1. Introduction
The recycling of concrete waste from construction and demolition is becoming more and more urgent as virgin aggregate scarcity is increasing. Most current applications only consider coarse fractions (particle size >2–4 mm) for high-end valorization such as in recycled aggregate concrete [
1,
2]. By recycling the coarse fraction of recycled aggregate, a significant negative impact on the environment can be mitigated [
3,
4], which in turn results in less consumption of natural resources. The coarse fraction is typically economically viable as it requires little to no adaptation for the production of new fresh concrete. Fine recycled concrete aggregate (FRCA), which concentrates the majority of the hardened paste, is typically downcycled and used in low-value applications [
5].
It is necessary to circumvent the challenges associated with the valorization of FRCA by employing innovative valorization technologies to convert waste concrete into secondary high-value raw materials and complete the recycling process [
4,
5,
6,
7]. To enhance the properties of FRCA, the inconvenient adhered cement paste of particles [
8,
9] may be densified by means of carbonation [
6]. This technique has been applied mostly to the coarse fraction [
10,
11,
12], and studies on FRCA are scarce so far. Differences in particle size can be an important factor in terms of the efficiency of carbonation treatments. As carbonation proceeds, the outer layer of aggregate particles becomes denser, and the reaction rate slows down. According to [
13], the relative humidity during the carbonation process plays an important role in the formation of this outer rim. With a relative humidity near saturation during the reaction, moisture accumulates at the surfaces of aggregate particles with liquid running-off and carrying dissolved ions (i.e., leaching of calcium). Porosity increases as the precipitation of carbonates does not occur within the pore structure of the hardened cement paste. For lower relative humidities, moisture forming during the carbonation reaction finds sufficient time to evaporate, and calcium accumulates near the surface where carbonation occurs. This results in a more efficient densification of the outer surfaces of particles [
13]. The effect of outer carbonation is exacerbated when carbonating aggregates with lower surface areas [
14]. Therefore, a different efficiency of the process can be expected for FRCA than for coarse RCA.
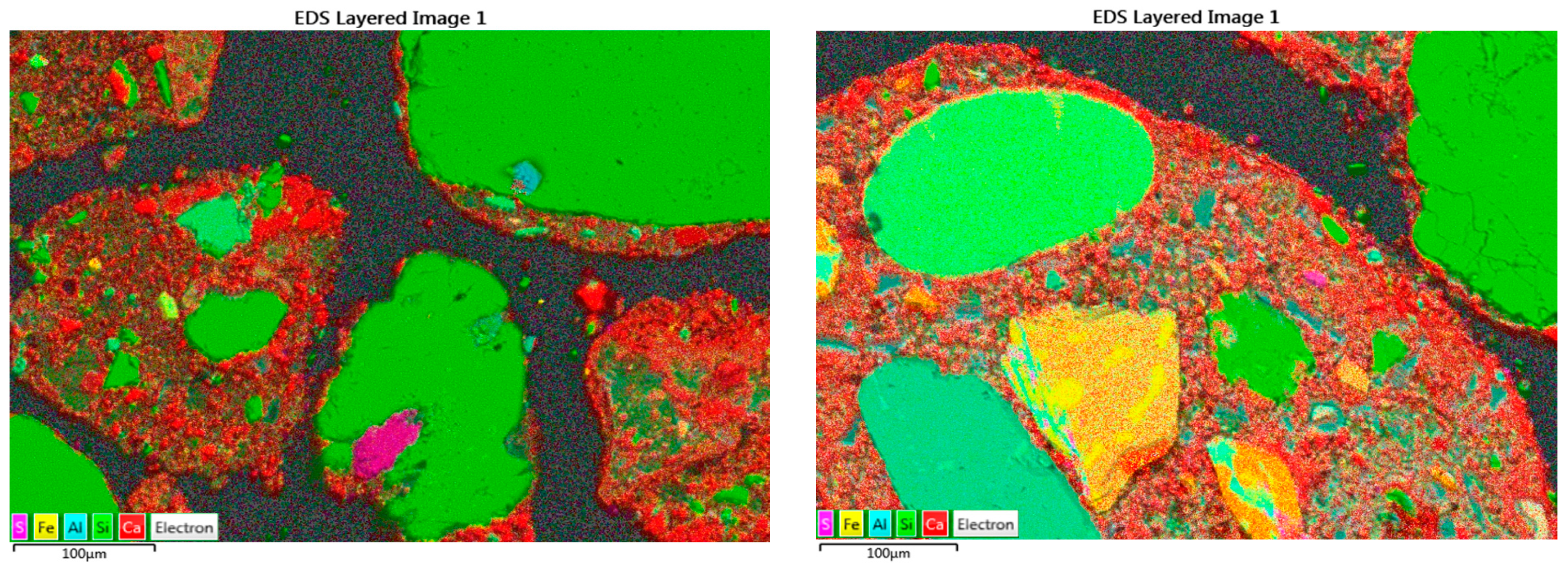
Upon carbonation, the microstructure of the FRCA would be densified as a result of the precipitation of calcium carbonate while the process is occurring [
15]. Such CO
2-enhancement techniques produce carbonation products that can cover not only the micropores of the attached mortar but also microcracks in recycled aggregates, potentially improving the aggregate performance. Both the new interfacial transition zone (ITZ) between the old and new mortar and the old ITZ between the old mortar and original aggregate are enhanced through CO
2 treatment [
16]. At the same time, CO
2 is being removed from the atmosphere, i.e., the FRCA has the potential to serve as a carbon sink during this carbon utilization strategy.
High-tech carbonation methods have been proposed earlier in the literature [
17], but such methods require significant investment and processing, which can reduce the competitiveness of the treated FRCA significantly. For example, provision of Ca
+-rich wastewater has been shown to be effective in treating coarse RCA for reduced water absorption [
18,
19]. A similar approach for fine fractions seems possible, but consideration to differences in size and composition should be given.
Because aggregate should remain a low-cost component of concrete mixes, it appears critical for potential upscaling that the treatment’s complexity is kept to a minimum and feasible in the field. In the context of concrete production plants, the use of wastewater and ambient pressure reaction chambers appear simple to implement.
Taking into account previous approaches in the literature, this current study investigated the feasibility of achieving significant improvements in accelerated carbonation using only minimally complicated technology. Furthermore, the variation in composition of FRCA with particle size and its impact on the efficiency of the carbonation treatment has not yet been addressed in the literature. In the present study, different size fractions of FRCA were subjected to an eco-efficient method of accelerated carbonation. Moreover, FRCA is frequently stored for long periods and suffers extensive natural carbonation. For this reason, in the present study, additional contribution of carbonatable material was pursued. FRCA was previously sprayed with wastewater from the production of mortar as an additional source of portlandite for further carbonation in an ambient pressure chamber. The findings revealed evidence of a limited relationship between the level of enhancement and the amount of precipitated calcium carbonate in the FRCA.
2. Materials and Methods
An industrial FRCA (Van Pelt Bouwmaterialen, Schoten, Belgium) was used (
Figure 1). The sum of crushed concrete, mortar and unbound natural aggregate in it was >90%. The FRCA, with the particle size distribution as shown in
Figure 2, was sieved into fractions <0.125 mm (F1), 0.125–0.25 mm (F2), 0.25–0.5 mm (F3), 0.5–1 mm (F4), 1–2 mm (F5) and 2–4 mm (F6) with the purpose of determining the variable efficiency of the proposed treatment on each fraction. Additionally, a CEN sand was used as reference material. The properties tested before and after carbonation included porosity by water absorption, analysis of SEM/BSE+EDS images, and composition by loss on ignition as well as TGA and XRD+Rietveld+PONKCS analysis [
20].
The electrical conductivity method (performed as explained in [
21]) was preferred over the NBN EN 1097-6 method [
22] as the latter tends to overestimate water absorption with respect to the electrical conductivity method.
Figure 3 exemplifies this, showing the collapse of the cone only occurring after over drying and for an undersaturated state according to the inflection point in conductivity of the skeleton (left branch in
Figure 3) as also indicated in [
21]. Scanning electron microscopy (SEM) was performed in a Jeol JSM-7600F Field Emission Scanning Electron Microscope, FESEM, with an in-lens Schottky electron source and elemental analysis with energy dispersive spectrometry (EDS). Samples were oven-dried at 40 °C and impregnated with epoxy in a vacuum chamber for a minimum of 2 h to remove most air in accessible pores and hence allow better infiltration of the resin. After hardening for 24 h, samples were polished with progressively finer abrasives (320–500–1200 grade SiC papers) followed by polishing with 3 and 1 µm diamond paste. Samples were afterwards carbon-coated with a 20 nm layer. SEM images were acquired at 250× magnification with a working distance of 8 mm, 15 keV. Energy-dispersive spectrometry (EDS) allowed us to make chemical mappings from the samples.
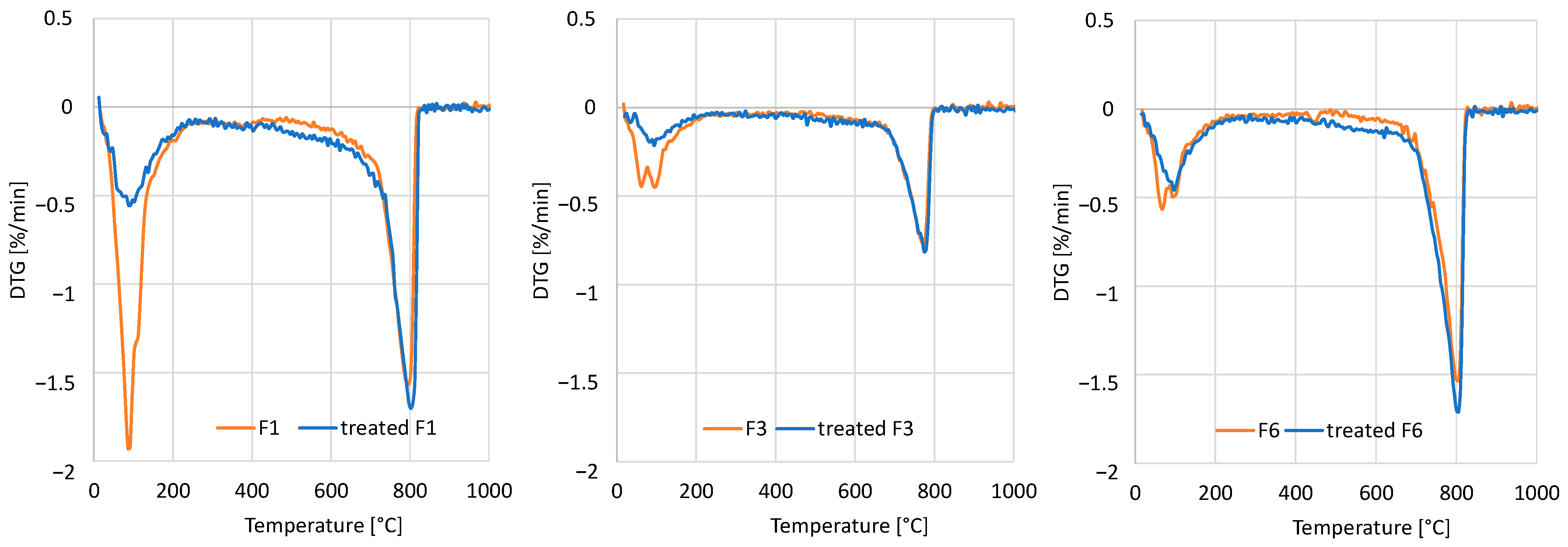
Loss on ignition was determined on duplicates for each sample. For each measurement, around (1 ± 0.1) g of sample, manually ground with mortar and pestle to <0.125 μm, was placed in pre-ignited crucibles and calcined at 950 °C for 2 h. The weight loss respective to the initial dry weight was computed as loss on ignition. Complementarily, thermogravimetric analysis was performed under a protective atmosphere of nitrogen in a NETZSCH STA 449F3 STA449F3A-0384-M device. Due to the features of the device, each measurement was performed on only 50 mg of sample, and for improved representativeness in terms of relative composition of cement paste and sand, the results were scaled to match those of the total loss on ignition resulting from larger samples. The recorded mass loss relative to the total weight was differentiated to obtain the derivative thermogravimetry (DTG).
For XRD measurements, powder samples (samples ground to <125 μm) were laterally loaded in the sample holders to mitigate the preferred orientation effects for XRD data collection. Only fractions F1, F3, and F6 were tested for XRD. A Thermo Scientific ARL X’tra diffractometer with a Peltier-cooled detector in BraggBrentano geometry was used for data collection with 30 mA and 40 kV tube operating conditions and a Cu X-ray source. Data were collected with a 0.02 2θ step size, from 5–70°, with a time per step of 1 s. The phase identification and Rietveld quantitative phase analysis were performed using the TOPAS Academic V6 software package.
The FRCA was almost fully naturally carbonated while stored in the plant. An additional source of portlandite was necessary for the treatment. Prior to carbonation, the FRCA fractions were pretreated by spraying them with wastewater (total solids = 0.23 wt.%, pH = 12.06) from mortar production. The sprayed amount was such that all particles were wet but not soaked (water/solid mass ratios ranged from 0.134 for F1 to 0.075 for F6). The content of calcium in the wastewater corresponded to that of an oversaturated limewater with additional content of solids. The average value determined by acid titration of the unfiltered solution was 1.5 g Ca2+/L solution. Considering this contribution, and the amounts of solution sprayed on the samples, the content of calcium incorporated into the samples was estimated at 0.203 (F1) to 0.113 (F6) g Ca2+/kg per sample. Once sprayed, the samples remained to dry in the laboratory environment for 24 h. Following this, they were placed in the carbonation chamber (20 °C, 60% RH, and 1% CO2) for a period of 2 weeks for full carbonation.
The untreated and treated FRCAs were also used in mortar mixes. Three mixes were made: one control mix with 100% siliceous sand as fine aggregates (NAC), one with 30 wt.% of the sand replaced by untreated FRCA (RAC), and one with 30 wt.% of treated FRCA (CRAC). The density of the aggregates was 2.65, 2.36, and 2.37 for the natural sand, FRCA, and carbonated FRCA, respectively. The whole size fraction was used for this replacement. For approaching realistic situations, the particle size distribution of the CEN sand was not mimicked, i.e., its volume was partially replaced with the FRCA with its own particle size distribution. For the RAC and CRAC mixes, compensation water was added to prevent water uptake from the mix by the dry FRCA, computed as 70% of the full water absorption capacity of the FRCA [
23]. The mix compositions are presented in
Table 1. The 28-day compressive strength and the water absorption under vacuum were tested on mortar mixes.
Finally, the potential of each mortar to contribute to global warming was estimated using the LCA software SimaPro (v. 8.5.2.0). The goal was to compare the environmental impacts of NAC and CRAC mortars. Because the effect of carbonation on recycled aggregates is the primary focus of this study, the equivalent CO
2 emissions were deemed the most important indicator to address. The functional unit was defined as a 1 kg mortar that met the same requirements in terms of workability, strength, and durability for all the mortar mixes. The NAC and CRAC mixes showed the same 28-day compressive strength (see values in the Results Section) and workability (with compensation water foreseen for CRAC based on the water absorption of the CFRCA). Regarding durability, it is assumed, based on the compressive strength values, that performance is comparable for both mortars exposed in similar non- or low-aggressive conditions [
24]. Final values might be rescaled to address potential different lifespans for each mortar but in the present study these were considered the same. Therefore, no rescaling was applied to the studied mortar mixes. A modified cradle-to-gate approach was followed. It was assumed that the sand was extracted in a quarry and transported to the concrete mixing plant from there. An integrated recycling plant was considered within the mortar production plant. Therefore, the construction and demolition waste was assumed to be transported from the demolition site to the production plant. The LCA regarded mortar mixing as the final stage. After all, for comparison purposes between NAC and CRAC, the transportation to the construction site after production would be the same considering the same unit weight. The different products and locations that were considered are presented in
Figure 4. Values for emissions and energy use of the different materials and processes were found in the literature, and the CML-IA baseline impact method was chosen.
The database Ecoinvent 3, cut-off by classification, was used [
25]. The primary production of materials was allocated to the primary user of the material. Hence, the effect of recycling was not attributed to this primary user. As a result, the recycled material bears no environmental burden from the primary material but only the impact of the recycling process itself. Moreover, unit processes were used. These contain links to other unit processes from which the inventory flows could be calculated with SimaPro. The approach has the potential to be used for detailed interpretation and uncertainty analysis using Monte Carlo [
26]. The effect of allocating (part of) the demolition to the construction and demolition waste was examined, with assignation rates of 0%, 50%, and 100% to the FRCA. The distance over which the construction and demolition waste had to be transported to the recycling plant was also investigated as the environmental impact of the recycling procedure is highly reliant on the transportation of the construction and demolition waste [
27]. Therefore, three scenarios were considered: no transport, transport from Ghent (Belgium) to Schoten (Belgium), and transport from Ostend (Belgium) to Schoten (Belgium).
Details for the life cycle inventory and additional analyses for indicators other than global warming potential are provided as
Supplementary Material.
4. Discussion
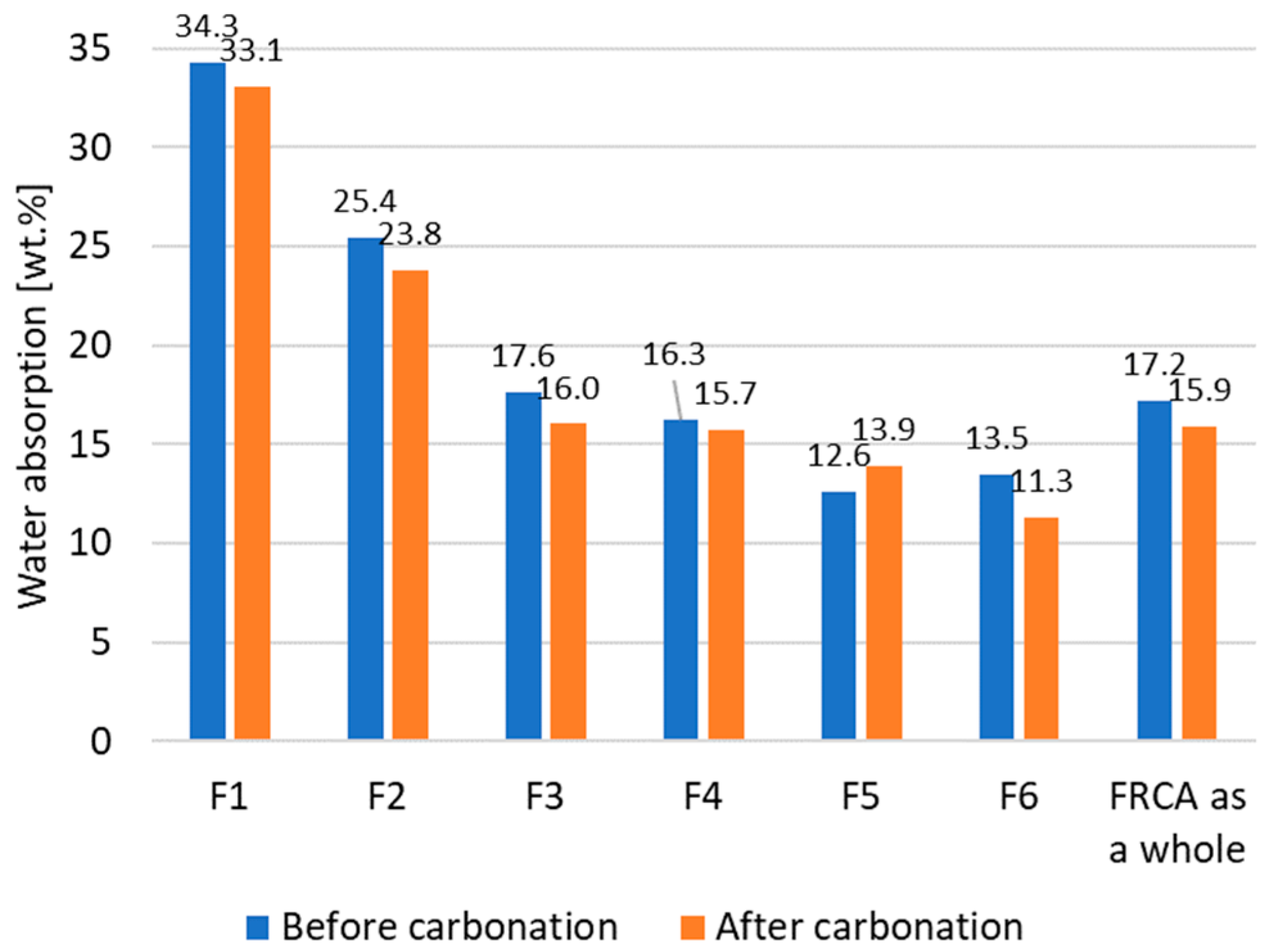
Water absorption capacity varies depending on the size range of the aggregate. Such variation can be attributed to differences in composition for each fraction. The high content of C-S-H in fraction F1 explains its high value for water absorption. Notably, the ratio between the amount of C-S-H and water absorption is much higher for F6 than for F3. This might be also due to the different quality of the cement paste in the different fractions. It seems logical that the cement paste in coarser fractions that survived the crushing process had a higher strength and lower porosity than the paste that was ground into the finest fractions and did not remain adhered to the natural aggregate particles. The contribution of the cement paste to the overall water absorption capacity might then be different depending on the quality of the corresponding paste, explaining the dissimilar correlation between the paste content and the water absorption for the different fractions.
Considering the water absorption, the treatment seems most effective for fractions F3 and F6 and least effective for fractions F1 and F4. Considering the loss on ignition, the treatment seems most effective for fractions F1, F4, and F6. An effective precipitation of calcium carbonate does not directly translate into a certain improvement degree of microstructure as this also depends on the initial pore size distribution. Hence, as the finest fraction includes the weakest cement paste with a large porosity and carbonatable phases, the large precipitation of calcium carbonate still seems insufficient to clog a large part of the pores, leading to a lesser relative reduction in water absorption. Therefore, the efficiency of the carbonation treatment depends on each particle size range.
The significant specific surface of FRCA makes it highly carbonatable when under atmospheric exposure. Carbonation treatments require the presence of carbonatable phases in the material. This seems more difficult to have in the field for FRCA compared to coarse RCA. Therefore, the strategy of carbonating FRCA to improve their properties will require (most of the time) the introduction of a carbonatable phase, as was the focus of this research in which we used wastewater from mortar preparation. Even then, the amount of precipitated carbonate might not be sufficient if the porosity of the FRCA is very high. It seems necessary to advance with additional studies at higher CO2 concentrations that might force higher carbonation degrees of additional phases, especially C-S-H, which even counteract the effect of other carbonatable products.