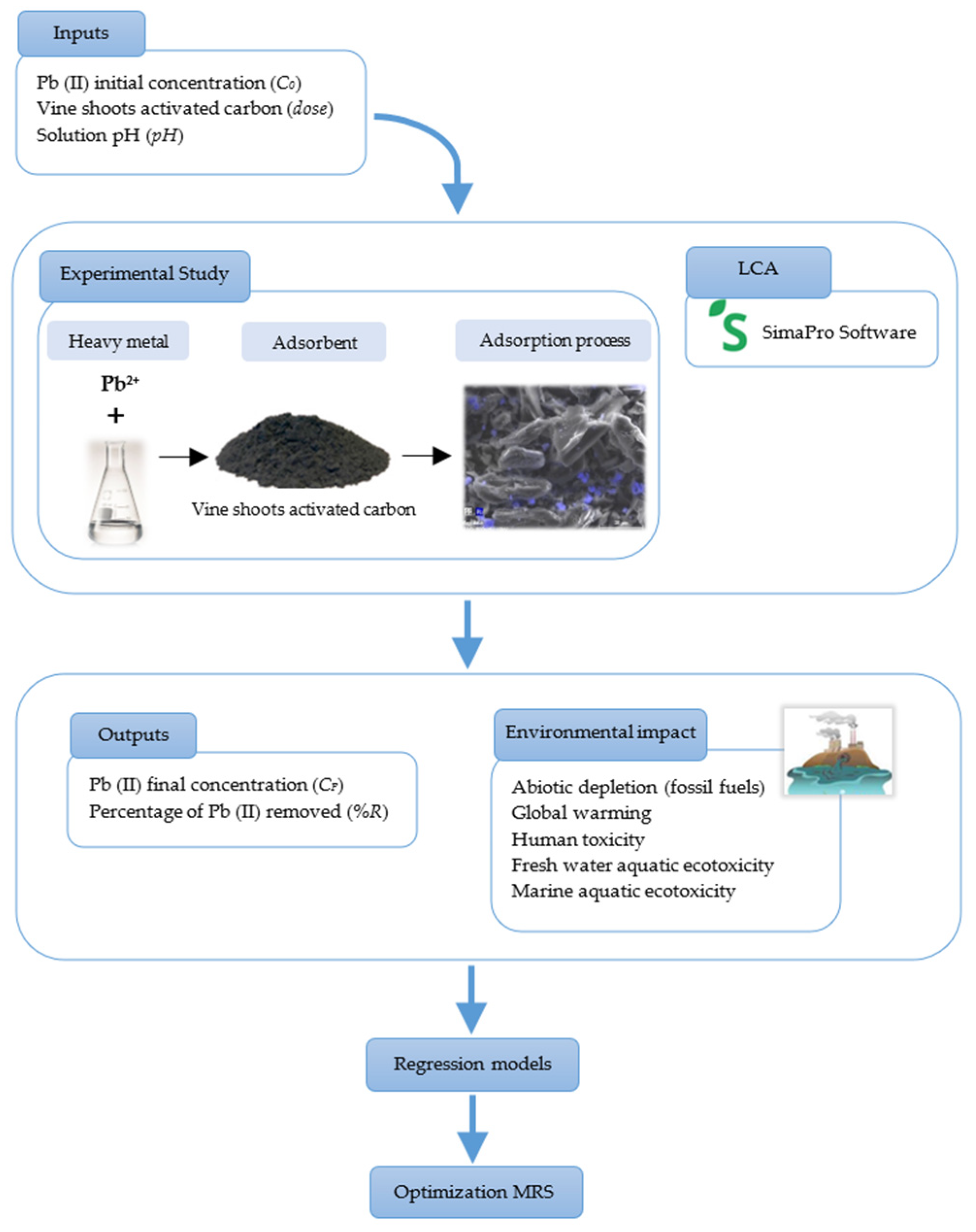
3.2. Analysis of Variance
Equation (2) was adjusted with the results that appear in
Table 4 and
Table 5, in order for the R Statistical Software to produce regression equations for the answers [
41]. For every response, a second-order polynomial model was constructed. The most accurate model was then selected by considering a variety of variables, such as the
p-value, RMSE, MAE, and correlation. Equations (7)–(12) gave second-degree polynomial functions for modelling the final Pb (II) concentration (
CF) and the following environmental impacts: abiotic depletion (
Ab_depl) and global warming (
Gl_War), human toxicity (
Hum_along with Tox), freshwater ecotoxicity (
Fresh_W), and marine aquatic ecotoxicity (
Mar_Aqu).
In addition,
Table 6,
Table 7,
Table 8,
Table 9,
Table 10 and
Table 11 provide the ANOVA results for the quadratic models that were developed, along with a determination of whether any interactions or other effects were statistically significant and had a
p-value (Pr (>F)). These tables show that most of the input variables’
p-values and their combinations were lower than 0.05.
Table 6 shows that the analysis of variance results support the conclusion that the initial lead concentration (
C0) (
p-value = 0.00030) directly influenced the output parameter of the final lead concentration (
CF). In contrast, the other input parameters, solution pH (
pH) (
p-value = 0.30083) and dose of vine shoot activated carbon (
dose) (
p-value = 0.91229), had no significant influence. However, they did when combined to create what are known as interaction effects (
pH·dose) with a
p-value of 0.01684. Furthermore, the pH (
pH) had a significant effect when it was squared (
pH2) (
p-value = 0.04003). Due to this disparity in the results for the
p-values of pH, which directly affect the final concentration, it is proposed that the dependence of these two variables be verified by the second-degree polynomial equation that was obtained (Equation (7)). Therefore,
Figure 2 shows the effect of the solution pH on Pb (II) biosorption at various initial lead concentrations (8, 19, and 30 ppm) and for different vine shoot activated carbon dosages (1.6 and 6 g).
Figure 2a shows that, at a low biosorbent dose (
dose = 1.6 g), the minimum final Pb (II) concentration was reached at a low initial lead concentration (
C0 = 8 ppm) and a high solution pH (
pH = 7.2). Additionally, the maximum final Pb (II) concentration was reached at a high initial concentration (
C0 = 30 ppm) and low solution pH (
pH = 2.8). However,
Figure 2b shows the opposite. It shows that with a high biosorbent dose (
dose = 6 g), the minimum final Pb (II) concentration was reached at a low initial lead concentration (
C0 = 8 ppm) and a low solution pH (
pH = 2.8). Additionally, it shows that the maximum final Pb (II) concentration was reached at a high initial concentration (
C0 = 30 ppm) and a high solution pH (
pH = 7.2). Thus, it can be concluded that the final lead concentration depends on the pH of the solutions, as stated in other references related to lead removal using activated carbon [
48,
49] and vine shoot activated carbon [
50,
51].
However, the dose of activated carbon (dose) had no significant effect when it was squared (dose2) (p-value = 0.06741) or combined with the initial concentration of lead (C0·dose) (p-value = 0.17789). In short, the input variable that had the most influence on the output variable (i.e., CF) was the initial concentration of Pb (II) (C0). That is, the final lead concentration depended mainly on the initial lead concentration contained in the solution. The input variable that influenced the final lead concentration (CF) the least was the dose of activated carbon (dose). This may be due to the fact that, if there is a saturation of vine shoot activated carbon, no further adsorption will occur during the process.
Table 7,
Table 8,
Table 9,
Table 10 and
Table 11 show that the input parameters of pH (
pH) and dose of vine shoot activated carbon (
dose) directly affected the effect of the process on the environment, except in the case of global warming (
Gl_War). In this case, in Equation (12), the dose of vine shoot activated carbon just appeared squared. The initial lead concentration (
C0) had no significant effect. There were very similar results for the remaining output parameters of approximately 0.98.
Table 7 shows that, for abiotic depletion (
Ab_depl), the dose of activated carbon (
dose) was the most significant variable (
p-value = 0.00007), followed by pH (
p-value = 0.00096). This was not true when it was squared (
dose2) (
p-value = 0.14172). The initial lead concentration (
C0) was the least significant variable (
p-value = 0.98458). Additionally, it was not a significant variable when squared (
C02) (
p-value = 0.15284). For the environmental impact outputs, there was only one combination of the input variables. It was formed by the dose of activated carbon (
dose) and the pH. In this case, for abiotic depletion (
Ab_depl), this combination (
pH·dose) had a significant influence (
p-value = 0.01066). In a similar fashion,
Table 8 shows that the dose of activated carbon (
dose) directly influenced the output variable of global warming (
Gl_War) (
p-value = 4.767 × 10
−6), as did the combination of input variables of pH squared (
pH2) (
p-value = 0.00019) and pH with the dose of activated carbon (
pH·dose) (
p-value = 0.00550). However, the initial lead concentration (
C0) had no significant influence (
p-value = 0.97908), even if it was squared (
C02) (
p-value = 0.17966). Similarly,
Table 9 shows that, for human toxicity (
Hum_Tox), the pH and the dose of activated carbon (
dose) had a significant influence (
p-value = 1.5726 × 10
−7 and
p-value = 8.595 × 10
−7, respectively). They also influenced human toxicity when they were combined (
pH·dose) (
p-value = 0.01426). Nevertheless, the initial lead concentration (
C0), its square (
C02), and the square of dose of activated carbon (
dose2) had no influence (
p-value = 0.98603,
p-value = 0.14921, and
p-value = 0.10496, respectively).
Table 10 shows that, for freshwater ecotoxicity (
Fresh_W), the pH (
pH) and dose of activated carbon (
dose) were the most significant variables (
p-value = 1.89 × 10
−6 and
p-value = 2.200 × 10
−6, respectively), just as the combination of both input variables (
pH·dose) (
p-value = 0.01986) was. Furthermore, the remaining input parameters had no influence on this environmental impact; the initial lead concentration (
C0) had a
p-value = 0.98927 even if it was squared (
C02) (
p-value = 0.17526), with other parameters as follows: (
pH2) (
p-value = 0.15476) and (
dose2) (
p-value = 0.12680). Finally,
Table 11 shows that the pH (
pH) and the dose of activated carbon directly influenced the environmental impact output of marine aquatic ecotoxicity (
Mar_Aqu), with
p-values of 3.7192 × 10
−5 and 8.306 × 10
−6, respectively, as did the combination of both (
pH·dose) (
p-value = 0.01159). However, the initial lead concentration (
C0), its square (
C02), and the square of the remaining input variable dose of activated carbon (
dose2) did not, with
p-values of 0.94399, 0.16971, and 0.13017, respectively.
The interaction effects, square effects, and main effects of the process inputs were considered in this study to be significant second-order regression model variables. The analysis of the ANOVA results show that the second-order models’ input process variables had statistically significant values. Additionally, we can also deduce from the analysis of variance that the input variables of pH (pH) and the dose of vine shoot activated carbon (dose) directly influenced the environmental impact of the sorption process. It is clear that this occurred because the LCA for the biosorption process in this research was covered gate-to-gate. Thus, neither the creation of activated carbon nor the subsequent treatments of the water emissions were considered in this work.
In this instance, the quadratic models that the RSM and n experiments developed are denoted by X
i Model, whereas the empirically observed outputs are denoted by X
i Test. The correlation and predicted errors are shown in
Table 12. One can see that the correlation coefficients almost reached 100%. This indicates that the approximation of the theoretical model, predicted by the quadratic regression models, to the experimentally derived values is very good. Environmental impacts had the highest correlation values, with the best result in the case of human toxicity (
Hum_Tox) (correlation = 98.335%), followed by the impact of freshwater ecotoxicity (
Fresh_W) (correlation = 98.207%). The lowest correlation corresponded to the final Pb (II) concentration (
CF) (correlation = 92.626%). The reason for this is that the
CF was determined experimentally using a spectrophotometer and the environmental effects using software (Simapro
®), which provided reliable correlation values. All of the variables that were examined also had very low MAE and RMSE values. This shows that the quadratic regression models properly reflect the experimental findings and have a high degree of generalizability. Similar to the analysis of correlation, it can be noted that, for the values that the RMSE and MAE provided, the final lead concentration was the output variable that had the highest MAE and RMSE (7.948% and 10.068%, respectively). In contrast, the lower MAE and RMSE were associated with human toxicity (
Hum_Tox) and freshwater ecotoxicity (
Fresh_W), whereas the MAE and RMSE were 3.426% and 4.611% for the first variable and 3.565% and 4.801% for the second variable, respectively.
3.3. Multiresponse Optimization
The optimal process variables within the required operating range were predicted using the desirability function tool in the R Statistical Software [
41]. By using the desirability function, it is possible to maximize many replies. As a result, a projected response becomes a scale-free value response with an optimal value that ranges from the lowest to the optimal (from 0 to 1) [
52].
Table 13,
Table 14,
Table 15 and
Table 16 provide the optimum results for the Pb (II) biosorption process in wastewater using activated carbon and MRS, which combined input and output parameters for four different optimization scenarios. These four scenarios reflect a tendency to minimize the final Pb (II) concentration in wastewater, the adsorbent dosage consumption, and the impact of the biosorption process on the environment. In this work, the input and output are both listed in the first column of each table. The goal is provided in the second column. The minimums and maximums (range) that were chosen for the adsorption optimization procedure appear in the third and fourth columns. The optimized values that were produced within the foregoing minimums and maximums are shown in the fifth column. The desirability values appear in the sixth column. The overall desirability was obtained from the maximization of individual desirability. It indicates how close a response is to the ideal value. If the desired value is reached, the coefficient will be equal to a single unit and will be nil otherwise.
Table 13 shows the first optimization scenario, in which the goal is to obtain the minimum final lead concentration, while the remaining variables are in range. The second optimization scenario is provided in
Table 14. This scenario involves the minimum vine shoot activated carbon dosage consumption. Thus, it minimizes the costs of the process to obtain the highest removal of Pb (II) (minimum final lead concentration), while the other input and output variables are set in range.
Table 15 shows the results of the third adsorption scenario with the objective of achieving the minimum environmental impact and final Pb (II) concentration, with the remaining variables in range. The third scenario’s output process parameter (minimum final lead concentration) and environmental effect were considered in the fourth scenario (
Table 16) in an attempt to minimize the activated carbon dosage consumption. After analyzing the results that appear in
Table 16, we can conclude that the values for the input of dose of vine shoot activated carbon were very similar for each of the studied optimization scenarios, whereas the values for overall desirability were extremely close to -1-. This indicates that when starting with that given amount of vine shoot activated carbon, its saturation will be produced without implying greater adsorption during the process. Thus, the minimum value of the final Pb (II) concentration was achieved in the first (
CF = 1.11 ppm) and the second (
CF = 1.12 ppm) optimization scenarios, whereas the maximum final lead concentration was achieved in the fourth scenario (
CF = 4.87 ppm). Additionally, using Equation (6), the lead removal percentage for all of the optimization scenarios can be calculated. Therefore, the Pb (II) removal percentages that were achieved were 92.12% for the first optimization scenario, 92.09% for the second optimization scenario, 52.66% for the third optimization scenario, and 39.23% for the fourth optimization scenario. This indicates that the highest removal of Pb (II) ions was achieved in the first and second optimization scenarios, in which environmental impacts were not considered. Moreover, better results were obtained in this work than in studies undertaken by other researchers in the removal of lead with activated carbon derived from coconut shells [
20] or residues of corn cobs [
21]. The results that were obtained in this study are similar to those obtained with activated carbon from
Juniperus procera leaves [
47].
The values reached in the first two optimization scenarios for the input and output variables were identical. The values in the third and the fourth optimization scenarios were also identical. These last two scenarios were those that produced the least environmental impact. The first two optimization scenarios produced the optimal value of pH at 7 and an initial lead concentration (
C0) of 14.14 ppm when the dose of vine shoot activated carbon (dose) was 1.6 g, achieving a final lead concentration (
CF) of 1.11 ppm. The other values achieved were an environmental impact of 0.218 MJ for abiotic depletion (
Ab_depl), 0.017 kg CO
2 eq. for global warming (
Gl_War), 0.009 kg 1.4 DB eq. for human toxicity (
Hum_Tox), 0.007 kg 1.4 DB eq. for freshwater ecotoxicity (
Fresh_W), and 23.04 kg 1.4 DB eq. for marine aquatic ecotoxicity (
Mar_Aqu). In the last two optimization scenarios, minimizing the environmental impact was an additional objective of the optimizations. For these scenarios, the optimal value of pH (
pH) was 2.8, whereas the initial lead concentration (
C0) was 8.01 ppm and the dose of activated carbon (
dose) was approximately 1.8 g, achieving a final lead concentration (
CF) of 3.8 and 4.87 ppm and an environmental impact of 0.047 MJ for abiotic depletion (
Ab_depl), 0.004 kg CO
2 eq. for global warming (
Gl_War), 0.002 kg 1.4 DB eq. for human toxicity (
Hum_Tox), 0.001 kg 1.4 DB eq. for freshwater ecotoxicity (
Fresh_W), and 4.86 kg 1.4 DB eq. for marine aquatic ecotoxicity (
Mar_Aqu). There are two reasons why the highest lead removal values were reached in the first two optimization scenarios. The first is that, as the adsorbent surface charges, the pH of the solution determines the adsorbent’s degree of ionization and specification. This factor greatly affects the number of heavy metal ions that are removed. Because lead precipitates in a solution at pH values higher than 6, lead adsorption is a process that is highly pH-dependent. The second reason is that the chemical structure of heavy metal ions often has a significant effect on the pH effect [
53]. However, a greater lead initial concentration increases the adsorption capacity. As the process continues, the higher Pb (II) ion concentration must contend with a finite number of active sites on the vine shoot activated carbon surface. As a result, more Pb (II) ions remain in the solution and are not adsorbed.
Figure 3 is a 3D graphic representation of
CF vs.
pH and
C0, for the maximum (6 g) and minimum (1.6 g) values of vine shoot activated carbon dose. The figure indicates that, when the
pH values are less than the study range (pH = 2.8), the
CF values that are achieved are always less. When the adsorbent dose is greater than any value of the
C0 study range, and when the
pH values are the largest of the study range (pH = 7.2), the
CF values achieved are always less when the adsorbent dose is less than any value of the
C0 study range. Starting from the results shown, it can be confirmed that, if one intends to eliminate the greatest possible quantity of Pb at the same time that the dose of adsorbent is the minimum possible, the pH of the water solution should have the maximum value of the study. Additionally, it can be deduced from
Figure 3 that, when the
pH is intermediate in the study range (from 4 to 6), the
CF values vary only in the study range of vine shoot activated carbon dose.