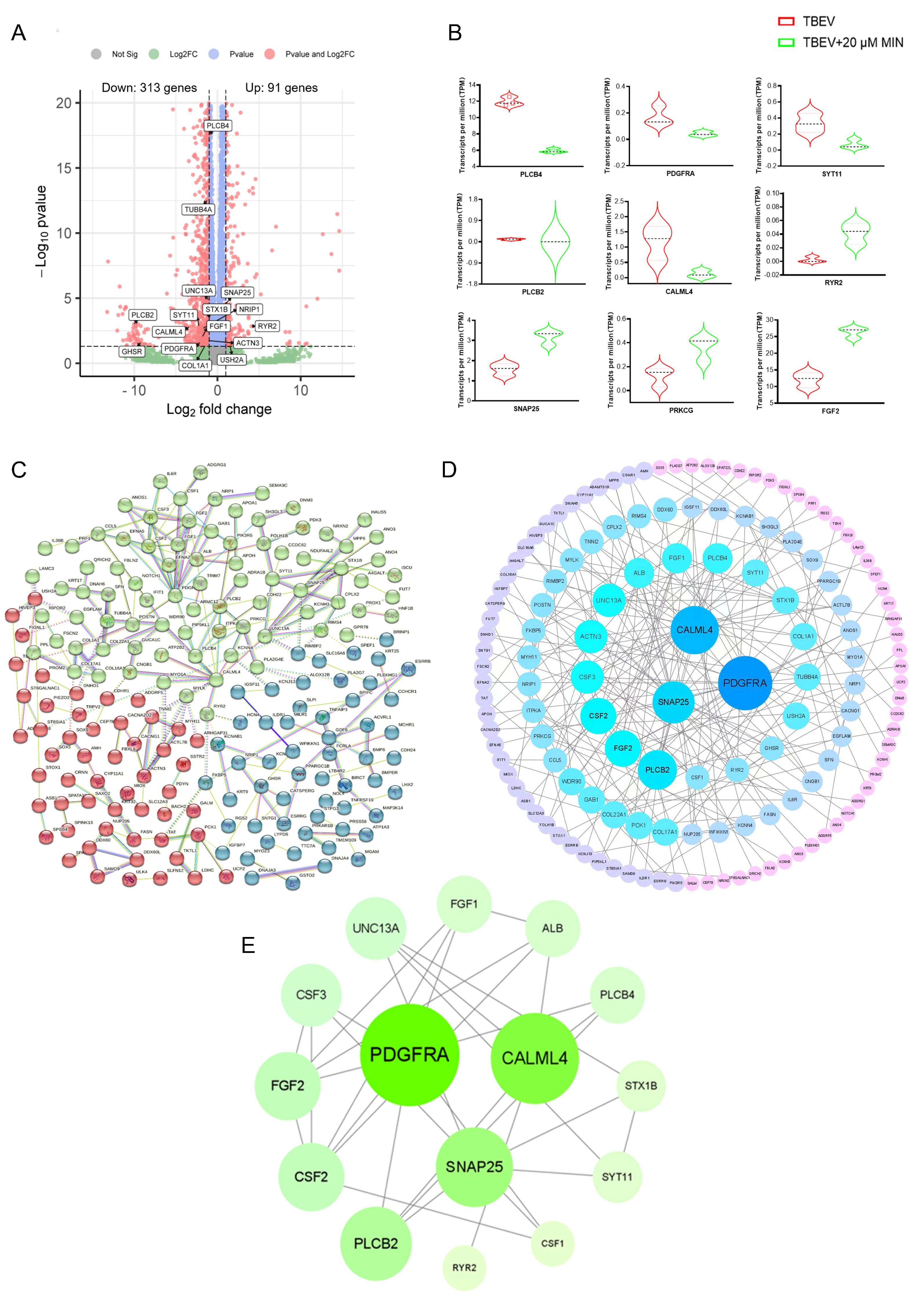
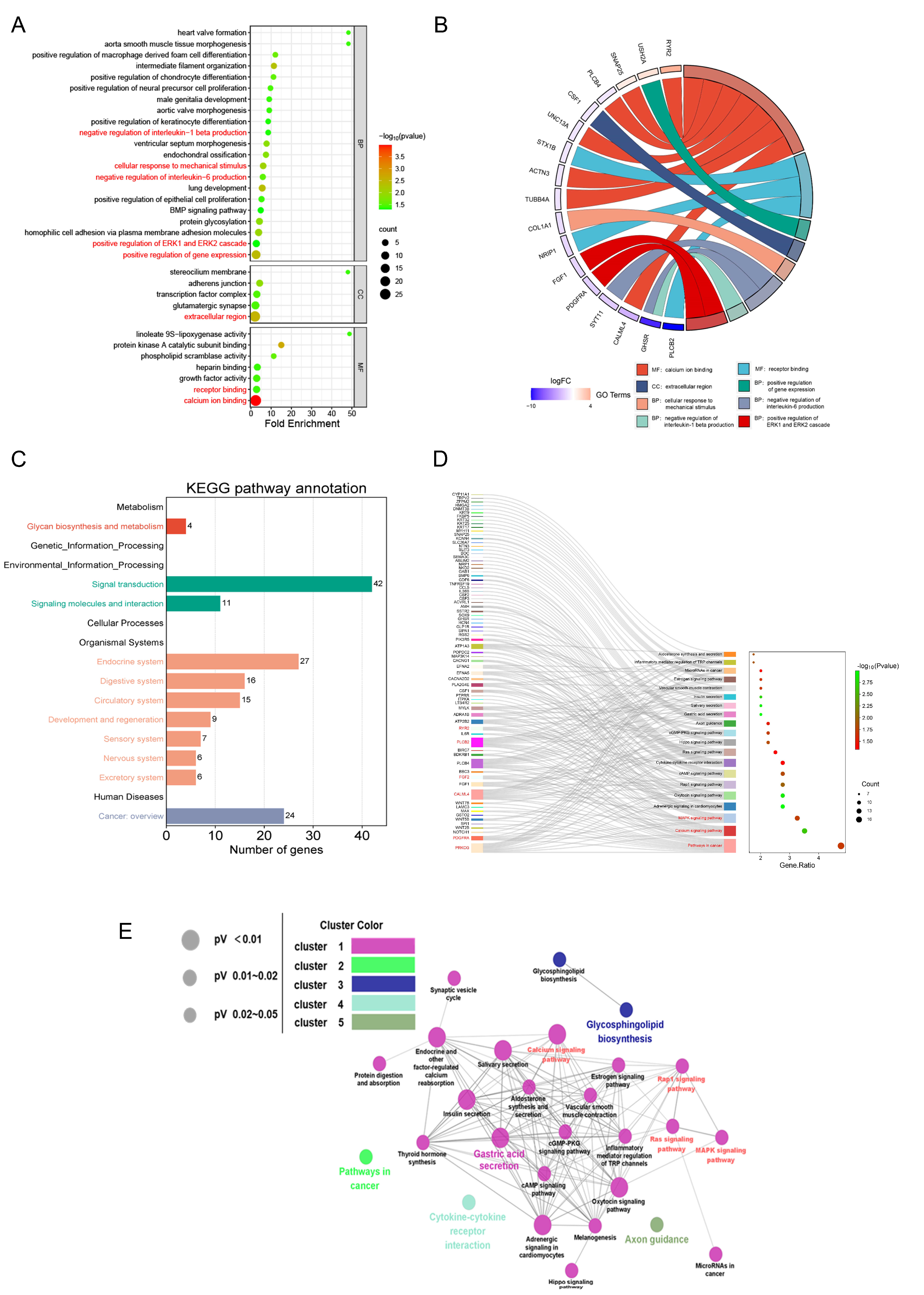
3.3. Transcriptomic Analysis Results
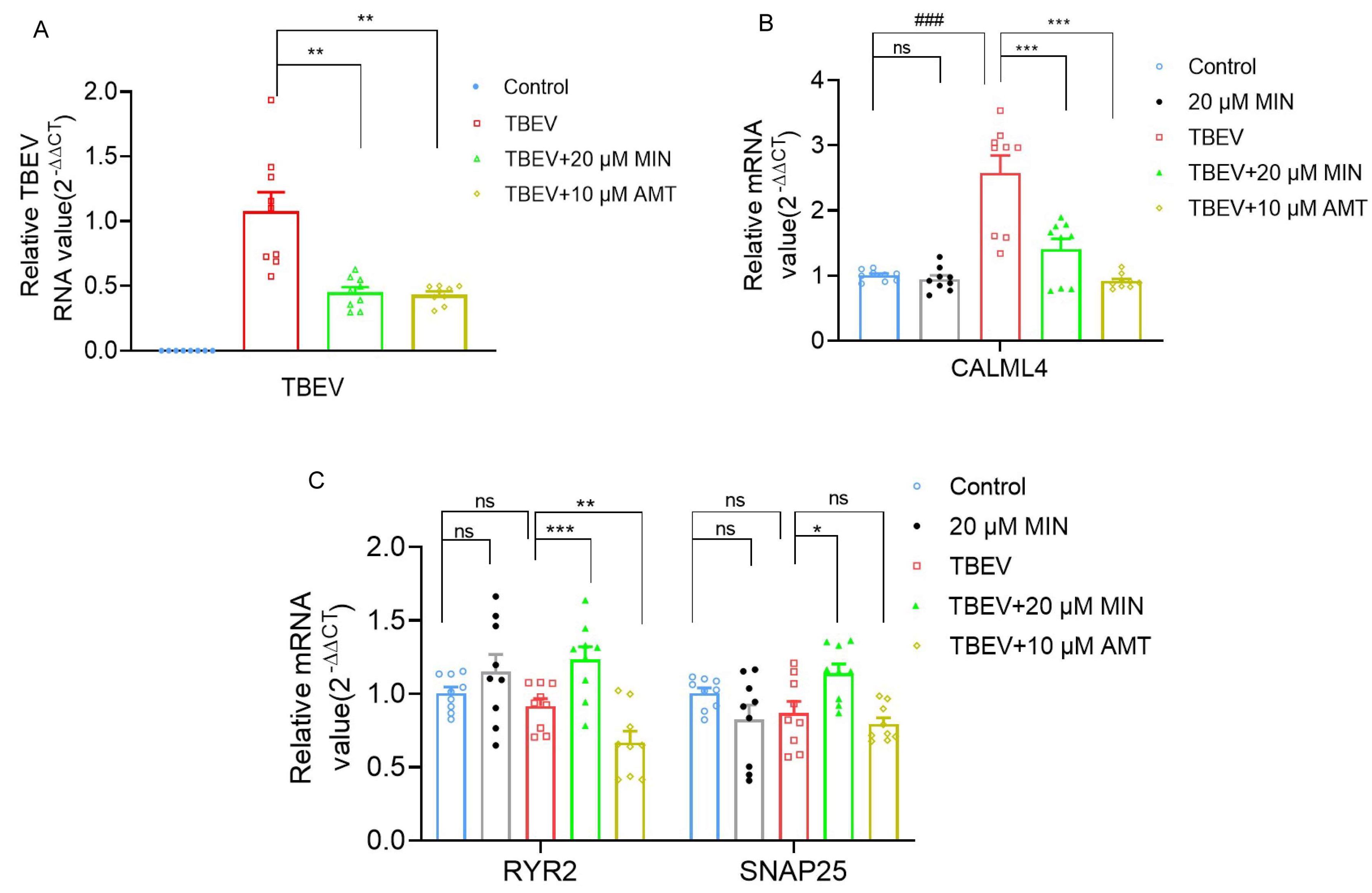
3.4. MIN Regulates Calcium Signaling Pathways in TBEV-Infected Vero Cells
3.5. MIN Inhibits the MAPK-ERK Signaling Pathway in TBEV-Infected Vero Cells
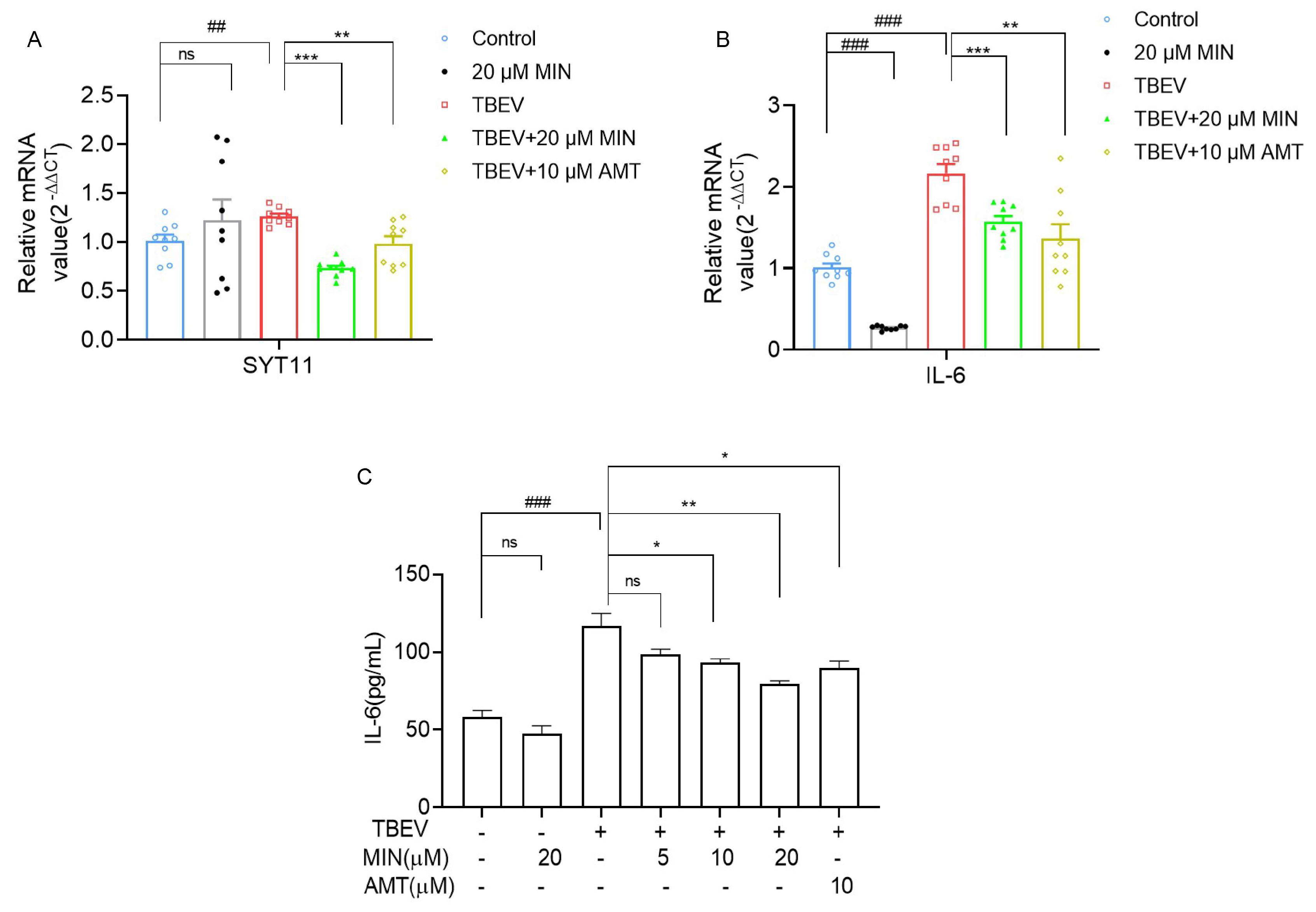
3.6. MIN Inhibits the Expression of Inflammatory Factor IL-6 in TBEV-Infected Vero Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Bogovic, P.; Strle, F. Tick-borne encephalitis: A review of epidemiology, clinical characteristics, and management. World J. Clin. Cases 2015, 3, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.M.; Mulkey, S.B.; Campos, J.M.; Debiasi, R.L.J.P.R. Laboratory diagnosis of CNS infections in children due to emerging and re-emerging neurotropic viruses. Pediatr. Res. 2024, 95, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Taba, P.; Schmutzhard, E.; Forsberg, P.; Lutsar, I.; Ljøstad, U.; Mygland, Å.; Levchenko, I.; Strle, F.; Steiner, I. EAN consensus review on prevention, diagnosis and management of Tick-borne encephalitis. Eur. J. Neurol. 2017, 24, 1214-e61. [Google Scholar] [CrossRef] [PubMed]
- Doñate-Macián, P.; Jungfleisch, J.; Pérez-Vilaró, G.; Rubio-Moscardo, F.; Perálvarez-Marín, A.; Diez, J.; Valverde, M.A. The TRPV4 channel links calcium influx to DDX3X activity and viral infectivity. Nat. Commun. 2018, 9, 2307. [Google Scholar] [CrossRef] [PubMed]
- Sreekanth, G.P.; Chuncharunee, A.; Sirimontaporn, A.; Panaampon, J.; Srisawat, C.; Morchang, A.; Malakar, S.; Thuwajit, P.; Kooptiwut, S.; Suttitheptumrong, A.; et al. Role of ERK1/2 signaling in dengue virus-induced liver injury. Virus Res. 2014, 188, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Formanova, P.P.; Palus, M.; Salat, J.; Hönig, V.; Stefanik, M.; Svoboda, P.; Ruzek, D. Changes in cytokine and chemokine profiles in mouse serum and brain, and in human neural cells, upon Tick-borne encephalitis virus infection. J. Neuroinflam. 2019, 16, 205. [Google Scholar] [CrossRef] [PubMed]
- Barrett, P.N.; Schober-Bendixen, S.; Ehrlich, H.J. History of TBE vaccines. Vaccine 2003, 21, S41–S49. [Google Scholar] [CrossRef] [PubMed]
- Ruzek, D.; Županc, T.A.; Borde, J.; Chrdle, A.; Eyer, L.; Karganova, G.; Kholodilov, I.; Knap, N.; Kozlovskaya, L.; Matveev, A. Tick-borne encephalitis in Europe and Russia: Review of pathogenesis, clinical features, therapy, and vaccines. Antivir. Res. 2019, 164, 23–51. [Google Scholar] [CrossRef] [PubMed]
- Stiasny, K.; Holzmann, H.; Heinz, F.X. Characteristics of antibody responses in Tick-borne encephalitis vaccination breakthroughs. Vaccine 2009, 27, 7021–7026. [Google Scholar] [CrossRef]
- Andersson, C.R.; Vene, S.; Insulander, M.; Lindquist, L.; Lundkvist, A.; Günther, G. Vaccine failures after active immunisation against Tick-borne encephalitis. Vaccine 2010, 28, 2827–2831. [Google Scholar] [CrossRef]
- Lotric-Furlan, S.; Bogovic, P.; Avsic-Zupanc, T.; Jelovsek, M.; Lusa, L.; Strle, F. Tick-borne encephalitis in patients vaccinated against this disease. J. Intern. Med. 2017, 3, 142–155. [Google Scholar] [CrossRef]
- Studahl, M.; Lindquist, L.; Eriksson, B.M.; Günther, G.; Aurelius, E. Acute Viral Infections of the Central Nervous System in Immunocompetent Adults: Diagnosis and Management. Drugs 2013, 73, 131–158. [Google Scholar] [CrossRef] [PubMed]
- Mosquera-Sulbaran, J.A.; Hernández-Fonseca, H. Tetracycline and viruses: A possible treatment for COVID-19? Arch. Virol. 2021, 166, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Garrido-Mesa, N.; Zarzuelo, A.; Gálvez, J. Minocycline: Far beyond an antibiotic. Br. J. Pharmacol. 2013, 169, 337–352. [Google Scholar] [CrossRef] [PubMed]
- Michaelis, M.; Kleinschmidt, M.C.; Doerr, H.W.; Cinatl, J., Jr. Minocycline inhibits West Nile virus replication and apoptosis in human neuronal cells. J. Antimicrob. Chemother. 2007, 60, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Mishra, M.K.; Basu, A. Minocycline neuroprotects, reduces microglial activation, inhibits caspase 3 induction, and viral replication following Japanese encephalitis. J. Neurochem. 2008, 105, 1582–1595. [Google Scholar] [CrossRef]
- Leela, S.L.; Srisawat, C.; Sreekanth, G.P.; Noisakran, S.; Yenchitsomanus, P.T.; Limjindaporn, T. Drug repurposing of minocycline against dengue virus infection. Biochem. Biophys. Res. Commun. 2016, 478, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Lei, C.; Yang, J.; Hu, J.; Sun, X. On the Calculation of TCID(50) for Quantitation of Virus Infectivity. Virol. Sin. 2021, 36, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Brinkley, C.; Nolskog, P.; Golovljova, I.; Lundkvist, Å.; Bergström, T. Tick-borne encephalitis virus natural foci emerge in western Sweden. Int. J. Med. Microbiol. 2008, 298, 73–80. [Google Scholar] [CrossRef]
- Lin, C.-C.; Chen, W.-C. Treatment Effectiveness of Amantadine Against Dengue Virus Infection. Am. J. Case Rep. 2016, 17, 921–924. [Google Scholar] [CrossRef]
- Suzuki, T.; Aoyama, T.; Suzuki, N.; Kobayashi, M.; Fukami, T.; Matsumoto, Y.; Tomono, K. Involvement of a proton-coupled organic cation antiporter in the blood-brain barrier transport of amantadine. Biopharm. Drug Dispos. 2016, 37, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Bezemer, B.; van Cleef, K.W.R.; Overheul, G.J.; Miesen, P.; van Rij, R.P. The calcium channel inhibitor lacidipine inhibits Zika virus replication in neural progenitor cells. Antivir. Res. 2022, 202, 105313. [Google Scholar] [CrossRef]
- Chen, W.C.; Simanjuntak, Y.; Chu, L.W.; Ping, Y.H.; Lee, Y.L.; Lin, Y.L.; Li, W.S. Benzenesulfonamide Derivatives as Calcium/Calmodulin-Dependent Protein Kinase Inhibitors and Antiviral Agents against Dengue and Zika Virus Infections. J. Med. Chem. 2020, 63, 1313–1327. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Min, T.; Dong, J.; Wang, X. Minocycline alleviates the symptoms of morphine withdrawal via the CaMKII-Ras-ERK signaling pathway. Neurosci. Lett. 2021, 752, 135825. [Google Scholar] [CrossRef] [PubMed]
- Palus, M.; Bílý, T.; Elsterová, J.; Langhansová, H.; Salát, J.; Vancová, M.; Růžek, D. Infection and injury of human astrocytes by Tick-borne encephalitis virus. J. Gen. Virol. 2014, 95, 2411–2426. [Google Scholar] [CrossRef] [PubMed]
- Eyer, L.; Seley-Radtke, K.; Ruzek, D. New directions in the experimental therapy of Tick-borne encephalitis. Antivir. Res. 2023, 210, 105504. [Google Scholar] [CrossRef] [PubMed]
- Bergold, P.J.; Furhang, R.; Lawless, S. Treating Traumatic Brain Injury with Minocycline. Neurotherapeutics 2023, 20, 1546–1564. [Google Scholar] [CrossRef] [PubMed]
- Kulprasertsri, S.; Aoshima, K.; Kobayashi, A.; Kimura, T. Minocycline prevents primary duck neurons from duck Tembusu virus-induced death. J. Vet. Med. Sci. 2021, 83, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Quick, E.D.; Seitz, S.; Clarke, P.; Tyler, K.L. Minocycline Has Anti-inflammatory Effects and Reduces Cytotoxicity in an Ex Vivo Spinal Cord Slice Culture Model of West Nile Virus Infection. J. Virol. 2017, 91, e00569-17. [Google Scholar] [CrossRef]
- Dutta, K.; Kumawat, K.L.; Nazmi, A.; Mishra, M.K.; Basu, A. Minocycline differentially modulates viral infection and persistence in an experimental model of Japanese encephalitis. J. Neuroimmune Pharmacol. Off. J. Soc. NeuroImmune Pharmacol. 2010, 5, 553–565. [Google Scholar] [CrossRef]
- Charlton, F.W.; Pearson, H.M.; Hover, S.; Lippiat, J.D.; Fontana, J.; Barr, J.N.; Mankouri, J. Ion Channels as Therapeutic Targets for Viral Infections: Further Discoveries and Future Perspectives. Viruses 2020, 12, 844. [Google Scholar] [CrossRef]
- Di Benedetto, G.; Scalzotto, E.; Mongillo, M.; Pozzan, T. Mitochondrial Ca2+ uptake induces cyclic AMP generation in the matrix and modulates organelle ATP levels. Cell Metab. 2013, 17, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Rossol, M.; Pierer, M.; Raulien, N.; Quandt, D.; Meusch, U.; Rothe, K.; Schubert, K.; Schöneberg, T.; Schaefer, M.; Krügel, U.; et al. Extracellular Ca2+ is a danger signal activating the NLRP3 inflammasome through G protein-coupled calcium sensing receptors. Nat. Commun. 2012, 3, 1329. [Google Scholar] [CrossRef]
- Choi, M.S.; Graves, M.J.; Matoo, S.; Storad, Z.A.; El Sheikh Idris, R.A.; Weck, M.L.; Smith, Z.B.; Tyska, M.J.; Crawley, S.W. The small EF-hand protein CALML4 functions as a critical myosin light chain within the intermicrovillar adhesion complex. J. Biol. Chem. 2020, 295, 9281–9296. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, C.; Roston, T.M.; van der Werf, C.; Sanatani, S.; Chen, S.R.W.; Wilde, A.A.M.; Krahn, A.D. RYR2-ryanodinopathies: From calcium overload to calcium deficiency. Europace 2023, 25, euad156. [Google Scholar] [CrossRef]
- Condliffe, S.B.; Corradini, I.; Pozzi, D.; Verderio, C.; Matteoli, M. Endogenous SNAP-25 regulates native voltage-gated calcium channels in glutamatergic neurons. J. Biol. Chem. 2010, 285, 24968–24976. [Google Scholar] [CrossRef]
- Sasaoka, M.; Ota, T.; Kageyama, M. Rotenone-induced inner retinal degeneration via presynaptic activation of voltage-dependent sodium and L-type calcium channels in rats. Sci. Rep. 2020, 10, 969. [Google Scholar] [CrossRef]
- Ashraf, U.; Ding, Z.; Deng, S.; Ye, J.; Cao, S.; Chen, Z. Pathogenicity and virulence of Japanese encephalitis virus: Neuroinflammation and neuronal cell damage. Virulence 2021, 12, 968–980. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Sun, X.; Chen, L.; Feng, X.; Yang, X.; Zou, P.; Wang, X.; Zhang, R. Hepatitis C Virus Core Protein Promotes the Metastasis of Human Hepatocytes by Activating the MAPK/ERK/PEA3-SRF/c-Fos/MMPs Axis. Arch. Med. Res. 2022, 53, 469–482. [Google Scholar] [CrossRef]
- Islam, M.S.; Greco, S.; Janjusevic, M.; Ciavattini, A.; Giannubilo, S.R.; D‘Adderio, A.; Biagini, A.; Fiorini, R.; Castellucci, M.; Ciarmela, P. Growth factors and pathogenesis, Best practice & research. Clin. Obstet. Gynaecol. 2016, 34, 25–36. [Google Scholar] [CrossRef]
- Kiyatkin, A.; van Alderwerelt van Rosenburgh, I.K.; Klein, D.E.; Lemmon, M.A. Kinetics of receptor tyrosine kinase activation define ERK signaling dynamics. Sci. Signal. 2020, 13, eaaz5267. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Persson, A.; Sun, Y.; Salford, L.G.; Nord, D.G.; Englund, E.; Jiang, T.; Fan, X. Better prognosis of patients with glioma expressing FGF2-dependent PDGFRA irrespective of morphological diagnosis. PLoS ONE 2013, 8, e61556. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Choi, E.J. Pathological roles of MAPK signaling pathways in human diseases. Biochim. Et Biophys. Acta 2010, 1802, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, Z.G.; Dhanasekaran, D.N. G protein regulation of MAPK networks. Oncogene 2007, 26, 3122–3142. [Google Scholar] [CrossRef]
- Hu, Y.Y.; Zhong, R.H.; Guo, X.J.; Li, G.T.; Zhou, J.Y.; Yang, W.J.; Ren, B.T.; Zhu, Y. Jinfeng pills ameliorate premature ovarian insufficiency induced by cyclophosphamide in rats and correlate to modulating IL-17A/IL-6 axis and MEK/ERK signals. J. Ethnopharmacol. 2023, 307, 116242. [Google Scholar] [CrossRef]
- Rose-John, S. Local and systemic effects of interleukin-6 (IL-6) in inflammation and cancer. FEBS Lett. 2022, 596, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Wang, Y.; Zhang, F.; Yan, S.; Guan, Y.; Gong, X.; Zhang, T.; Cui, X.; Wang, X.; Zhang, C.X. Synaptotagmin-11 inhibits cytokine secretion and phagocytosis in microglia. Glia 2017, 65, 1656–1667. [Google Scholar] [CrossRef]
- Marušić, M.; Kopitar, A.N.; Korva, M.; Knap, N.; Bogovič, P.; Strle, F.; Ihan, A.; Avšič-Županc, T. Dendritic cell activation and cytokine response in vaccine breakthrough TBE patients after in vitro stimulation with TBEV. Front. Immunol. 2023, 14, 1190803. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, M.; Yang, W.; Yang, J.; Zhao, Y.; Hu, X.; Xu, X.; Tian, J.; Chen, Y.; Jiang, H.; Ren, R.; et al. Minocycline Inhibits Tick-Borne Encephalitis Virus and Protects Infected Cells via Multiple Pathways. Viruses 2024, 16, 1055. https://doi.org/10.3390/v16071055
Cao M, Yang W, Yang J, Zhao Y, Hu X, Xu X, Tian J, Chen Y, Jiang H, Ren R, et al. Minocycline Inhibits Tick-Borne Encephalitis Virus and Protects Infected Cells via Multiple Pathways. Viruses. 2024; 16(7):1055. https://doi.org/10.3390/v16071055
Chicago/Turabian StyleCao, Mengtao, Wei Yang, Jintao Yang, Yanli Zhao, Xiaoyu Hu, Xiaoli Xu, Jing Tian, Yue Chen, Hongxia Jiang, Ruiwen Ren, and et al. 2024. "Minocycline Inhibits Tick-Borne Encephalitis Virus and Protects Infected Cells via Multiple Pathways" Viruses 16, no. 7: 1055. https://doi.org/10.3390/v16071055




