Insights on Cadmium Removal by Bioremediation: The Case of Haloarchaea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Information Processing to Carry out Bibliometric Analysis
2.2. Search Strategy and Information Processing to Carry out Bibliographic Analysis
3. Results and Discussion
3.1. Bibliometric Review
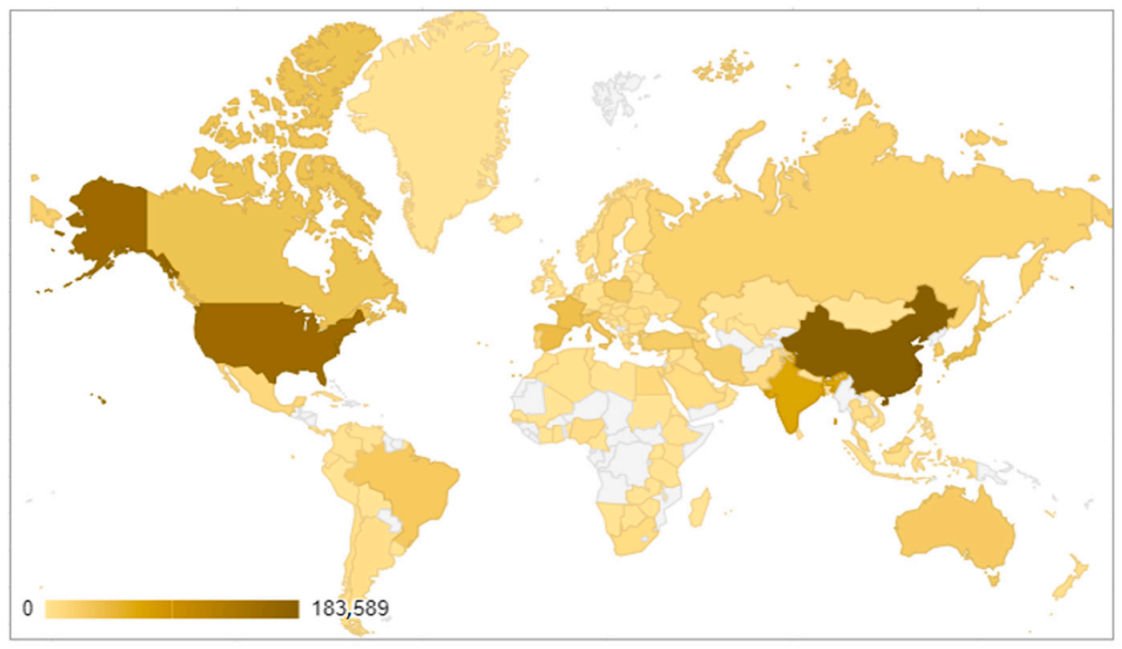
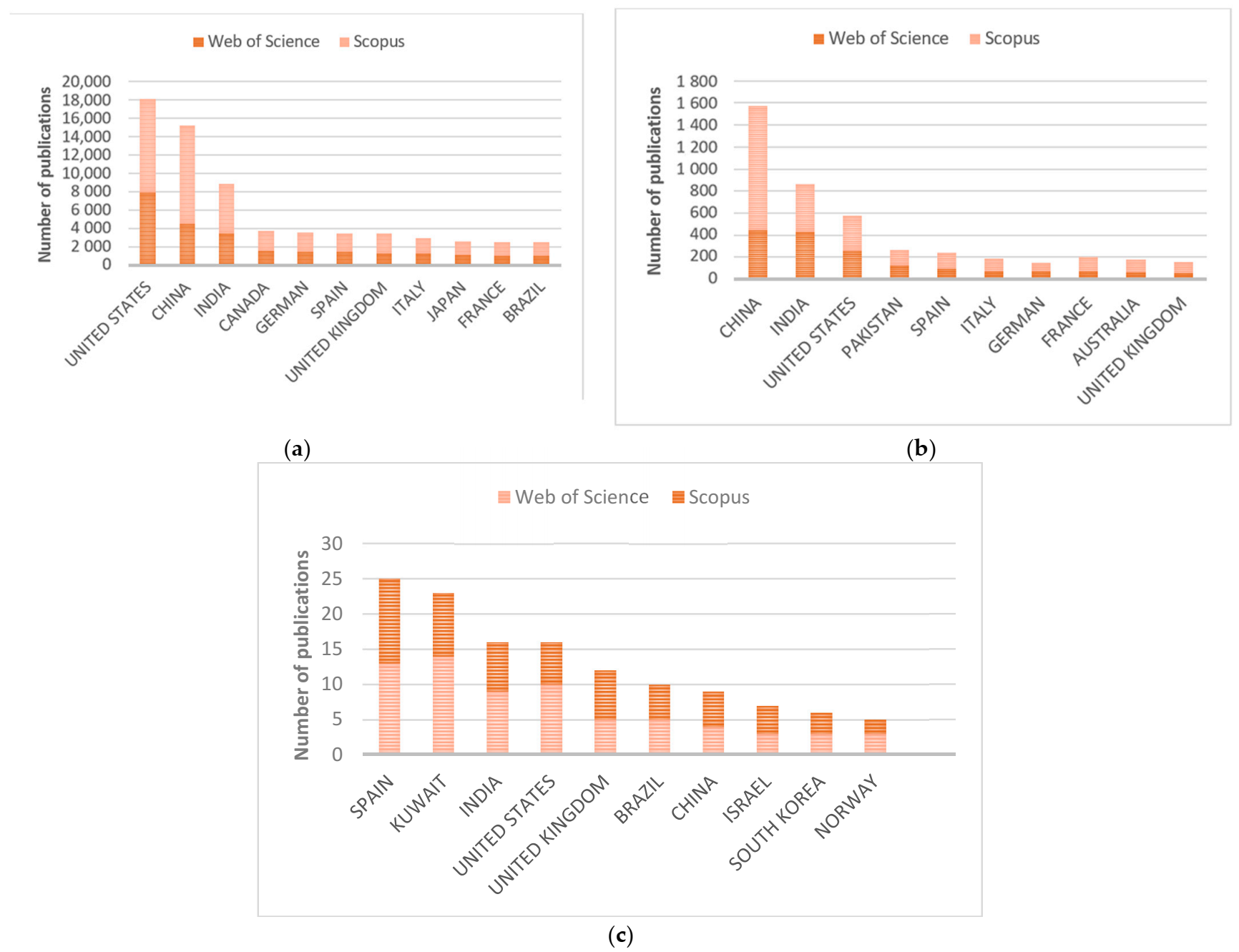
3.1.1. Study of the Number of Publications According to Their Distribution by Country
3.1.2. Number of Publications Focused on Bioremediation of Cadmium Contaminated Samples/Sites
3.1.3. Publications Related to the Use of Haloarchaea in Bioremediation. The Case of Haloferax mediterranei
3.2. Bibliographic Review
3.2.1. Bioremediation: Possible Solution to Contamination of Soils and Water by Heavy Metals
3.2.2. Description of the Main Molecular Mechanisms Sustaining Heavy Metals Resistance in Microorganisms
Biosorption
Bioaccumulation
P-Type ATPases
RND Transporters Family
Cation Diffusion Facilitators (CDF)
3.2.3. Environmental Contamination by Cadmium. Adverse Effects on Human and Animal Health
- Oral route: through contaminated food or water. The United States Environmental Protection Agency (EPA) has established a reference dose as a limit value for daily cadmium consumption to avoid adverse health effects: concentration in water up to 0.5 μg/L, and in feeding up to 1 μg/kg (ATSDR, 1999) (available at https://www.atsdr.cdc.gov/toxprofiles/tp5.pdf, accessed on 16 February 2020). In some Europe and North America countries the intake of this metal can be up to 40 µg per day through the diet [86].
- Respiratory route: it occurs when people are exposed to several industrial activities, reaching inhalation values of up to 50 µg/L [87].
3.2.4. Cadmium Removal in the Presence of Other Heavy Metals: Synergistic and Antagonistic Effects
- pH of the media: at low pH values, the functional groups located in the cell wall are fully protonated, so the metal ions adsorption does not take place. If pH value increases, these groups become deprotonate, and the metal binding sites would be free to join heavy metals [92].
- Hydrated ion radius: it is the amount of water surrounding the ions, and this depends on each element. Compounds with a lower hydration radius will present a higher biomass adsorption affinity than those with a higher hydration radius. In the study conducted by Sulaymon and co-workers, the removal efficiency of Pb, Cr and Cd metals found in synthetic wastewater was tested using a heterogeneous culture containing protozoa, yeast, and anaerobic bacteria [93]. In this study, the element with the highest adsorption capacity was Pb, followed by Cr, and finally Cd (Pb > Cr > Cd). This order correlated with the values of the hydration radius showed by each element (Pb for instance shows the smaller hydration radius (4.01 Å) and the highest adsorption capacity to biomass) [93].
- Metal electronegativity: this is the ability of an atom to attract the electrons belonging to another atom. As the electronegativity of the atom increases, the ionic form can be easily adsorbed by the sorbent [93]. Thus, the preference for Pb adsorption is also enhanced by its high electronegativity (2.33). Cadmium, however, is the one with the lowest biosorption capacity, coinciding with its low electronegativity (1.69); Cr for instance has an intermediate electronegativity value (1.66) [93].
- Solute solubility: solubility of heavy metals in water is in general low thus negatively affecting biosorption increases. If several heavy metals are in a solution, biosorption of Cd is lower than biosorption of other heavy metals like Pb (examples of solubility values: Pb (52 g/mL), Cr (81 g/mL) and Cd (136 g/mL)). Consequently, this factor has a negative impact on bioremediation of cadmium when it is joining other heavy metals [93].
- Ionic radii and molecular weight: compounds characterized by higher ionic radii and higher the molecular weight shows greater biosorption [94]. Studies like the one conducted by Moreira using the macroalgae Fucus vesiculosus demonstrated greater adsorption capacity for Pb followed by cadmium and Ni, respectively. This order coincides with the ionic radii values of each element: the ionic radii value for Pb, Cd and Ni are 119 p.m.; Cd, 95 p.m. and 60 p.m., respectively [94].
3.2.5. Haloarchaea as Model Organisms for Bioremediation of Heavy Metals Contaminated Sites: The Case of Cadmium
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of heavy metals- concepts and applications. Chemosphere 2013, 91, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Li, Z.; Han, C.; Liu, L.; Teng, Y.; Sun, X.; Pan, C.; Huang, Y.; Luo, Y.; Christie, P. Phytoremediation of soil contaminated with cadmium, copper and polychlorinated biphenyls. Int. J. Phytoremed. 2012, 14, 570–584. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Li, X.; Song, J.; Jiang, W.; Liu, Y.; Fan, W. Bioremediation of cadmium and zinc contaminated soil using Rhodobacter sphaeroides. Chemosphere 2018, 197, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Khatiwada, B.; Hasan, M.T.; Sun, A.; Kamath, K.S.; Mirzaei, M.; Sunna, A.; Nevalainen, H. Proteomic response of Euglena gracilis to heavy metal exposure—Identification of key proteins involved in heavy metal tolerance and accumulation. Algal Res. 2020, 45, 101764. [Google Scholar] [CrossRef]
- Londoño-Franco, L.F.; Londoño-Muñoz, P.T.; Muñoz-García, F.G. Risk of heavy metals in human and animal health. Biotecnol. Sect. Agropecu. Agroind. 2016, 14, 145–153. [Google Scholar]
- Hernández, A.; Hansen, A. Uso de plaguicidas en dos zonas agrícolas de México y evaluación de la contaminación de agua y sedimentos. Rev. Int. Contam. Ambient. 2012, 27, 115–127. [Google Scholar]
- Molina, C.; Ibañez, C.; Gibon, F.M. Proceso de biomagnificación de metales pesados en un lago hiperhalino (Poopó, Oruro, Bolivia): Posible riesgo en la salud de consumidores. Ecología 2013, 47, 99–118. [Google Scholar]
- Sarwar, N.; Ishaq, W.; Farid, G.; Shaheen, M.R.; Muhammad, R.; Imran, M.; Geng, M.; Hussain, S. Zinc-cadmium interactions: Impact on wheat physiology and mineral acquisition. Ecotoxicol. Environ. Saf. 2015, 122, 528–536. [Google Scholar] [CrossRef]
- Sarwar, N.; Imran, M.; Shaheen, M.R.; Ishaque, W.; Kamran, M.A.; Matloob, A.; Rehim, A.; Hussain, S. Phytoremediation strategies for soils contaminated with heavy metals: Modifications and future perspectives. Chemosphere 2017, 171, 710–721. [Google Scholar] [CrossRef]
- Mudipalli, A. Metals (micronutrients or toxicans) and global health. Indian J. Med Res. 2008, 128, 331–334. [Google Scholar]
- Das, K.; Das, S.; Dhundasi, S. Nickel, its adverse health effects and oxidative stress. Indian J. Med Res. 2008, 128, 412–425. [Google Scholar]
- Sánchez-Chardi, A.; Ribeiro, C.A.O.; Nadal, J. Metals in liver and kidneys and the effects of chronic exposure to pyrite mine pollution in the shrew Crocidura russula inhabiting the protected wetland of Doñana. Chemosphere 2009, 76, 387–394. [Google Scholar] [CrossRef]
- Malayeri, B.E.; Cheheregani, A.; Yousefi, N.; Lorestani, B. Identification of the hyper accumulator plants in copper and iron mine in Iran. Pak. J. Biol. Sci. 2008, 11, 490–492. [Google Scholar] [CrossRef] [Green Version]
- Luo, L.Y.; Xie, L.L.; Jin, D.C.; Mi, B.B.; Wang, D.H.; Li, X.F.; Dai, X.Z.; Zou, X.X.; Zhang, Z.; Ma, Y.Q.; et al. Bacterial community response to cadmium contamination of agricultural paddy soil. Appl. Soil Ecol. 2019, 139, 100–106. [Google Scholar] [CrossRef]
- Khan, S.; Hesham, A.E.L.; Qiao, M.; Rehman, S.; He, J.Z. Effects of Cd and Pb on soil microbial community structure and activities. Environ. Sci. Pollut. Res. 2010, 17, 288–296. [Google Scholar] [CrossRef]
- Sheng, W.; Wei, W.; Li, J.; Qi, X.; Zuo, G.; Chen, Q.; Pan, X.; Dong, W. Amine-functionalized magnetic mesoporous silica nanoparticles for DNA separation. Appl. Surf. Sci. 2016, 387, 1116–1124. [Google Scholar] [CrossRef]
- Lazar, M.M.; Dinu, I.A.; Silion, M.; Dragan, E.S.; Dinu, M.V. Could the porous chitosan-based composite materials have a chance to a “NEW LIFE” after Cu (II) ion binding? Int. J. Biol. Macromol. 2019, 131, 134–146. [Google Scholar] [CrossRef]
- Zeng, Q.; Qi, X.; Zhang, M.; Tong, X.; Jiang, N.; Pan, W.; Xiong, W.; Li, Y.; Xu, J.; Shen, J.; et al. Efficient decontamination of heavy metals from aqueous solution using pullulant/polydopamine hydrogels. Int. J. Biol. Macromol. 2020, 145, 1049–1058. [Google Scholar] [CrossRef]
- Garba, Z.N.; Lawan, I.; Zhou, W.; Zhang, M.; Wang, L.; Yuan, Z. Microcrystalline cellulose (MCC) based materials as emerging adsorbents for the removal of dyes and heavy metals—A review. Sci. Total Environ. 2019, 717, 135070. [Google Scholar] [CrossRef]
- Bonete, M.J.; Martínez-Espinosa, R.M.; Pire, C.; Zafrilla, B.; Richardson, D.J. Nitrogen metabolism in haloarchaea. Saline Syst. 2008, 4, 9. [Google Scholar] [CrossRef] [Green Version]
- Sato, T.; Atomi, H. Novel metabolic pathways in archaea. Curr. Opin. Microbiol. 2011, 14, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, L.; Maklad, H.R.; Bervoets, I.; Peeters, E. Transcription regulators in Archaea: Homologies and differences with bacterial regulators. J. Mol. Biol. 2019, 431, 4132–4146. [Google Scholar] [CrossRef] [PubMed]
- Torregrosa-Crespo, J.; Martínez-Espinosa, R.M.; Esclapez, J.; Bautista, V.; Pire, C.; Camacho, M.; Richardson, D.J.; Bonete, M.J. Anaerobic metabolism in Haloferax genus: Denitrification as case of study. Advances in Microbial Physiology 68. Adv. Microb. Physiol. 2016, 68, 41–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia, M.T.; Mellado, E.; Ostos, J.C.; Ventosa, A. Halomonas organivorans sp. nov., a moderate halophile able to degrade aromatic compounds. Int. J. Syst. Evol. Microbiol. 2004, 54, 1723–1728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pieper, U.; Kapadia, G.; Mevarech, M.; Herzberg, O. Structural features of halophilicity derived from the crystal structure of dihydrofolate reductase from the Dead Sea halophilic archaeon, Haloferax volcanii. Structure 1998, 6, 75–88. [Google Scholar] [CrossRef] [Green Version]
- Ventosa, A.; Nieto, J.J.; Oren, A. Biology of moderately halophilic aerobic bacteria. Microbiol. Mol. Biol. Rev. 1998, 62, 504–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mevarech, M.; Frolow, F.; Gloss, L.M. Halophilic enzymes: Proteins with a grain of salt. Biophys. Chem. 2000, 86, 155–164. [Google Scholar] [CrossRef]
- Madigan, M.T.; Martinko, J.M.; Bender, K.S.; Buckley, D.H.; Stahl, D.A. BROCK. Biología de los microorganismos, 14th ed.; Editorial Pearson Educación: Madrid, Spain, 2015; p. 1131. [Google Scholar]
- Liu, H.L.; Han, J.; Liu, X.Q.; Zhou, J.; Xiang, H. Development of pyrF-based gene knockout systems for genome-wide manipulation of the archaea Haloferax mediterranei and Haloarcula hispanica. J. Genet. Genom. 2011, 38, 261–269. [Google Scholar] [CrossRef]
- Martínez-Espinosa, R.M.; Zafrilla, B.; Camacho, M.; Bonete, M.J. Nitrate and nitrite removal from salted water by Haloferax mediterranei. Biocatal. Biotransformation 2007, 25, 295–300. [Google Scholar] [CrossRef]
- Martínez-Espinosa, R.M.; Richardson, D.J.; Bonete, M.J. Characterisation of chlorate reduction in the haloarchaeon Haloferax mediterranei. Biochim. Biophys. Acta 2015, 1850, 587–594. [Google Scholar] [CrossRef] [Green Version]
- Wiesman, F.; Hasman, A.; Herik, H.V.D. Information retrieval: An overview of system characteristics. Int. J. Med Inform. 1997, 47, 5–26. [Google Scholar] [CrossRef]
- Oxenrider, K.A.; Kennelly, P.J. A protein-serine phosphatase from the halophilic archaeon Haloferax volcanii. Biochem. Biophys. Res. Commun. 1993, 194, 1330–1335. [Google Scholar] [CrossRef]
- Durrant, L.R.; Bonfá, M.; Piubelli, F.; Zaballos, M.P.; Cuadros-Orellana, S. Biodegradation of aromatic compounds and PAHs by halophilic archaea. In Proceedings of the 9th International In Situ and On-Site Bioremediation Symposium, Baltimore, MD, USA, 7–10 May 2011; pp. 496–503. [Google Scholar]
- Bonete, M.J.; Bautista, V.; Esclapez, J.; García-Bonete, M.J.; Pire, C.; Camacho, M.; Torregrosa-Crespo, J.; Martínez-Espinosa, R.M. New uses of haloarchaeal species in bioremediation processes. Advances in bioremediation of wastewater and polluted soil. Intech 2015, 2, 23–49. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Espinosa, R.M.; Richardson, D.J.; Butt, J.N.; Bonete, M.J. Respiratory nitrate and nitrite pathway in the denitrifier haloarchaeon Haloferax mediterranei. Biochem. Soc. Trans. 2006, 34, 115–117. [Google Scholar] [CrossRef]
- Nájera-Fernández, C.; Zafrilla, B.; Bonete, M.J.; Martínez-Espinosa, R.M. Role of the denitrifying Haloarchaea in the treatment of nitrite-brines. Int. Microbiol. 2012, 15, 111–119. [Google Scholar] [CrossRef] [Green Version]
- Oren, A.; Gurevich, P.; Henis, Y. Reduction of nitrosubstituted aromatic compounds by the halophilic anaerobic eubacteria Haloanaerobium praevalens and Sporohalobacter marismortui. Appl. Eviron. Microbiol. 1991, 57, 3367–3370. [Google Scholar] [CrossRef] [Green Version]
- Yang, H. China’s soil plan needs strong support. Nature 2016, 536, 375. [Google Scholar] [CrossRef] [Green Version]
- Harris, T. Rains or not, India Faces Drinking Water Crisis. 2016. Available online: https://phys.org/news/2016–06-india-crisis.html (accessed on 10 April 2020).
- Aracil-Gisbert, S.; Torregrosa-Crespo, J.; Martínez-Espinosa, R.M. Recent trend on bioremediation of polluted salty soils and waters using haloarchaea. In Advances in Bioremediation and Phytoremediation; Shiomi, N., Ed.; IntechOpen: London, UK, 2018; Chapter 4. [Google Scholar] [CrossRef] [Green Version]
- Le Borgne, S.; Paniagua, D.; Vazquez-Duhalt, R. Biodegradation of organic pollutants by halophilic bacteria and archaea. J. Mol. Microbiol. Biotechnol. 2008, 15, 7492. [Google Scholar] [CrossRef]
- Falb, M.; Müller, K.; Königsmaier, L.; Oberwinkler, T.; Horn, P.; von Gronau, S.; González, O.; Pfeiffer, F.; Bornberg-Bauer, E.; Oesterhelt, D. Metabolism of halophilic archaea. Extremophiles 2008, 12, 177–196. [Google Scholar] [CrossRef] [Green Version]
- Quero, J.L.; Maestre, F.T.; Ochoa, V.; García-Gómez, M.; Delgado-Baquerizo, M. On the importance of shrub encroachment by sprouters, climate, species richness and anthropic factors for ecosystem multifunctionality in semi-arid Mediterranean ecosystems. Ecosystems 2013, 16, 1248–1261. [Google Scholar] [CrossRef] [Green Version]
- Berdugo, M.; Delgado-Baquerizo, M.; Soliveres, S.; Hernández-Clemente, R.; Zhao, Y.; Gaitán, J.J.; Gross, N.; Saiz, H.; Maire, V.; Lehmann, A.; et al. Global ecosystem thresholds driven by aridity. Science 2020, 367, 787–790. [Google Scholar] [CrossRef] [Green Version]
- Gupta, R.S.; Naushad, S.; Baker, S. Phylogenomic analyses and molecular signatures for the class Halobacteria and its two major clades: A proposal for division of the class Halobacteria into an emended order Halobacteriales and two new orders, Haloferacales ord. nov. and Natrialbales ord. nov., containing the novel families Haloferacaceae fam. nov. and Natrialbaceae fam. nov. Int. J. Syst. Evol. Microbiol. 2015, 65, 1050–1069. [Google Scholar] [CrossRef]
- Rodríguez-Valera, F.; Ruiz-Berraquero, F.; Ramos-Cormenzana, A. Isolation of extremely halophilic bacteria able to grow in defined inorganic media with single carbon sources. J. Gen. Microbiol. 1980, 119, 535–538. [Google Scholar] [CrossRef] [Green Version]
- Oren, A. Industrial and environmental applications of halophilic microorganisms. Environ. Technol. 2010, 31, 825–834. [Google Scholar] [CrossRef] [Green Version]
- Mojica, F.J.M.; Díez-Villaseñor, C.; García-Martínez, J.; Almendros, C. Short motif sequences determine the targets of the prokaryotic CRISPR defence system. Microbiology 2009, 155, 733–740. [Google Scholar] [CrossRef] [Green Version]
- Adams, G.; Fufeyin, P.; Okoro, S.; Ehinomen, I. Bioremediation, biostimulation and bioaugmentation: A review. Int. J. Environ. Bioremediation Biodegrad. 2015, 3, 28–39. [Google Scholar] [CrossRef]
- Cycon, M.; Mrozik, A.; Piotrowska-Seget, Z. Bioaugmentation as a strategy for the remediation of pesticide-polluted soil: A review. Chemosphere 2017, 172, 52–71. [Google Scholar] [CrossRef]
- Li, X.N.; Song, H.L.; Li, W.; Xi, O. An integrated ecological floating-bed employing plant freshwater clam and biofilm carrier for purification of eutrophic water. Ecol. Eng. 2010, 36, 382–390. [Google Scholar] [CrossRef]
- Garzón, J.M.; Rodríguez-Miranda, J.P.; Hernández-Gómez, C. Revisión del aporte de la biorremediación para solucionar problemas de contaminación y su relación con el desarrollo sostenible. Rev. Univ. Y Salud 2017, 19, 309–318. [Google Scholar] [CrossRef] [Green Version]
- El-Bestawy, E. Efficiency of immobilized cyanobacteria in heavy metals removal from industrial effluents. Desalination Water Treat. 2019, 159, 66–78. [Google Scholar] [CrossRef] [Green Version]
- Das, M.; Adholeya, A. Potential uses of immobilized bacteria, fungi, algae, and their aggregates for treatment of organic and inorganic pollutants in wastewater. Acs Symp. Ser. 2015, 1206, 319–337. [Google Scholar]
- Vimala, R.; Das, N. Biosorption of cadmium (II) and lead (II) from aqueous solutions using mushrooms: A comparative study. J. Hazard. Mater. 2009, 168, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Viraraghavan, T. Fungal biosorption an alternative treatment option for heavy metal cleaning wastewaters: A review. Bioresour. Technol. 1995, 53, 195–206. [Google Scholar]
- Marrero-Coto, J.; Díaz-Valdivia, A.; Coto-Pérez, O. Mecanismos moleculares de resistencia a metales pesados en bacterias y sus aplicaciones en biorremediación. Cenic Cienc. Biológicas 2010, 41, 67–78. [Google Scholar]
- Smets, B.F.; Morrow, J.B.; Pinedo, C.A. Plasmid introduction in metal-stressed, sub-surface-derived microcosms: Plasmid fate and community response. Appl. Environ. Microbiol. 2003, 69, 4087–4097. [Google Scholar] [CrossRef] [Green Version]
- Das, D.; Salgaonkar, B.B.; Mani, K.; Braganca, J.M. Cadmium resistance in extremely halophilic archaeon Haloferax strain BBK2. Chemosphere 2014, 112, 385–392. [Google Scholar] [CrossRef]
- Pagnanelli, F.; Mainelli, S.; Bornoroni, L.; Dionisi, D.; Toro, L. Mechanisms of heavy-metal removal by activated sludge. Chemosphere 2009, 75, 1028–1034. [Google Scholar] [CrossRef]
- Nies, D.H. Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol. Rev. 2003, 27, 313–339. [Google Scholar] [CrossRef]
- Cañizares-Villanueva, R.O. Biosorción de metales pesados mediante el uso de biomasa microbiana. Rev. Latinoam. Microbiol. 2000, 42, 131–143. [Google Scholar]
- Rajesh, M.V.; Kumar, A.S.K.; Rajesh, N. Biosorption of cadmium using a novel bacterium isolated from an electronic industry effluent. Chem. Eng. J. 2014, 235, 176–185. [Google Scholar]
- Rajesh, M.V.; Rajesh, N. An indigenous Halomonas BVR1 strain immobilized in crosslinked chitosan for adsorption of lead and cadmium. Int. J. Biol. Macromol. 2015, 79, 300–308. [Google Scholar]
- Showalter, A.R.; Szymanowski, J.E.S.; Fein, J.B.; Bunker, B.A. An X-ray absorption spectroscopy study of Cd binding onto a halophilic archaeon. J. Phys. 2016, 712, 012079. [Google Scholar] [CrossRef] [Green Version]
- Klaassen, C.D.; Liu, J.; Choudhuri, S. Metallothionein: An intracellular protein to protect against cadmium toxicity. Pharmacol. Toxicol. 1999, 39, 267–294. [Google Scholar] [CrossRef] [Green Version]
- Olafson, R.W.; Abel, K.; Sim, R.G. Prokaryotic metallothionein, preliminary characterization of a blue-green alga heavy metal-binding protein. Biochem. Biophys. Res. Commun. 1979, 89, 36–43. [Google Scholar] [CrossRef]
- Nanda, M.; Kumar, V.; Sharma, D.K. Multimetal tolerance mechanisms in bacteria: The resistance strategies acquired by bacteria that can be exploited to “clean-up” heavy metal contaminants from water. Aquat. Toxicol. 2019, 212, 1–10. [Google Scholar] [CrossRef]
- Haferburg, G.; Kothe, E. Microbes and metals: Interactions in the environment. J. Basic Microbiol. 2007, 47, 453–467. [Google Scholar] [CrossRef]
- Nies, D.H.; Silver, S. Ion efflux systems involved in bacterial metal resistances. J. Ind. Microbiol. 1995, 14, 186–199. [Google Scholar] [CrossRef]
- De Hertogh, B.; Lantin, A.C.; Baret, P.V.; Goffeau, A. The archaeal P-type ATPases. J. Bioenerg. Biomembr. 2004, 36, 135–142. [Google Scholar] [CrossRef]
- Bredeston, L.M.; González Flecha, F.L. The promiscuous phosphomonoestearase activity of Archaeoglobus fulgidus CopA, a thermophilic Cu+ transport ATPase. Biochim. Biophys. Acta 2016, 1858, 1471–1478. [Google Scholar] [CrossRef]
- Völlmecke, C.; Drees, S.L.; Reimann, J.; Albers, S.V.; Lübben, M. The ATPases CopA and CopB both contribute to copper resistance of the thermoacidophilic archaeon Sulfolobus solfataricus. Microbiology 2012, 158 Pt 6, 1622–1633. [Google Scholar] [CrossRef]
- Silver, S.; Phung, L.T. A bacterial view of the periodic table: Genes and proteins for toxic inorganic ions. J. Ind. Microbiol. Biotechnol. 2005, 32, 587–605. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. The microbiology of acidic mine waters. Res. Microbiol. 2003, 154, 466–473. [Google Scholar] [CrossRef]
- Legatzki, A.; Franke, S.; Lucke, S.; Hoffmann, T.; Anton, A.; Neumann, D. First step towards a quantitative model describing czc-mediated heavy metal resistance in Ralstonia metallidurans. Biodegradation 2003, 14, 153–168. [Google Scholar] [CrossRef]
- Nikaido, H. RND transporters in the living world. Res. Microbiol. 2018, 169, 363–371. [Google Scholar] [CrossRef]
- Russell, D.; Soulimane, T. Evidence for zinc and cadmium binding in a CDF transporter lacking the cytoplasmatic domain. FEBS Lett. 2012, 586, 4332–4338. [Google Scholar] [CrossRef]
- Mockaitis, G.; Rodrigues, J.A.D.; Foresti, E.; Zaiat, M. Toxic effects of cadmium on anaerobic biomass: Kinetic and metabolic implications. J. Environ. Manag. 2012, 106, 75–84. [Google Scholar] [CrossRef]
- Correa-García, C. Ecotoxicología del Cadmio. Riesgo Para la Salud Por la Utilización de Suelos Ricos en Cadmio. Trabajo de Fin de Grado Universidad Complutense de Madrid, España. 2016. Available online: https://eprints.ucm.es/49137/ (accessed on 30 September 2020).
- Hattab, S.; Boussetta, H.; Banni, M. Influence of nitrate fertilization on Cd uptake and oxidative stress parameters in alfalfa plants cultivated in presence of Cd. J. Soil Sci. Plant. Nutr. 2014, 14. [Google Scholar] [CrossRef] [Green Version]
- Pérez, P.E.; Azcona, M.I. Los efectos del cadmio en la salud. Rev. Espec. Médico-Quirúrgicas 2012, 17, 199–205. [Google Scholar]
- Mancilla-Villa, O.R.; Ortega-Excobar, H.M.; Ramírez-Ayala, C.; Uscanga-Mortera, E.; Ramos-Bello, R.; Reyes-Ortigoza, A.L. Metales pesados totales y arsénico en el agua para riego de Puebla y Veracruz, México. Rev. Int. Contam. Ambient. 2012, 28. [Google Scholar]
- Repetto-Jiménez, M.; Repetto-Kuhn, G. Toxicología fundamental. In Desarrollo y Evolución Histórica de la Toxicología, 4th ed.; Editorial Díaz de Santos: Madrid, Spain, 2009; p. 35. [Google Scholar]
- Nava-Ruíz, C.; Méndez-Armenta, M. Efectos neurotóxicos de metales pesados (cadmio, plomo, arsénico y talio). Arch. Neurocienc. 2011, 16, 140–147. [Google Scholar]
- Reyes, Y.C.; Vergara, I.; Torres, O.E.; Díaz, M.; González, E.E. Contaminación por metales pesados: Implicaciones en salud, ambiente y seguridad alimentaria. Ing. Investig. Desarro. 2016, 16, 66–67. [Google Scholar] [CrossRef]
- Gaur, N.; Flora, G.; Yadav, M.; Tiwari, A. A review with recent advancements on bioremediation-based abolition of heavy metals. Environ. Sci. Process. Impacts 2014, 16, 180. [Google Scholar] [CrossRef]
- Angeletti, R.; Binato, G.; Guidotti, M.; Morelli, S.; Pastorelli, A.A.; Sagratella, E.; Ciardullo, S.; Stacchini, P. Cadmium bioaccumulation in Mediterranean spider crab (Maya squinado): Human consumption and health implications for exposure in Italian population. Chemosphere 2014, 100, 83–88. [Google Scholar] [CrossRef]
- Arroyo, V.; Flores, K.; Ortiz, L.; Gómez-Quioz, L.; Gutiérrez-Ruiz, M.J. Liver and cadmium toxicity. J. Drug Metab. Toxicol. 2012, S5. [Google Scholar]
- Esposito, A.; Pagnanelli, F.; Vegliòa, F. pH-related equilibria models for biosorption in single metal systems. Chem. Eng. Sci. 2002, 57, 307–313. [Google Scholar] [CrossRef]
- Fowle, D.A.; Fein, J.B. Competitive adsorption of metal cations onto two gram positive bacteria: Testing the chemical equilibrium model. Geochim. Cosmochim. Acta 1999, 63, 3059–3067. [Google Scholar] [CrossRef]
- Sulaymon, A.H.; Ebrahim, S.E.; Mohammed-Ridha, M.J. Equilibrium, kinetic and thermodynamic biosorption of Pb (II), Cr (II) and Cd (II) ions by dead anaerobic biomass from synthetic wastewater. Environ. Sci. Pollut. Res. 2013, 20, 175–187. [Google Scholar] [CrossRef]
- Moreira, V.R.; Lebron, Y.A.R.; Lange, L.C.; Santos, L.V.S. Simultaneous biosorption of Cd (II), Ni (II) and Pb (II) onto a brown macroalgae Fucus vesiculosus: Mono- and multi-component isotherms, kinetics and thermodynamics. J. Environ. Manag. 2019, 251, 109587. [Google Scholar]
- Giovanella, P.; Cabral, L.; Costa, A.P.; Camargo, F.A.O.; Gianello, C.; Bento, F.M. Metal resistance mechanisms in Gram-negative bacteria and their potential to remove Hg in the presence of other metals. Ecotoxicol. Environ. Saf. 2017, 140, 162–169. [Google Scholar] [CrossRef]
- Krzmarzick, M.J.; Taylor, D.K.; Fu, X.; McCutchan, A.L. Diversity and Niche of Archaea in Bioremediation. Archaea 2018, 3194108. [Google Scholar] [CrossRef] [Green Version]
- Auernik, K.S.; Cooper, C.R.; Kelly, R.M. Life in hot acid: Pathway analyses in extremely thermoacidophilic archaea. Curr. Opin. Biotechnol. 2008, 19, 445–453. [Google Scholar] [CrossRef] [Green Version]
- Donati, E.R.; Castro, C.; Urbieta, M.S. Thermophilic microorganisms in biomining. World J. Microbiol. Biotechnol. 2016, 32, 179. [Google Scholar] [CrossRef]
- Orell, A.; Remonsellez, F.; Arancibia, R.; Jerez, C.A. Molecular characterization of copper and cadmium resistance determinants in the biomining thermoacidophilic archaeon Sulfolobus metallicus. Archaea 2013, 289236. [Google Scholar] [CrossRef] [Green Version]
- Castillo-Carvajal, L.C.; Sanz-Martín, J.L.; Barragán-Huerta, B.E. Biodegradation of organic pollutants in saline wastewater by halophilic microorganisms: A review. Environ. Sci. Pollut. Res. Int. 2014, 21, 9578–9588. [Google Scholar] [CrossRef]
- Cuadros-Orellana, S.; Pohlschröder, M.; Grossman, M.; Durrant, L. Biodegradation of aromatic compounds by a halophilic archaeon isolated from the dead sea. Chem. Eng. Trans. 2012, 27, 13–18. [Google Scholar] [CrossRef]
- Bonfá, M.R.; Grossman, M.J.; Mellado, E.; Durrant, L.R. Biodegradation of aromatic hydrocarbons by Haloarchaea and their use for the reduction of the chemical oxygen demand of hypersaline petroleum produced water. Chemosphere 2011, 84, 1671–1676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosal, D.; Ghosh, S.; Dutta, T.; Ahn, Y. Current state of knowledge in microbial degradation of polycyclic aromatic hydrocarbons (PAHs): A review. Front. Microbiol. 2016, 7, 1369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arora, S.; Vanza, M.; Mehta, R.; Bhuva, C.; Patel, P. Halophilic microbes for bio-remediation of salt affected soils. Afr. J. Microbiol. Res. 2014, 8, 3070–3078. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Zhou, J.; Li, M.; Xiang, H.; Zhao, D. Insights into the metabolism pathway and functional genes of long-chain aliphatic alkane degradation in haloarchaea. Extremophiles 2020, 24, 475–483. [Google Scholar] [CrossRef]
- Zhao, D.; Kumar, S.; Zhou, J.; Wang, R.; Li, M.; Xiang, H. Isolation and complete genome sequence of Halorientalis hydrocarbonoclasticus sp. nov., a hydrocarbon-degrading haloarchaeon. Extremophiles 2017, 21, 1081–1090. [Google Scholar] [CrossRef]
- Mukherji, S.; Ghosh, A.; Bhattacharyya, C.; Mallick, I.; Bhattacharyya, A.; Mitra, S.; Ghosh, A. Molecular and culture-based surveys of metabolically active hydrocarbon-degrading archaeal communities in Sundarban mangrove sediments. Ecotoxicol. Environ. Saf. 2020, 195, 110481. [Google Scholar] [CrossRef]
- Bader, M.; Müller, K.; Foerstendorf, H.; Schmidt, M.; Simmons, K.; Swanson, J.S.; Reed, D.T.; Stumpf, T.; Cherkouk, A. Comparative analysis of uranium bioassociation with halophilic bacteria and archaea. PLoS ONE 2018, 13, e0190953. [Google Scholar] [CrossRef] [Green Version]
- Bader, M.; Rossberg, A.; Steudtner, R.; Drobot, B.; Großmann, K.; Schmidt, M.; Musat, N.; Stumpf, T.; Ikeda-Ohno, A.; Cherkouk, A. Impact of Haloarchaea on Speciation of Uranium-A Multispectroscopic Approach. Environ. Sci. Technol. 2018, 52, 12895–12904. [Google Scholar] [CrossRef]
- Bader, M.; Moll, H.; Steudtner, R.; Lösch, H.; Drobot, B.; Stumpf, T.; Cherkouk, A. Association of Eu(III) and Cm(III) onto an extremely halophilic archaeon. Environ. Sci. Pollut. Res. Int. 2019, 26, 9352–9364. [Google Scholar] [CrossRef]
- Kiadehi, M.S.H.; Amoozegar, M.A.; Asad, S.; Siroosi, M. Exploring the potential of halophilic archaea for the decolorization of azo dyes. Water Sci. Technol. 2018, 77, 1602–1611. [Google Scholar] [CrossRef]
- Liao, Y.; Williams, T.J.; Walsh, J.C.; Ji, M.; Poljak, A.; Curmi, P.M.; Duggin, I.G.; Cavicchioli, R. Developing a genetic manipulation system for the Antarctic archaeon, Halorubrum lacusprofundi: Investigating acetamidase gene function. Sci. Rep. 2016, 6, 34639. [Google Scholar] [CrossRef]
- Al-Mailem, D.M.; Al-Awadh, H.; Sorkhoh, N.A.; Eliyas, M.; Radwan, S.S. Mercury resistance and volatilization by oil utilizing haloarchaea under hypersaline conditions. Extremophiles 2011, 15, 39–44. [Google Scholar] [CrossRef]
- Abdollahnia, M.; Makhdoumi, A.; Mashreghi, M.; Eshghi, H. Exploring the potentials of halophilic prokaryotes from a solar saltern for synthesizing nanoparticles: The case of silver and selenium. PLoS ONE 2020, 15, e0229886. [Google Scholar] [CrossRef]
- Nieto, J.J.; Fernandez-Castillo, R.; Marquez, M.C.; Ventosa, A.; Quesada, E.; Ruiz-Berraquero, F. Survey of metal tolerance in moderately halophilic Eubacteria. Appl. Environ. Microbiol. 1989, 55, 2385–2390. [Google Scholar] [CrossRef] [Green Version]
- Nieto, J.J.; Ventosa, A.; Ruiz-Berraquero, F. Susceptibility of Halobacteria to heavy metals. Appl. Environ. Microbiol. 1987, 53, 1199–1202. [Google Scholar] [CrossRef] [Green Version]
- Braganca, J.M.; Furtado, I. Resistance of Halobacterium strain R1 to cadmium during growth in mineral salts medium devoid of growth factors. Asian J. Microbiol. Biotechnol. Environ. Sci. 2013, 15, 299–302. [Google Scholar]
- Chaudhary, A.; Pasha, M.I.; Salgaonkar, B.B.; Braganca, J.M. Cadmium Tolerance by Haloarchaeal Strains Isolated from Solar Salterns of Goa, India. Int. J. Biosci. Biochem. Bioinform. 2014, 1. [Google Scholar] [CrossRef]
- Ng, W.V.; Kennedy, S.P.; Mahairas, G.G.; Berquistc, B.; Pana, M.; Shuklac, H.D.; Laskya, S.R.; Baligac, N.S.; Thorssona, V.; Sbrognac, J.; et al. Genome sequence of Halobacterium species NRC-1. Proc. Natl. Acad. Sci. USA 2000, 97, 12176–12181. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Kennedy, S.P.; Fasiludeen, S.; Rensing, C.; DasSarma, S. Arsenic resistance in Halobacterium sp. strain NRC-1 examined by using an improved gene knockout system. J. Bacteriol. 2004, 186, 3187–3194. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, P.; Kowshik, M. Mechanisms of metal resistance and homeostasis in haloarchaea. Archaea 2013, 732864. [Google Scholar] [CrossRef] [Green Version]
- Al-Mailem, D.M.; Eliyas, M.; Radwan, S.S. Ferric sulfate and proline enhance heavy-metal tolerance of halophilic/halotolerant soil microorganisms and their bioremediation potential for spilled-oil under multiple stresses. Front. Microbiol. 2018, 9, 394. [Google Scholar] [CrossRef] [Green Version]
- Bai, J.; Yu, P.; Wen, X.; Wang, W.; Jia, J.; Wang, X. Effects of cadmium addition on net nitrogen mineralization processes in the urban constructed wetland soils of a Chinese delta. Environ. Geochem. Health 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chen, S.; Chen, L.; Wang, D. Saline stress modifies the effect of cadmium toxicity on soil archaeal communities. Ecotoxicol. Environ. Saf. 2019, 182, 109431. [Google Scholar] [CrossRef] [PubMed]
| Metabolic Capacity | Haloraceal Species | Poluant | Concentration | Degradation/ Resistance | Coexisting Compounds | Condition | References |
|---|---|---|---|---|---|---|---|
| Mineralization of aliphatic alkane. Aromatic hydrocarbon/Hydrocarbon degradation | Halorientalis hydrocarbonoclasticus IM1011 Halorientalis sp. | Hexadecane | 5 g/L | Degradation 57% | Eicosane, Duodecane | 3.6 M NaCl; 37 °C; 7/18 days 3.6 M NaCl; 37 °C; 24 days | [105,106] |
| p-Hydroxybenzoic acid degradation | Haloferax sp. Haladaptatus sp. | p-hydroxybenzoic acid | 0.4 mM | Degradation 50% | Naphthalene, Anthracene, Phenanthrene, Pyrene and Benzo[a]anthracene Naphthalene, Benzoic acid, and o-Phthalate | 20% NaCl, 40 °C, 7 days 10% salt medium; 37 °C; 7–14 days | [102,107] |
| Removal of nitrogenous compounds (nitrate and nitrite) | Haloferax mediterranei R4 | Nitrite Nitrate | 40 mM 50 mM | Degradation 75% 60% | Na, Ca, Mg, Chlorides, Sulphates | 25% (w/v) mixture of inorganic salts; 42 °C | [35,37] |
| Uranium biomineralization | Halobacterium noricense DSM 15987 | U (VI) | 30 μM 85 μM | U (IV) carbonate compound formation | - | Modified DSM372 medium; 30 °C; dark conditions; 5 min–360 h | [108,109] |
| Curium and europium complexation | Halobacterium noricense DSM 15987 | Eu (III) Cm (III) | 30 μM 300 nM | Complexation with Eu(III) and Cm(III) by phosphate species and carboxylic groups, respectively | - | DSM372 medium; 30 °C; dark conditions | [110] |
| Decolorization of azo dyes | Halogeometricum sp. strain A Haloferax sp. strain B | Remazol black B (di-azo dye) and Acid blue 161 (mono-azo dye) | 50 mg/L | Degradation 70% Acid blue 161,95% Remazol black B 68% Acid blue 161, 91% Remazol black B | - | MGM broth with 3 M NaCl; 40 °C; 7 days | [111] |
| Acetamide and formamide degradation | Halorubrum lacusprofundi | [112] | |||||
| Heavy metals resistance | Haloferax strain BBK2 | Cd | 0.5 mM 1 mM | Accumulation 21.08% 15.19% | - | Complex (NTYE) or minimal media (NGSM); 5–30% NaCl; 37 °C; 10 days | [60] |
| Heavy metals resistance | Haloferax sp. Halobacterium Halococcus | HgCl2 | 0–300 ppm | Resistance 100 and 200 ppm 200 ppm 100 ppm | - | Solid mineral medium; 0–4 M NaCl; 40 °C for Haloferax and 45 °C for Halobacterium and Halococcus; dark conditions; 10 days | [113,114,115,116,117,118] |
| Synthesis of nanoparticles involving metals (silver) | Haloferax sp. | AgNO3 | 0.5 mM | Intracellular silver nanoparticles formation | - | SW broth medium; 40 °C; 3–7 days | [114] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vera-Bernal, M.; Martínez-Espinosa, R.M. Insights on Cadmium Removal by Bioremediation: The Case of Haloarchaea. Microbiol. Res. 2021, 12, 354-375. https://doi.org/10.3390/microbiolres12020024
Vera-Bernal M, Martínez-Espinosa RM. Insights on Cadmium Removal by Bioremediation: The Case of Haloarchaea. Microbiology Research. 2021; 12(2):354-375. https://doi.org/10.3390/microbiolres12020024
Chicago/Turabian StyleVera-Bernal, Mónica, and Rosa María Martínez-Espinosa. 2021. "Insights on Cadmium Removal by Bioremediation: The Case of Haloarchaea" Microbiology Research 12, no. 2: 354-375. https://doi.org/10.3390/microbiolres12020024
APA StyleVera-Bernal, M., & Martínez-Espinosa, R. M. (2021). Insights on Cadmium Removal by Bioremediation: The Case of Haloarchaea. Microbiology Research, 12(2), 354-375. https://doi.org/10.3390/microbiolres12020024







