1. Introduction
The predominant method of disposing of municipal solid waste (MSW) in many countries involves burying it in landfills. This practice leads to the generation of landfill leachate (LL), which is formed when rainwater infiltrates through various layers of waste in the landfill [
1]. Within the landfill, a combination of microbial, chemical, and physical processes occur, transferring contaminants from the solid waste materials to the percolating or infiltrated water [
2].
Landfills typically receive significant amounts of municipal, commercial, and mixed industrial waste, all of which are disposed of in one location [
3]. This accumulation contributes to the production of substantial quantities of leachate and the contamination of groundwater. If a landfill contaminates the groundwater, a variable plume of contamination may form [
4]. The composition of LL can vary significantly and is primarily influenced by factors such as the type of waste present in the landfill, the age of the landfill, biochemical processes involved in waste decomposition, ambient temperature, and rainfall [
5]. Based on the age of the landfill, LL can be classified into three categories: young (<1 year), medium (1–5 years), and mature (>5 years) [
6].
The current state of LL treatment can be categorized into three main groups: physical–chemical processes, biological processes, and a combination of biological and physical–chemical processes [
4,
7,
8,
9]. Previous studies have reported chemical oxygen demand (COD) removal ranging from 40% in aerated lagoons to 81% in moving bed biofilm reactors, while biochemical oxygen demand (BOD
5) removal varied from 61% in activated sludge systems to 84% in sequencing batch reactors [
4]. Removals of up to 97% were reached for ammonium–nitrogen (NH
4-N) by using an MBBR process [
10]. Currently, the predominant method for leachate treatment involves a combination of aerobic, chemical, and physical processes [
11]. Within the realm of physical–chemical processes, the commonly employed techniques for LL treatment encompass coagulation–flocculation, chemical precipitation, ion exchange, adsorption, and advanced oxidation processes (AOPs) [
12,
13,
14].
Although biological treatments utilizing aerobic ponds have been used for leachate treatment, they have certain limitations, including the lower removal efficiency of organic matter (OM) and certain toxic pollutants. In contrast, anaerobic treatments of wastewater have gained significant attention from researchers and sanitary engineers, primarily because of their economic advantages over conventional aerobic methods [
15].
Biological methods are commonly employed for the treatment of LL because of their simplicity, reliability, and cost-effectiveness [
16]. Biological treatment approaches have been successful in reducing concentrations of COD and BOD
5 in LL. Younger LL (less than 5 years old) with COD/BOD
5 ratios ranging from 0.4 to 0.6 is particularly suitable for biological treatment because of the presence of biodegradable organic compounds in these leachates [
17,
18]. Most biological techniques utilized for LL treatment are based on suspended growth systems such as sequencing batch reactors (SBRs), activated sludge methods, aerated lagoons, and biofilm reactors. Jery EA et al., for example, studied the treatment of sanitary wastewater by using microorganisms and the microalgae Chlorella vulgaris in conjunction with sequencing batch reactors (SBRs), obtaining a removal percentage for COD of 94% [
19]. Numerous scientific studies have described the successful application of these methods for LL treatment [
18,
20]. However, anaerobic alternatives are better suited for treating LL with high concentrations of contaminants. In contrast to aerobic methods, anaerobic treatments require less energy, produce fewer solids, and can operate at lower ambient temperatures [
21]. Additionally, anaerobic processes have the potential to convert the organic matter in LL into biogas, a valuable renewable energy source, along with by-products such as O
2, N
2, CO
2, CH
4, and sulfides.
Over the past decade, MBBRs have been successfully employed for the treatment of various industrial effluents, including waste from the pulp and paper industry, poultry processing wastewater, cheese factory waste, refinery and slaughterhouse waste, and phenolic wastewater. They have also been utilized for biological phosphorus removal [
22,
23,
24,
25,
26,
27,
28].
Researchers have demonstrated that MBBRs and advanced oxidation processes offer several advantageous characteristics, such as high biomass concentration, the ability to handle high COD loadings, resilience to fluctuating load impacts, a relatively compact reactor design, and no sludge bulking issues [
28,
29].
Chen et al. (2008) utilized a moving bed biofilm reactor (MBBR) system with an anaerobic/aerobic configuration for the treatment of LL. In the anaerobic treatment stage, high concentrations of COD were removed by 90% for applied organic loading rates (OLRs) ranging from 4 to 15 kg COD m
−3 d
−1. The aerobic stage of the MBBR system served as a polishing phase for COD removal and a phase for the removal of ammonia (NH
3) from the leachate [
10]. The biological treatment of landfill leachate usually results in low nutrient removal because of high COD, high ammonium-N content, and the presence of toxic compounds such as heavy metals [
30]. Solely relying on a single treatment process is typically insufficient for effectively treating LL. Success in LL treatment often hinges on the integration of various physical–chemical and biological methods [
31].
The advancement of bio-adsorbents in recent years has been driven by characteristics such as a large surface area, high porosity, and thermostability, which make them highly effective in the removal of various organic and inorganic pollutants. Bio-adsorbents have shown enormous potential in removing ammoniacal nitrogen and COD, with removal efficiencies of up to 91%.
Biochar is a solid carbonaceous material produced through the thermochemical conversion, specifically, pyrolysis, of several types of biomasses. This conversion process occurs at temperatures ranging from 300 to 800 °C in the absence of or with limited oxygen. A wide range of biomass sources can be used to produce biochar, including forest residues, agricultural by-products (both woody and herbaceous), animal manures, and the organic fraction of municipal solid waste [
32].
The properties of biochar, such as its physical and chemical characteristics, are influenced by several factors, including the type of biomass used, the pyrolysis temperature, and the residence time. Consequently, a diverse range of biochars can be produced, each exhibiting unique functionalities based on their intended applications [
33].
Biochar’s porous structure and adsorptive properties make it suitable for the removal and immobilization of contaminants from both wastewater and soil environments. Numerous studies have demonstrated the promising potential of biochar in the remediation of contaminants, highlighting its ability to adsorb and sequester harmful substances from wastewater and soil systems.
Thus, biochar can serve as a versatile material in mitigating environmental pollution by adsorbing and immobilizing organic and inorganic contaminants in wastewater and soil [
33,
34,
35,
36].
In the existing literature, LL treatment primarily revolves around physical–chemical and biological methods. These approaches have demonstrated various levels of success in removing contaminants like COD, BOD5, and NH4-N. However, they often fall short of effectively treating LL with high concentrations of contaminants. The anaerobic treatment of LL has been explored as a more energy-efficient alternative, but its potential in LL treatment remains relatively uncharted territory. Furthermore, the application of biochar as an adsorbent material for removing contaminants from both wastewater and soil environments has gained attention because of its unique properties, yet its full potential in LL treatment has not been thoroughly investigated.
This work introduces a novel approach to efficiently remove organic matter and nutrients from landfill leachate. It employs a unique combination of anaerobic and aerobic MBBR systems, alongside biochar adsorption, to target contaminants like COD, NH4-N, and total phosphorus (TP). To the best of the authors’ knowledge, this marks the first-ever investigation of the anaerobic/aerobic MBBR process followed by biochar adsorption for the simultaneous removal of COD, NH4-N, and TP. The MBBR system is meticulously designed to create an ideal environment for the growth of microorganisms capable of degrading organic matter and nutrients, while biochar adsorption serves as an innovative way to eliminate any residual non-biodegradable pollutants. Furthermore, this study delves into the system’s performance under varying operating conditions, such as hydraulic retention time and organic loading rate, aiming to optimize both its efficiency and sustainability. Additionally, a comprehensive kinetic study of the adsorption process of biochar is conducted, involving the modeling and fitting of pseudo-first-order (PFO), pseudo-second-order (PSO), and Elovich kinetic equations.
2. Materials and Methods
2.1. Experimental Setup and LL Samples
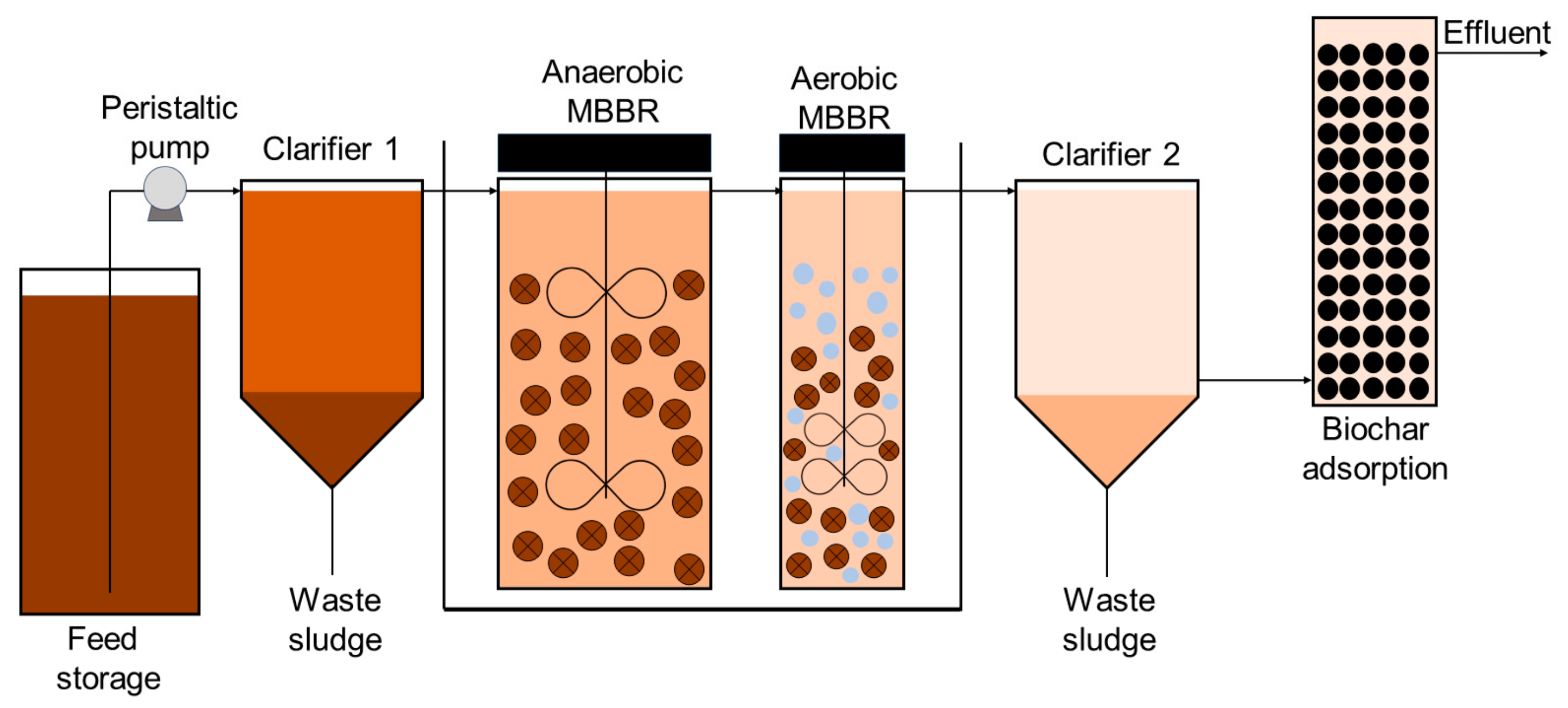
The pilot plant consisted of an effluent equalization and acclimatization tank with a volume of 25 L, one primary clarifier with a volume of 2 L, one anaerobic MBBR with a volume of 15 L, one aerobic MBBR with a volume of 6 L, one secondary clarifier with a volume of 2 L, and one adsorption column packed with raw biochar (
Figure 1) with hydraulic retention times (HRTs) of 4 h. The process was performed in continuous mode.
The volume of LL to be treated was 22 L, and the HRTs for the clarifiers were set at 10 h. The anaerobic reactor was a cylinder with an inside diameter of 62 mm and a working volume of 12 L, and the filling of the bio-carrier was about 40 vol%. The aerobic reactor was cylindrical, the working volume was 6 L, and the filling of the bio-carrier was about 60 vol%. It should be noted that the HRT of the aerobic MBBR was half of the HRT of the anaerobic MBBR because the two reactors were connected in a series.
The LL was procured from a municipal solid waste (MSW) landfill, aged 7 years, situated in the State of Pará within the Amazon region of northern Brazil. LL is typically stored for up to 12 h prior to its conveyance to a centralized wastewater treatment plant (WWTP). Subsequently, the LL samples employed in all experiments were transported to the laboratory and promptly refrigerated at 4 °C upon arrival, adhering to the standard methods outlined in the APHA (1998) guidelines for the examination of water and wastewater. These LL samples were systematically collected over the span of one year, with monthly collection intervals spanning from January to early September. This collection frequency allowed for an assessment of variations that may be linked to seasonal changes, including those associated with the rainy season. Wastewater characterization was repeated before each purification test. The most pertinent characteristics of the LL samples are presented in
Table 1. The characteristics of the LL used in this work agree with the characteristics found in the literature for an intermediate LL (age between 5 and 10 years) [
37,
38,
39,
40,
41].
2.2. Operating Procedures
The untreated LL is stored at a temperature of 4 °C in a refrigerator and is pumped into the primary clarifier using a peristaltic pump. From there, it is directed to the anaerobic MBBR (moving bed biofilm reactor) from the bottom. Inside the anaerobic MBBR, the influent undergoes degradation caused by anaerobic microbes. The resulting supernatant is then directed into the aerobic reactor. Subsequently, the effluent overflows out of the reactor and moves on to the secondary clarifier.
Upon reaching the secondary clarifier, the effluent is further processed and eventually transferred to the adsorption column. The anaerobic reactor is maintained at a temperature of 35 ± 1 °C, regulated by an electronic wire heater. Both reactors operate in continuous stirred tank reactor (CSTR) mode, without the need for sludge-returning equipment for sludge accumulation. In addition to the attached biomass in the biofilm, suspended sludge naturally accumulates based on the hydraulic conditions.
This study was conducted in three stages over a period of 90 days. During step I (from day 1 to day 30), the hydraulic retention time (HRT) of the anaerobic MBBR was maintained at a constant 4 days. Various concentrations of leachate (in terms of COD), ranging from 1226 to 2356 mg/L, were obtained by diluting the raw leachate and were used as the feed for the system.
During step II (from day 31 to day 60), the feed COD of the anaerobic MBBR was kept constant at approximately 3300 mg/L. However, the HRT of the anaerobic MBBR was altered to 3 days.
During step III (from day 61 to day 90), the concentration of the raw leachate was kept constant at about 4700 mg/L (without dilution). At this concentration, an additional shorter HRT of 2 days was evaluated.
2.3. Analytical Methods
BOD₅, TN, TP, and NH
3-N were analyzed following standard methods (CNR-IRSA 5120, CNR-IRSA 4060, and CNR-IRSA 4110), as per IRSA-CNR (2003) guidelines [
42]. COD was measured using the spectrophotometric method proposed by Li et al. [
43]
Temperature, pH, and electrical conductivity were continuously monitored using the pH/EC/TDS/Temperature Meter Hanna HI9813-6. BOD₅ was determined through manometric respiration tests following OECD301F guidelines, using the OxiTop
® Control measuring system [
44].
TN and TP were measured by oxidizing N-containing compounds and P-containing compounds, respectively, with a mixture of peroxydisulfate, boric acid, and sodium hydroxide, transforming these compounds into nitrate and orthophosphate simultaneously. The nitrate content was determined by measuring the absorbance at 220 nm. Orthophosphate was measured using the molybdenum blue spectrophotometric method.
NH4-N was quantified using the nesslerization method, and the absorbance was measured at a wavelength of 425 nm.
For volatile suspended solids (VSSs), the biomass adhered to the bio-carriers was initially dislodged using ultrasonic vibration for 30 min. Subsequently, the mixed liquid was filtered through a 0.45 μm Millipore filter and then dried at 105 °C to determine the dry weight.
All the analytical procedures were based on IRSA-CNR standard methods [
42].
2.4. Biochar Characterization
Fourier-transform infrared spectroscopy (FTIR) spectra were obtained using a Bruker Vertex70 spectrometer (Germany). To prepare the samples, a mixture of the powdered sample and FTIR-grade KBr was used to create potassium bromide disks. These disks were employed for transmittance-mode measurements in a spectral range of 600–4000 cm−1, with a resolution of 4 cm−1. Prior to spectral acquisition, all samples were ground into fine powders. All the collected spectra were presented in transmittance units. X-ray diffraction (XRD) analyses on the piano leads were conducted using a Bruker D8 Advance automatic diffractometer with nickel-filtered CuKα radiation. Data were recorded in a 2θ range of 10–70° with a resolution of 0.02°.
2.5. Kinetic Modeling
Experimental data were fitted to well-known kinetic empirical models, that is, nonlinear PFO (Equation (1)), PSO (Equation (2)), and the Elovich equation (Equation (3)) [
45].
where
qe = adsorptive capacity (mg·g−1) at the equilibrium;
qt = adsorptive capacity (mg·g−1) at time t (min);
k1, k2 = kinetic constants for PFO and PSO, respectively;
α, β = Elovich constants.
The removal efficiencies for all the parameters were calculated according to the following equation (Equation (4)) [
46]:
where
R% = removal percentage;
C0 = concentration at time zero;
Ct = concentration at time t.
The experiments were performed in batch mode, varying the HRT of the process. The model equations were implemented with MATLAB R2023a, and the fitting was performed using the built-in optimization routine “lsqcurvefit”. The results of the fitting were evaluated in terms of correlation coefficient (R2) and root mean square error (RMSE).
3. Results and Discussion
3.1. Biochar Characterization
In this study, FTIR was employed to analyze the biochar sample. The FTIR spectrum provided valuable insights into the chemical composition and functional groups present in the biochar material [
47,
48,
49].
In particular, the following peaks were detected (
Figure 2):
3445 cm−1: The presence of a broad peak at 3445 cm−1 suggests the presence of O-H or N-H functional groups, indicative of potential hydrogen bonding or the presence of hydroxyl or amine groups.
2920 cm−1 and 2850 cm−1: Peaks at 2920 cm−1 and 2850 cm−1 are associated with C-H stretching vibrations in aliphatic chains, indicating the presence of methyl or methylene groups within the biochar structure.
1625 cm−1: The peak at 1625 cm−1 may correspond to carbonyl (C=O) groups, which could be associated with ketonic/aldehydic functionalities within the biochar.
1380 cm−1: A peak at 1380 cm−1 may correspond to C-H bending vibrations, possibly indicating the presence of methyl or methylene groups, similar to the peaks at 2920 cm−1 and 2850 cm−1. They can be also associated with the C=C unsaturated bonds of the aromatic structure of biochar.
880 cm−1: The peak at 880 cm−1 is generally associated with C-H bending vibrations in cyclic or aromatic structures.
Figure 2.
FTIR spectrum of biochar.
Figure 2.
FTIR spectrum of biochar.
The XRD analysis (
Figure 3) of the biochar unveiled crucial insights into its structural properties and mineralogical constituents. Specifically, the peaks situated within a 2θ range of 20°–30° are indicative of the stacking arrangement of aromatic layers, akin to the graphite 002 plane. The broadening of these peaks can be attributed to the limited dimensions of crystallites perpendicular to the aromatic layers, as has been elucidated in previous studies [
50]. Furthermore, conspicuous peaks present in the biochar’s XRD spectrum, though not explicitly labeled, signify the presence of diverse inorganic elements. While a detailed discussion of these components is beyond the scope of this analysis, their prevalence aligns with the heightened concentrations of SiO
2, CaO, and MgO detected within the biochar by the seller. Notably, the most prominent and sharply defined peak observed at 2θ = 26° can be unequivocally attributed to crystalline SiO
2, thus corroborating its existence within the biochar matrix [
51].
3.2. COD Removal
The assessment of COD removal was conducted at various stages (
Figure 4). Phase I was regarded as the start-up phase aimed at acclimatizing the microbes to the leachate and accommodating the increase in loading.
As the feed COD was incrementally elevated, the effluent COD from both the anaerobic and aerobic MBBRs was reduced. The maximum COD removal obtained with the anaerobic system in phase I was about 65%, while a maximum of about 30% was reached with the aerobic step with a total COD removal of about 77%. In phase I, the COD organic loading rate (OLR) varied between 0.33 kgCOD/(m3 day) and 0.63 kgCOD/(m3 day).
From phase II to the end of the experimental period, the process could be considered stable. In this phase, the OLR rose to about 0.88 kgCOD/(m3 day), while the anaerobic HRT decreased to 3 days. The average COD of the anaerobic MBBR effluent remained fairly constant with an increase in COD removal from an average of 58% in phase I to about an average of 79% in phase II. In the same way, the COD of the aerobic MBBR effluent did not change significantly, but in this case, the removal efficiency decreased slightly (from about 26% to about 24%).
In phase III, the OLR rose to about 1.25 kgCOD/(m3 day), and the HRT decreased to 2 days. Again, the COD of the effluents did not change, and the removal percentage of COD increased to about 86% for the anaerobic process and slightly decreased to about 22% for the aerobic process. The overall COD removal efficiency of the system remained constant (about 89%), indicating that HRT had minimal impact on the performance of the entire system given the complementary function of the anaerobic and aerobic reactor. In the anaerobic–aerobic MBBR system, the anaerobic reactor played a pivotal role in effectively removing COD, while the aerobic MBBR served as a vital component in enhancing the treatment efficiency and ensuring the generation of high-quality effluent. The anaerobic reactor displayed impressive performance in significantly reducing the elevated COD concentration in the leachate, even in the presence of a substantial amount of readily degradable substrate.
The effect of HRT on biological processes was evaluated by lowering it during phases II and III. In phase I, it was kept constant at 4 days to promote the acclimatization of the microbial communities. During phase II and phase III, which can be considered quasi-stationary phases, HRT was varied from 3 to 2 days. During phase II and phase III, COD removal increased from about 77% to about 88%, with an average of 85% during phase II and an average of 86% during phase III. Thus, the optimal HRT of the process turned out to be 2 days, as it allowed for the faster treatment of the effluent without loss of efficiency.
The effluent of the anaerobic MBBR was directly used as the feed of the aerobic process. In this way, the OLR of the aerobic reactor was determined by using the removal efficiency of the anaerobic MBBR. The COD removal in the whole aerobic/anaerobic process was not influenced by the differences in the OLR, as is well known in the literature for MBBR systems [
10,
52]. Even with the OLR of the anaerobic MBBR in a high range, >1.2 kgCOD/(m
3 d), the average COD removal efficiency of the anaerobic MBBR was still up to 86%, which indicated that the anaerobic MBBR could tolerate a high OLR.
After the biological process, the average COD of the effluent was 514 ± 24 mg/L, and (from an initial average value of 4698 mg/L) to ensure further purification of the effluent, it was treated via adsorption onto biochar. The biochar adsorption was permitted to reach a maximum removal of 99% at the beginning of phase I, but the efficiency decreased during the experiment until reaching an average removal of 85.5% during phase III (from about 514 mg/L to 75 mg/L).
The results of the experiment demonstrate the effectiveness of the MBBR system in treating leachate and reducing COD levels. The phased approach allowed for gradual adjustments to the system, ensuring the successful acclimatization of microorganisms and the optimization of the treatment performance.
The anaerobic and aerobic reactors showed complementary functions, with the anaerobic reactor achieving higher COD removal rates during the initial phases, while the aerobic reactor enhanced treatment efficiency. The overall COD removal efficiency remained relatively stable, indicating the robustness of the combined anaerobic–aerobic MBBR system. The subsequent adsorption onto biochar further improved the effluent quality, achieving significant COD removal. However, it was observed that the adsorption efficiency decreased slightly over time, possibly because of biochar saturation or changes in adsorption characteristics. In conclusion, the anaerobic–aerobic MBBR system, followed by biochar adsorption, proved to be a successful and efficient approach to treating leachate and reducing COD levels. This research highlights the potential of such integrated treatment systems for wastewater treatment, with the findings contributing to the development of sustainable and effective leachate treatment strategies. Future studies could explore optimization techniques to maintain high adsorption efficiency and investigate the long-term stability and performance of the system under varying operating conditions.
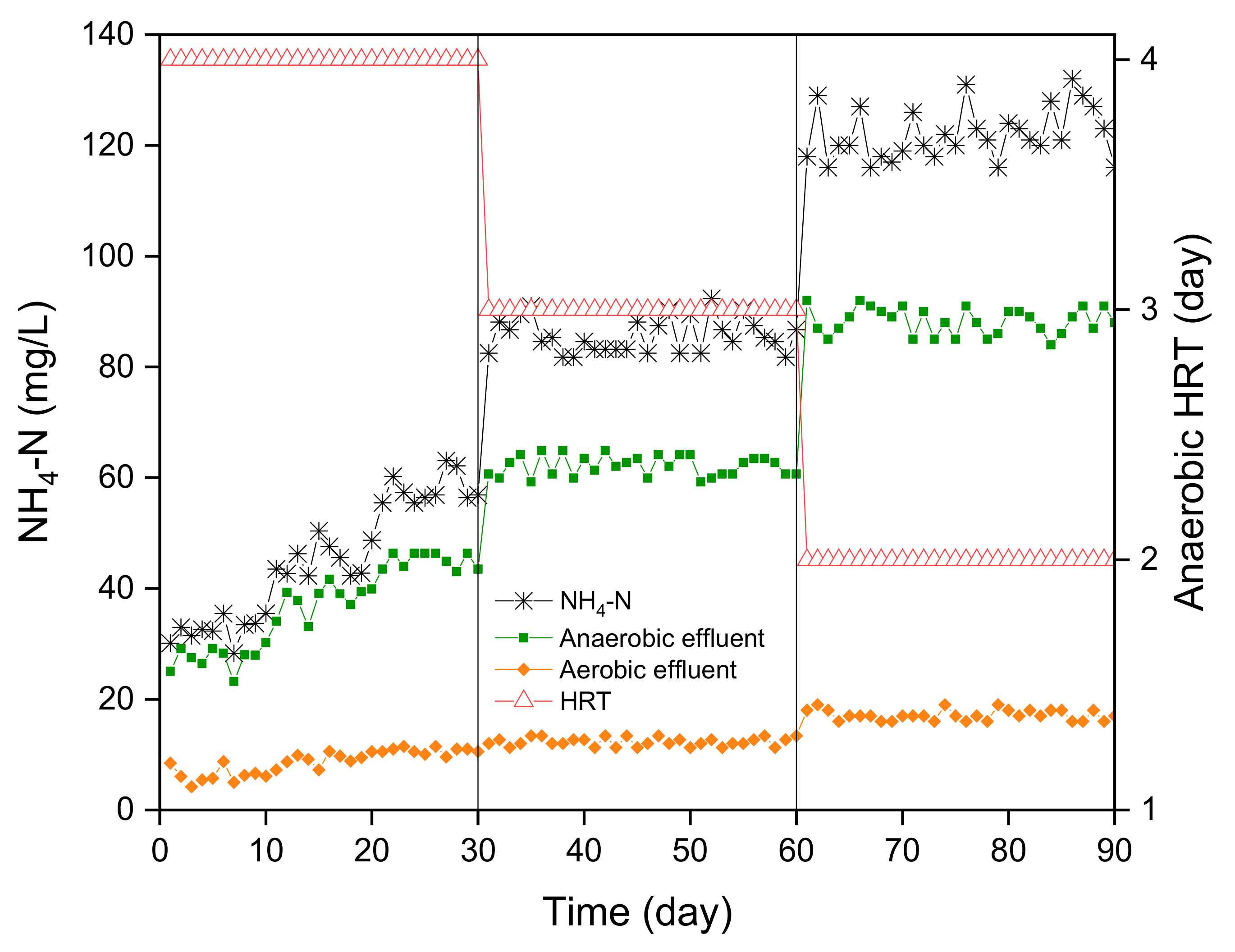
3.3. NH4-N Removal
The removal of ammonium with the anaerobic–aerobic MBBR system was evaluated in this study (
Figure 5). The average concentration of NH
4-N in the LL was 123 mg/L. The main process that contributed to the ammonium removal was the aerobic MBBR because the main mechanism for this elimination is aerobic nitrification [
10].
During the start-up phase, the system exhibited a gradual increase in NH
4-N removal, culminating at day 30 when the start-up phase concluded. This observation suggests the establishment of nitrifying bacterial populations in the aerobic biofilm, which contributed significantly to ammonium conversion into nitrate via nitrification. However, once the system entered phase II, which encompassed the experimental period, the ammonium removal efficiency plateaued at approximately 80%. This stabilization in ammonium removal could be attributed to the establishment of a dynamic equilibrium between the microbial communities and the available substrate. The anaerobic process, instead, allowed for only a little ammonium removal (about 25% in phase III). The removal observed in the anaerobic MBBR can be attributed to the utilization of NH
4-N by the anaerobic microbial assimilation [
10]. The total removal efficiency, encompassing both aerobic and anaerobic contributions, displayed an improvement from approximately 75% during phase I to around 87% during phase III. Despite this positive trend, it is pertinent to note that the overall efficiencies achieved in this study were slightly lower compared with the work of Chen et al. [
10], which reported a remarkable 97% removal. This discrepancy could be attributed to the specific characteristics of the influent wastewater employed in this study, particularly its higher COD content.
Again, HRT showed a negligible effect on the removal percentage of NH4-N. Unlike COD, however, the removal efficiency remained almost constant from phase I to phases II and III with an increase from about 80% in phase I to about 85% in phases II and III. The lower efficiency in phase I—despite the HRT of the higher aerobic process, which should favor the nitrification process—may be due to the gradual acclimatization of the nitrifying species to the quasi-steady state of phases II and III. After the biological process, the average NH4-N of the effluent was 17 mg/L (from an initial average value of 123 mg/L). The biochar adsorption reached a maximum removal of 94% (from about 17 mg/L to 0.97 mg/L).
The influence of high COD content on the efficiency of nitrification processes is well documented. When wastewater contains elevated levels of organic compounds and potential nitrification inhibitors, heterotrophic bacteria tend to outcompete nitrifiers for resources. The relatively rapid growth rate of heterotrophic bacteria allows them to dominate the biofilm surface, limiting the colonization and proliferation of autotrophic nitrifiers. Consequently, only a restricted number of nitrifying bacteria can establish themselves on the outer surface of the biofilm, leading to suboptimal conditions for ammonium removal. After the anaerobic–aerobic process, the average NH4-N of the effluent was 17.33 mg/L. Moreover, the incorporation of biochar adsorption in the study, after phase III, demonstrated promising results, achieving a maximum ammonium removal of 94%. This observation raises questions regarding the underlying mechanisms driving enhanced ammonium removal through biochar adsorption and its synergistic interaction with the MBBR system.
3.4. TP Removal
The anaerobic–aerobic MBBR eliminated a maximum of about 60% of the TP from LL (
Figure 6). As in the case of NH
4-N, the aerobic process is predominant in TP removal. In fact, it is well known that biologically enhanced phosphorus removal is dominated by anaerobic phosphorus release and aerobic phosphorus uptake [
53]. The lower efficiency for TP removal compared with that of NH
4-N can be explained by the high efficiency obtained for COD removal. In fact, when, in a biological process, the removal of COD is high, the microorganisms responsible for the COD removal are in competition with a phosphorus-accumulating organism (PAO), limiting TP removal [
54]. In the same way, the nitrifying bacteria are potential competitors for the PAO [
55]. After the biological process, the average TP of the effluent was 8.2 mg/L (from an initial average value of 22 mg/L). TP removal via adsorption onto biochar reached a removal of 92% (from an average of 8.2 mg/L to an average of 0.68 mg/L during phase III). In the case of TP removal, the removal efficiency remained quite low for the entire duration. The HRT-lowering showed, for the aerobic process, a quite negative effect; in fact, during phases II and III, the removal percentage, because of aerobic processes, never reached a stable condition, with widely varying values (from about 7% to about 60%). Overall, however, the loss of efficiency in the aerobic process was compensated for by the increase in efficiency in the anaerobic one and vice versa. This behavior may be due to the influence of the high concentrations of COD and NH
4-N in phases II and III, which, as found in the literature, are competitive against biological phosphorous removal. In fact, high organic loads favor the growth of non-polyphosphate-accumulating organisms and lead to polyphosphate-accumulating organisms being screened out of the system [
56,
57]. Our study found that the integrated anaerobic–aerobic MBBR and biochar adsorption method was highly effective in removing TP from landfill leachate, with a total maximum removal rate of 99.9%. This is a significant finding because TP is a major pollutant in landfill leachate and can cause eutrophication in receiving waterbodies, leading to harmful algal blooms and other ecological problems. The results showed that the integrated system was highly effective in removing TP from landfill leachate, which is a significant step toward developing more effective and sustainable methods of treating this type of wastewater.
3.5. Adsorption Kinetics
To examine the adsorption kinetics of the biochar, PFO, PSO, and Elovich kinetic models were carefully employed and finely tuned to the experimental data obtained through batch mode experiments (as depicted in
Figure 7 for COD,
Figure 8 for NH
4-N, and
Figure 9 for TP). BOD
5-removal kinetics were not performed because, with the respirometric method used, the error is rather high, and therefore, we preferred to measure only the BOD
5 of the raw effluent and the effluent after treatment to assess the initial and final BOD
5/COD ratio. In general, the adsorption process exhibited an initial rapid increase, followed by a slower approach to a plateau, where the amount of adsorbed contaminant remained nearly constant. The majority of contaminants were adsorbed within the first 30 min of the experiment (71.4%, 77.6%, and 75.6% for COD, NH
4-N, and TP, respectively). This observation can be attributed to the availability of adsorption sites on the material’s surface at the beginning of the experiment [
58]. The adsorption capacity increases rapidly during the initial phase, and it takes approximately 60 min to reach a steady state (84.4%, 93.7%, and 87.8% for COD, NH
4-N, and TP respectively). The rapid removal rates of the adsorbate and the quick attainment of equilibrium are key characteristics that indicate the effectiveness of the adsorbent [
59].
Table 2 presents the kinetic parameters obtained by fitting empirical kinetic models to the experimental data. To assess the goodness of fit of the kinetic models to the experimental data, various parameters were considered, including adjusted R
2 values and root mean square error (RMSE) values. Among the kinetic models evaluated (PFO, PSO, and Elovich), the fitting results of both the first-order and second-order models demonstrated a remarkable similarity in terms of R
2, Q
max, and RMSE. This suggests that the adsorption process on biochar may exhibit kinetics that are intermediate between the first and second orders (
Table 2).
The resemblance in R2 values indicates that both models adequately describe the experimental data, while the Qmax values imply that the maximum adsorption quantity is similar for both models. This observation could imply that the adsorption process involves both an external diffusion component (as suggested by the first order) and a chemical adsorption component (as indicated by the second order). Furthermore, the comparable RMSE values between the two models suggest that both models fit the experimental data with similar accuracy. However, it should be noted that this similarity might mask further intricacies in the adsorption process.
The adaptability of the Elovich model to our experimental data was significantly poorer, with a low R2 and a significantly higher RMSE. This discrepancy suggests that the Elovich model may not be the best choice for describing our activated carbon adsorption system. This result could be associated with the complexity of our system and its surface heterogeneity.
The discrepancy between the Elovich model and the other models can be interpreted in terms of mass transfer. While the first- and second-order models showed good adaptability, suggesting the importance of both external diffusion and chemical adsorption reactions, the Elovich model, with its varying adsorption rate over time and assumption of surface heterogeneity, might not adequately capture the details of our adsorption system. This discrepancy could reflect greater complexity in mass transfer, potentially involving surface phenomena and variations in the adsorption rate during the process. Further research and analyses are required to fully unveil the specific mass transfer mechanisms at play in our activated carbon adsorption system.
In conclusion, while the first- and second-order models appear to provide a more accurate representation of our adsorption process, the Elovich model does not align with our experimental data and suggests the need for further investigations to fully understand mass transfer in this complex system.
The investigation of adsorption kinetics using empirical models (PFO, PSO, and Elovich) provided valuable insights into the behavior of biochar as an adsorbent for COD, NH
4-N, and TP removal. The experimental data demonstrated that the biochar rapidly adsorbed contaminants from the wastewater, achieving equilibrium within a short duration. These findings contribute to the understanding of biochar’s adsorption capacity and its potential applicability in wastewater treatment for efficient contaminant removal.
Figure 10 and
Table 3 show a summary of the removal percentages and the final quality of the effluent achieved with each process for each parameter investigated.
4. Conclusions
In conclusion, the integrated anaerobic–aerobic MBBR and biochar adsorption method proposed in this study offers a promising solution for the efficient removal of organic matter and nutrients from landfill leachate. The MBBR provides a suitable environment for the growth of microorganisms that can degrade organic matter and nutrients, while biochar adsorption removes any remaining pollutants that are not biodegradable. The results showed that the integrated system achieved high removal efficiencies for COD, NH4-N, and TP, with maximum removal rates of 98.5%, 91.2%, and 99.9%, respectively. Removal percentages of about 77%, 87%, and 92% after using the anaerobic–aerobic MBBR system for COD, NH4-N, and TP were reached, respectively (from 4696 to 514 mg/L, from 123 to 17 mg/L, and from 22 to 8.2 mg/L, respectively). The removal adsorption contributions of the biochar were about 85.4%, 95.9%, and 91.7% for COD, NH4-N, and TP (from 514 to 75 mg/L, from 17 to 0.97 mg/L, and from 8.2 to 0.68 mg/L, respectively).
It is crucial to emphasize the significance of developing more effective and sustainable landfill leachate treatment methods given their substantial impacts on both public health and the environment. Compared with conventional treatments, our proposed method boasts advantages such as lower energy consumption, reduced sludge generation, and heightened removal efficiency. However, the long-term performance and sustainability of this integrated system across various conditions, as well as its economic viability, require further investigation.
This study sheds light on the development of advanced and sustainable approaches to addressing the global issue of landfill leachate treatment. By combining an anaerobic–aerobic MBBR and biochar adsorption, we took a significant step toward mitigating the environmental challenges posed by landfill leachate. Future research endeavors should focus on optimizing this method, exploring its applicability in diverse settings, and assessing its economic feasibility to pave the way for a cleaner and healthier environment.