Dynamic Habitat Indices and Climatic Characteristics Explain Species Richness Patterns on the Mongolian Plateau
Abstract
1. Introduction
2. Materials and Methods
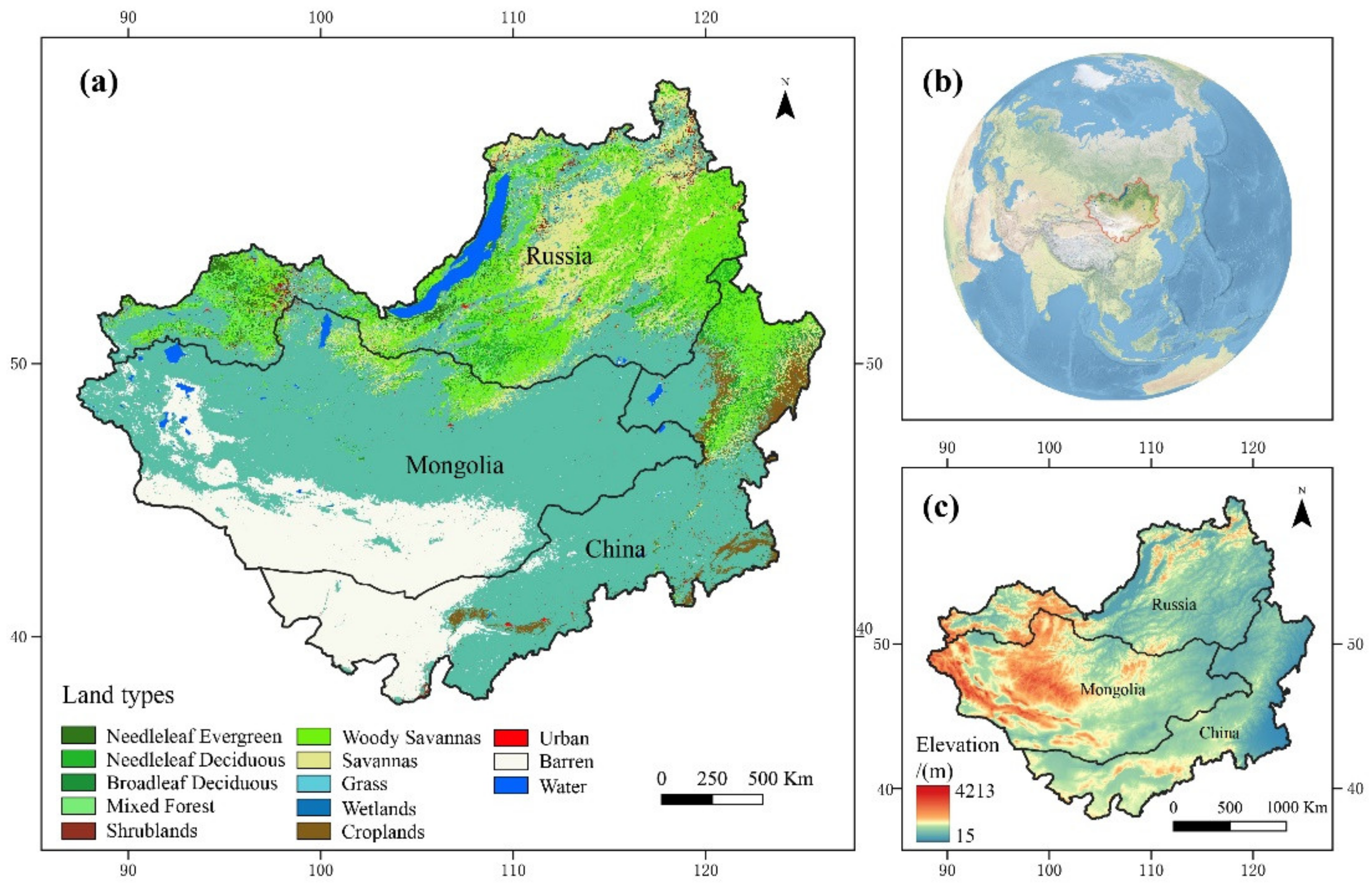
2.1. Study Area
2.2. Data
2.2.1. GLASS MODIS FAPAR
2.2.2. Species Richness
2.2.3. Climate Variables
2.3. Methods
2.3.1. Calculation of Dynamic Habitat Indices (DHIs)
2.3.2. Statistical Analysis
3. Results
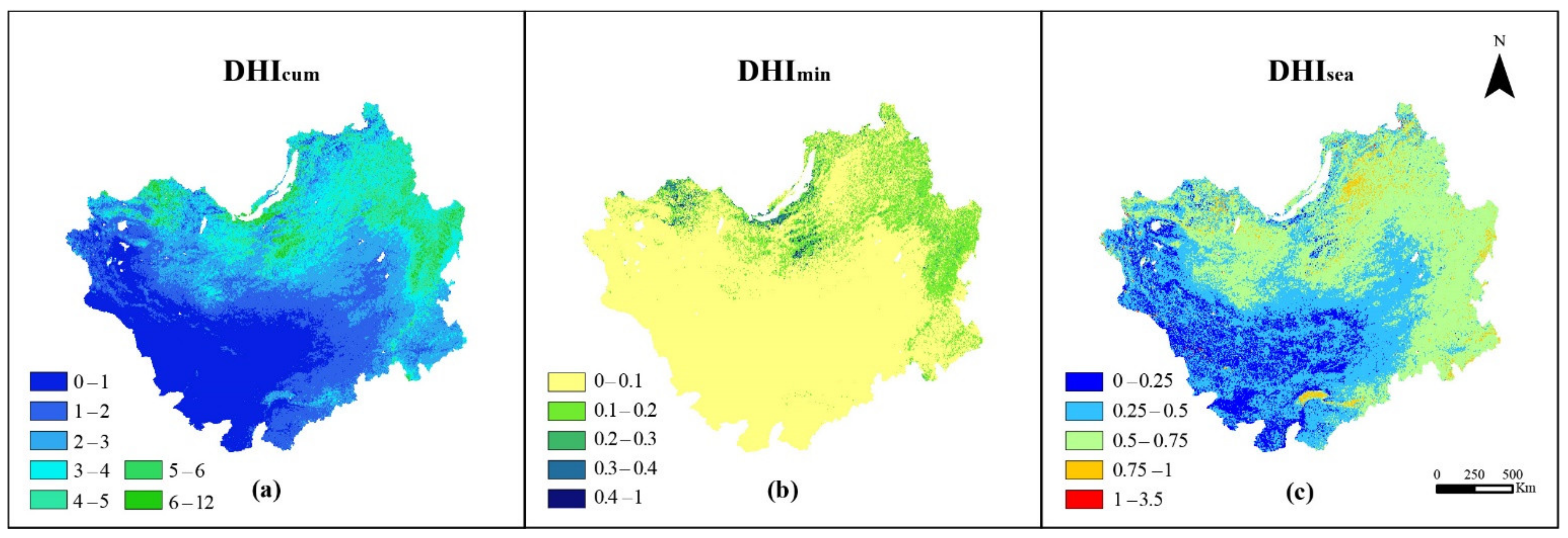
3.1. Distribution Patterns of DHIs
3.2. Geographic Distribution of Species Richness
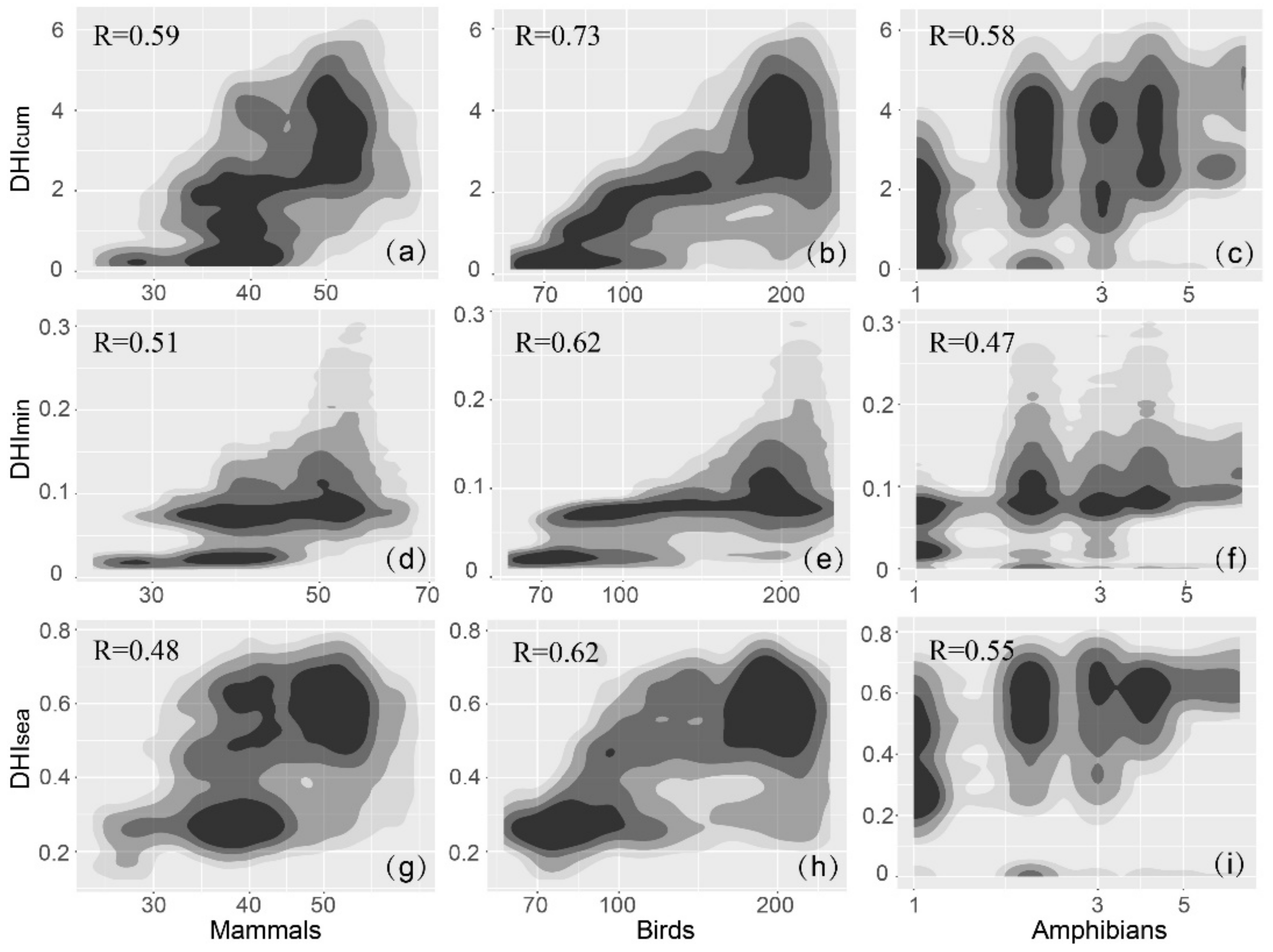
3.3. DHIs and Species Richness
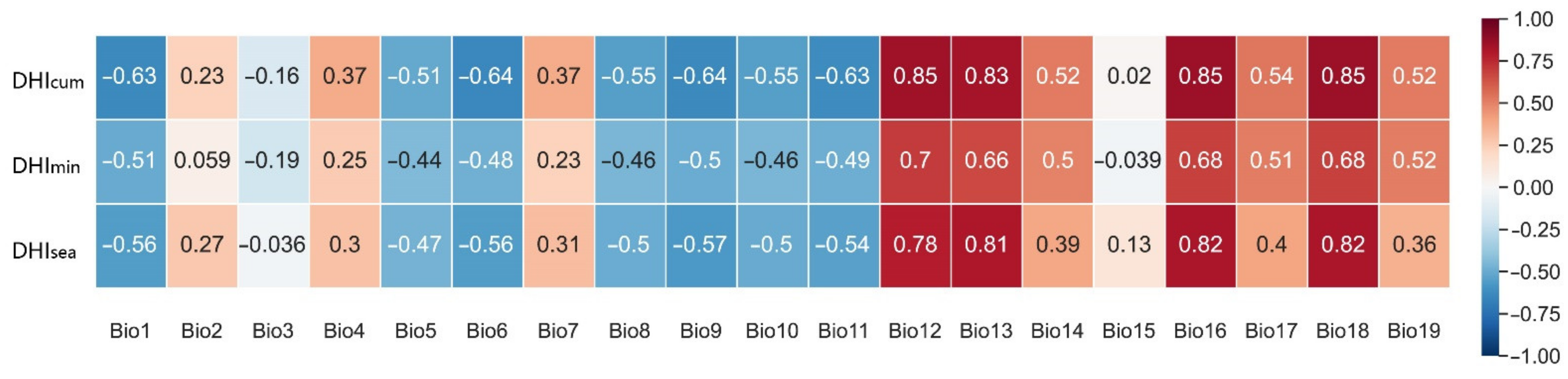
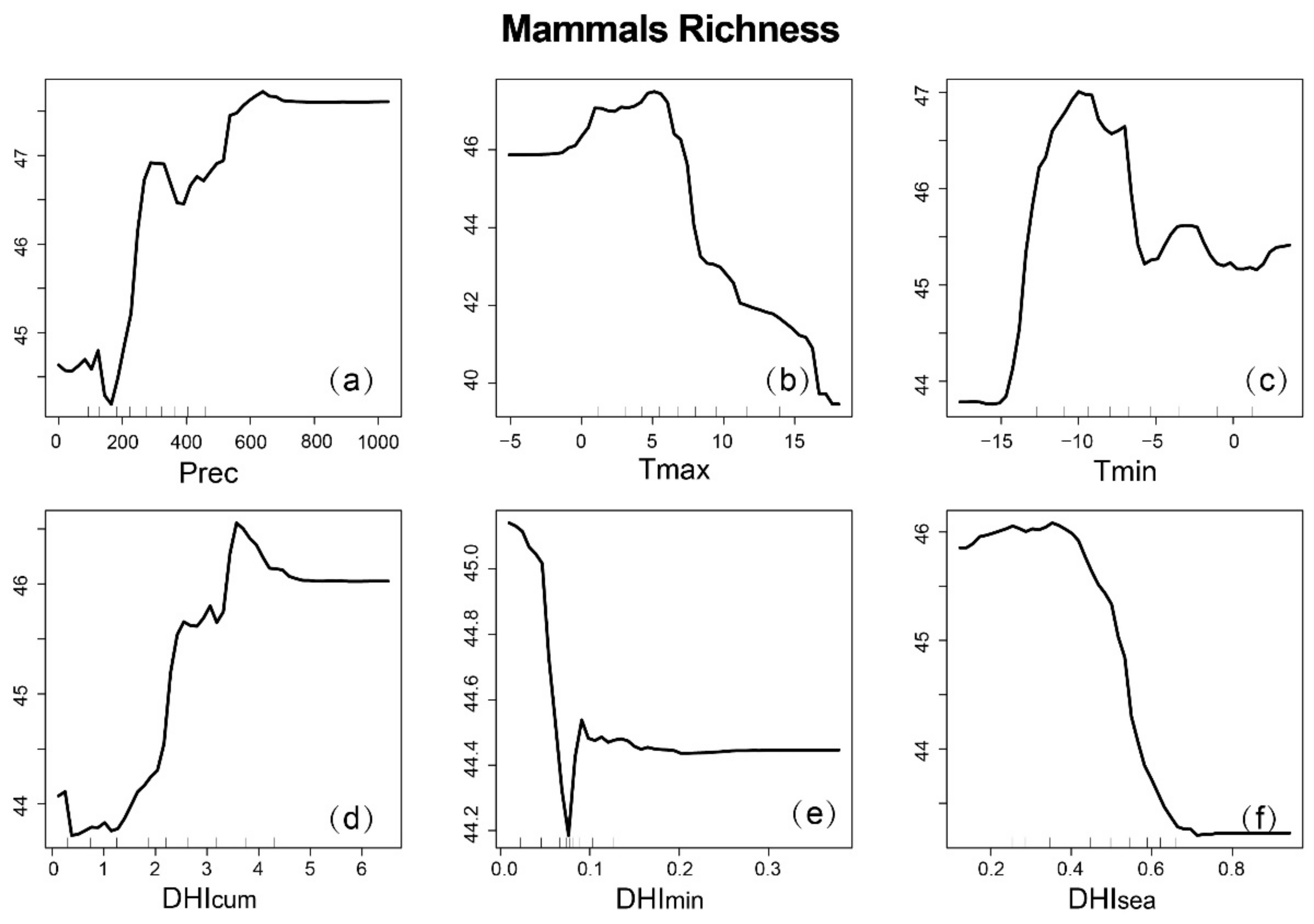
3.4. Interpretative Analysis of Species Richness Using DHIs
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hooper, D.U.; Chapin Iii, F.S.; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of Biodiversity on Ecosystem Functioning: A Consensus of Current Knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Loreau, M.; Naeem, S.; Inchausti, P.; Bengtsson, J.; Grime, J.P.; Hector, A.; Hooper, D.U.; Huston, M.A.; Raffaelli, D.; Schmid, B.; et al. Biodiversity and Ecosystem Functioning: Current Knowledge and Future Challenges. Science 2001, 294, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Chapin Iii, F.S.; Zavaleta, E.S.; Eviner, V.T.; Naylor, R.L.; Vitousek, P.M.; Reynolds, H.L.; Hooper, D.U.; Lavorel, S.; Sala, O.E.; Hobbie, S.E.; et al. Consequences of changing biodiversity. Nature 2000, 405, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Young, B.E.; Gill, M.J.; Hamilton, H.; Vergara, S.G. Data on Indicators used in Southeast Asian nations’ 4th and 5th National Reports to the Convention on Biological Diversity. Data Brief 2020, 31, 105705. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.A.; Barrio Froján, C.; Gunn, V.; Johnson, D.E. A role for UNEP’s Regional Seas Programme under the post-2020 global biodiversity framework. Mar. Policy 2022, 136, 104930. [Google Scholar] [CrossRef]
- Sillmann, J.; Thorarinsdottir, T.; Keenlyside, N.; Schaller, N.; Alexander, L.V.; Hegerl, G.; Seneviratne, S.I.; Vautard, R.; Zhang, X.; Zwiers, F.W. Understanding, modeling and predicting weather and climate extremes: Challenges and opportunities. Weather Clim. Extrem. 2017, 18, 65–74. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Pimm, S.L.; Jenkins, C.N.; Abell, R.; Brooks, T.M.; Gittleman, J.L.; Joppa, L.N.; Raven, P.H.; Roberts, C.M.; Sexton, J.O. The biodiversity of species and their rates of extinction, distribution, and protection. Science 2014, 344, 1246752. [Google Scholar] [CrossRef]
- Sala, O.E.; Stuart Chapin, F.; Iii, N.; Armesto, J.J.; Berlow, E.; Bloomfield, J.; Dirzo, R.; Huber-Sanwald, E.; Huenneke, L.F.; Jackson, R.B.; et al. Global Biodiversity Scenarios for the Year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef]
- Zhang, S.; Zhou, Y.; Yu, R.; Xu, X.; Xu, M.; Li, G.; Wang, W.; Yang, Y. China’s biodiversity conservation in the process of implementing the sustainable development goals (SDGs). J. Clean. Prod. 2022, 338, 130595. [Google Scholar] [CrossRef]
- IUCN. The IUCN Red List of Threatened Species. Available online: https://www.iucnredlist.org/en (accessed on 1 August 2022).
- D’Amen, M.; Bombi, P. Global warming and biodiversity: Evidence of climate-linked amphibian declines in Italy. Biol. Conserv. 2009, 142, 3060–3067. [Google Scholar] [CrossRef]
- Erdelen, W.R. Shaping the Fate of Life on Earth: The Post-2020 Global Biodiversity Framework. Glob. Policy 2020, 11, 347–359. [Google Scholar] [CrossRef]
- Nagendra, H. Using remote sensing to assess biodiversity. Int. J. Remote Sens. 2001, 22, 2377–2400. [Google Scholar] [CrossRef]
- Turner, W.; Spector, S.; Gardiner, N.; Fladeland, M.; Sterling, E.; Steininger, M. Remote sensing for biodiversity science and conservation. Trends Ecol. Evol. 2003, 18, 306–314. [Google Scholar] [CrossRef]
- Zarnetske, P.L.; Read, Q.D.; Record, S.; Gaddis, K.D.; Pau, S.; Hobi, M.L.; Malone, S.L.; Costanza, J.; Dahlin, K.M.; Latimer, A.M.; et al. Towards connecting biodiversity and geodiversity across scales with satellite remote sensing. Glob. Ecol. Biogeogr. 2019, 28, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Dietz, H.; Steinlein, T. Determination of plant species cover by means of image analysis. J. Veg. Sci. 1996, 7, 131–136. [Google Scholar] [CrossRef]
- Ehrlich, D.; Estes, J.; Scepan, J. Improving crop type determination using satellite imagery: A study for the Regione del Veneto, Italy. Geocarto Int. 1990, 5, 35–47. [Google Scholar] [CrossRef]
- Duro, D.C.; Coops, N.C.; Wulder, M.A.; Han, T. Development of a large area biodiversity monitoring system driven by remote sensing. Prog. Phys. Geogr. 2007, 31, 235–260. [Google Scholar] [CrossRef]
- Pettorelli, N.; Laurance, W.F.; O’Brien, T.G.; Wegmann, M.; Nagendra, H.; Turner, W. Satellite remote sensing for applied ecologists: Opportunities and challenges. J. Appl. Ecol. 2014, 51, 839–848. [Google Scholar] [CrossRef]
- Olff, H.; Ritchie, M.E.; Prins, H.H.T. Global environmental controls of diversity in large herbivores. Nature 2002, 415, 901–904. [Google Scholar] [CrossRef]
- Berry, S.; Mackey, B.; Brown, T. Potential applications of remotely sensed vegetation greenness to habitat analysis and the conservation of dispersive fauna. Pac. Conserv. Biol. 2007, 13, 120–127. [Google Scholar] [CrossRef]
- Coops, N.C.; Wulder, M.A.; Duro, D.C.; Han, T.; Berry, S. The development of a Canadian dynamic habitat index using multi-temporal satellite estimates of canopy light absorbance. Ecol. Indic. 2008, 8, 754–766. [Google Scholar] [CrossRef]
- Tucker, C.J. Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens. Environ. 1979, 8, 127–150. [Google Scholar] [CrossRef]
- Bawa, K.; Rose, J.; Ganeshaiah, K.N.; Barve, N.; Kiran, M.C.; Umashaanker, R. Assessing Biodiversity from Space: An Example from the Western Ghats, India. Conserv. Ecol. 2002, 6, 1–5. [Google Scholar] [CrossRef]
- Buckley, L.B.; Hurlbert, A.H.; Jetz, W. Broad-scale ecological implications of ectothermy and endothermy in changing environments. Glob. Ecol. Biogeogr. 2012, 21, 873–885. [Google Scholar] [CrossRef]
- Seto, K.C.; Fleishman, E.; Fay, J.P.; Betrus, C.J. Linking spatial patterns of birds and butterfly species richness with Landsat TM derived NDVI. Int. J. Remote Sens. 2004, 25, 4309–4324. [Google Scholar] [CrossRef]
- McFarland, T.M.; Van Riper, C.; Johnson, G.E. Evaluation of NDVI to assess avian abundance and richness along the upper San Pedro River. J. Arid Environ. 2012, 77, 45–53. [Google Scholar] [CrossRef]
- Nightingale, J.M.; Coops, N.C.; Waring, R.H.; Hargrove, W.W. Comparison of MODIS gross primary production estimates for forests across the U.S.A. with those generated by a simple process model, 3-PGS. Remote Sens. Environ. 2007, 109, 500–509. [Google Scholar] [CrossRef]
- Asner, G.P.; Braswell, B.H.; Schimel, D.S.; Wessman, C.A. Ecological Research Needs from Multiangle Remote Sensing Data. Remote Sens. Environ. 1998, 63, 155–165. [Google Scholar] [CrossRef]
- Coops, N.C.; Wulder, M.A.; Iwanicka, D. Demonstration of a satellite-based index to monitor habitat at continental-scales. Ecol. Indic. 2009, 9, 948–958. [Google Scholar] [CrossRef]
- Wright, D. Species-Energy Theory: An Extension of Species-Area Theory. Oikos 1983, 41, 496. [Google Scholar] [CrossRef]
- Currie, D.J.; Mittelbach, G.G.; Cornell, H.V.; Field, R.; Guégan, J.F.; Hawkins, B.A.; Kaufman, D.M.; Kerr, J.T.; Oberdorff, T.; O’Brien, E.; et al. Predictions and tests of climate-based hypotheses of broad-scale variation in taxonomic richness. Ecol. Lett. 2004, 7, 1121–1134. [Google Scholar] [CrossRef]
- Klopfer, P.H.; MacArthur, R.H. Niche Size and Faunal Diversity. Am. Nat. 1960, 94, 293–300. [Google Scholar] [CrossRef]
- Mackey, B.; Bryan, J.; Randall, L. Australia’s Dynamic Habitat Template 2003. 2004. Available online: http://hdl.handle.net/1885/41959 (accessed on 13 February 2023).
- Coops, N.C.; Waring, R.H.; Wulder, M.A.; Pidgeon, A.M.; Radeloff, V.C. Birds diversity: A predictable function of satellite-derived estimates of seasonal variation in canopy light absorbance across the United States. J. Biogeogr. 2009, 36, 905–918. [Google Scholar] [CrossRef]
- Nelson, T.A.; Coops, N.C.; Wulder, M.A.; Perez, L.; Fitterer, J.; Powers, R.; Fontana, F. Predicting Climate Change Impacts to the Canadian Boreal Forest. Diversity 2014, 6, 133–157. [Google Scholar] [CrossRef]
- Hobi, M.L.; Dubinin, M.; Graham, C.H.; Coops, N.C.; Clayton, M.K.; Pidgeon, A.M.; Radeloff, V.C. A comparison of Dynamic Habitat Indices derived from different MODIS products as predictors of avian species richness. Remote Sens. Environ. 2017, 195, 142–152. [Google Scholar] [CrossRef]
- Radeloff, V.C.; Dubinin, M.; Coops, N.C.; Allen, A.M.; Brooks, T.M.; Clayton, M.K.; Costa, G.C.; Graham, C.H.; Helmers, D.P.; Ives, A.R.; et al. The Dynamic Habitat Indices (DHIs) from MODIS and global biodiversity. Remote Sens. Environ. 2019, 222, 204–214. [Google Scholar] [CrossRef]
- Zhang, C.; Cai, D.; Guo, S.; Guan, Y.; Fraedrich, K.; Nie, Y.; Liu, X.; Bian, X. Spatial-Temporal Dynamics of China’s Terrestrial Biodiversity: A Dynamic Habitat Index Diagnostic. Remote Sens. 2016, 8, 227. [Google Scholar] [CrossRef]
- Zhu, L.; Guo, Y. Remotely Sensed Winter Habitat Indices Improve the Explanation of Broad-Scale Patterns of Mammal and Birds Species Richness in China. Remote Sens. 2022, 14, 794. [Google Scholar] [CrossRef]
- Suttidate, N.; Hobi, M.L.; Pidgeon, A.M.; Round, P.D.; Coops, N.C.; Helmers, D.P.; Keuler, N.S.; Dubinin, M.; Bateman, B.L.; Radeloff, V.C. Tropical birds species richness is strongly associated with patterns of primary productivity captured by the Dynamic Habitat Indices. Remote Sens. Environ. 2019, 232, 111306. [Google Scholar] [CrossRef]
- Mahanand, S.; Behera, M.D.; Roy, P.S.; Kumar, P.; Barik, S.K.; Srivastava, P.K. Satellite Based Fraction of Absorbed Photosynthetically Active Radiation Is Congruent with Plant Diversity in India. Remote Sens. 2021, 13, 159. [Google Scholar] [CrossRef]
- Angerer, J.; Han, G.; Fujisaki, I.; Havstad, K. Climate change and ecosystems of Asia with emphasis on inner Mongolia and Mongolia. Rangelands 2008, 30, 46–51. [Google Scholar] [CrossRef]
- Chen, J.; John, R.; Sun, G.; Fan, P.; Henebry, G.M.; Fernández-Giménez, M.E.; Zhang, Y.; Park, H.; Tian, L.; Groisman, P.; et al. Prospects for the sustainability of social-ecological systems (SES) on the Mongolian plateau: Five critical issues. Environ. Res. Lett. 2018, 13, 123004. [Google Scholar] [CrossRef]
- Qi, J.; Chen, J.; Wan, S.; Ai, L. Understanding the coupled natural and human systems in Dryland East Asia. Environ. Res. Lett. 2012, 7, 015202. [Google Scholar] [CrossRef]
- Chen, H.; Shao, L.; Zhao, M.; Zhang, X.; Zhang, D. Grassland conservation programs, vegetation rehabilitation and spatial dependency in Inner Mongolia, China. Land Use Policy 2017, 64, 429–439. [Google Scholar] [CrossRef]
- Tan, M.; Li, X. Does the Green Great Wall effectively decrease dust storm intensity in China? A study based on NOAA NDVI and weather station data. Land Use Policy 2015, 43, 42–47. [Google Scholar] [CrossRef]
- Friedl, M.A.; Sulla-Menashe, D.; Tan, B.; Schneider, A.; Ramankutty, N.; Sibley, A.; Huang, X. MODIS Collection 5 global land cover: Algorithm refinements and characterization of new datasets. Remote Sens. Environ. 2010, 114, 168–182. [Google Scholar] [CrossRef]
- Xiao, Z.; Liang, S.; Sun, R.; Wang, J.; Jiang, B. Estimating the fraction of absorbed photosynthetically active radiation from the MODIS data based GLASS leaf area index product. Remote Sens. Environ. 2015, 171, 105–117. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Pimm, S.L.; Joppa, L.N. Global patterns of terrestrial vertebrate diversity and conservation. Proc. Natl. Acad. Sci. USA 2013, 110, E2602–E2610. [Google Scholar] [CrossRef]
- Brasil, L.S.; Silverio, D.V.; Cabette, H.S.R.; Batista, J.D.; Vieira, T.B.; Dias-Silva, K.; Oliveira-Junior, J.M.B.d.; Carvalho, F.G.d.; Calvão, L.B.; Macedo, M.N.; et al. Net primary productivity and seasonality of temperature and precipitation are predictors of the species richness of the Damselflies in the Amazon. Basic Appl. Ecol. 2019, 35, 45–53. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Sheldon, K.S.; Tewksbury, J.J. The impact of seasonality in temperature on thermal tolerance and elevational range size. Ecology 2014, 95, 2134–2143. [Google Scholar] [CrossRef]
- Coops, N.C.; Bolton, D.K.; Hobi, M.L.; Radeloff, V.C. Untangling multiple species richness hypothesis globally using remote sensing habitat indices. Ecol. Indic. 2019, 107, 105567. [Google Scholar] [CrossRef]
- Zhang, C.; Li, L.; Guan, Y.; Cai, D.; Chen, H.; Bian, X.; Guo, S. Impacts of vegetation properties and temperature characteristics on species richness patterns in drylands: Case study from Xinjiang. Ecol. Indic. 2021, 133, 108417. [Google Scholar] [CrossRef]
- Tews, J.; Brose, U.; Grimm, V.; Tielbörger, K.; Wichmann, M.C.; Schwager, M.; Jeltsch, F. Animal species diversity driven by habitat heterogeneity/diversity: The importance of keystone structures. J. Biogeogr. 2004, 31, 79–92. [Google Scholar] [CrossRef]
- Mason, N.W.H.; Mouillot, D.; Lee, W.G.; Wilson, J.B. Functional richness, functional evenness and functional divergence: The primary components of functional diversity. Oikos 2005, 111, 112–118. [Google Scholar] [CrossRef]
- Williams, S.E.; Middleton, J. Climatic seasonality, resource bottlenecks, and abundance of rainforest birdss: Implications for global climate change. Divers. Distrib. 2008, 14, 69–77. [Google Scholar] [CrossRef]
- Lasmar, C.J.; Rosa, C.; Queiroz, A.C.M.; Nunes, C.A.; Imata, M.M.G.; Alves, G.P.; Nascimento, G.B.; Ázara, L.N.; Vieira, L.; Louzada, J.; et al. Temperature and productivity distinctly affect the species richness of ectothermic and endothermic multitrophic guilds along a tropical elevational gradient. Oecologia 2021, 197, 243–257. [Google Scholar] [CrossRef]
- Hawkins, B.A.; Field, R.; Cornell, H.V.; Currie, D.J.; Guégan, J.-F.; Kaufman, D.M.; Kerr, J.T.; Mittelbach, G.G.; Oberdorff, T.; O’Brien, E.M.; et al. Energy, Water, and Broad-Scale Geographic Patterns of Species Richness. Ecology 2003, 84, 3105–3117. [Google Scholar] [CrossRef]
- Mimiller-Rushing, A.J.; Lloyd-Evans, T.L.; Primack, R.B.; Satzinger, P. Bird migration times, climate change, and changing population sizes. Glob. Change Biol. 2008, 14, 1959–1972. [Google Scholar] [CrossRef]






| Name | Description | Data Source | Spatial Resolution |
|---|---|---|---|
| GLASS MODIS FAPAR | Calculate DHIs | National Earth System Science Data Center (http://www.geodata.cn/, accessed on 15 February 2022) | 500 m |
| Species distribution data | Includes mammal, bird, and amphibian species richness | The BiodiversityMapping.org site (https://biodiversitymapping.org/, accessed on 15 June 2022) | 10 km |
| BIOCLIM climate variable | Includes 19 biologically meaningful variables (Table 2) | WorldClim (https://worldclim.org/data/worldclim21.html, accessed on 1 August 2022) | 21 km² |
| Temperature | Includes maximum and minimum temperature | 21 km² | |
| Precipitation | Annual cumulative precipitation | 21 km² |
| Variables | Description | Variables | Description |
|---|---|---|---|
| Bio1 | Annual Mean Temperature (°C) | Bio11 | Mean Temperature of Coldest Quarter (°C) |
| Bio2 | Mean Diurnal Range (°C) | Bio12 | Annual Precipitation (mm) |
| Bio3 | Isothermality (Bio2/Bio7 × 100) | Bio13 | Precipitation of Wettest Month (mm) |
| Bio4 | Temperature Seasonality | Bio14 | Precipitation of Driest Month (mm) |
| Bio5 | Max Temperature of Warmest Month (°C) | Bio15 | Precipitation Seasonality (mm) |
| Bio6 | Min Temperature of Coldest Month (°C) | Bio16 | Precipitation of Wettest Quarter (mm) |
| Bio7 | Temperature Annual Range (°C) | Bio17 | Precipitation of Driest Quarter (mm) |
| Bio8 | Mean Temperature of Wettest Quarter (°C) | Bio18 | Precipitation of Warmest Quarter (mm) |
| Bio9 | Mean Temperature of Driest Quarter (°C) | Bio19 | Precipitation of Coldest Quarter (mm) |
| Bio10 | Mean Temperature of Warmest Quarter (°C) | - | - |
| Model | Grouping of Variables | Mammals | Birds | Amphibians |
|---|---|---|---|---|
| LR | DHI | 0.35 | 0.54 | 0.36 |
| DHI + Climate variables | 0.51 | 0.72 | 0.68 | |
| GAM | DHI | 0.54 | 0.68 | 0.51 |
| DHI + Climate variables | 0.71 | 0.82 | 0.80 | |
| RF | DHI | 0.62 | 0.74 | 0.61 |
| DHI + Climate variables | 0.89 | 0.94 | 0.91 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Yang, Y.; Yue, X.; Chen, X.; Liu, Y. Dynamic Habitat Indices and Climatic Characteristics Explain Species Richness Patterns on the Mongolian Plateau. Remote Sens. 2023, 15, 1092. https://doi.org/10.3390/rs15041092
Liu Y, Yang Y, Yue X, Chen X, Liu Y. Dynamic Habitat Indices and Climatic Characteristics Explain Species Richness Patterns on the Mongolian Plateau. Remote Sensing. 2023; 15(4):1092. https://doi.org/10.3390/rs15041092
Chicago/Turabian StyleLiu, Yingbin, Yaping Yang, Xiafang Yue, Xiaona Chen, and Yangxiaoyue Liu. 2023. "Dynamic Habitat Indices and Climatic Characteristics Explain Species Richness Patterns on the Mongolian Plateau" Remote Sensing 15, no. 4: 1092. https://doi.org/10.3390/rs15041092
APA StyleLiu, Y., Yang, Y., Yue, X., Chen, X., & Liu, Y. (2023). Dynamic Habitat Indices and Climatic Characteristics Explain Species Richness Patterns on the Mongolian Plateau. Remote Sensing, 15(4), 1092. https://doi.org/10.3390/rs15041092







