Preliminary Study of Bioelectricity Generation Using Lettuce Waste as Substrate by Microbial Fuel Cells
Abstract
:1. Introduction
2. Materials and Methods
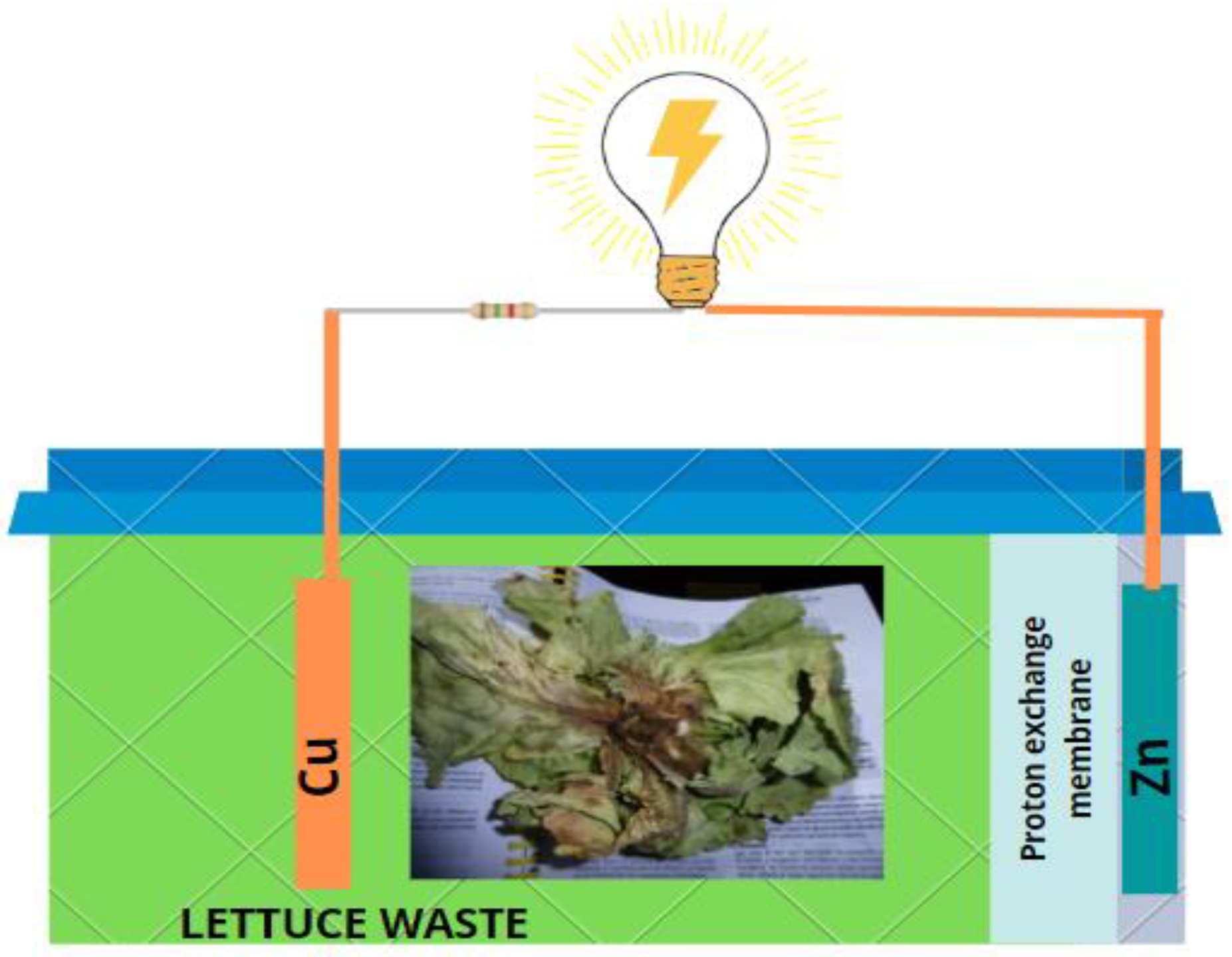
2.1. Design and Construction of scMFCs
2.2. Samples of Lettuce Waste
2.3. Characterization of Microbial Fuel Cells
2.4. Isolation of Anodic Microbes
2.5. Molecular Identification
2.6. Statistics Analysis
3. Results and Analysis
3.1. Electrical Parameters Measurement
3.2. Isolation and Identification of the Electrogenic Microorganisms
3.3. Bioelectricity Generation from Lettuce Waste by scMFC
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ganesh, K.S.; Sridhar, A.; Vishali, S. Utilization of Fruit and Vegetable Waste to Produce Value-Added Products: Conventional Utilization and Emerging Opportunities-A Review. Chemosphere 2022, 287, 132221. [Google Scholar] [CrossRef] [PubMed]
- United Nations World Population Prospects 2019: Highlights|Naciones Unidas. Available online: https://www.un.org/es/desa/world-population-prospects-2019-highlights (accessed on 25 May 2023).
- Rifna, E.J.; Misra, N.N.; Dwivedi, M. Recent Advances in Extraction Technologies for Recovery of Bioactive Compounds Derived from Fruit and Vegetable Waste Peels: A Review. Crit. Rev. Food Sci. Nutr. 2023, 63, 719–752. [Google Scholar] [CrossRef] [PubMed]
- Mortula, M.M.; Ahmed, A.; Fattah, K.P.; Zannerni, G.; Shah, S.A.; Sharaby, A.M. Sustainable Management of Organic Wastes in Sharjah, UAE through Co-Composting. Methods Protoc. 2020, 3, 76. [Google Scholar] [CrossRef] [PubMed]
- Introduction—Food Loss. Available online: http://www.fao.org/platform-food-loss-waste/food-loss/introduction/en/ (accessed on 25 May 2023).
- Salihoglu, G.; Salihoglu, N.K.; Ucaroglu, S.; Banar, M. Food Loss and Waste Management in Turkey. Bioresour. Technol. 2018, 248, 88–99. [Google Scholar] [CrossRef]
- Esparza, I.; Jiménez-Moreno, N.; Bimbela, F.; Ancín-Azpilicueta, C.; Gandía, L.M. Fruit and Vegetable Waste Management: Conventional and Emerging Approaches. J. Environ. Manag. 2020, 265, 110510. [Google Scholar] [CrossRef]
- Gonçalves Neto, J.; Vidal Ozorio, L.; Campos de Abreu, T.C.; Ferreira dos Santos, B.; Pradelle, F. Modeling of Biogas Production from Food, Fruits and Vegetables Wastes Using Artificial Neural Network (ANN). Fuel 2021, 285, 119081. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Rosello, C.; Bélanger, R.; Ratti, C. Fate of Residual Pesticides in Fruit and Vegetable Waste (FVW) Processing. Foods 2020, 9, 1468. [Google Scholar] [CrossRef]
- Coman, V.; Teleky, B.-E.; Mitrea, L.; Martău, G.A.; Szabo, K.; Călinoiu, L.-F.; Vodnar, D.C. Bioactive Potential of Fruit and Vegetable Wastes. Adv. Food Nutr. Res. 2020, 91, 157–225. [Google Scholar] [CrossRef]
- Mao, G.; Wu, D.; Wei, C.; Tao, W.; Ye, X.; Linhardt, R.J.; Orfila, C.; Chen, S. Reconsidering Conventional and Innovative Methods for Pectin Extraction from Fruit and Vegetable Waste: Targeting Rhamnogalacturonan I. Trends Food Sci. Technol. 2019, 94, 65–78. [Google Scholar] [CrossRef]
- Kumar, H.; Bhardwaj, K.; Sharma, R.; Nepovimova, E.; Kuča, K.; Dhanjal, D.S.; Verma, R.; Bhardwaj, P.; Sharma, S.; Kumar, D. Fruit and Vegetable Peels: Utilization of High Value Horticultural Waste in Novel Industrial Applications. Molecules 2020, 25, 2812. [Google Scholar] [CrossRef]
- Han, R.; Truco, M.J.; Lavelle, D.O.; Michelmore, R.W. A Composite Analysis of Flowering Time Regulation in Lettuce. Front. Plant Sci. 2021, 12, 632708. [Google Scholar] [CrossRef]
- Ahmed, H.A.; Tang, Y.-X.; Yang, Q.-C. Optimal Control of Environmental Conditions Affecting Lettuce Plant Growth in a Controlled Environment with Artificial Lighting: A Review. S. Afr. J. Bot. 2020, 130, 75–89. [Google Scholar] [CrossRef]
- Lian, J.; Liu, W.; Meng, L.; Wu, J.; Chao, L.; Zeb, A.; Sun, Y. Foliar-Applied Polystyrene Nanoplastics (PSNPs) Reduce the Growth and Nutritional Quality of Lettuce (Lactuca sativa L.). Environ. Pollut. 2021, 280, 116978. [Google Scholar] [CrossRef]
- Zhou, X.; Sun, J.; Tian, Y.; Lu, B.; Hang, Y.; Chen, Q. Hyperspectral Technique Combined with Deep Learning Algorithm for Detection of Compound Heavy Metals in Lettuce. Food Chem. 2020, 321, 126503. [Google Scholar] [CrossRef]
- Gao, M.; Liu, Y.; Dong, Y.; Song, Z. Effect of Polyethylene Particles on Dibutyl Phthalate Toxicity in Lettuce (Lactuca sativa L.). J. Hazard. Mater. 2021, 401, 123422. [Google Scholar] [CrossRef]
- Boas, J.V.; Oliveira, V.B.; Simões, M.; Pinto, A.M.F.R. Review on Microbial Fuel Cells Applications, Developments and Costs. J. Environ. Manag. 2022, 307, 114525. [Google Scholar] [CrossRef]
- Ramya, M.; Senthil Kumar, P. A Review on Recent Advancements in Bioenergy Production Using Microbial Fuel Cells. Chemosphere 2022, 288, 132512. [Google Scholar] [CrossRef]
- Chaturvedi, V.; Verma, P. Microbial Fuel Cell: A Green Approach for the Utilization of Waste for the Generation of Bioelectricity. Bioresour. Bioprocess. 2016, 3, 38. [Google Scholar] [CrossRef] [Green Version]
- Santoro, C.; Arbizzani, C.; Erable, B.; Ieropoulos, I. Microbial Fuel Cells: From Fundamentals to Applications. A Review. J. Power Sources 2017, 356, 225–244. [Google Scholar] [CrossRef]
- Javed, M.M.; Nisar, M.A.; Ahmad, M.U.; Yasmeen, N.; Zahoor, S. Microbial Fuel Cells as an Alternative Energy Source: Current Status. Biotechnol. Genet. Eng. Rev. 2018, 34, 216–242. [Google Scholar] [CrossRef]
- Obileke, K.; Onyeaka, H.; Meyer, E.L.; Nwokolo, N. Microbial Fuel Cells, a Renewable Energy Technology for Bio-Electricity Generation: A Mini-Review. Electrochem. Commun. 2021, 125, 107003. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ibrahim, M.N.M.; Guerrero-Barajas, C. Modern Trend of Anodes in Microbial Fuel Cells (MFCs): An Overview. Environ. Technol. Innov. 2021, 23, 101579. [Google Scholar] [CrossRef]
- Vera Natalia Ginting, C.; Sari Nasution, J.; Alfatah Sembiring, M.; Simorangkir, M. The Effect of Composition and Substrate Fermentation Duration on Microbial Fuel Cell Electrical Energy. J. Pendidik. Kim. 2019, 11, 116–121. [Google Scholar] [CrossRef] [Green Version]
- Kalagbor, I.A.; Azunda, B.I.; Igwe, B.C.; Akpan, B.J. Electricity Generation from Waste Tomatoes, Banana, Pineapple Fruits and Peels Using Single Chamber Microbial Fuel Cells (SMFC). Open Access J. Waste Manag. Xenobiotics 2020, 3, 141. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ibrahim, M.N.M.; Rodríguez-Couto, S. Development and Modification of Materials to Build Cost-Effective Anodes for Microbial Fuel Cells (MFCs): An Overview. Biochem. Eng. J. 2020, 164, 107779. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ibrahim, M.N.M.; Umar, K. Biomass-Derived Composite Anode Electrode: Synthesis, Characterizations, and Application in Microbial Fuel Cells (MFCs). J. Environ. Chem. Eng. 2021, 9, 106111. [Google Scholar] [CrossRef]
- Palanisamy, G.; Jung, H.-Y.; Sadhasivam, T.; Kurkuri, M.D.; Kim, S.C.; Roh, S.-H. A Comprehensive Review on Microbial Fuel Cell Technologies: Processes, Utilization, and Advanced Developments in Electrodes and Membranes. J. Clean. Prod. 2019, 221, 598–621. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; De La Cruz-Noriega, M.; Cabanillas-Chirinos, L.; Benites, S.M.; Nazario-Naveda, R.; Delfín-Narciso, D.; Gallozzo-Cardemas, M.; Díaz, F.; Murga-Torres, E.; Rojas-Villacorta, W. Use of Kiwi Waste as Fuel in MFC and Its Potential for Use as Renewable Energy. Fermentation 2023, 9, 446. [Google Scholar] [CrossRef]
- Yang, W.; Du, M.; Liu, H.; Bao, J.; Tang, J.; Li, J. Full Cell Mathematical Models of Air Cathode Microbial Fuel Cells. Exp. Comput. Multiph. Flow 2023, 5, 111–121. [Google Scholar] [CrossRef]
- Safwat, S.M.; Khaled, A.; Elawwad, A.; Matta, M.E. Dual-Chamber Microbial Fuel Cells as Biosensors for the Toxicity Detection of Benzene, Phenol, Chromium, and Copper in Wastewater: Applicability Investigation, Effect of Various Catholyte Solutions, and Life Cycle Assessment. Process Saf. Environ. Prot. 2023, 170, 1121–1136. [Google Scholar] [CrossRef]
- Ray, S.; Pandey, S.; Mohanty, M.; Padhee, S. Comparative Analysis of Power Management System for Microbial Fuel Cell. In Communications in Computer and Information Science; Springer Nature: Cham, Switzerland, 2022; pp. 127–133. ISBN 9783031217494. [Google Scholar]
- Dziegielowski, J.; Mascia, M.; Metcalfe, B.; Di Lorenzo, M. Voltage Evolution and Electrochemical Behaviour of Soil Microbial Fuel Cells Operated in Different Quality Soils. Sustain. Energy Technol. Assess. 2023, 56, 103071. [Google Scholar] [CrossRef]
- Abazarian, E.; Gheshlaghi, R.; Mahdavi, M.A. Interactions between Sediment Microbial Fuel Cells and Voltage Loss in Series Connection in Open Channels. Fuel 2023, 332, 126028. [Google Scholar] [CrossRef]
- Bhattacharya, R.; Bose, D.; Yadav, J.; Sharma, B.; Sangli, E.; Patel, A.; Mukherjee, A.; Ashutosh Singh, A. Bioremediation and Bioelectricity from Himalayan Rock Soil in Sediment-Microbial Fuel Cell Using Carbon Rich Substrates. Fuel 2023, 341, 127019. [Google Scholar] [CrossRef]
- Din, M.I.; Ahmed, M.; Ahmad, M.; Iqbal, M.; Ahmad, Z.; Hussain, Z.; Khalid, R.; Samad, A. Investigating the Activity of Carbon Fiber Electrode for Electricity Generation from Waste Potatoes in a Single-Chambered Microbial Fuel Cell. J. Chem. 2023, 2023, 8520657. [Google Scholar] [CrossRef]
- Abubackar, H.N.; Biryol, İ.; Ayol, A. Yeast Industry Wastewater Treatment with Microbial Fuel Cells: Effect of Electrode Materials and Reactor Configurations. Int. J. Hydrogen Energy 2023, 48, 12424–12432. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, X.; Lin, H. Effects of PH on Simultaneous Cr(VI) and p-Chlorophenol Removal and Electrochemical Performance in Leersia Hexandra Constructed Wetland-Microbial Fuel Cell. Environ. Technol. 2022, 1–12. [Google Scholar] [CrossRef]
- Vemuri, B.; Chilkoor, G.; Dhungana, P.; Islam, J.; Baride, A.; Koratkar, N.; Ajayan, P.M.; Rahman, M.M.; Hoefelmeyer, J.D.; Gadhamshetty, V. Oxygen Reduction Reaction with Manganese Oxide Nanospheres in Microbial Fuel Cells. ACS Omega 2022, 7, 11777–11787. [Google Scholar] [CrossRef]
- Sharma, R.; Kumari, R.; Pant, D.; Malaviya, P. Bioelectricity Generation from Human Urine and Simultaneous Nutrient Recovery: Role of Microbial Fuel Cells. Chemosphere 2022, 292, 133437. [Google Scholar] [CrossRef]
- Zonfa, T.; Kamperidis, T.; Falzarano, M.; Lyberatos, G.; Polettini, A.; Pomi, R.; Rossi, A.; Tremouli, A. Two-Stage Process for Energy Valorization of Cheese Whey through Bio-Electrochemical Hydrogen Production Coupled with Microbial Fuel Cell. Fermentation 2023, 9, 306. [Google Scholar] [CrossRef]
- Segundo, R.-F.; De La Cruz-Noriega, M.; Milly Otiniano, N.; Benites, S.M.; Esparza, M.; Nazario-Naveda, R. Use of Onion Waste as Fuel for the Generation of Bioelectricity. Molecules 2022, 27, 625. [Google Scholar] [CrossRef]
- Agrahari, R.; Bayar, B.; Abubackar, H.N.; Giri, B.S.; Rene, E.R.; Rani, R. Advances in the Development of Electrode Materials for Improving the Reactor Kinetics in Microbial Fuel Cells. Chemosphere 2022, 290, 133184. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Calay, R.K.; Eregno, F.E. Role and Important Properties of a Membrane with Its Recent Advancement in a Microbial Fuel Cell. Energies 2022, 15, 444. [Google Scholar] [CrossRef]
- Potrykus, S.; León-Fernández, L.F.; Nieznański, J.; Karkosiński, D.; Fernandez-Morales, F.J. The Influence of External Load on the Performance of Microbial Fuel Cells. Energies 2021, 14, 612. [Google Scholar] [CrossRef]
- Rossi, R.; Logan, B.E. Unraveling the Contributions of Internal Resistance Components in Two-Chamber Microbial Fuel Cells Using the Electrode Potential Slope Analysis. Electrochim. Acta 2020, 348, 136291. [Google Scholar] [CrossRef]
- Koók, L.; Nemestóthy, N.; Bélafi-Bakó, K.; Bakonyi, P. The Influential Role of External Electrical Load in Microbial Fuel Cells and Related Improvement Strategies: A Review. Bioelectrochemistry 2021, 140, 107749. [Google Scholar] [CrossRef]
- Al Lawati, M.J.; Jafary, T.; Baawain, M.S.; Al-Mamun, A. A Mini Review on Biofouling on Air Cathode of Single Chamber Microbial Fuel Cell; Prevention and Mitigation Strategies. Biocatal. Agric. Biotechnol. 2019, 22, 101370. [Google Scholar] [CrossRef]
- Tremouli, A.; Karydogiannis, I.; Pandis, P.K.; Papadopoulou, K.; Argirusis, C.; Stathopoulos, V.N.; Lyberatos, G. Bioelectricity Production from Fermentable Household Waste Extract Using a Single Chamber Microbial Fuel Cell. Energy Procedia 2019, 161, 2–9. [Google Scholar] [CrossRef]
- Daud, N.N.M.; Ahmad, A.; Yaqoob, A.A.; Ibrahim, M.N.M. Application of Rotten Rice as a Substrate for Bacterial Species to Generate Energy and the Removal of Toxic Metals from Wastewater through Microbial Fuel Cells. Environ. Sci. Pollut. Res. Int. 2021, 28, 62816–62827. [Google Scholar] [CrossRef]
- Din, M.I.; Iqbal, M.; Hussain, Z.; Khalid, R. Bioelectricity Generation from Waste Potatoes Using Single Chambered Microbial Fuel Cell. Energy Sources Recovery Util. Environ. Eff. 2020, 2, 1–11. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; De La Cruz-Noriega, M.; Cabanillas-Chirinos, L.; Nazario-Naveda, R.; Gallozzo-Cardenas, M.; Diaz, F.; Murga-Torres, E. Potential Use of Coriander Waste as Fuel for the Generation of Electric Power. Sustainability 2023, 15, 896. [Google Scholar] [CrossRef]
- Wang, H.; Chai, G.; Zhang, Y.; Wang, D.; Wang, Z.; Meng, H.; Jiang, C.; Dong, W.; Li, J.; Lin, Y.; et al. Copper Removal from Wastewater and Electricity Generation Using Dual-Chamber Microbial Fuel Cells with Shrimp Shell as the Substrate. Electrochim. Acta 2023, 441, 141849. [Google Scholar] [CrossRef]
- Hirose, S.; Inukai, K.; Nguyen, D.T.; Taguchi, K. Use of Loofah Electrodes Coated with Rice Husk Smoked Charcoal and Japanese Ink in a Microbial Fuel Cell for Muddy Water Treatment. Energy Rep. 2023, 9, 160–167. [Google Scholar] [CrossRef]
- Fadhil, S.H.; Ismail, Z.Z. Bioremediation of Real-Field Slaughterhouse Wastewater Associated with Power Generation in Algae-Photosynthetic Microbial Fuel Cell. Bioremediat. J. 2021, 27, 75–83. [Google Scholar] [CrossRef]
- Mulyono, T.; Misto; Cahyono, B.E.; Fahmidia, N.H. The Impact of Adding Vegetable Waste on the Functioning of Microbial Fuel Cell. In Proceedings of the 3rd International Conference on Physical Instrumentation and Advanced Materials (ICPIAM) 2021, Jember, Indonesia, 27 October 2021. [Google Scholar]
- Yaakop, A.S.; Ahmad, A.; Hussain, F.; Oh, S.-E.; Alshammari, M.B.; Chauhan, R. Domestic Organic Waste: A Potential Source to Produce the Energy via a Single-Chamber Microbial Fuel Cell. Int. J. Chem. Eng. 2023, 2023, 2425735. [Google Scholar] [CrossRef]
- Venkata Mohan, S.; Mohanakrishna, G.; Sarma, P.N. Composite Vegetable Waste as Renewable Resource for Bioelectricity Generation through Non-Catalyzed Open-Air Cathode Microbial Fuel Cell. Bioresour. Technol. 2010, 101, 970–976. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; Cabanillas-Chirinos, L.; Nazario-Naveda, R.; Gallozzo-Cardenas, M.; Diaz, F.; Delfin-Narciso, D.; Rojas-Villacorta, W. Use of Tangerine Waste as Fuel for the Generation of Electric Current. Sustainability 2023, 15, 3559. [Google Scholar] [CrossRef]
- Segundo, R.-F.; Benites, S.M.; De La Cruz-Noriega, M.; Vives-Garnique, J.; Otiniano, N.M.; Rojas-Villacorta, W.; Gallozzo-Cardenas, M.; Delfín-Narciso, D.; Díaz, F. Impact of Dragon Fruit Waste in Microbial Fuel Cells to Generate Friendly Electric Energy. Sustainability 2023, 15, 7316. [Google Scholar] [CrossRef]
- Said, M.S.; Tirthani, E.; Lesho, E. Stenotrophomonas Maltophilia. Available online: https://www.ncbi.nlm.nih.gov/books/NBK572123/ (accessed on 22 May 2023).
- An, S.-Q.; Berg, G. Stenotrophomonas maltophilia. Trends Microbiol. 2018, 26, 637–638. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, P.; Roy, P. Genomic Potential of Stenotrophomonas maltophilia in Bioremediation with an Assessment of Its Multifaceted Role in Our Environment. Front. Microbiol. 2016, 7, 967. [Google Scholar] [CrossRef]
- Bonaventura, G.D.; Stepanović, S.; Picciani, C.; Pompilio, A.; Piccolomini, R. Effect of Environmental Factors on Biofilm Formation by Clinical Stenotrophomonas maltophilia isolates. Folia Microbiol. 2007, 52, 86–90. [Google Scholar] [CrossRef]
- Greenman, J.; Gajda, I.; You, J.; Mendis, B.A.; Obata, O.; Pasternak, G.; Ieropoulos, I. Microbial Fuel Cells and Their Electrified Biofilms. Biofilm 2021, 3, 100057. [Google Scholar] [CrossRef] [PubMed]
- Malvankar, N.S.; Lovley, D.R. Microbial Nanowires for Bioenergy Applications. Curr. Opin. Biotechnol. 2014, 27, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Reguera, G. Microbial Nanowires and Electroactive Biofilms. FEMS Microbiol. Ecol. 2018, 94, fiy086. [Google Scholar] [CrossRef] [PubMed]
- Juang, D.F.; Yang, P.C.; Lee, C.H.; Hsueh, S.C.; Kuo, T.H. Electrogenic Capabilities of Gram Negative and Gram Positive Bacteria in Microbial Fuel Cell Combined with Biological Wastewater Treatment. Int. J. Environ. Sci. Technol. 2011, 8, 781–792. [Google Scholar] [CrossRef] [Green Version]
- Hemdan, B.A.; El-Taweel, G.E.; Naha, S.; Goswami, P. Bacterial Community Structure of Electrogenic Biofilm Developed on Modified Graphite Anode in Microbial Fuel Cell. Sci. Rep. 2023, 13, 1255. [Google Scholar] [CrossRef]
- Segundo, R.-F.; De La Cruz-Noriega, M.; Nazario-Naveda, R.; Benites, S.M.; Delfín-Narciso, D.; Angelats-Silva, L.; Díaz, F. Golden Berry Waste for Electricity Generation. Fermentation 2022, 8, 256. [Google Scholar] [CrossRef]
- Venkidusamy, K.; Megharaj, M. Identification of Electrode Respiring, Hydrocarbonoclastic Bacterial Strain Stenotrophomonas maltophilia MK2 Highlights the Untapped Potential for Environmental Bioremediation. Front. Microbiol. 2016, 7, 1965. [Google Scholar] [CrossRef] [Green Version]
- Galai, S.; Pérez de los Ríos, A.; Hernández-Fernández, F.J.; Kacem, S.H.; Ramírez, F.M.; Quesada-Medina, J. Microbial Fuel Cell Application for Azoic Dye Decolorization with Simultaneous Bioenergy Production Using Stenotrophomonas sp. Chem. Eng. Technol. 2015, 38, 1511–1518. [Google Scholar] [CrossRef]
- Silva-Palacios, F.; Salvador-Salinas, A.; Quezada-Alvarez, M.A.; Rodriguez-Yupanqui, M.; Segundo, R.F.; Renny, N.N.; Cabanillas-Chirinos, L. Bioelectricity generation through Microbial Fuel Cells using Serratia fonticola bacteria and Rhodotorula glutinis yeast. Energy Rep. 2023, 9, 295–301. [Google Scholar] [CrossRef]
- Montero, D.A.; Arellano, C.; Pardo, M.; Vera, R.; Gálvez, R.; Cifuentes, M.; Berasain, M.A.; Gómez, M.; Ramírez, C.; Vidal, R.M. Antimicrobial Properties of a Novel Copper-Based Composite Coating with Potential for Use in Healthcare Facilities. Antimicrob. Resist. Infect. Control 2019, 8, 3. [Google Scholar] [CrossRef]
- Baudler, A.; Schmidt, I.; Langner, M.; Greiner, A.; Schröder, U. Does It Have to Be Carbon? Metal Anodes in Microbial Fuel Cells and Related Bioelectrochemical Systems. Energy Environ. Sci. 2015, 8, 2048–2055. [Google Scholar] [CrossRef] [Green Version]
- Destouni, G.; Frank, H. Renewable Energy. Ambio 2010, 39 (Suppl. S1), 18–21. [Google Scholar] [CrossRef]
- Seetharaman; Moorthy, K.; Patwa, N.; Saravanan; Gupta, Y. Breaking Barriers in Deployment of Renewable Energy. Heliyon 2019, 5, e01166. [Google Scholar] [CrossRef] [Green Version]
- Dhanya, B.S.; Mishra, A.; Chandel, A.K.; Verma, M.L. Development of Sustainable Approaches for Converting the Organic Waste to Bioenergy. Sci. Total Environ. 2020, 723, 138109. [Google Scholar] [CrossRef]
- Fagunwa, O.E.; Olanbiwoninu, A.A. Accelerating the Sustainable Development Goals through Microbiology: Some Efforts and Opportunities. Access Microbiol. 2020, 2, acmi000112. [Google Scholar] [CrossRef]




| Organic Waste | MIFC Type | Electrodes | Voltage (V) | Current (mA) | Power Density | Ref. |
|---|---|---|---|---|---|---|
| Vegetable (spinach waste) | dual-chamber MFC | graphite fiber electrodes | 0.804 | 2.37 | 211 mW/m2 | [57] |
| Domestic organic substrate | single-chamber MFC | carbon | 0.110 | 0.80 | 0.1047 mW/m2 | [58] |
| Vegetable waste from a market | single-chamber MFC | non-catalyzed graphite plates | 0.208 | ------- | 87.14 mW/m2 | [59] |
| Potato | single-chamber MFC | copper anode and zinc cathode | 1.120 | 12.45 | ------ | [37] |
| Tangerine fruit | single-chamber MFC | copper anode and zinc cathode | 1.191± 0.035 | 1.439 ± 0.055 | 475.32 ± 24.56 mW/cm2 | [60] |
| Kiwi fruit | single-chamber MFC | copper anode and zinc cathode | 0.993 ± 0.061 | 3.807 ± 0.102 | 212.68 ± 26.84 mW/cm2 | [30] |
| Pitahaya fruit | single-chamber MFC | copper anode and zinc cathode | 0.460 ± 0.03 | 2.86 ± 0.07 | 304.33 ± 16.51 mW/cm2 | [61] |
| Parameters (Lettuce Waste) | Mean | SD | 95% CI |
|---|---|---|---|
| Voltage (V) | 0.959 | 0.026 | 0.9–1.0 |
| Current (mA) | 5.697 | 0.065 | 5.5–5.9 |
| Power density (mW/m2) | 378.145 | 5.417 | 364.7–391.6 |
| Identified Species | Base Pairs (bp) | Percentage of Identity (%) | Access Number | Lineage |
|---|---|---|---|---|
| Stenotrophomonas maltophilia | 1458 | 99.59 | NR_041577.1 | Cellular organisms; Bacteria; Pseudomonadota; Gammaproteobacteria; Xanthomonadales; Xanthomonadaceae; Stenotrophomonas; Stenotrophomonas maltophilia group |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojas-Villacorta, W.; Rojas-Flores, S.; Benites, S.M.; Nazario-Naveda, R.; Romero, C.V.; Gallozzo-Cardenas, M.; Delfín-Narciso, D.; Díaz, F.; Murga-Torres, E. Preliminary Study of Bioelectricity Generation Using Lettuce Waste as Substrate by Microbial Fuel Cells. Sustainability 2023, 15, 10339. https://doi.org/10.3390/su151310339
Rojas-Villacorta W, Rojas-Flores S, Benites SM, Nazario-Naveda R, Romero CV, Gallozzo-Cardenas M, Delfín-Narciso D, Díaz F, Murga-Torres E. Preliminary Study of Bioelectricity Generation Using Lettuce Waste as Substrate by Microbial Fuel Cells. Sustainability. 2023; 15(13):10339. https://doi.org/10.3390/su151310339
Chicago/Turabian StyleRojas-Villacorta, Walter, Segundo Rojas-Flores, Santiago M. Benites, Renny Nazario-Naveda, Cecilia V. Romero, Moisés Gallozzo-Cardenas, Daniel Delfín-Narciso, Félix Díaz, and Emzon Murga-Torres. 2023. "Preliminary Study of Bioelectricity Generation Using Lettuce Waste as Substrate by Microbial Fuel Cells" Sustainability 15, no. 13: 10339. https://doi.org/10.3390/su151310339







