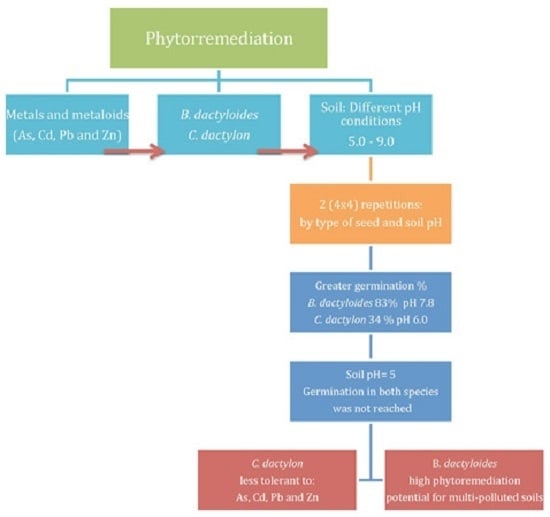
3.3. Seeds Germination
The standard germination test demonstrated that B. dactyloides germinated at 82.5% and C. dactylon at 35%.
The germination time with the contaminated soil for both seeds was from 3 to 21 days.
Figure 1 and
Figure 2 show the behavior over 21 days of seed germination for
B. dactyloides and
C. dactylon by the type of soil.
B. dactyloides presented a greater germination percentage from the fifth day in Soil Samples A, B, C, and F.
The grass
B. dactyloides presented a germination percentage from 68% to 83% in soils with pH between 7 and 8 (B, C, and F). In the soil with a pH of 6 (Soil Sample A), germination declined to 50%. The soil with an increased pH of 9 (Soil Sample E) caused a germination reduction to 13%, and the soil with acid conditions (pH of 5, Soil Sample D) did not germinate (
Figure 1). The germination percentage of
C. dactylon was from 28% to 34% in soils with pH of 6 and 7 (Soil Samples A and C). In the soil with pH > 7 (Soil Samples B and F), a germination reduction to 17% and 18% was presented. In the soil with pH values of 5.0 (Soil Sample D) and 9.0 (Soil Sample E), germination was not reached (
Figure 2). Despite the differences between germination percentages, both species showed a high germination compared with the standard germination test.
According to Bartlett’s test, the data exhibit variance homogeneity (p > 0.296). PROC GLM model demonstrated that B. dactyloides and C. dactylon germination were significantly different (R2 = 0.9315, p < 0.05).
In the LSD test (
p < 0.05) the least significant difference was 3.9473. For
B. dactyloides demonstrated that germination in Soil Samples B and C was not significantly different (pH of 7.8 and 7.0, respectively), nor was that of Soil Samples C and F (pH of 7.0 and pH of 8.0, respectively) (
Table 4), so Soil Sample C soil had similar germination development than the uncontaminated soil. Soil Sample A was not similar to any other; and Soil Samples B, C, and F had a higher germination mean than A.
For
C. dactylon, germination in Soil Samples A and C (pH 6 and 7, respectively) was not significantly different (soils with the higher germination for this species). Additionally, Soil Samples B and F soils (pH >7) were not significantly different, so Soil Sample B had similar germination development than that of the uncontaminated soil (
Table 4).
Table 5 and
Table 6 show the germination percentage, mean of germinated seeds, and variation coefficient (V
c) of each grass species and soil type.
As far as the results of V
c, the greatest homogeneity on grass germination with
B. dactyloides was in Soil Samples B and C with a V
c of 13.8% and 10.5%, respectively. Meanwhile, with
C. dactylon, it was Soil Sample C with a V
c of 11.6% (
Table 4 and
Table 5).
Grass germination reached a peak and stabilized, and this was determinate with a regression of orthogonal contrast, so the model proved being square (p < 0.0001).
3.4. Discussion
B. dactyloides showed a better germination percentage than
C. dactylon. Both species had higher germination percentages with soil with an approximately neutral pH, similar percentages than the standard test, and lower variation. In both grasses, there was no germination in Soil Sample D (pH of 5). Although Soil Sample D did not present a higher concentration of total metals than the other soils, it had a lower pH and a higher bioavailability of Cd and Zn; in fact, Cd content was higher than the Rc (
Table 3). This metal solubility could cause a lack of development in both species, because soil pH has a strong influence on the bioavailability of metals and metalloids. Acidic soil pH makes metals more soluble; therefore, their bioavailability is greater for plants [
22].
Cd has been known to interfere with the water uptake of plants, which is why germination does not occur [
10]. Cd has also been reported to cause chromosomal aberration, with cytogenetic analysis of
Pisum sativum root from seeds germinated on different concentrations of Cd, which demonstrated an increased frequency of abnormalities. The total number of aberrations increased with Cd concentration [
8].
On the other hand, Zn is one of the essential micronutrients in plants; it is necessary for normal plant growth and development and is required in several metabolic processes. Nevertheless, high concentrations of Zn are toxic and hamper plant growth [
3]. The excess of Zn affects the uptake of other nutrients, inhibiting seed germination, plant growth, and root development [
10].
Rapeseed exposed to 1.12 mM Zn caused plant growth inhibition and chlorosis in leaves. The contents of Fe, Cu, Mg, and Mn decreased in roots and leaves. Additionally, it was demonstrated that Mg and Cu content decreased with increased Zn concentrations in ryegrass. These results indicated that an excess of Zn might suppress the uptake of these elements due to the competition among metals [
23]. Moreover, a study with sunflower determined that seeds germinated at high Zn concentrations, while germination decreased to below 50% after exposure to Cd, Cu, and Pb [
8].
Marichali et al. (2014) achieved germination of
C. sativum seedlings irrigated with high Zn concentrations at a pH of 6.8, but root length was reduced. They showed that, for
C. sativum, Zn has low toxic effects on germination, and roots are more sensitive than seeds to metal stress [
3]. This result does not agree with the present research possibly because the pH and the germination conditions were different.
The bioavailability of Cd, Pb, and Zn decreases while soil pH increases due to its precipitation as insoluble hydroxides, carbonates, and organic complexes; opposite As bioavailability increases at a basic pH [
24]. This can explain why
B. dactyloides and
C. dactylon reduced germination in Soil Sample E (
Table 4) with a similar metal pollution level compared with the other soils (
Table 2), but a basic pH induced more As bioavailability (
Table 3). The most common genotoxic effects reported for As exposure involve mitotic spindle disturbance. In
Hordeum vulgare seeds, the frequency of chromosomal abnormalities is proportional to the concentration of As [
8].
Metal tolerance is an important factor for the use of plants and seeds for phytoremediation, so the capacity of seeds to germinate in the presence of metals and metalloids could be a primary concern [
8]. This research supported the hypothesis that metals generally inhibit germination and seedling growth [
25], but the metal distribution in seeds and affectation degree differs depending on the metal involved, the plant species, and the seed anatomy. Additionally, the soil pH interferes in the bioavailability of the metals and, with it, in the germination response [
24]. Thus, seeds of metal-tolerant plants may have a higher threshold for toxicity than non-tolerant ones [
8]. After permeation through the seed coat, germination relies on the seed reserves for metabolite supply for respiration, Metals can cause stress and disrupt the process, and interfere with the enzymes involved in the germination [
10].