Abstract
The valorization study of the largely available corn stover waste biomass after pretreatment with deep eutectic solvent (DES) for biomethane production in one-liter glass bioreactors by anaerobic digestion for 21 days was presented. Ammonium thiocyanate and urea deep eutectic solvent pretreatments under different conditions in terms of the components ratio and temperature were examined on corn stover waste biomass. The lignocellulose biomass was characterized in detail for its chemistry and morphology to determine the effect of the pretreatment on the natural biocomposite. Furthermore, the implications on biomethane production through anaerobic digestion with different loadings of corn stover biomass at 35 g/L and 50 g/L were tested. The results showed an increase of 48% for a cumulative biomethane production for a DES-pretreated biomass, using a solid-to-liquid ratio of 1:2 at 100 °C for 60 min, which is a strong indication that DES-pretreatment significantly enhanced biomethane production.
1. Introduction
The bio-based economy, an emerging concept that advances new uses of bioresources, is one of the main future drivers of sustainable economic growth, a big contributor to the 2030 UN Sustainable development goals agenda, and the focus in the transition to a fossil-free society [1]. In December 2018, a new renewable energy (RE) directive (Directive, 2018/2001/EU) entered into force, setting a target minimum 32% share of RE by 2030 [2]. Agricultural waste and forest residues, as well as municipal waste, are the drivers of biorefinery technology due to their wide availability and accessibility. Still, only in Europe, 60 million tons of organic solid waste is mismanaged and displaced in landfills, which presents a loss in potential energy [3,4]. Anaerobic digestion (AD) is highly preferred for energy generation and waste disposal because of its adaptability and sustainability [5]. After AD of the biomass, two major products are obtained: biogas, consisting mainly of CH4 and CO2 (60–40%), and digestates. In order to obtain biomethane, biogas upgrading—a process of removing CO2—is required. Currently, six main upgrading techniques are used: water scrubbing, physical scrubbing, chemical scrubbing, pressure swing adsorption, membrane technology, and cryogenic separation, all well-analyzed by Carranza-Abid et al. [6]. Further, when CO2 is separated from biomethane, various carbon capture and storage techniques and more sustainable carbon capture and utilization techniques have been proposed. Recently, Baena-Moreno et al. searched for an added-value product from CO2 and obtained CaCO3 through the precipitation of FGD gypsum from a power station [7]. The second product of AD is also known to be good fertilizers for agriculture or horticulture [8]. Rezaee et al. 2020 suggested that these two products would play some significant roles in shaping the future of the circular economy/bioeconomy through resource recovery enhancement and nutrient recycling [9]. The circular economy, which at present is in its infancy, is confronted with some challenges in reaching its full potentials. AD could be a veritable tool in actualizing this goal and could serve as an alternative to the linear or take-make-waste economy approach, which is both exploitative and environmentally unfriendly [10]. Biomethane production via AD has the capability of reducing greenhouse emissions through the utilization of wastes that would otherwise have ended up in the landfill or be burned, which, under uncontrolled natural anaerobic fermentation, could have led to the generation of methane and CO2 (nearly 150 million tons in India) that would be released directly into the atmosphere [11,12]. Furthermore, the use of biofertilizers from the AD process can help in mitigating the adverse effects of the mineral fertilizers on the environment, which includes contamination of groundwater, surface water, and the atmosphere [13].
Various analyses on the economical feasibility of biomethane production, taking into account marketing biogas, as well as digestates, showed that currently, governmental incentives and subsidies play key roles in expanding to the wider market and paving the way to a fossil-free society [2,8,14].
Lignocellulosic materials are recalcitrant, and a barrier to their optimization for bio-based solutions is in the material preparation. Pretreatment is therefore imperative to improve biodegradability, thus making the lignocellulosic materials more amenable for microbial digestion [15]. Many pretreatment methods on raw biomass have been researched, employing mechanical pretreatment, physico-chemical pretreatment, biological pretreatment, chemical pretreatment, and a combination of these methods [16,17,18]. The chemical pretreatment method, in comparison to other methods, has the advantage of easiness, fastness, and efficiency [19]. The physical structures and chemical compositions of lignocellulosic materials could be altered or modified through various chemical pretreatments, making the compositions in lignocellulosic materials more accessible and more readily biodegradable to anaerobic microorganisms, thus increasing digestion efficiency and biogas production [20]. The chemical pretreatment of biomass, recently reported also together with sonification, has been reported to have a positive significant effect on the native structure of lignocellulosic biomass and, thus, their influence on biogas production [17,21]. However, some of these commonly used chemicals, such as acids, alkalis, and organic solvents, have their drawbacks such as the formation of inhibitors, high pretreatment severity, safety concerns, and high energy requirements. The need to mitigate these knotty issues has encouraged research endeavors to develop or produce solvents that would be more environmentally friendly, less toxic or nontoxic, and significantly improve the biomass structure for downstream processes [22]. Ionic liquids (ILs) were preferred to the conventional solvents because of their low melting point, high thermal stability, high reaction rates, and low volatility. The use of ionic liquids for the pretreatment of biomass prior to anaerobic digestion (AD) has been reported to notably increase the production of biogas [23,24]. Some of the commonly used ionic liquids are N-methylmorphine-N-oxide (NMMO), 1-ethyl-3-methylimidazolium acetate [EMIM]-[OAc], and 1-butyl-3-methylimidazolium acetate [BMIM]-[OAc]. Karp et al. [25] noted that the ability of ILs to solubilize polysaccharides in biomass has given them a prominent place among researchers. Padrino et al. [26] reported 28% and 80% increases in methane yield when [BMIM]-[OAc]-pretreated barley was anaerobically digested at mesophilic and thermophilic temperatures, respectively, for 35 days. However, toxicity and high prices have been identified as some of the drawbacks in using ILs for biomass pretreatment [27,28]. As a new class of ILs, deep eutectic solvents (DES) are the preferred alternative for the pretreatment of lignocellulosic biomass in recent times [29]. These green solvents are formed by the selection of appropriate hydrogen bond donors and hydrogen bond acceptors, which are both typically natural compounds, making this type of solvent biocompatible and biodegradable among the most noticeable green factors [30]. Furthermore, its ease of synthesis without the need for purification, as well as its lower cost, recyclability, and environmental benignity, are the most pronounced benefits compared to ILs [31]. DESs have been used in the pretreatment of lignocellulosic biomass with varying degrees of impact on the chemical and physical compositions of the biomass [32,33]. Procentese et al. [22] reported the processing of lettuce leaves with choline chloride–glycerol as an efficient pretreatment in biobutanol production, which is energetically more economical than alkaline treatment. Very recently, Lima et al. [34] studied the pretreatment of lignocellulosic biomass with choline chloride–oxalic acid DES, and demonstrated the positive effect on biogas production at lower concentrations, while concentrations of DES as high as 19.8 g/L showed strong toxic effects. However, to the best of the authors’ knowledge, there are no reports on the use of ammonium thiocyanate:urea DES-pretreated biomass for biomethane production via AD. Therefore, the objectives of this work were to evaluate the physico-chemical properties of ammonium thiocyanate:urea DES-pretreated corn stover, and to determine its biomethane potentials at different loading rates through AD.
2. Materials and Methods
2.1. Materials
Analytical-grade ammonium thiocyanate (NH4SCN) and urea (NH2CONH2) were purchased from Sigma-Aldrich (Darmstadt, Germany). H2SO4 (95–97%) was bought from Merck, and CaCO3 for neutralization from Riedel-de Haën. The chemicals were used as received without further purification.
2.2. Preparation of Deep Eutectic Solvent
Deep eutectic solvent was prepared from ammonium thiocyanate (HBA) with urea (HBD) in molar ratios 1:1 and 1:2, respectively. The components were measured in the desired ratio and placed in a sealed beaker heated on a hotplate with magnetic stirring at the rate of 500 rpm at 80 °C for one hour, when the clear homogeneous solvent was obtained.
2.3. Sample Collection and Preparation
Corn stover (CS) was obtained from Irrua, Esan Central, Edo State, Nigeria. The sample was cleaned of adhering soil, ground, and passed through a 600 micron sieve. The sample was kept in an air-tight plastic bottle at room temperature before pretreatment.
2.4. Deep Eutectic Solvent Pretreatment of Corn Stover
Corn stover was pretreated with DES at 80 and 100 °C for one hour in an oven at solid-to-liquid ratios of 1:2 and 1:4. Then, 5 g and 10 g of the biomass were thoroughly mixed with 20 g of DES. The pretreated samples were cooled to room temperature and washed with water to remove all the DES components, until the pH approached neutral. The washed biomass was then dried at 70 °C for 3 h. The effect of varying temperature, DES molar ratios, and solid-to-liquid ratios on the samples was analyzed to evaluate the structural and chemical changes of the lignocellulosic biomass. In all eight corn stover biomass samples labeled from A to H, with the raw sample used as the control, labeled I, the biomass recovery was calculated as described by Procentese and Rehmann [35].
2.5. Biomass Characterization
2.5.1. Determination of Lignin, Organic Carbon, Ash, and Moisture Content
Acid-soluble lignin (ASL), acid-insoluble lignin (AIL), ash content, and organic carbon content (CHO) were determined according to the standard procedure for the determination of structural carbohydrates and lignin in biomass [36]. Moisture content (MC) was determined with a Moisture Analyzer (Mettler Toledo, Greifensee, Switzerland).
2.5.2. Fourier-Transform Infrared Spectroscopy Analysis
Fourier-transform infrared spectroscopy (FT-IR) was performed with FT-IR Spectrum Two (PerkinElmer, Waltham, Massachusetts, USA). The spectra were recorded at room temperature, with wavenumbers ranging from 4000 cm−1 to 400 cm−1, a resolution of 4 cm−1, and 32 scans being performed for each measurement.
2.5.3. X-ray Powder Diffraction Analysis
Crystallinity was assessed through XRD analysis with the PANanalytical XPert PRO (Malvern Panalytical, UK) high-resolution diffractometer using a Cu-κ1 radiation source (1.5406 Å). The analysis was performed at an energy of 40 kV and electric current of 40 mA, in the 2θ range from 10° to 80° (100 s per step at step size 0.034°). The crystallinity index (CrI) was calculated through the Segal method [37] according to Equation (1):
CrI = (I200 − Iam)/I200 × 100
I200 is the maximum intensity of the crystalline plane (200) at 2θ = 22.8° and Iam is the minimum intensity of the amorphous region that is measured at 2θ = 18°.
2.5.4. Morphology of the Corn Stover Biomass
The samples for SEM imaging were taped onto carbon tape and observed by the scanning electron microscope SUPRA 35VP (Carl Zeiss, Jena, Germany).
2.5.5. Elemental Analysis
The carbon, nitrogen, hydrogen, and sulfur (CHNS) content was determined. Approximately 15 mg of sample was weighted and analyzed using the Elemental Analyzer vario EL Cube (Elementar, Langenselbold, Hesse, Germany). All samples were measured at least three times.
2.6. Calculation of Higher and Lower Heating Values
Higher heating values (HHV) and lower heating values (LHV) of raw and DES-pretreated corn stover were calculated through Equations (2) and (3), as described by [38].
where HHV and LHV are expressed in kJ·kg−1; C, H, O, and S present the weight percentages on a dry basis of carbon, hydrogen, oxygen, and sulfur, respectively, while Mw is a product of the fraction of hydrogen in the sample in 9 kg.
HHV = 337 C + 1428 ((H − O)/8) + 95 S
LHV = HHV − 2465 Mw
2.7. Anaerobic Digestion Process
Based on the physico-chemical properties and morphology of the samples, sample G was used in the production of biomethane in a batch-mode digester at an average ambient temperature of 28 ± 2 °C for a retention time of 21 days. Digesters were one-liter glass reactors labeled accordingly, i.e., I35 and I50 represented raw samples at the feeding regimes of 35 and 50 g/L, respectively, while G35 and G50 represented pretreated samples at the same feeding regimes. Inoculum was obtained from the effluent from a laboratory digester run on plantain peels for 21 days under mesophilic conditions. The inoculum was kept for about two months under anaerobic conditions prior to the time it was used in the anaerobic digestion (AD) process. Each of the digesters was seeded with 30 mL of inoculum. Biomethane productions were determined by the method described by Fernández-Cegrí, Ángeles De la Rubia, Raposo, and Borja [39].
2.8. Statistical Analysis
Statistical analysis was carried out using one-way ANOVA with the confidence level of 95% (p < 0.05) in conjunction with Tukey’s honestly significant difference post hoc test. All experiments were performed in duplicate or triplicate and the results were expressed as the mean ± standard deviation.
3. Results and Discussion
3.1. Effect of Deep Eutectic Solvent Pretreatment on Yield Recovery
The yield recoveries of the DES-treated corn stover samples are shown in Table 1, which ranged from 81.8% to 86.3% with sample H having the lowest value (81.8%) while sample A had the highest (86.3%) followed by sample B (85.6%). The range in our study is higher than the 50% reported by Procentese and Rehmann [35] for coffee silverskin (CS) pretreated with choline chloride/glycerol DES at 150 °C for three hours.

Table 1.
Pretreatment conditions and the obtained yield of the recovered DES-treated corn stover.
3.2. Phsicochemical Properties of Raw and Deep Eutectic Solvent Pretreated Samples
The physicochemical properties of raw (I) and pretreated samples (A–G) are shown in Table 2.

Table 2.
Pretreatment conditions and obtained yield of the recovered DES-treated corn stover.
The ash content of pretreated samples was lower than that of the raw sample, which could be an indication of the high solubility of inorganic compounds in ammonium thiocyanate:urea deep eutectic solvents [40]. The organic carbon contents of all the pretreated samples were all significantly higher than those of the raw samples. The AIL ranged from 12.84% to 17.31% with sample A having the highest while sample G had the lowest value. These values are lower than the 21.7% reported for corn stover by Zhu et al. [23]. For the ASL, sample A recorded the lowest value of 1.00%, while samples D and G had the highest values of 1.63% and 1.62%, respectively. The values for ASL from samples D and G are higher than the 1.5% reported for wheat straw by [41]. The AIL degradation ranged from 2.35% to 23.53%. Sample G had the highest lignin fraction reduction while sample C had the lowest (Table 3). On the other hand, samples A and E had an increase in lignin fraction after pretreatment; this observed phenomenon has been attributed to the formation of pseudo-lignin during pretreatment [42]. A decrease in AIL could improve the biogas production capability of the samples as the lignin-carbohydrates complex would have been broken, making it more amenable to enzymatic attack [43]. Regarding MC, TS, and VC, only small and inconsistent changes were detected; therefore, it is impossible to make a reliable conclusion about the effect of DES pretreatment on these parameters. The statistical analysis using one-way ANOVA showed statistical significance of the results in Table 2 (p < 0.05). Further analysis with Tukey’s honestly significant difference post hoc test revealed significant differences of the treated samples compared to the initial sample I shown in Table 2.

Table 3.
Degradation of acid-insoluble and acid-soluble lignin after DES pretreatment.
The ASL degradation for samples A, B, and C ranged from 5.15% to 26.47% with sample A having the highest followed by sample B with 7.35%. On the other hand, samples C, D, F, G, and H had an increase in ASL after pretreatment. Table 2 revealed that there was an increase in carbohydrate contents of the samples after pretreatment except for samples D and E.
3.2.1. Elemental Analysis
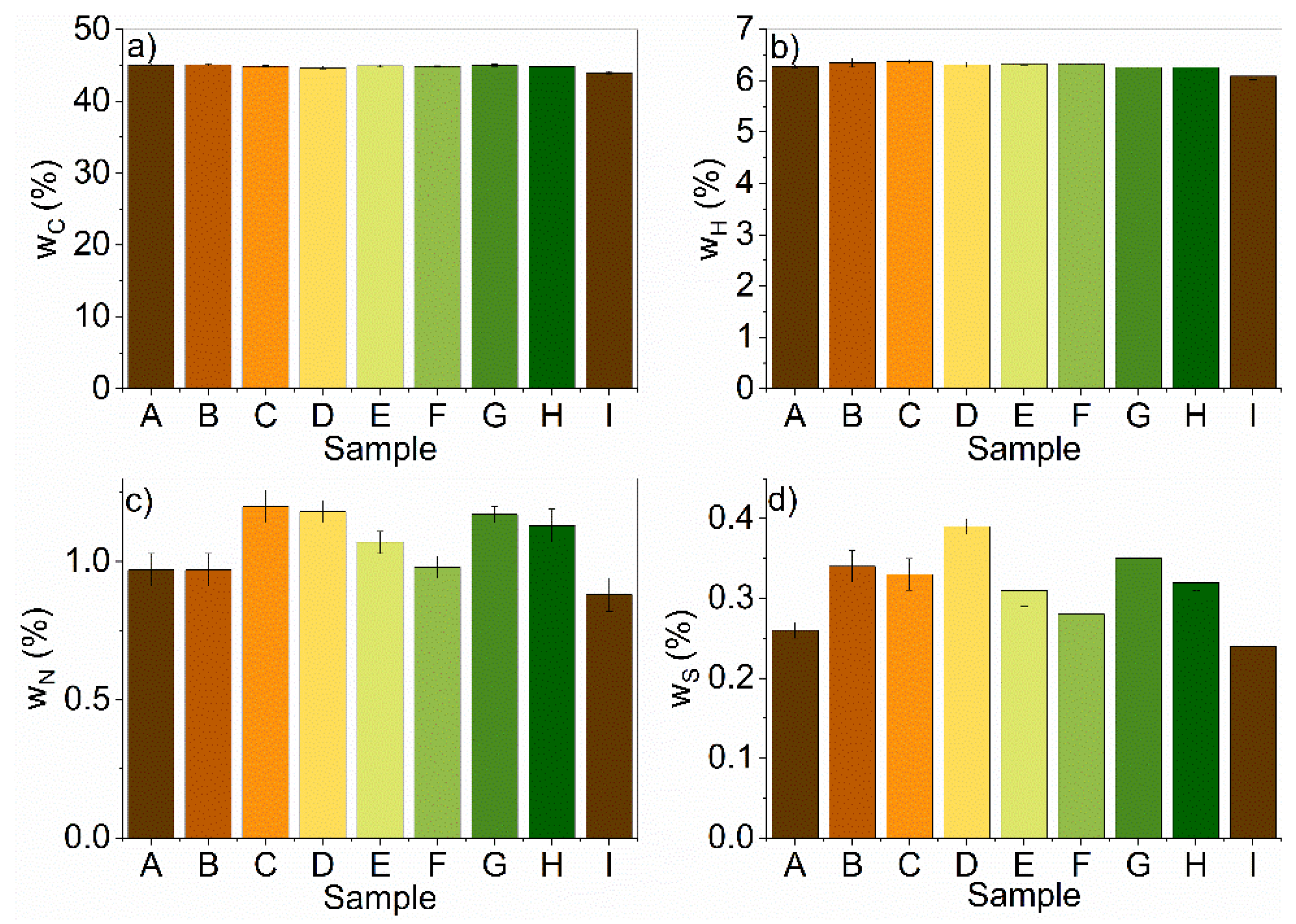
Elemental analysis of the samples revealed that carbon, hydrogen, and sulfur contents increased with treatment, unrelated to the treatment conditions, while the nitrogen content decreased (Figure 1a–d). The decrease in nitrogen could be attributed to the removal of proteins, where treatments with an A:U ratio of 1:1 and a temperature of 80 °C were most efficient. Moreover, the removal of nitrogen can influence the composition ratios between other elements as well.
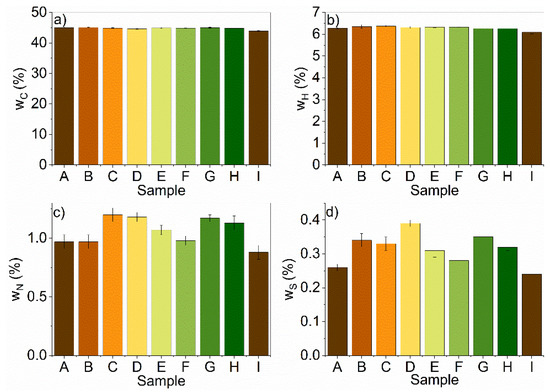
Figure 1.
Elemental analysis of the pretreated (A–H) and untreated (I) samples for (a) carbon (C); (b) hydrogen (H); (c) nitrogen (N); (d) sulfur (S).
The nitrogen content values ranged between 0.88% and 1.20% with the raw sample having the lowest value while sample C had the highest value. There was a significant increase in nitrogen content after DES pretreatment, due to the selective removal of the nitrogen-rich materials (proteins) form the corn stover biomass. The C/N ratio was calculated, which ranged from 37.39% to 49.95%. This range was higher by 15–30%, which was recommended for the AD [44].
3.2.2. Fourier-Transform Infrared Spectroscopy Analysis
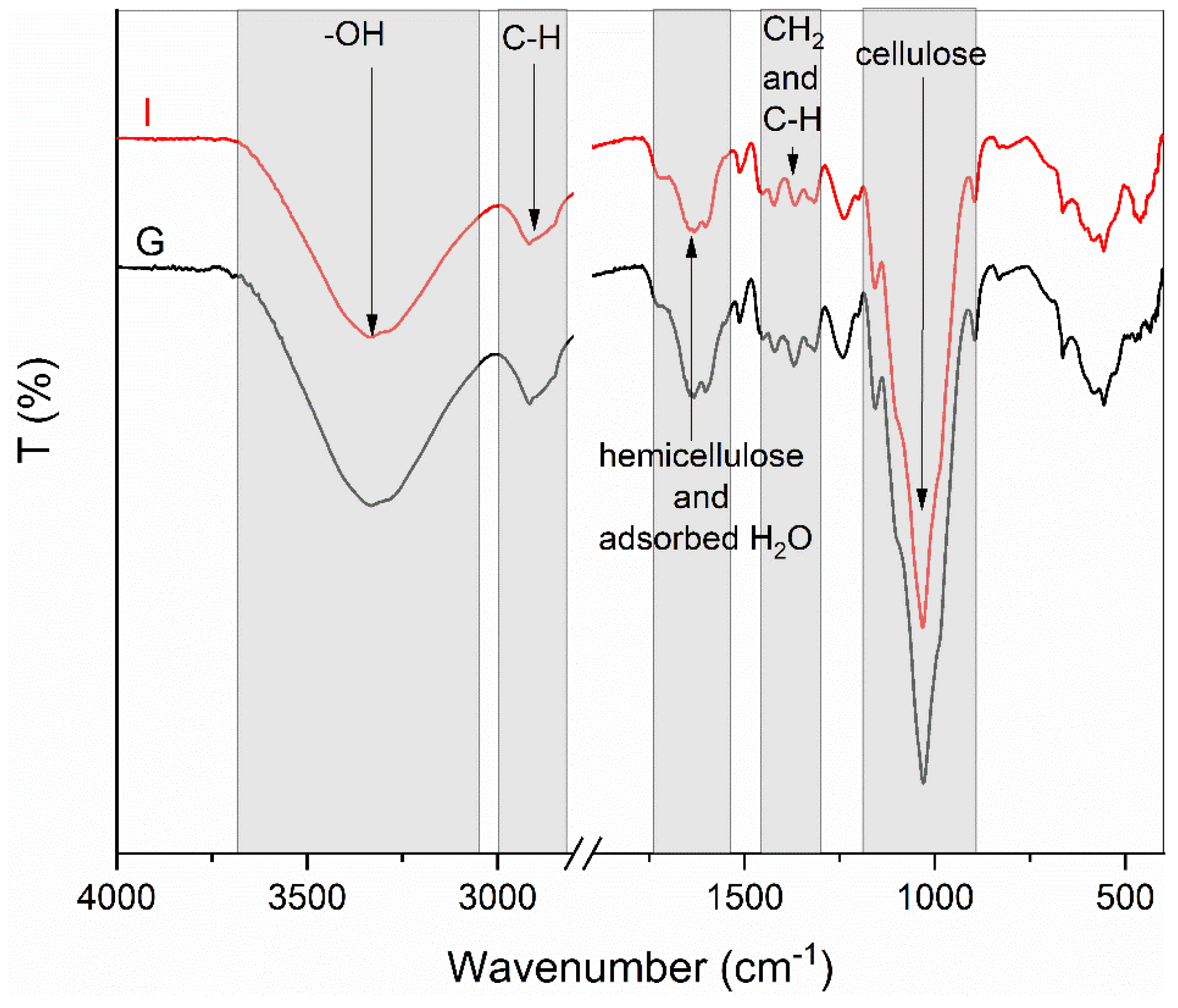
FT-IR analysis revealed the same structural characteristics in all samples. In Figure 2, the spectra of the untreated sample (I) and sample (G) are presented. The broadbands observed at the wavelength of 3330 cm−1 are characteristic for the stretching vibration of the hydroxyl group in polysaccharides. The band at 2900 cm−1 is attributed to the stretching vibration of the C–H bond. The adsorption bands between 1756 cm−1 and 1546 cm−1 are associated with the hemicellulose complex [45]. The stretching in the range from 1700 cm−1 to 1550 cm−1 may be correlated to the water adsorption. The bands at 1418 cm−1, 1369 cm−1, and 1319 cm−1 are attributed to CH2, in-plane CH deformation, and CH2 wagging, respectively. The bands at wavelengths 1156 cm−1 and 1029 cm−1 are characteristic for cellulose and are associated with the asymmetric C–O–C bridge stretching of the anhydroglucose ring and C–O–C pyranose ring skeletal vibration that are present in cellulose [46,47].
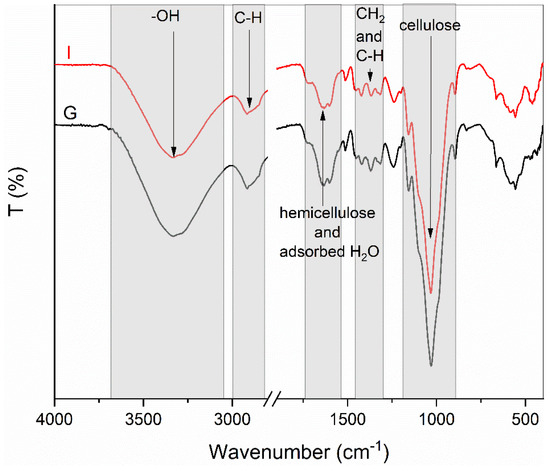
Figure 2.
FT-IR spectra of pretreated (A–H) and raw (I) samples, grouped according to the pretreatment temperature.
3.2.3. X-ray Powder Diffraction Analysis
In XRD spectra, peaks for different crystal planes characteristic of cellulose I were observed. The peaks at 14.9° and 16.7°, which represent (110) and (110) planes, respectively, are overlapped in all spectra, which would point to a diamond-shaped cross-section of the crystallites [48]. There were no major differences in the characteristics of the XRD spectra of samples, which point to the fact that cellulose does not undergo structural changes during pretreatment. No shift in peaks was detected, which means that cellulose I did not transition to cellulose II during the dissolution [49]. The crystallinity indexes (CrI) of the treated and untreated samples are presented in Table 3. The CrI of the untreated material was 54.9%. It was observed that samples treated with DES had some increase in crystallinity except samples A and C. The highest CrI was reached by sample F with 62.1% (Table 4). The increase in CrI is in agreement with the results reported for cotton stalk pretreated with different pretreatment methods by Zhang et al. [50]. They proposed that the increase was a result of the removal of the noncrystalline portions (amorphous regions) of the samples by the DES pretreatments and may not necessarily mean an increase in cellulose crystallinity.

Table 4.
Crystallinity indexes of treated and untreated samples.
3.2.4. Morphology
The samples were observed with a scanning electron microscope. Regarding morphology, no differences were observed between samples. In Figure 3, untreated sample (I) and sample B are presented. Both samples appeared rod-shaped with a rough surface and were structured out of thinner, fiber-like particles.

Figure 3.
SEM images of: (a,b) surface of untreated sample I, (c) treated sample B; (d–g) surface and morphology of sample G, which was used for further production of biomethane.
Furthermore, sample G was more thoroughly investigated as it was used for the production of biogas. The micrographs of sample G are presented in Figure 3d–g. It is possible to observe that the samples consisted of fiber-like particles, but their surface was smoother than in sample B. The surface seemed more cracked than in the untreated sample.
3.3. Determination of Higher and Lower Heating Values
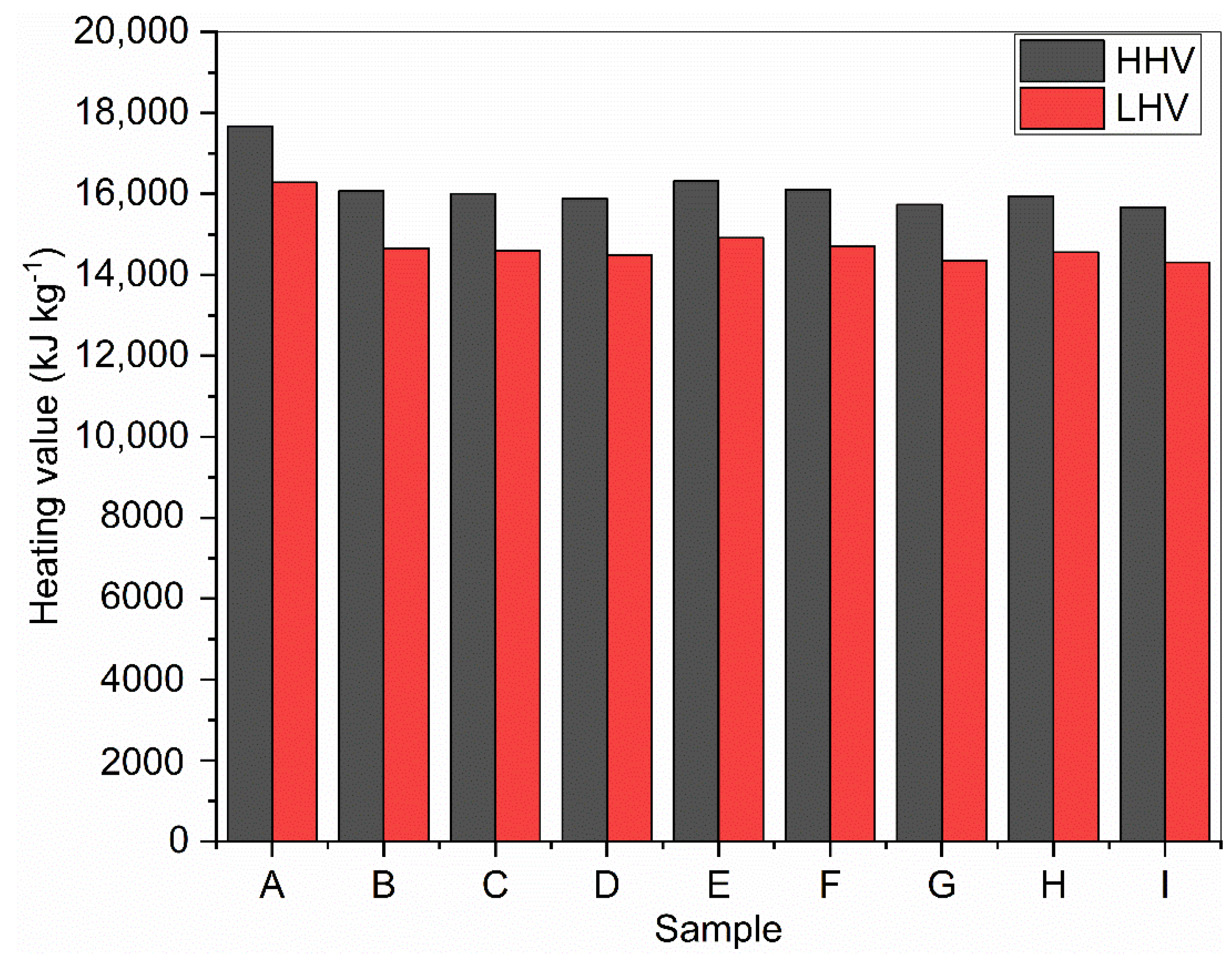
Using Equations (2) and (3), the calculation of HHV and LHV for corn stover biomass samples was performed. HHV values contain the latent heat of the water vapor products of combustion, while the LHV represents the correction to HHV due to moisture in the fuel (biomass) or water vapor formed during the combustion of hydrogen in the fuel [51]. The calculated values for higher and lower heating values are presented in Figure 4. The higher heating values ranged from 15,659 to 17,676 kJ kg−1, while the lower heating values ranged from 14,308 to 16,282 kJ kg−1. The two samples (A and E) with the highest lignin contents had the highest values for both HHV and LHV (17,676 kJ kg−1 and 16,282 kJ kg−1; 16,321 kJ kg−1 and 14,917 kJ kg−1). A direct correlation between lignin content and HHV has been already reported in the literature [52]. The HHV for sample A was comparable to 17,530 kJ kg−1 reported for palm oil mill effluent (POME), while its LHV is higher than 13,872 kJ kg−1 reported for the same sample of POME by Jekayinfa and Omisakin [53].
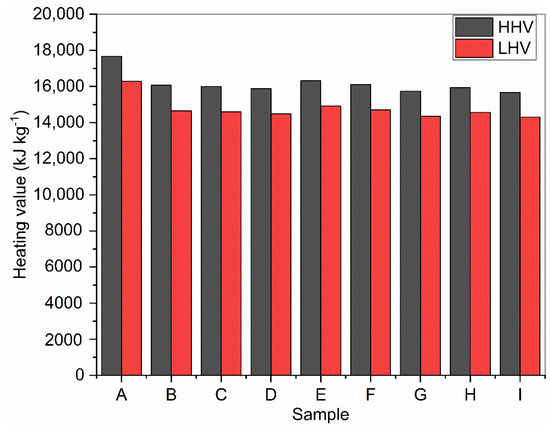
Figure 4.
Calculated higher and lower heating values of raw and DES-pretreated corn stover.
3.4. Daily and Cumulative Biomethane Production
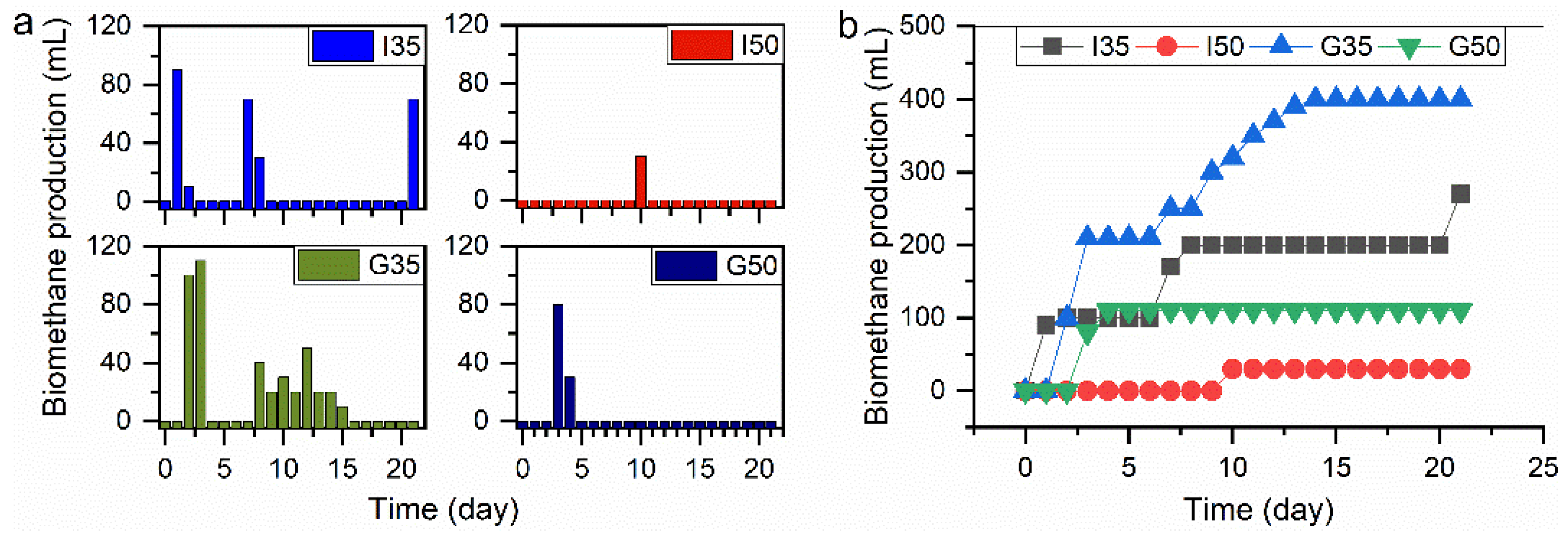
Daily biomethane production is shown in Figure 5a for raw samples I and DES-pretreated samples G in digesters with 35 g/L and 50 g/L of biomass loadings. The digester I35 production commenced on day 1, while in digester I50, there was no production recorded until day 10, when the production reached 30 mL. For the DES-pretreated digesters, G35 had started its production one day after I35, while digester G50 experienced a delay in production for two days compared to I35. The latter had its peak value of 90 mL on day 1; digester G35 attained its peak value of 110 mL on day 3, while G50 recorded 80 mL on day 3.
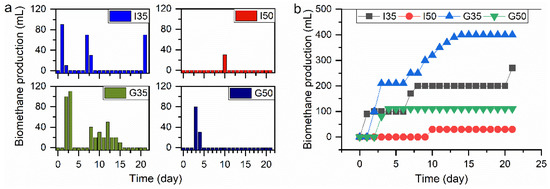
Figure 5.
(a) Daily biomethane production from raw and DES-pretreated corn stover and (b) the cumulative biomethane production from raw and DES-pretreated corn stover.
Generally, there was a fast acclimation of microorganisms in digesters to the substrates, as evident in the lag phase day(s), except digesters G50 with a lag phase of two days. Digester I35 had no lag phase, while digester G35 had a lag phase of one day. Lag phase is an important factor in AD because it shows the rapidity with which the microorganisms acclimatize to the substrates, and from an economic point of view, the less lag phase that is present, the more beneficial it is for the AD process. The cumulative biomethane production is shown in Figure 5b, which revealed that a lower feeding regime produced more biogas than the higher feeding regime. It is imperative to determine the appropriate feeding regime for any biomass to be used for biogas production as there is no fit-it-all feeding regime for potential feedstocks; therefore, each has to be treated on its own merit.
The biomethane production of digester G35 was 48% higher than its counterpart (I35), a strong indication that DES-pretreatment significantly enhanced biomethane production at this feeding regime. This was due largely to the changes in the physical and chemical composition of the substrate after treatment with ammonium thiocyanate-urea-based DES at 100 °C (G). The percentage increase of 48% recorded in this study is higher than the previously reported 15.5% increase for wheat plant pretreated with dilute sulfuric acid by Taherdanak, Zilouei, and Karimi [54].
Digester G50 had a lower biomethane yield than I35 did, which showed that the 35 g L−1 feeding regime was better for biomethane production for the DES-pretreated sample. The lower feeding regime performed better in both the raw and DES-pretreated digesters. Digester I50 produced only 30 mL of biomethane and, afterward, there was no production until the end of the experiment (day 21). It has been reported previously that overloading a digester could lead to low methane yield or digester failure due to the accumulation of volatile fatty acid [55]. Although the complete study of the DES pretreatment effects on the production of biomethane is not shown, the collected experimental data showed a high potential for further exploration on the topic of waste corn stover biomethane production employing AD.
The values obtained by different DES treatments of corn stover in Table 2 are statistically significant (p < 0.005); however, due to the single measurement of the anaerobic corn stover, digestion cannot be claimed the same with a high degree of certainty; therefore, further studies on that topic also including the techno economics should be considered.
4. Conclusions
The pretreatment of corn stover with ammonium thiocyanate:urea DES under varying conditions revealed changes in physicochemical and structural properties compared to the untreated sample. The implications of these changes on biomethane production from the samples were discussed. The most promising of the samples was used in a novel attempt to determine the effect of ammonium thiocyanate:urea-based DES pretreatment on the biomethane potential of corn stover at two feeding regimes of 35 g L−1 and 50 g L−1. DES pretreatment significantly enhanced methane production at a 35 g L−1 loading rate with a 48% increase over the untreated sample.
In the future, a larger study of the DES pretreatment effects on the production of biomethane should be carried out, as the collected experimental data showed a high potential for further exploration on the topic of waste corn stover biomethane production employing AD, in addition to the study of digestate composition, its utilization, and its role in resources circulation. Furthermore, there is a need to explore other DESs for the pretreatment of different substrates in order to evaluate their biomethane potentials and optimize the process efficiency for an improved economy of AD.
Author Contributions
Conceptualization, A.D.O. and B.L.; methodology, A.O. and U.N.; formal analysis, A.D.O., A.O. and U.N.; investigation, A.D.O., A.O. and U.N.; resources, A.D.O., U.N. and B.L.; data curation, A.D.O., A.O. and U.N.; writing—original draft preparation, A.D.O. and A.O.; writing—review and editing, A.D.O., U.N. and B.L.; visualization, A.O.; supervision, U.N. and B.L.; funding acquisition, A.D.O. and B.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ph.D. research grant (Ana Oberlintner) and Slovenian Research Agency (Program P2-0152).
Acknowledgments
The authors wish to thank Joshua Isaac Oludiran and his colleagues for their technical support. Anže Prašnikar is acknowledged for preparation of the SEM images.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bennich, T.; Belyazid, S.; Stjernquist, I.; Diemer, A.; Seifollahi-Aghmiuni, S.; Kalantari, Z. The bio-based economy, 2030 Agenda, and strong sustainability—A regional-scale assessment of sustainability goal interactions. J. Clean. Prod. 2021, 283, 125174. [Google Scholar] [CrossRef]
- D’Adamo, I.; Falcone, P.M.; Gastaldi, M.; Morone, P. RES-T trajectories and an integrated SWOT-AHP analysis for biomethane. Policy implications to support a green revolution in European transport. Energy Policy 2020, 138. [Google Scholar] [CrossRef]
- Hladnik, L.; Vicente, F.A.; Novak, U.; Grilc, M.; Likozar, B. Solubility assessment of lignin monomeric compounds and organosolv lignin in deep eutectic solvents using in situ Fourier-transform infrared spectroscopy. Ind. Crop. Prod. 2021, 164, 113359. [Google Scholar] [CrossRef]
- Kohli, K.; Prajapati, R.; Sharma, B.K. Bio-based chemicals from renewable biomass for integrated biorefineries. Energies 2019, 12, 233. [Google Scholar] [CrossRef] [Green Version]
- Wainaina, S.; Awasthi, M.K.; Sarsaiya, S.; Chen, H.; Singh, E.; Kumar, A.; Ravindran, B.; Awasthi, S.K.; Liu, T.; Duan, Y.; et al. Resource recovery and circular economy from organic solid waste using aerobic and anaerobic digestion technologies. Bioresour. Technol. 2020, 301, 122778. [Google Scholar] [CrossRef] [PubMed]
- Carranza-Abaid, A.; Wanderley, R.R.; Knuutila, H.K.; Jakobsen, J.P. Analysis and selection of optimal solvent-based technologies for biogas upgrading. Fuel 2021, 303, 121327. [Google Scholar] [CrossRef]
- Baena-Moreno, F.M.; le Saché, E.; Price, C.A.H.; Reina, T.R.; Navarrete, B. From biogas upgrading to CO2 utilization and waste recycling: A novel circular economy approach. J. CO2 Util. 2021, 47, 101496. [Google Scholar] [CrossRef]
- D’Adamo, I.; Falcone, P.M.; Huisingh, D.; Morone, P. A circular economy model based on biomethane: What are the opportunities for the municipality of Rome and beyond? Renew. Energy 2021, 163, 1660–1672. [Google Scholar] [CrossRef]
- Rezaee, M.; Gitipour, S.; Sarrafzadeh, M.-H. Different pathways to integrate anaerobic digestion and thermochemical processes: Moving toward the circular economy concept. Environ. Energy Econ. Res. 2020, 4, 57–67. [Google Scholar] [CrossRef]
- Rodríguez, R.W.; Pomponi, F.; Webster, K.; D’Amico, B. The future of the circular economy and the circular economy of the future. Built Environ. Proj. Asset Manag. 2020, 10, 529–546. [Google Scholar] [CrossRef]
- Selvaggi, R.; Valenti, F.; Pecorino, B.; Porto, S.M.C. Assessment of tomato peels suitable for producing biomethane within the context of circular economy: A GIS-based model analysis. Sustainability 2021, 13, 5559. [Google Scholar] [CrossRef]
- Porichha, G.K.; Hu, Y.; Rao, K.T.; Xu, C.C. Crop residue management in India: Stubble burning vs. other utilizations including bioenergy. Energies 2021, 14, 4281. [Google Scholar] [CrossRef]
- Suhartini, S.; Heaven, S.; Banks, C.J. Can anaerobic digestion of sugar beet pulp support the circular economy? A study of biogas and nutrient potential. IOP Conf. Series Earth Environ. Sci. 2018, 131, 012048. [Google Scholar] [CrossRef] [Green Version]
- Baena-Moreno, F.M.; Malico, I.; Marques, I.P. Promoting sustainability: Wastewater treatment plants as a source of biomethane in regions far from a high-pressure grid. A real Portuguese case study. Sustainability 2021, 13, 8933. [Google Scholar] [CrossRef]
- Mirmohamadsadeghi, S.; Karimi, K.; Azarbaijani, R.; Yeganeh, L.P.; Angelidaki, I.; Nizami, A.-S.; Bhat, R.; Dashora, K.; Vijay, V.K.; Aghbashlo, M.; et al. Pretreatment of lignocelluloses for enhanced biogas production: A review on influencing mechanisms and the importance of microbial diversity. Renew. Sustain. Energy Rev. 2021, 135, 110173. [Google Scholar] [CrossRef]
- Thompson, D.N.; Campbell, T.; Bals, B.; Runge, T.; Teymouri, F.; Ovard, L.P. Chemical preconversion: Application of low-severity pretreatment chemistries for commoditization of lignocellulosic feedstock. Biofuels 2013, 4, 323–340. [Google Scholar] [CrossRef]
- Agbor, V.B.; Cicek, N.; Sparling, R.; Berlin, A.; Levin, D.B. Biomass pretreatment: Fundamentals toward application. Biotechnol. Adv. 2011, 29, 675–685. [Google Scholar] [CrossRef]
- Isikgor, F.H.; Becer, C.R. Lignocellulosic biomass: A sustainable platform for the production of bio-based chemicals and polymers. Polym. Chem. 2015, 6, 4497–4559. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Beltrán, J.U.; Hernández-De Lira, I.; Cruz-Santos, M.; Saucedo-Luevanos, A.; Hernández-Terán, F.; Balagurusamy, N. Insight into Pretreatment Methods of Lignocellulosic Biomass to Increase Biogas Yield: Current State, Challenges, and Opportunities. Appl. Sci. 2019, 9, 3721. [Google Scholar] [CrossRef] [Green Version]
- Ariunbaatar, J.; Panico, A.; Esposito, G.; Pirozzi, F.; Lens, P.N.L. Pretreatment methods to enhance anaerobic digestion of organic solid waste. Appl. Energy 2014, 123, 143–156. [Google Scholar] [CrossRef]
- Ouahabi, Y.R.; Bensadok, K.; Ouahabi, A. Optimization of the biomethane production process by anaerobic digestion of wheat straw using chemical pretreatments coupled with ultrasonic disintegration. Sustainability 2021, 13, 7202. [Google Scholar] [CrossRef]
- Procentese, A.; Raganati, F.; Olivieri, G.; Russo, M.E.; Rehmann, L.; Marzocchella, A. Low-energy biomass pretreatment with deep eutectic solvents for bio-butanol production. Bioresour. Technol. 2017, 243, 464–473. [Google Scholar] [CrossRef]
- Zhu, J.; Wan, C.; Li, Y. Enhanced solid-state anaerobic digestion of corn stover by alkaline pretreatment. Bioresour. Technol. 2010, 101, 7523–7528. [Google Scholar] [CrossRef]
- Gao, J.; Chen, L.; Yuan, K.; Huang, H.; Yan, Z. Ionic liquid pretreatment to enhance the anaerobic digestion of lignocellulosic biomass. Bioresour. Technol. 2013, 150, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Karp, E.M.; Donohoe, B.S.; O’Brien, M.H.; Ciesielski, P.N.; Mittal, A.; Biddy, M.J.; Beckham, G.T. Alkaline pretreatment of corn stover: Bench-scale fractionation and stream characterization. ACS Sustain. Chem. Eng. 2014, 2, 1481–1491. [Google Scholar] [CrossRef]
- Padrino, B.; Serrano, M.L.; Morales-Delarosa, S.; Campos-Martín, J.M.; Fierro, J.L.G.; Martínez, F.; Melero, J.A.; Puyol, D. resource recovery potential from lignocellulosic feedstock upon lysis with ionic liquids. Front. Bioeng. Biotechnol. 2018, 6, 119. [Google Scholar] [CrossRef] [PubMed]
- Grilc, M.; Likozar, B.; Levec, J. Kinetic model of homogeneous lignocellulosic biomass solvolysis in glycerol and imidazolium-based ionic liquids with subsequent heterogeneous hydrodeoxygenation over NiMo/Al2O3 catalyst. Catal. Today 2015, 256, 302–314. [Google Scholar] [CrossRef]
- Weerachanchai, P.; Lee, J.-M. Effect of organic solvent in ionic liquid on biomass pretreatment. ACS Sustain. Chem. Eng. 2013, 1, 894–902. [Google Scholar] [CrossRef]
- Bjelić, A.; Hočevar, B.; Grilc, M.; Novak, U.; Likozar, B. A review of sustainable lignocellulose biorefining applying (natural) deep eutectic solvents (DESs) for separations, catalysis and enzymatic biotransformation processes. Rev. Chem. Eng. 2020, 2019077. [Google Scholar] [CrossRef]
- Elgharbawy, A.A.M.; Hayyan, M.; Hayyan, A.; Basirun, W.J.; Salleh, H.M.; Mirghani, M.E.S. A grand avenue to integrate deep eutectic solvents into biomass processing. Biomass Bioenergy 2020, 137, 105550. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep eutectic solvents (DESs) and their applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, R.; Rahman, S.; Raynie, D.E. Recent advances of greener pretreatment technologies of lignocellulose. Curr. Res. Green Sustain. Chem. 2020, 3, 100035. [Google Scholar] [CrossRef]
- Xue, B.; Yang, Y.; Tang, R.; Xue, D.; Sun, Y.; Li, X. Efficient dissolution of lignin in novel ternary deep eutectic solvents and its application in polyurethane. Int. J. Biol. Macromol. 2020, 164, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Lima, F.; Branco, L.C.; Lapa, N.; Marrucho, I.M. Beneficial and detrimental effects of choline chloride–oxalic acid deep eutectic solvent on biogas production. Waste Manag. 2021, 131, 368–375. [Google Scholar] [CrossRef]
- Procentese, A.; Rehmann, L. Fermentable sugar production from a coffee processing by-product after deep eutectic solvent pretreatment. Bioresour. Technol. Rep. 2018, 4, 174–180. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass; NREL/TP-510-42618; National Renewable Energy Laboratory: Golden, CO, USA, 2004. [Google Scholar]
- Segal, L.; Creely, J.J.; Martin, A.E.; Conrad, C.M. An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text. Res. J. 1959, 29, 786–794. [Google Scholar] [CrossRef]
- Podolski, W.F.; Conrad, V.; Lowenhaupt, D.E.; Winschel, R.A. Materials of construction. In Perry’s Chemical Engineers’ Handbook; Perry, R.H., Green, D.W., Eds.; The McGraw-Hill Companies, Inc.: New York, NY, USA, 2008; pp. 24–25. [Google Scholar] [CrossRef]
- Fernández-Cegrí, V.; de la Rubia, M.A.; Raposo, F.; Borja, R. Effect of hydrothermal pretreatment of sunflower oil cake on biomethane potential focusing on fibre composition. Bioresour. Technol. 2012, 123, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Suopajärvi, T.; Ricci, P.; Karvonen, V.; Ottolina, G.; Liimatainen, H. Acidic and alkaline deep eutectic solvents in delignification and nanofibrillation of corn stalk, wheat straw, and rapeseed stem residues. Ind. Crop. Prod. 2020, 145, 111956. [Google Scholar] [CrossRef]
- Zhang, X.; Nghiem, N.P. Pretreatment and fractionation of wheat straw for production of fuel ethanol and value-added co-products in a biorefinery. AIMS Bioeng. 2014, 1, 40–52. [Google Scholar] [CrossRef]
- Mulat, D.G.; Dibdiakova, J.; Horn, S.J. Microbial biogas production from hydrolysis lignin: Insight into lignin structural changes. Biotechnol. Biofuels 2018, 11, 61. [Google Scholar] [CrossRef] [Green Version]
- Sasmal, S.; Mohanty, K. Pretreatment of lignocellulosic biomass toward biofuel production. In Biorefining of Biomass to Biofuels; Springer: Cham, Switzerland, 2018; pp. 203–221. [Google Scholar]
- Petravić-Tominac, V.; Nastav, N.; Buljubašić, M.; Šantek, B. Current state of biogas production in Croatia. Energy Sustain. Soc. 2020, 10. [Google Scholar] [CrossRef]
- Kubovský, I.; Kačíková, D.; Kačík, F. Structural changes of oak wood main components caused by thermal modification. Polymers 2020, 12, 485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poletto, M.; Pistor, V.; Zeni, M.; Zattera, A.J. Crystalline properties and decomposition kinetics of cellulose fibers in wood pulp obtained by two pulping processes. Polym. Degrad. Stab. 2011, 96, 679–685. [Google Scholar] [CrossRef]
- Lao, W.; Li, G.; Zhou, Q.; Qin, T. Quantitative analysis of biomass in three types of wood-plastic composites by FTIR spectroscopy. Bioresources 2014, 9, 6073–6086. [Google Scholar] [CrossRef] [Green Version]
- Duchemin, B. Size, shape, orientation and crystallinity of cellulose Iβ by X-ray powder diffraction using a free spreadsheet program. Cellulose 2017, 24. [Google Scholar] [CrossRef]
- Moyer, P.; Kim, K.; Abdoulmoumine, N.; Chmely, S.; Long, B.K.; Carrier, D.J.; Labbé, N. Structural changes in lignocellulosic biomass during activation with ionic liquids comprising 3-methylimidazolium cations and carboxylate anions. Biotechnol. Biofuels 2018, 11, 265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ning, Z.; Khalid, H.; Zhang, R.; Liu, G.; Chen, C. Enhancement of methane production from Cotton Stalk using different pretreatment techniques. Sci. Rep. 2018, 8, 3463. [Google Scholar] [CrossRef] [Green Version]
- Demirbas, B. Biomass business and operating. Energy Educ. Sci. Technol. Part A 2010, 26, 37–47. [Google Scholar]
- Sujan, S.; Kashem, M.; Fakhruddin, A. Lignin: A valuable feedstock for biomass pellet. Bangladesh J. Sci. Ind. Res. 2020, 55, 83–88. [Google Scholar] [CrossRef]
- Jekayinfa, S.O.; Omisakin, O.S. The energy potentials of some agricultural wastes as local fuel materials in Nigeria. Agric. Eng. Int. CIGR EJ. 2005, 7, 1–10. [Google Scholar]
- Taherdanak, M.; Zilouei, H.; Karimi, K. The influence of dilute sulfuric acid pretreatment on biogas production form wheat plant. Int. J. Green Energy 2016, 13, 1129–1134. [Google Scholar] [CrossRef]
- Mirmohamadsadeghi, S.; Karimi, K.; Horváth, I.S. Improvement of solid-state biogas production from wood by concentrated phosphoric acid pretreatment. Bioresources 2016, 11, 3230–3243. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).