High-Intensity Intermittent Exercise Increases Fat Oxidation Rate and Reduces Postprandial Triglyceride Concentrations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Design
2.3. Preliminary Measurements
2.3.1. Running Economy
2.3.2. Submaximal Oxygen Uptake
2.4. Protocol
2.4.1. The HIIE Intervention
2.4.2. The MICE Intervention
2.5. Oral Fat Tolerance Test
2.6. Blood Sample Collection and Analysis
2.7. Statistical Analyses
3. Results
3.1. Exercise Indices
3.2. The Fasted State on the Morning of Day 2 of Each Main Trial
3.3. Postprandial Fat Oxidation
3.4. Blood Biochemical Indices
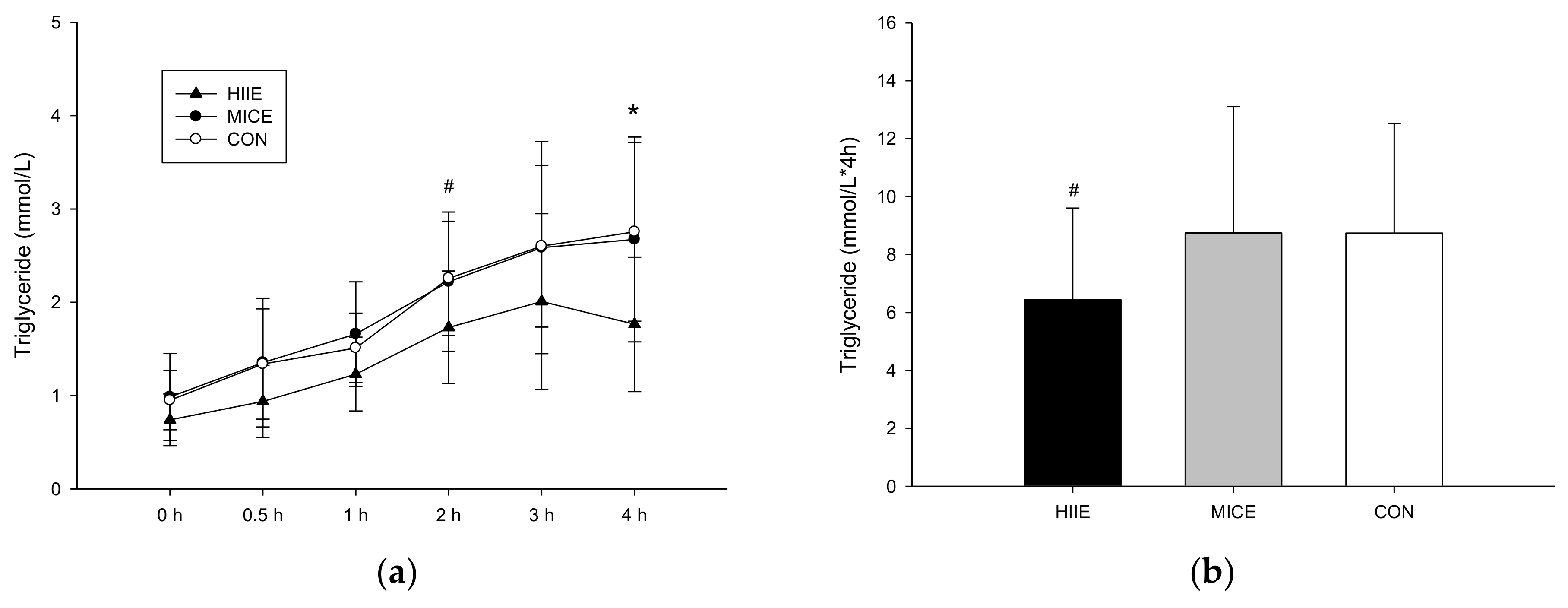
3.3.1. TG
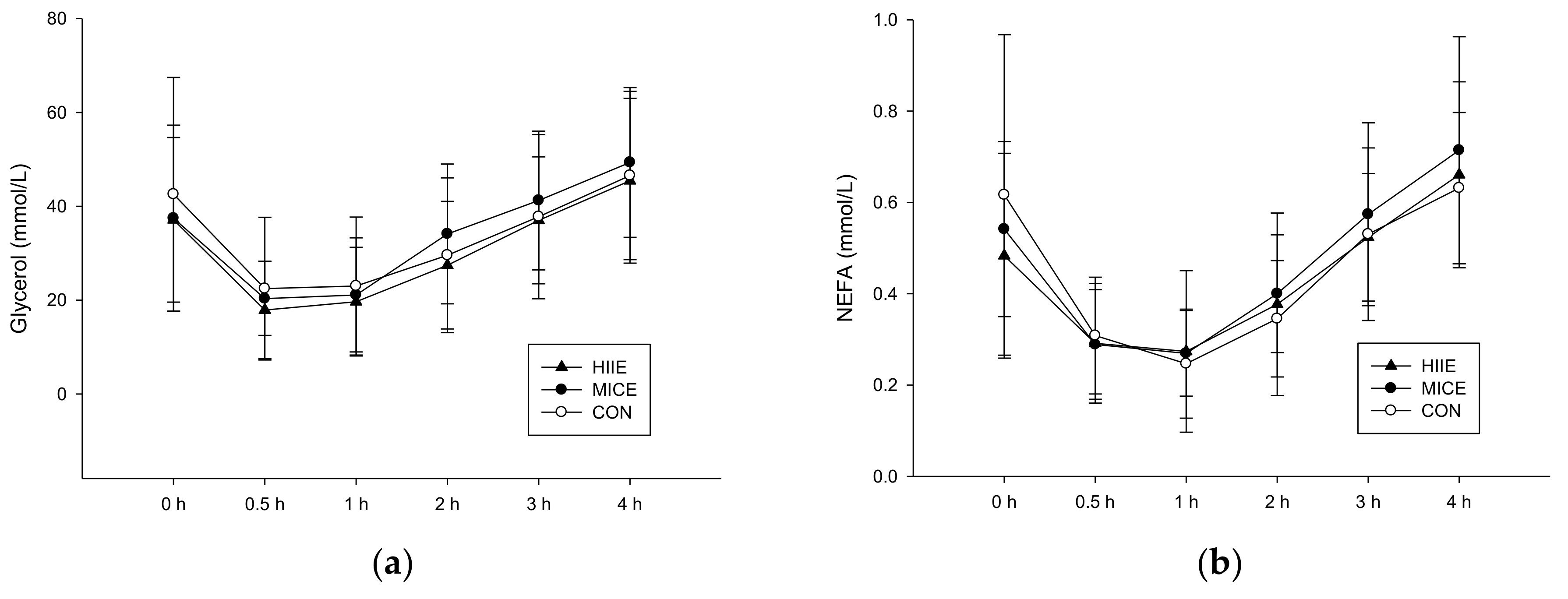
3.3.2. NEFA and Glycerol
3.3.3. CK
4. Discussion
5. Limitations
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker, P.M. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA 2007, 298, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Patsch, J.R.; Miesenbock, G.; Hopferwieser, T.; Muhlberger, V.; Knapp, E.; Dunn, J.K.; Gotto, A.M., Jr.; Patsch, W. Relation of triglyceride metabolism and coronary artery disease. Studies in the postprandial state. Arterioscler. Thromb. Vasc. Biol. 1992, 12, 1336–1345. [Google Scholar] [CrossRef]
- Sharrett, A.R.; Chambless, L.E.; Heiss, G.; Paton, C.C.; Patsch, W. Association of postprandial triglyceride and retinyl palmitate responses with asymptomatic carotid artery atherosclerosis in middle-aged men and women. The Atherosclerosis Risk in Communities (ARIC) Study. Arterioscler. Thromb. Vasc. Biol. 1995, 15, 2122–2129. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.; Pang, J.; Romic, G.; Watts, G.F. Postprandial hypertriglyceridemia and cardiovascular disease: Current and future therapies. Curr. Atheroscler. Reports 2013, 15, 309. [Google Scholar] [CrossRef] [PubMed]
- Aldred, H.E.; Perry, I.C.; Hardman, A.E. The effect of a single bout of brisk walking on postprandial lipemia in normolipidemic young adults. Metabolism 1994, 43, 836–841. [Google Scholar] [CrossRef]
- Tsetsonis, N.V.; Hardman, A.E.; Mastana, S.S. Acute effects of exercise on postprandial lipemia: A comparative study in trained and untrained middle-aged women. Am. J. Clin. Nutr. 1997, 65, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Gill, J.M.; Herd, S.L.; Vora, V.; Hardman, A.E. Effects of a brisk walk on lipoprotein lipase activity and plasma triglyceride concentrations in the fasted and postprandial states. Eur. J. Appl. Phys. 2003, 89, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Seip, R.L.; Mair, K.; Cole, T.G.; Semenkovich, C.F. Induction of human skeletal muscle lipoprotein lipase gene expression by short-term exercise is transient. Am. J. Physiol. 1997, 272, E255–E261. [Google Scholar] [CrossRef] [PubMed]
- Gill, J.M.; Hardman, A.E. Exercise and postprandial lipid metabolism: An update on potential mechanisms and interactions with high-carbohydrate diets (review). J. Nutr. Biochem. 2003, 14, 122–132. [Google Scholar] [CrossRef]
- Freese, E.C.; Levine, A.S.; Chapman, D.P.; Hausman, D.B.; Cureton, K.J. Effects of acute sprint interval cycling and energy replacement on postprandial lipemia. J. Appl. Phys. 2011, 111, 1584–1589. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, B.; Ratkevicius, A.; Gray, P.; Frenneaux, M.P.; Gray, S.R. High-intensity exercise attenuates postprandial lipaemia and markers of oxidative stress. Clin. Sci. 2012, 123, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Fat, R.C.M.; Boutcher, Y.N.; Boutcher, S.H. Effect of high-intensity intermittent exercise on postprandial plasma triacylglycerol in sedentary young women. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Moro, T.; Marcolin, G.; Neri, M.; Bianco, A.; Palma, A.; Grimaldi, K. High-Intensity Interval Resistance Training (HIRT) influences resting energy expenditure and respiratory ratio in non-dieting individuals. J. Transl. Med. 2012, 10, 237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, B.A. High-Intensity Interval Training: Efficient, Effective, and a Fun Way to Exercise Brought to you by the American College of Sports Medicine www.acsm.org. ACSMS Health Fit. J. 2013, 17, 3. [Google Scholar] [CrossRef]
- Burns, S.F.; Miyashita, M.; Stensel, D.J. High-intensity interval exercise and postptandial triacylglycerol. Sports Med. 2015, 45, 957–968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabata, I.; Nishimura, K.; Kouzaki, M.; Hirai, Y.; Ogita, F.; Miyachi, M.; Yamamoto, K. Effects of moderate-intensity endurance and high-intensity intermittent training on anaerobic capacity and VO2max. Med. Sci. Sports Exerc. 1996, 28, 1327–1330. [Google Scholar] [CrossRef] [PubMed]
- Emberts, T.; Porcari, J.; Dobers-Tein, S.; Steffen, J.; Foster, C. Exercise intensity and energy expenditure of a tabata workout. J. Sports Sci. Med. 2013, 12, 612–613. [Google Scholar] [PubMed]
- Yang, T.J.; Chiu, C.H.; Tseng, M.H.; Chang, C.K.; Wu, C.L. The influence of pre-exercise glucose versus Fructose ingestion on subsequent postprandial Lipemia. Nutrients 2018, 10, 149. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G* Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef] [PubMed]
- Trombold, J.R.; Christmas, K.M.; Machin, D.R.; Kim, I.Y.; Coyle, E.F. Acute high-intensity endurance exercise is more effective than moderate-intensity exercise for attenuation of postprandial triglyceride elevation. J. Appl. Phys. 2013, 114, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Harrison, M.; O’Gorman, D.J.; McCaffrey, N.; Hamilton, M.T.; Zderic, T.W.; Carson, B.P.; Moyna, N.M. Influence of acute exercise with and without carbohydrate replacement on postprandial lipid metabolism. J. Appl. Phys. 2009, 106, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L.J.; Greenhaff, P.L.; Constantin-Teodosiu, D.; Saris, W.H.; Wagenmakers, A.J. The effects of increasing exercise intensity on muscle fuel utilisation in humans. J. Physiol. 2001, 536, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Romijn, J.A.; Coyle, E.F.; Sidossis, L.S.; Gastaldelli, A.; Horowitz, J.F.; Endert, E.; Wolfe, R.R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol. 1993, 265, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Kiens, B.; Richter, E.A. Utilization of skeletal muscle triacylglycerol during postexercise recovery in humans. Am. J. Physiol. 1998, 275, E332–E337. [Google Scholar] [CrossRef] [PubMed]
- Kiens, B.; Lithell, H. Lipoprotein metabolism influenced by training-induced changes in human skeletal muscle. J. Clin. Investig. 1989, 83, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Thornton, M.K.; Potteiger, J.A. Effects of resistance exercise bouts of different intensities but equal work on EPOC. Med. Sci. Sports Exerc. 2002, 34, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Treuth, M.S.; Hunter, G.R.; Williams, M. Effects of exercise intensity on 24-h energy expenditure and substrate oxidation. Med. Sci. Sports Exerc. 1996, 28, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Melby, C.; Scholl, C.; Edwards, G.; Bullough, R. Effect of acute resistance exercise on postexercise energy expenditure and resting metabolic rate. J. Appl. Phys. 1993, 75, 1847–1853. [Google Scholar] [CrossRef] [PubMed]
- Gillette, C.A.; Bullough, R.C.; Melby, C.L. Postexercise energy expenditure in response to acute aerobic or resistive exercise. Int. J. Sport Nutr. 1994, 4, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Freese, E.C.; Gist, N.H.; Cureton, K.J. Effect of prior exercise on postprandial lipemia: An updated quantitative review. J. Appl. Phys. 2014, 116, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Elliot, D.L.; Goldberg, L.; Kuehl, K.S. Effect of Resistance Training on Excess Post-exercise Oxygen Consumption. J. Strength Cond. Res. 1992, 6, 77–81. [Google Scholar]
- Foureaux, G.; Pinto, K.M.D.C.; Dâmaso, A. Effects of excess post-exercise oxygen consumption and resting metabolic rate in energetic cost. Rev. Bras. Med. Esporte 2006, 12, 393–398. [Google Scholar] [CrossRef]
- Preiss-Landl, K.; Zimmermann, R.; Hammerle, G.; Zechner, R. Lipoprotein lipase: The regulation of tissue specific expression and its role in lipid and energy metabolism. Curr. Opin. Lipidol. 2002, 13, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Mead, J.R.; Irvine, S.A.; Ramji, D.P. Lipoprotein lipase: Structure, function, regulation, and role in disease. J. Mol. Med. 2002, 80, 753–769. [Google Scholar] [CrossRef] [PubMed]
- Herd, S.L.; Kiens, B.; Boobis, L.H.; Hardman, A.E. Moderate exercise, postprandial lipemia, and skeletal muscle lipoprotein lipase activity. Metabolism 2001, 50, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.T.; Hamilton, D.G.; Zderic, T.W. Exercise physiology versus inactivity physiology: An essential concept for understanding lipoprotein lipase regulation. Exerc. Sport Sci. Rev. 2004, 32, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Petitt, D.S.; Arngrimsson, S.A.; Cureton, K.J. Effect of resistance exercise on postprandial Lipemia. J. Appl. Phys. 2003, 94, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Byrnes, W.C.; McCormick, K.M.; Turcotte, L.P.; White, J.S. Muscle soreness and serum creatine kinase activity following isometric, eccentric, and concentric exercise. Int. J. Sports Med. 1986, 7, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Ebbeling, C.B.; Clarkson, P.M. Exercise-induced muscle damage and adaptation. Sports Med. 1989, 7, 207–234. [Google Scholar] [CrossRef] [PubMed]
- Burns, S.F.; Broom, D.R.; Miyashita, M.; Ueda, C.; Stensel, D.J. Increased postprandial triacylglycerol concentrations following resistance exercise. Med. Sci. Sports Exerc. 2006, 38, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Pafili, Z.K.; Bogdanis, G.C.; Tsetsonis, N.V.; Maridaki, M. Postprandial lipemia 16 and 40 hours after low-volume eccentric resistance exercise. Med. Sci. Sports Exerc. 2009, 41, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Dolezal, B.A.; Potteiger, J.A.; Jacobsen, D.J.; Benedict, S.H. Muscle damage and resting metabolic rate after acute resistance exercise with an eccentric overload. Med. Sci. Sports Exerc. 2000, 32, 1202–1207. [Google Scholar] [CrossRef] [PubMed]


| HIIE | MICE | |
|---|---|---|
| Body fat (%) | 21.8 ± 6.2 | |
| VO2max (mL/min/kg) | 42.2 ± 7.86 | |
| Mean heart rate (bpm) | 140.1 ± 3.7 | 127.8 ± 1.9 |
| Max heart rate (bpm) | 171.0 ± 9.1 | 138.7 ± 2.0 |
| Exercise time (min) | 20 | 36.6 ± 4.8 |
| Slope (%) | NA | 4.9 ± 2.6 |
| Speed (km/h) | NA | 5.6 ± 0.2 |
| EE (kcal) | 384.4 ± 69.9 | 376.7 ± 76.9 |
| HIIE | MICE | CON | p | |
|---|---|---|---|---|
| TG (mmol/L) | 0.74 ± 0.27 | 0.99 ± 0.46 | 0.95 ± 0.31 | 0.174 |
| NEFA (mmol/L) | 0.54 ± 0.19 | 0.48 ± 0.22 | 0.62 ± 0.35 | 0.499 |
| Glycerol (µmol/L) | 37.11 ± 17.53 | 37.44 ± 19.84 | 42.56 ± 24.90 | 0.721 |
| CK (mmol/L) | 242.11 ± 108.62 1 | 164.78 ± 61.03 | 127.78 ± 33.55 | 0.04 |
| Fat oxidation (g/min) | 0.07 ± 0.04 | 0.06 ± 0.05 | 0.07 ± 0.04 | 0.403 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, T.-J.; Wu, C.-L.; Chiu, C.-H. High-Intensity Intermittent Exercise Increases Fat Oxidation Rate and Reduces Postprandial Triglyceride Concentrations. Nutrients 2018, 10, 492. https://doi.org/10.3390/nu10040492
Yang T-J, Wu C-L, Chiu C-H. High-Intensity Intermittent Exercise Increases Fat Oxidation Rate and Reduces Postprandial Triglyceride Concentrations. Nutrients. 2018; 10(4):492. https://doi.org/10.3390/nu10040492
Chicago/Turabian StyleYang, Tsung-Jen, Ching-Lin Wu, and Chih-Hui Chiu. 2018. "High-Intensity Intermittent Exercise Increases Fat Oxidation Rate and Reduces Postprandial Triglyceride Concentrations" Nutrients 10, no. 4: 492. https://doi.org/10.3390/nu10040492
APA StyleYang, T.-J., Wu, C.-L., & Chiu, C.-H. (2018). High-Intensity Intermittent Exercise Increases Fat Oxidation Rate and Reduces Postprandial Triglyceride Concentrations. Nutrients, 10(4), 492. https://doi.org/10.3390/nu10040492





