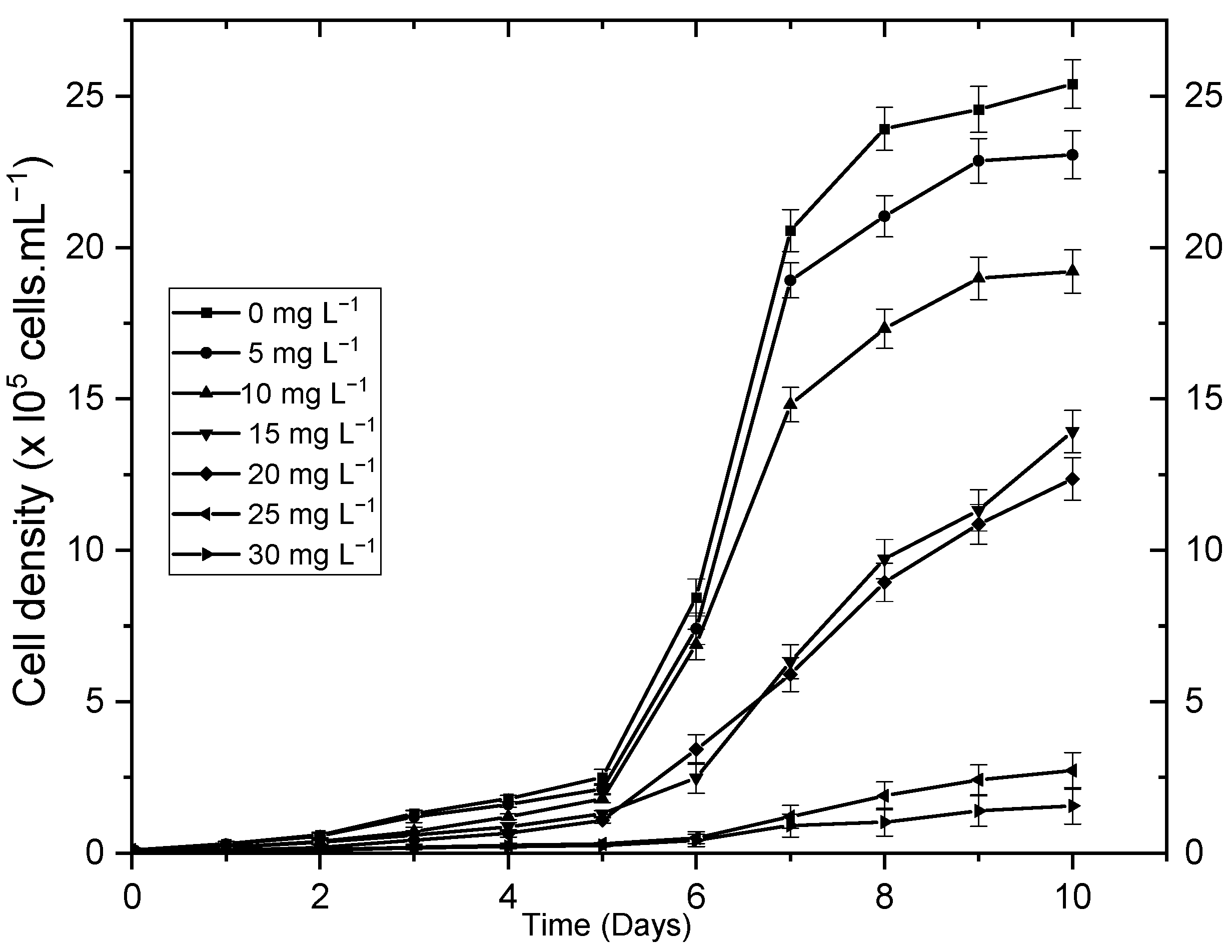
3.2.1. Single Factor Experiments
- a.
Effect of initial pH
Because the biosorbent’s protonation determines the functional ion exchange sites and surface charge, the solution’s starting pH is critical for hexavalent chromium uptake [
41] (Sibi, 2016). The pH also regulates the adsorbate’s charge. The microalgae contain hydrolyzable groups such as amines and aldehydes, as was discussed in the FTIR analysis. These two groups have more binding sites when acidified biomass and are easily protonated [
42]. As a result, the pH of the solution was varied from 0.5 to 5.0 to study the influence of the initial pH on Cr(VI) adsorption while other parameters were kept constant. Different initial pHs were used to evaluate the Cr(VI) uptake effectiveness and capacity during the pH change experiment.
Figure 2 illustrates the uptake capacity and efficiency plot with starting solution pH. At pH 1.0, the highest uptake capacity was 277.48 mg g
−1. The pH increased consistently from 1.0 to 5.0, attaining 90.29 mg g
−1 at pH 5.0.
The highest Cr(VI) fixed on the protonated microalgae surface at acidic pH is favored by anionic adsorption [
53]. At pH-1.0, the highest biosorption rate was 95.27%. At low pH values, amide, carboxyl, halide, and hydroxyl groups on biomass surfaces become protonated and positively charged [
54]. Simultaneously, anionic Cr(VI) species, for example, dichromate (Cr2O
72−), chromate (CrO
42−), and tetraoxohydrochromate (HCrO
4−) ions formed under lower pH in the acidic solution [
55]. The positively charged cell surface electrostatically adsorbed the anionic Cr(VI) species at lower pH ranges, causing significant physisorption of Cr(VI) on the microalgae [
54]. As the proton concentration decreased as the pH of the solution increased, the fixation sites of the microalgae developed to be negatively charged. Due to electrostatic repulsion, negatively charged cells, compared with anionic chromate ions, cause lower uptake capacity at higher pH ranges [
53]. The outcomes of the investigation into the influence of pH levels on the microalgae’s ability to remove Cr(VI) from its environment indicate an optimal removal at notably acidic pH conditions.
This suggests that the algae eliminate this element through adsorption rather than absorption at this pH, which is unfavorable for the algae’s survival. Consequently, as a continuation of this work, we conclude that the elimination of Cr(VI) of this microalgae is achieved through adsorption. Given this optimum, an implication arises for industrial wastewater treatment processes that typically exhibit a very low pH range around one, particularly in effluents from tanneries, as reported by Muthukkauppan and Parthiban [
56], and around three, as mentioned by Lissaneddine et al. [
57]. Although this is slightly more alkaline than the identified optimum of our study, it remains within a range conducive to high biosorption rates. This suggests that while industrial effluents may benefit from some degree of acidification, excessive pH modification may not be necessary. In addition, the environmental costs of such modifications, including potential ecological disruptions and compliance with discharge regulations, must be weighed against the incremental gains in decontamination efficiency. Hence, in the broader context of practical application, it becomes crucial to balance the theoretical maximal efficiency obtained under controlled experimental conditions with the pragmatic, economic, and environmental realities of industrial operations.
- b.
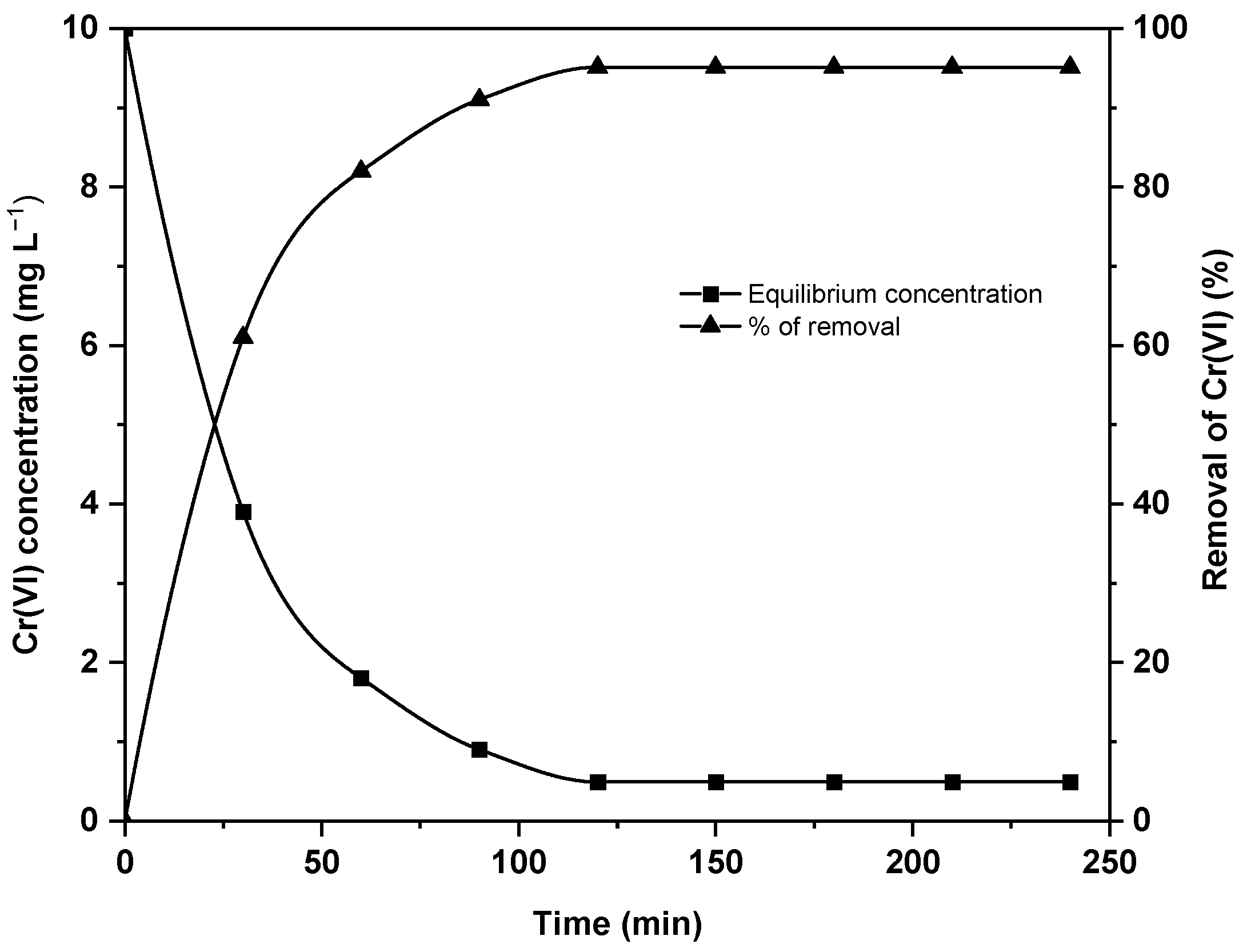
Effect of contact time
Various contact times, ranging from 30 to 240 min, were conducted for the adsorption of Cr(VI) employing the
C. subminuscula biomass while maintaining other constant parameters. The graph in
Figure 3 illustrates the relationship between the contact time, the concentration of Cr(VI), and the percentage of its removal.
The bioremediation rate was extremely rapid, reaching a maximum of 90 min, with an adsorption percentage of just around 91%. Extending the exposure time to 240 min added just 4% additional Cr(VI). The rapid rate contributed to 91% of Cr(VI) uptake in just 90 min. After 90 min, the adsorption rate increased significantly, accounting for 95.1% removal up to 120 min. Hence, the equilibrium point of the adsorption experiment was thought to be at 120 min of biosorption. The biomass’s finer particle size may cause a quick equilibrium time [
58]. Therefore, the remaining adsorption tests modifying all parameters were performed for 120 min.
The first, quick phase could involve ions adhering physically or exchanging at the cell’s surface, which precedes a more prolonged phase where other processes, such as compound formation, micro-precipitation, or the saturation of receptor sites, may occur. This two-stage process illustrates the diverse mechanisms at play when removing substances, showcasing the complexity of the interaction between the material and the cell [
59]. Also notable is that various factors influence the adsorption rate, including the sorbate’s and biosorbent’s structural characteristics (e.g., composition of proteins and carbohydrates, surface charge density, topography, and surface area). Additionally, the quantity of biosorbent, the initial concentration of metal ions, and the presence of competing ions can impact the adsorption rate on active sites [
59].
- c.
Effect of biosorbent dosage
The biosorbent quantity varied from 2.5, 5, 10, 15, and 20 mg L
−1.
Figure 4 shows an adsorption capacity and efficiency graph plotted against algal biomass. With increased algae biomass from 2.5 to 10 mg L
−1, the removal efficiency of Cr(VI) increased rapidly from 49 to 95.29% and increased considerably to 20 mg L
−1, somewhat increasing to 96.3%. According to Bermudez et al. [
60], the increased chromium found in smaller doses of biomass is beneficial for industrial applications. Adding additional biosorbent increased the biosorption rate as algal biomass increased, creating a larger surface area.
In contrast, the flat increase in the biosorption rate observed with biosorbent doses of more than 10 mg L
−1 was due to incomplete aggregation of the biomass particles because of a reduction in the effective surface area [
59]. Maleki et al. [
32] reported that a generally decreased biphasic interface causes biosorbent overlap or aggregation. Therefore, a biomass alga of 10 mg L
−1 was selected as the optimal biosorbent quantity for Cr(VI) uptake. The adsorption capacity gradually reduced from 588 to 144.45 mg g
−1 with increased algae biomass from 2.5 to 20 mg L
−1, respectively. The uptake rate decreased as the dosage of the algae biomass was increased (
Figure 4), as determined by the quantity of hexavalent chromium uptake per gram of algae biomass (Equation (1)).
- d.
Effect of initial Cr(VI) concentration
The starting quantity of Cr(VI) as an adsorbate is significant in controlling the uptake rate because biosorption is a physical mass diffusion process that occurs at the interface of two phases [
58]. The curve in
Figure 5 shows an initial rapid adsorption that gradually slows down, suggesting adsorption is controlled by a diffusion process, such as physical mass diffusion. Although the initial increase in Cr(VI) concentration (5–10 mg L
−1) had no significant effect, the percentage uptake decreased from 98.3% to 95.3%. However, the starting Cr(VI) concentration rose from 10 to 30 mg L
−1, progressively reducing from 95.3% to 32.26%. Therefore, the optimal initial Cr(VI) concentration was 10 mg/L. When the initial Cr(VI) concentration was raised from 5 to 10 mg L
−1, the adsorption capacity doubled from 126.9 to 277.57 mg g
−1. When the concentration was elevated further up to 30 mg L
−1, practically nothing was modified. This could be due to a lack of binding sites in the microalgae to allow the growing quantity of Cr(VI) to diffuse, or their simultaneous contacts either restricted propagation of the two phases interface or their competitive attraction between them [
61]. Bermdez et al. [
59] suggest that the algal surface possesses a finite number of adsorptive sites. Upon saturation of these sites, further accumulation of Cr becomes unattainable.
- e.
Effect of temperature
Physical adsorption starts thermodynamically at the interface of two phases based on the adsorption process [
60]. It is determined by randomizing adsorbed molecules on the biosorbent’s surface. Consequently, the temperature change is important in the randomness determination at the two-phase system interface. To assess the effect of the temperature variation for binding Cr(VI) to diatom biomass, the temperature was changed from 15 to 35 °C.
Figure 6 depicts a graph that plots the relationship between temperature and adsorption efficacy and capacity, demonstrating an upward trend in biosorption performance as the temperature rises. As the temperature increased from 15 to 35 °C, the biosorption rate continuously increased from 75 to 96.9%. This might be because of the endothermic nature of the biosorption process [
59]. The percentage of biosorption was enhanced with rising temperatures, potentially attributable to a surge in active groups on the biosorbent’s surface or a reduction in the boundary layer’s density around the microalgae, thereby making Cr(VI) more accessible to the active sites for binding, or possibly a combination of these factors [
60].
3.2.2. Optimization of Cr(VI) Removal Conditions for C. subminuscula
Based on the batch experiments, three important conditions (biosorbent dosage, treatment time, and pH) were evaluated for more optimization. The RSM designed 15 treatments for the optimization experiments (
Table 3). We notice that in treatment 8, the maximum Cr(VI) uptake efficiency was found with a Cr(VI) uptake efficiency of 95.32%. Using Design Expert 13.0 software, the experimental data were analyzed using multiple regression to create a quadratic multiple regression equation, shown in Equation (8).
where Y is f Cr(VI) removal efficiency (%), A is the biosorbent dosage concentration (mg L
−1), B is the treatment time (min), and C is the pH.
The analysis conducted to evaluate the validity of the quadratic multiple regression equation is detailed in
Table 4. Based on the analysis, the equation proved extremely significant, as evidenced by an F-value of 99.13 and a
p-value of less than 0.0001. It is essential for the model to present statistical significance (
p-value less than 0.05) to fit the experimental design adequately. Conversely, a nonsignificant
p-value (greater than 0.05) should correspond to the lack of fit factor [
62].
In line with these criteria, the equation’s “lack of fit” was deemed nonsignificant given its F-value of 1947.54 and
p-value greater than 0.05, confirming a strong alignment between the regression equation and observed experimental results. A precision ratio of 30.577, significantly exceeding the threshold of 4, ensured a reliable signal, affirming the equation’s capability to navigate the design space efficiently. The R
2 determination coefficient reaffirmed the significant relevance of the independent variables concerning the dependent variable, with a narrow gap observed between the predicted R
2 of 0.9112 and the adjusted R
2 of 0.9844, well under the 0.2 margin. In terms of individual factors, the regression analysis highlighted the importance of the biosorbent dosage (A), treatment time (B), and pH level (C) as significant contributors to the Cr(VI) removal efficiency, each with a
p-value less than 0.05. However, the interactions amongst biosorbent dosage (A) and treatment time (B), dosage (A) and pH (C), as well as treatment time (B) and pH (C), did not demonstrate significance, as evidenced by
p-values exceeding 0.05. On the other hand, the quadratic terms A
2, B
2, and C
2 each showed significant influence with
p-values less than 0.01, as noted in
Table 4.
The effects and appropriate concentration of the three parameters for maximum Cr(VI) elimination efficiency were examined using a 3D response surface (
Figure 7). The plots of the response surface illustrated the impact of each variable on the efficiency of chromium (VI) removal. By optimizing two factors and maintaining a third at its median z-axis value, we can observe their effects. In
Figure 7i, you can see how the interplay between treatment duration (B) and pH level (C) unfolds when the biosorbent amount (A) is held constant at 10.91 mg L
−1. The combination of pH (C) and treatment duration had a significant effect on Cr(VI) biosorption capacity (B). Among increasing pH and time, the Cr(VI) uptake capacity primarily increased and then reduced due to excessively low or high pH levels that may reduce microalgae’s ability to biosorbent metals [
63].
When the treatment duration (B) was maintained at a mean of 128.47 min, the influence of the pH (C) and biosorbent quantity (A) on the efficiency of chromium (VI) removal was minor, as illustrated in
Figure 7ii. An increase in pH and biosorbent levels either raised or lowered the efficiency of chromium (VI) removal, paralleling findings by Moreira et al. [
63].
Figure 7iii further shows that holding the pH (C) at a median value of 1.09, the correlations between treatment time (B), biosorbent dosage (A), and chromium (VI) removal were marginal. Using Equation (8), optimal conditions for chromium absorption by
C. subminuscula were determined. The peak removal efficiency for chromium (VI) was estimated at 95.30%, achieved at an ideal pH of 1.09, a biosorbent quantity of 10.91 mg/L, and a treatment span of 129.47 min. Verification of the optimization was conducted through a set of triplicate experiments at these optimal parameters. The findings showed negligible differences between the experimental and predicted values (
p-value > 0.05), as reported in
Table 5, affirming the model’s reliability and possible replication.
Biosorption experiments in batch culture were performed to maximize physicochemical factors (pH, temperature, agitation time, adsorbent dose, initial chromium concentration, and contact time). To optimize the chromium binding on the algae surface and to situate the performance of our tested species,
Table 6 compares the uptake efficiency of Cr(VI) in the study with other studies in the literature. The Cr(VI) uptake efficiency by
C. subminuscula was consistent with that by
Parachlorella kessleri (96.1%) [
64] and
Scenedesmus sp. (92.89%) [
65], higher than that by
Planothidium lanceolatum (about 87%) [
26],
Chlamydomonas sp. (91.31%) [
66],
Scenedesmus quadricauda (47.6%) [
67], and
Chlorella vulgaris (43%) [
41], and lower than that by
Chlorella sorokiniana (99.67%) [
68], and
Scenedesmus quadricauda (98.1%) [
69]. It is clear from the table above that this alga has high metal binding capacities.