Cu(II) and Cr(VI) Removal in Tandem with Electricity Generation via Dual-Chamber Microbial Fuel Cells
Abstract
1. Introduction
2. Materials and Methods
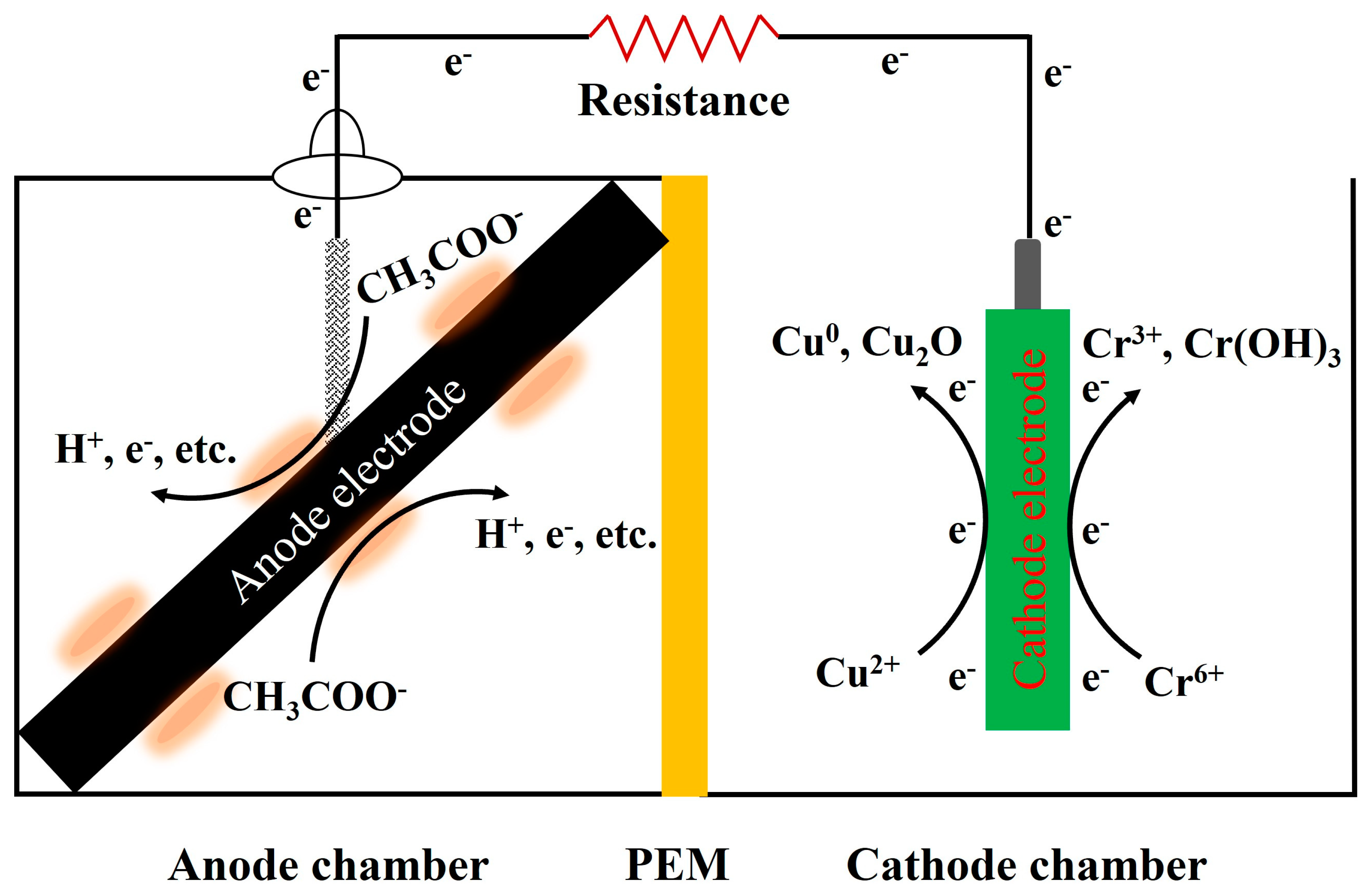
2.1. MFC Construction, Inoculation, and Operation
2.2. Experiment Design
2.3. Analytical Methods and Calculations
2.3.1. Electrical Properties of MFCs
2.3.2. Concentration and Morphology of Heavy Metals
3. Results and Discussion
3.1. The Performance of Electricity Generation
3.2. Heavy Metals Removal in MFCs
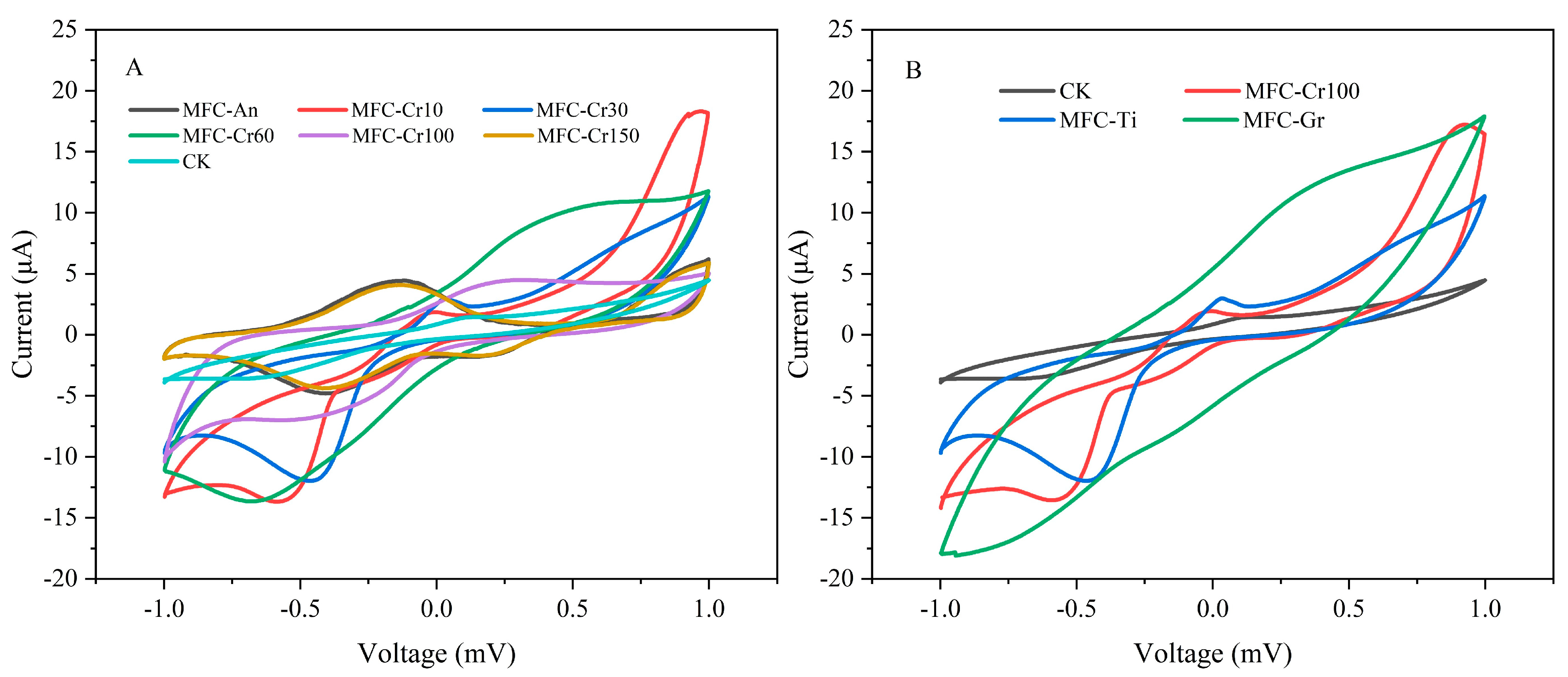
3.3. Cyclic Voltammetry Analysis and Reduction Products on the Cathode Electrode
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, S.-H.; Huang, W.-J.; Lin, C.-W.; Zhu, T.-J. Enhanced copper removal and bioelectricity generation in sediment microbial fuel cells through biostimulation and bioaugmentation. J. Clean. Prod. 2022, 350, 131458. [Google Scholar] [CrossRef]
- Matsena, M.T.; Chirwa, E.M.N. Hexavalent chromium-reducing microbial fuel cell modeling using integrated Monod kinetics and Butler-Volmer equation. Fuel 2022, 312, 122834. [Google Scholar] [CrossRef]
- Rajendran, S.; Priya, T.A.K.; Khoo, K.S.; Hoang, T.K.A.; Ng, H.-S.; Munawaroh, H.S.H.; Karaman, C.; Orooji, Y.; Show, P.L. A critical review on various remediation approaches for heavy metal contaminants removal from contaminated soils. Chemosphere 2022, 287, 132369. [Google Scholar] [CrossRef]
- Yu, Y.; Ali, J.; Yang, Y.; Kuang, P.; Zhang, W.; Lu, Y.; Li, Y. Synchronous Cr(VI) Remediation and Energy Production Using Microbial Fuel Cell from a Subsurface Environment: A Review. Energies 2022, 15, 1989. [Google Scholar] [CrossRef]
- Yu, H.; Zhao, Q.; Liu, X.; Meng, F.; Ruan, L.; Sun, T.; Liu, W.; Zhu, Y.; Li, W.; Meng, F.; et al. Enhanced chromium recovery and simultaneous sludge degradation in a novel bioelectrochemical system assembled with bio/abio-cathodes. Sep. Purif. Technol. 2020, 250, 117229. [Google Scholar] [CrossRef]
- Wang, H.; Ren, Z.J. Bioelectrochemical metal recovery from wastewater: A review. Water Res. 2014, 66, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Hemdan, B.; Garlapati, V.K.; Sharma, S.; Bhadra, S.; Maddirala, S.; Varsha, K.M.; Motru, V.; Goswami, P.; Sevda, S.; Aminabhavi, T.M. Bioelectrochemical systems-based metal recovery: Resource, conservation and recycling of metallic industrial effluents. Environ. Res. 2022, 204, 112346. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.-M.; Ong, S.-A.; Ho, L.-N.; Wong, Y.-S.; Thung, W.-E.; Teoh, T.-P. The reaction of wastewater treatment and power generation of single chamber microbial fuel cell against substrate concentration and anode distributions. J. Environ. Health Sci. Eng. 2020, 18, 793–807. [Google Scholar] [CrossRef]
- Qiu, R.; Zhang, B.; Li, J.; Lv, Q.; Wang, S.; Gu, Q. Enhanced vanadium (V) reduction and bioelectricity generation in microbial fuel cells with biocathode. J. Power Sources 2017, 359, 379–383. [Google Scholar] [CrossRef]
- Wang, H.; Long, X.; Zhang, J.; Cao, X.; Liu, S.; Li, X. Relationship between bioelectrochemical copper migration, reduction and electricity in a three-chamber microbial fuel cell. Chemosphere 2020, 241, 125097. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, L.; Pan, Y.; Quan, X.; Li Puma, G. Impact of Fe(III) as an effective electron-shuttle mediator for enhanced Cr(VI) reduction in microbial fuel cells: Reduction of diffusional resistances and cathode overpotentials. J. Hazard. Mater. 2017, 321, 896–906. [Google Scholar] [CrossRef]
- Gangadharan, P.; Nambi, I.M. The performance of Cu(2+)as dissolved cathodic electron-shuttle mediator for Cr(6+)reduction in the microbial fuel cell. Sustain. Environ. Res. 2020, 30, 19. [Google Scholar] [CrossRef]
- Zhang, B.; Feng, C.; Ni, J.; Zhang, J.; Huang, W. Simultaneous reduction of vanadium (V) and chromium (VI) with enhanced energy recovery based on microbial fuel cell technology. J. Power Sources 2012, 204, 34–39. [Google Scholar] [CrossRef]
- Mier, A.A.; Olvera-Vargas, H.; Mejía-López, M.; Longoria, A.; Verea, L.; Sebastian, P.J.; Arias, D.M. A review of recent advances in electrode materials for emerging bioelectrochemical systems: From biofilm-bearing anodes to specialized cathodes. Chemosphere 2021, 283, 131138. [Google Scholar] [CrossRef] [PubMed]
- Rusli, S.F.N.; Abu Bakar, M.H.; Loh, K.S.; Mastar, M.S. Review of high-performance biocathode using stainless steel and carbon-based materials in Microbial Fuel Cell for electricity and water treatment. Int. J. Hydrog. Energy 2019, 44, 30772–30787. [Google Scholar] [CrossRef]
- Li, M.; Zhou, S.; Xu, Y. Performance of Pb(II) reduction on different cathodes of microbial electrolysis cell driven by Cr(VI)-reduced microbial fuel cell. J. Power Sources 2019, 418, 1–10. [Google Scholar] [CrossRef]
- Watanabe, K. Recent Developments in Microbial Fuel Cell Technologies for Sustainable Bioenergy. J. Biosci. Bioeng. 2008, 106, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Chang, I.S.; Kim, B.H. Continuous electricity production from artificial wastewater using a mediator-less microbial fuel cell. Bioresour. Technol. 2006, 97, 621–627. [Google Scholar] [CrossRef]
- Tandukar, M.; Huber, S.J.; Onodera, T.; Pavlostathis, S.G. Biological chromium(VI) reduction in the cathode of a microbial fuel cell. Environ. Sci. Technol. 2009, 43, 8159–8165. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, I.; Ossieur, W.; Verhaege, M.; Verstraete, W. Continuous microbial fuel cells convert carbohydrates to electricity. Water Sci. Technol. 2005, 52, 515–523. [Google Scholar] [CrossRef]
- Gustave, W.; Yuan, Z.; Liu, F.; Chen, Z. Mechanisms and challenges of microbial fuel cells for soil heavy metal(loid)s remediation. Sci Total Environ. 2021, 756, 143865. [Google Scholar] [CrossRef]
- Heijne, A.T.; Liu, F.; Weijden, R.v.d.; Weijma, J.; Buisman, C.J.N.; Hamelers, H.V.M. Copper Recovery Combined with Electricity Production in a Microbial Fuel Cell. Environ. Sci. Technol. 2010, 44, 4376–4381. [Google Scholar] [CrossRef]
- Oh, S.-E.; Logan, B.E. Proton exchange membrane and electrode surface areas as factors that affect power generation in microbial fuel cells. Appl. Microbiol. Biotechnol. 2006, 70, 162–169. [Google Scholar] [CrossRef]
- Ouitrakul, S.; Sriyudthsak, M.; Charojrochkul, S.; Kakizono, T. Impedance analysis of bio-fuel cell electrodes. Biosens. Bioelectron. 2007, 23, 721–727. [Google Scholar] [CrossRef]
- Wang, J.; Song, X.; Wang, Y.; Abayneh, B.; Ding, Y.; Yan, D.; Bai, J. Microbial community structure of different electrode materials in constructed wetland incorporating microbial fuel cell. Bioresour. Technol. 2016, 221, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Gil, G.-C.; Chang, I.-S.; Kim, B.H.; Kim, M.; Jang, J.-K.; Park, H.S.; Kim, H.J. Operational parameters affecting the performannce of a mediator-less microbial fuel cell. Biosens. Bioelectron. 2003, 18, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yuan, Y.; Li, F.-b.; Feng, C.-h. In-situ Cr(VI) reduction with electrogenerated hydrogen peroxide driven by iron-reducing bacteria. Bioresour. Technol. 2011, 102, 2468–2473. [Google Scholar] [CrossRef] [PubMed]
- Cecconet, D.; Callegari, A.; Capodaglio, A.G. Bioelectrochemical Systems for Removal of Selected Metals and Perchlorate from Groundwater: A Review. Energies 2018, 11, 2643. [Google Scholar] [CrossRef]
- Li, M.; Zhou, S. Efficacy of Cu(II) as an electron-shuttle mediator for improved bioelectricity generation and Cr(VI) reduction in microbial fuel cells. Bioresour. Technol. 2019, 273, 122–129. [Google Scholar] [CrossRef]
- Hidayat, A.R.P.; Widyanto, A.R.; Asranudin, A.; Ediati, R.; Sulistiono, D.O.; Putro, H.S.; Sugiarso, D.; Prasetyoko, D.; Purnomo, A.S.; Bahruji, H.; et al. Recent development of double chamber microbial fuel cell for hexavalent chromium waste removal. J. Environ. Chem. Eng. 2022, 10, 107505. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, X.; Lei, L. Electricity production during the treatment of real electroplating wastewater containing Cr6+ using microbial fuel cell. Process Biochem. 2008, 43, 1352–1358. [Google Scholar] [CrossRef]
- Wu, X.; Ren, X.; Owens, G.; Brunetti, G.; Zhou, J.; Yong, X.; Wei, P.; Jia, H. A Facultative Electroactive Chromium(VI)-Reducing Bacterium Aerobically Isolated From a Biocathode Microbial Fuel Cell. Front. Microbiol. 2018, 9, 2883. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Huang, L.; Zhou, P.; Quan, X.; Li Puma, G. Correlation between circuital current, Cu(II) reduction and cellular electron transfer in EAB isolated from Cu(II)-reduced biocathodes of microbial fuel cells. Bioelectrochemistry 2017, 114, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Beretta, G.; Daghio, M.; Tofalos, A.E.; Franzetti, A.; Mastorgio, A.F.; Saponaro, S.; Sezenna, E. Microbial Assisted Hexavalent Chromium Removal in Bioelectrochemical Systems. Water 2020, 12, 466. [Google Scholar] [CrossRef]



| MFC Type | Control Conditions | Average Current (mA) | Maximum Current Density (mA/m2) | Coulombic Efficiency (%) | Ohmic Internal Resistance (Ω) |
|---|---|---|---|---|---|
| MFC-An | Anaerobic cathode chamber, 100 mg/L Cr, stainless steel | 0.045 | 16.97 | 19.21 | 5.26 |
| MFC-Cr10 | 10 mg/L Cr, stainless steel | 0.17 | 93.35 | 28.63 | 3.92 |
| MFC-Cr30 | 30 mg/L Cr, stainless steel | 0.064 | 50.92 | 26.92 | 7.24 |
| MFC-Cr60 | 60 mg/L Cr, stainless steel | 0.061 | 33.95 | 22.87 | 8.67 |
| MFC-Cr100 | 100 mg/L Cr, stainless steel | 0.066 | 28.29 | 21.41 | 19.45 |
| MFC-Cr150 | 150 mg/L Cr, stainless steel | 0.050 | 11.32 | 18.97 | 81.01 |
| MFC-Ti | 100 mg/L Cr, titanium sheet | 0.20 | 33.95 | 38.18 | 8.24 |
| MFC-Gr | 100 mg/L Cr, graphite plate | 0.29 | 62.23 | 50.71 | 6.47 |
| CK | MFC-An | MFC-Cr10 | MFC-Cr100 | MFC-Cr150 | MFC-Ti | MFC-Gr | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Element | WP 1 (%) | AP 2 (%) | WP 1 (%) | AP 2 (%) | WP 1 (%) | AP 2 (%) | WP 1 (%) | AP 2 (%) | WP 1 (%) | AP 2 (%) | WP 1 (%) | AP 2 (%) | WP 1 (%) | AP 2 (%) |
| Ni | 17.03 | 14.94 | 0 | 0 | 0 | 0 | 3.12 | 2.21 | 1.84 | 1.08 | 0 | 0 | 0 | 0 |
| O | 3.24 | 8.36 | 18.3 | 20.73 | 19.43 | 32.92 | 25.09 | 30.58 | 28.59 | 34.96 | 29.99 | 59.45 | 21.71 | 21.21 |
| Ca | 0 | 0 | 3.85 | 6.47 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Cl | 0 | 0 | 6.13 | 1.66 | 2.77 | 2.15 | 0.81 | 0.78 | 0 | 0 | 3.27 | 2.92 | 2.89 | 1.39 |
| C | 6.99 | 14.03 | 0.69 | 0.91 | 0.54 | 0.53 | 4.09 | 4.16 | 4.86 | 3.92 | 0 | 0 | 6.41 | 3.12 |
| Cr | 7.3 | 3.73 | 18.32 | 18.34 | 19.04 | 15.09 | 22.34 | 17.83 | 24.34 | 26.49 | 38.95 | 23.76 | 30.47 | 31.2 |
| Fe | 63.45 | 46.89 | 14.67 | 14.35 | 13.49 | 16.72 | 3.3 | 11.95 | 2.02 | 4.48 | 0 | 0 | 0.13 | 0.11 |
| Cu | 0 | 0 | 38.05 | 37.54 | 39.18 | 27.66 | 40.09 | 31.87 | 35.52 | 26.93 | 27.79 | 13.87 | 37.94 | 42.6 |
| P | 0 | 0 | 0 | 0 | 5.55 | 4.94 | 4.09 | 4.16 | 2.83 | 2.15 | 0 | 0 | 0.45 | 0.37 |
| Si | 0.78 | 1.14 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Mn | 1.22 | 10.92 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ta | 0 | 0 | 0 | 0 | 0 | 0 | 0.74 | 0.61 | 0 | 0 | 0 | 0 | 0 | 0 |
| total | 100% | 100% | 100% | 100% | 100% | 100% | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Li, Y.; Mi, Y.; Wang, D.; Wang, Z.; Meng, H.; Jiang, C.; Dong, W.; Li, J.; Li, H. Cu(II) and Cr(VI) Removal in Tandem with Electricity Generation via Dual-Chamber Microbial Fuel Cells. Sustainability 2023, 15, 2388. https://doi.org/10.3390/su15032388
Wang H, Li Y, Mi Y, Wang D, Wang Z, Meng H, Jiang C, Dong W, Li J, Li H. Cu(II) and Cr(VI) Removal in Tandem with Electricity Generation via Dual-Chamber Microbial Fuel Cells. Sustainability. 2023; 15(3):2388. https://doi.org/10.3390/su15032388
Chicago/Turabian StyleWang, Hui, Yu Li, Yue Mi, Dongqi Wang, Zhe Wang, Haiyu Meng, Chunbo Jiang, Wen Dong, Jiake Li, and Huaien Li. 2023. "Cu(II) and Cr(VI) Removal in Tandem with Electricity Generation via Dual-Chamber Microbial Fuel Cells" Sustainability 15, no. 3: 2388. https://doi.org/10.3390/su15032388
APA StyleWang, H., Li, Y., Mi, Y., Wang, D., Wang, Z., Meng, H., Jiang, C., Dong, W., Li, J., & Li, H. (2023). Cu(II) and Cr(VI) Removal in Tandem with Electricity Generation via Dual-Chamber Microbial Fuel Cells. Sustainability, 15(3), 2388. https://doi.org/10.3390/su15032388












