An In Vitro Study on the Combination Effect of Metformin and N-Acetyl Cysteine against Hyperglycaemia-Induced Cardiac Damage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Metformin and N-Acetylcysteine Preparation
2.2. Effect of MET and NAC on H9c2 Cells Exposed to High Glucose
2.3. Measurement of Metabolic Activity
2.4. Measurement of 2-Deoxy-[3H]-D-Glucose (DOG) Uptake
2.5. Quantification of Intracellular Lipid Content
2.6. Measurement of Intracellular Reactive Oxygen Species (ROS)
2.7. Measurements of Superoxide Dismutase (SOD) Activity
2.8. Measurements of Glutathione (GSH) Content
2.9. Lipid Peroxidation Analysis
2.10. Evaluating Mitochondrial Membrane Potential (ΔΨm)
2.11. The Activity of Caspase 3/7
2.12. Western Blot Analysis
3. Results
3.1. The Combination Effect of MET and NAC on ATP Activity
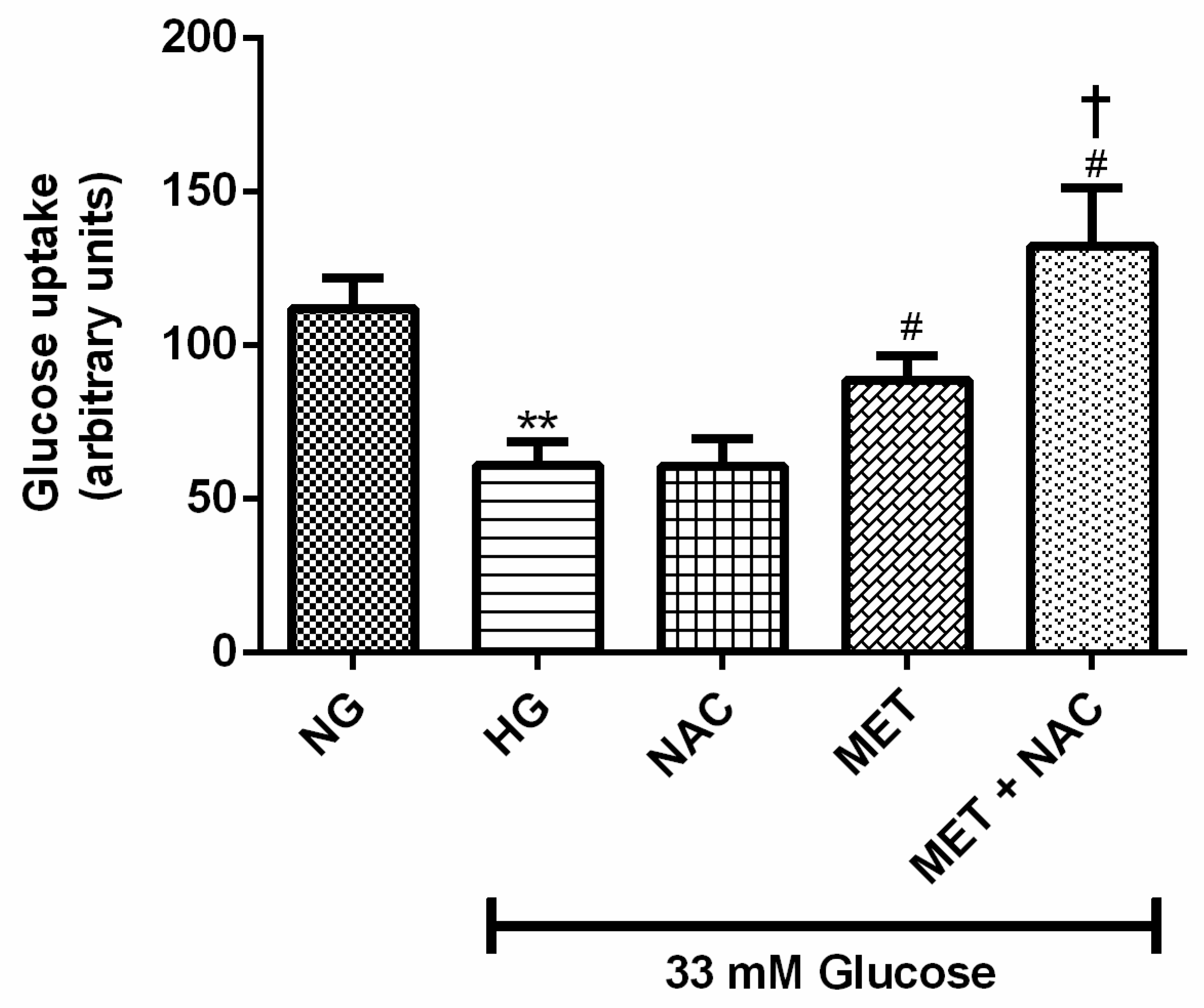
3.2. The Combination Effect of MET and NAC on Glucose Uptake (GU)
3.3. The Combination Effect of MET and NAC on Lipid Content
3.4. The Combination Effect of MET and NAC to Attenuate ROS Production
3.5. The Combination Effect of MET and NAC to Diminish Lipid Peroxidation
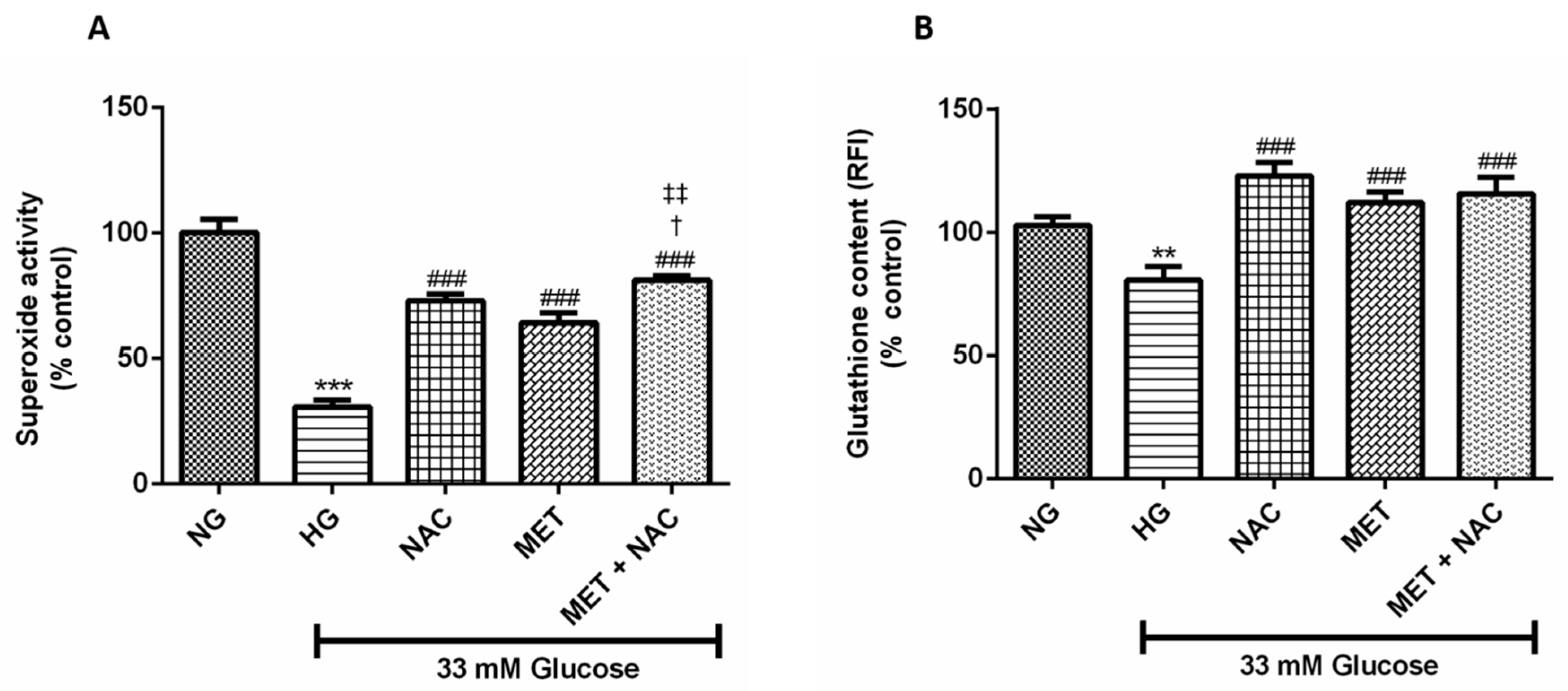
3.6. The Combination Effect of MET and NAC to Augment SOD Activity And GSH Content
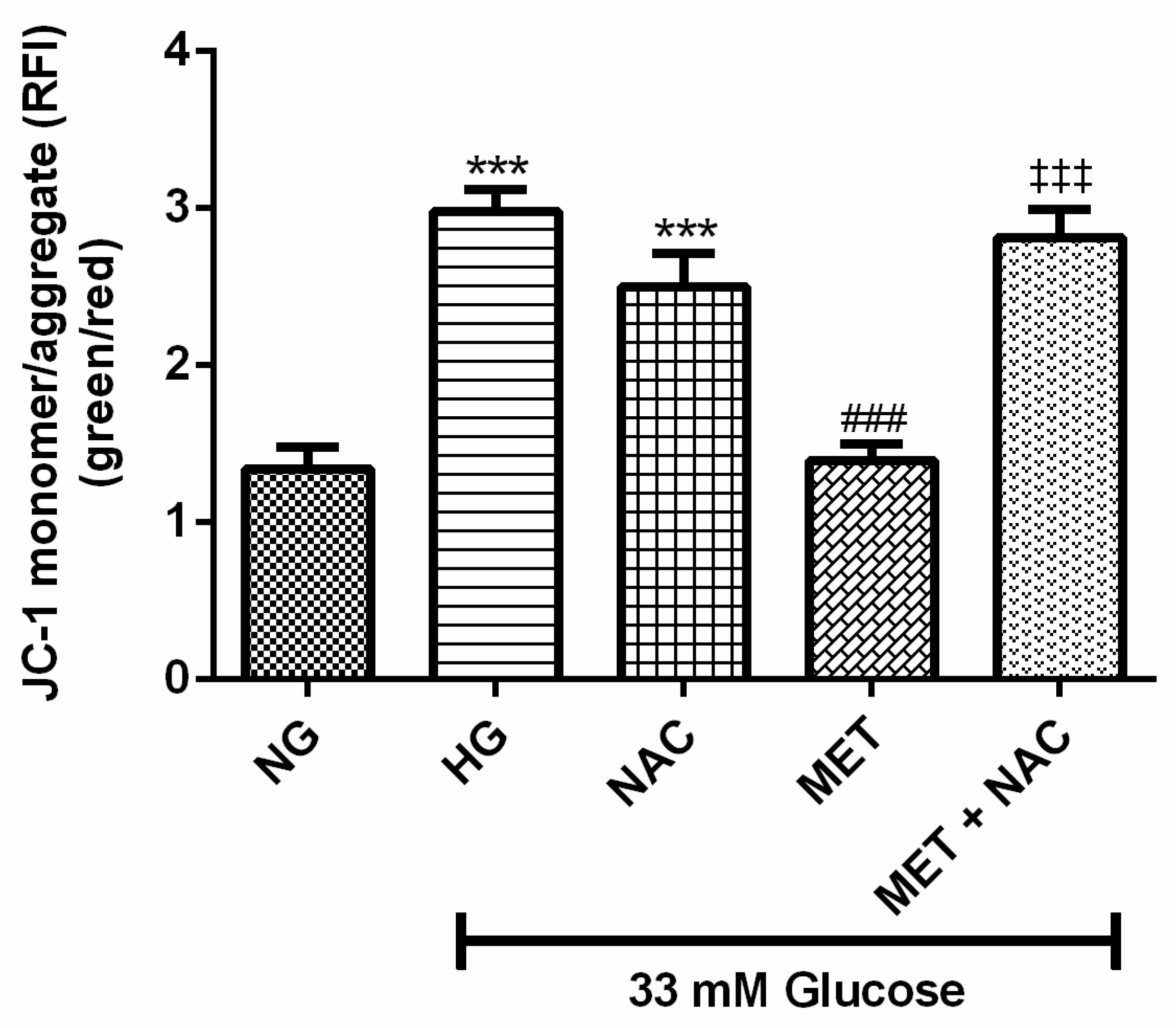
3.7. The Combination Effect of MET and NAC to Modify Mitochondrial Membrane Potential (ΔΨm)
3.8. The Effect of MET and NAC on DNA Fragmentation
3.9. The Effect of MET and NAC on Caspase 3/7 Activity
3.10. Gene and Protein Expression
3.10.1. The effect of MET and NAC on phosphorylated 5’ AMP-activated protein kinase (AMPK) and nuclear factor-kappa B phosphorylation (p-NF-kB) expression
3.10.2. The effect of MET and NAC to increased glucose uptake, through the inhibition of phosphorylated protein kinase C (PKC(Ser660))/insulin receptor substrate-1 (IRS-1(Ser307)) inhibition
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Federation, I. IDF Diabetes Atlas, 8th ed.; IDF: Brussels, Belgium, 2017. [Google Scholar]
- Keates, A.K.; Mocumbi, A.O.; Ntsekhe, M.; Sliwa, K.; Stewart, S. Cardiovascular disease in Africa: Epidemiological profile and challenges. Nat. Rev. Cardiol. 2017, 14, 273. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, B.K.; Pandey, K.B.; Abidi, A.B.; Rizvi, S.I. Markers of Oxidative Stress during Diabetes Mellitus. J. Biomark. 2013, 2013, 378790. [Google Scholar] [CrossRef] [PubMed]
- Davargaon, R.S.; Sambe, A.D.; Muthangi, V.V.S. Toxic effect of high glucose on cardiomyocytes, H9c2 cells: Induction of oxidative stress and ameliorative effect of trolox. J. Biochem. Mol. Toxicol. 2019, 33, e22272. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Kayama, Y.; Raaz, U.; Jagger, A.; Adam, M.; Schellinger, I.N.; Sakamoto, M.; Suzuki, H.; Toyama, K.; Spin, J.M.; Tsao, P.S. Diabetic Cardiovascular Disease Induced by Oxidative Stress. Int. J. Mol. Sci. 2015, 16, 25234–25263. [Google Scholar] [CrossRef]
- Johnson, R.; Dludla, P.; Joubert, E.; February, F.; Mazibuko, S.; Ghoor, S.; Muller, C.; Louw, J. Aspalathin, a dihydrochalcone C-glucoside, protects H9c2 cardiomyocytes against high glucose induced shifts in substrate preference and apoptosis. Mol. Nutr. Food Res. 2016, 60, 922–934. [Google Scholar] [CrossRef]
- Schulze, P.C.; Drosatos, K.; Goldberg, I.J. Lipid Use and Misuse by the Heart. Circ. Res. 2016, 118, 1736–1751. [Google Scholar] [CrossRef]
- Ayala, A.; Munoz, M.F.; Arguelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Batchuluun, B.; Inoguchi, T.; Sonoda, N.; Sasaki, S.; Inoue, T.; Fujimura, Y.; Miura, D.; Takayanagi, R. Metformin and liraglutide ameliorate high glucose-induced oxidative stress via inhibition of PKC-NAD(P)H oxidase pathway in human aortic endothelial cells. Atherosclerosis 2014, 232, 156–164. [Google Scholar] [CrossRef]
- Dludla, P.V.; Dias, S.C.; Obonye, N.; Johnson, R.; Louw, J.; Nkambule, B.B. A Systematic Review on the Protective Effect of N-Acetyl Cysteine Against Diabetes-Associated Cardiovascular Complications. Am. J. Cardiovasc. Drugs Drugs Devices Other Interv. 2018, 18, 283–298. [Google Scholar] [CrossRef]
- Talasa, A.H. Potential role of N-acetylcysteine in cardiovascular disorder. Clin. Pract. 2011, 8, 237. [Google Scholar]
- Rasoli, S.; Kakouros, N.; Harling, L.; Gukop, P.; Soni, M.; Athanasiou, T.; Kourliouros, A. Antioxidant vitamins in the prevention of atrial fibrillation: What is the evidence? Cardiol. Res. Pract. 2011, 2011, 164078. [Google Scholar] [CrossRef] [PubMed]
- Ozaydin, M.; Peker, O.; Erdogan, D.; Kapan, S.; Turker, Y.; Varol, E.; Ozguner, F.; Dogan, A.; Ibrisim, E. N-acetylcysteine for the prevention of postoperative atrial fibrillation: A prospective, randomized, placebo-controlled pilot study. Eur. Heart J. 2008, 29, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Mariappan, N.; Elks, C.M.; Sriramula, S.; Guggilam, A.; Liu, Z.; Borkhsenious, O.; Francis, J. NF-κB-induced oxidative stress contributes to mitochondrial and cardiac dysfunction in type II diabetes. Cardiovasc. Res. 2009, 85, 473–483. [Google Scholar] [CrossRef]
- Jadaun, P.; Yadav, D.; Bisen, P.S. Spirulina platensis prevents high glucose-induced oxidative stress mitochondrial damage mediated apoptosis in cardiomyoblasts. Cytotechnology 2018, 70, 523–536. [Google Scholar] [CrossRef]
- Xia, Y.; Gong, L.; Liu, H.; Luo, B.; Li, B.; Li, R.; Li, B.; Lv, M.; Pan, J.; An, F. Inhibition of prolyl hydroxylase 3 ameliorates cardiac dysfunction in diabetic cardiomyopathy. Mol. Cell. Endocrinol. 2015, 403, 21–29. [Google Scholar] [CrossRef]
- Dludla, P.V.; Joubert, E.; Muller, C.J.F.; Louw, J.; Johnson, R. Hyperglycemia-induced oxidative stress and heart disease-cardioprotective effects of rooibos flavonoids and phenylpyruvic acid-2-O-beta-D-glucoside. Nutr. Metab. 2017, 14, 45. [Google Scholar] [CrossRef]
- Dludla, P.V.; Muller, C.J.; Joubert, E.; Louw, J.; Essop, M.F.; Gabuza, K.B.; Ghoor, S.; Huisamen, B.; Johnson, R. Aspalathin Protects the Heart against Hyperglycemia-Induced Oxidative Damage by Up-Regulating Nrf2 Expression. Molecules 2017, 22, 129. [Google Scholar] [CrossRef]
- Johnson, R.; Dludla, P.V.; Muller, C.J.; Huisamen, B.; Essop, M.F.; Louw, J. The Transcription Profile Unveils the Cardioprotective Effect of Aspalathin against Lipid Toxicity in an In Vitro H9c2 Model. Molecules 2017, 22, 219. [Google Scholar] [CrossRef]
- Shi, R.; Huang, C.C.; Aronstam, R.S.; Ercal, N.; Martin, A.; Huang, Y.W. N-acetylcysteine amide decreases oxidative stress but not cell death induced by doxorubicin in H9c2 cardiomyocytes. BMC Pharmacol. 2009, 9, 7. [Google Scholar] [CrossRef]
- Sanderson, M.; Mazibuko, S.E.; Joubert, E.; de Beer, D.; Johnson, R.; Pheiffer, C.; Louwa, J.; Mullera, C.J.F. Effects of fermented rooibos (Aspalathus linearis) on adipocyte differentiation. Phytomedicine Int. J. Phytother. Phytopharm. 2014, 21, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Shabalala, S.C.; Dludla, P.V.; Muller, C.J.F.; Nxele, X.; Kappo, A.P.; Louw, J.; Johnson, R. Aspalathin ameliorates doxorubicin-induced oxidative stress in H9c2 cardiomyoblasts. Toxicol. In Vitro An Int. J. Publ. Assoc. BIBRA 2019, 55, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart Disease and Stroke Statistics-2017 Update: A Report from the American Heart Association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Guo, Z.; Nagareddy, P.R.; Yuen, V.; Yeung, E.; McNeill, J.H. Antioxidant N-acetylcysteine restores myocardial Mn-SOD activity and attenuates myocardial dysfunction in diabetic rats. Eur. J. Pharmacol. 2006, 544, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Wu, W.; Chen, J.; Guo, R.; Lin, J.; Liao, X.; Feng, J. Exogenous hydrogen sulfide protects H9c2 cardiac cells against high glucose-induced injury by inhibiting the activities of the p38 MAPK and ERK1/2 pathways. Int. J. Mol. Med. 2013, 32, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Li, R.; Ma, Y.; Wang, X.; Li, C.; Zhang, X.; Mab, R.; Ding, Z.; Liu, L. Alpha-Lipoic acid increases tolerance of cardiomyoblasts to glucose/glucose oxidase-induced injury via ROS-dependent ERK1/2 activation. Biochim. Biophys. Acta 2012, 1823, 920–929. [Google Scholar] [CrossRef]
- Cai, H.; Zhang, G.; Chen, W.; Zhang, B.; Zhang, J.; Chang, J.; Tang, C.; Qi, Y.; Yin, X. Metformin protects the myocardium against isoproterenol-induced injury in rats through alleviating endoplasmic reticulum stress. Die Pharm. 2014, 69, 64–69. [Google Scholar]
- Ren, J.; Dominguez, L.J.; Sowers, J.R.; Davidoff, A.J. Metformin but not glyburide prevents high glucose-induced abnormalities in relaxation and intracellular Ca2+ transients in adult rat ventricular myocytes. Diabetes. 1999, 48, 2059–2065. [Google Scholar] [CrossRef]
- Wang, G.Y.; Bi, Y.G.; Liu, X.D.; Zhao, Y.; Han, J.F.; Wei, M.; Zhang, Q.Y. Autophagy was involved in the protective effect of metformin on hyperglycemia-induced cardiomyocyte apoptosis and Connexin43 downregulation in H9c2 cells. Int. J. Med. Sci. 2017, 14, 698–704. [Google Scholar] [CrossRef]
- Kajbaf, F.; De Broe, M.E.; Lalau, J.D. Therapeutic Concentrations of Metformin: A Systematic Review. Clin. Pharmacokinet. 2016, 55, 439–459. [Google Scholar] [CrossRef]
- Goyal, V.; Bews, H.; Cheung, D.; Premecz, S.; Mandal, S.; Shaikh, B.; Best, R.; Bhindi, R.; Chaudhary, R.; Ravandi, A.; et al. The Cardioprotective Role of N-Acetyl Cysteine Amide in the Prevention of Doxorubicin and Trastuzumab-Mediated Cardiac Dysfunction. Can. J. Cardiol. 2016, 32, 1513–1519. [Google Scholar] [CrossRef] [PubMed]
- Nagoor Meeran, M.F.; Mainzen Prince, P.S. Protective effects of N-acetyl cysteine on lipid peroxide metabolism on isoproterenol-induced myocardial infarcted rats. J. Biochem. Mol. Toxicol. 2011, 25, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Wongjaikam, S.; Kumfu, S.; Khamseekaew, J.; Chattipakorn, S.C.; Chattipakorn, N. Restoring the impaired cardiac calcium homeostasis and cardiac function in iron overload rats by the combined deferiprone and N-acetyl cysteine. Sci. Rep. 2017, 7, 44460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerksick, C.; Willoughby, D. The antioxidant role of glutathione and N-acetyl-cysteine supplements and exercise-induced oxidative stress. J. Int. Soc. Sports Nutr. 2005, 2, 38–44. [Google Scholar] [CrossRef] [Green Version]
- Martinez, P.F.; Bonomo, C.; Guizoni, D.M.; Junior, S.A.O.; Damatto, R.L.; Cezar, M.D.M.; Lima, A.R.R.; Pagan, L.U.; Seiva, F.R.; Fernandes, D.C.; et al. Influence of N-acetylcysteine on oxidative stress in slow-twitch soleus muscle of heart failure rats. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2015, 35, 148–159. [Google Scholar] [CrossRef]
- Abdel Baky, N.A.; Mohamed, A.M.; Faddah, L.M. Protective effect of N-acetyl cysteine and/or pro vitamin A against monosodium glutamate-induced cardiopathy in rats. J. Pharmacol. Toxicol. 2009, 4, 178–193. [Google Scholar]
- Basha, R.H.; Priscilla, D.H. An in vivo and in vitro study on the protective effects of N-acetylcysteine on mitochondrial dysfunction in isoproterenol treated myocardial infarcted rats. Exp Toxicol Pathol.: Off. J. Ges. Fur Toxikol. Pathol. 2013, 65, 7–14. [Google Scholar] [CrossRef]
- Ke, R.; Xu, Q.; Li, C.; Luo, L.; Huang, D. Mechanisms of AMPK in the maintenance of ATP balance during energy metabolism. Cell Biol. Int. 2018, 42, 384–392. [Google Scholar] [CrossRef] [Green Version]
- Zhi, L.; Yuzhang, Z.; Tianliang, H.; Hisatome, I.; Yamamoto, T.; Jidong, C. High Uric Acid Induces Insulin Resistance in Cardiomyocytes In Vitro and In Vivo. PLoS ONE 2016, 11, e0147737. [Google Scholar] [CrossRef]
- Hu, M.; Ye, P.; Liao, H.; Chen, M.; Yang, F. Metformin protects h9c2 cardiomyocytes from high-glucose and hypoxia/reoxygenation injury via inhibition of reactive oxygen species generation and inflammatory responses: Role of ampk and jnk. J. Diabetes Res. 2016, 2016, 2961954. [Google Scholar] [CrossRef] [Green Version]








© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, R.; Sangweni, N.F.; Mabhida, S.E.; Dludla, P.V.; Mabasa, L.; Riedel, S.; Chapman, C.; Mosa, R.A.; Kappo, A.P.; Louw, J.; et al. An In Vitro Study on the Combination Effect of Metformin and N-Acetyl Cysteine against Hyperglycaemia-Induced Cardiac Damage. Nutrients 2019, 11, 2850. https://doi.org/10.3390/nu11122850
Johnson R, Sangweni NF, Mabhida SE, Dludla PV, Mabasa L, Riedel S, Chapman C, Mosa RA, Kappo AP, Louw J, et al. An In Vitro Study on the Combination Effect of Metformin and N-Acetyl Cysteine against Hyperglycaemia-Induced Cardiac Damage. Nutrients. 2019; 11(12):2850. https://doi.org/10.3390/nu11122850
Chicago/Turabian StyleJohnson, Rabia, Nonhlakanipho F. Sangweni, Sihle E. Mabhida, Phiwayinkosi V. Dludla, Lawrence Mabasa, Sylvia Riedel, Charna Chapman, Rebamang A. Mosa, Abidemi P. Kappo, Johan Louw, and et al. 2019. "An In Vitro Study on the Combination Effect of Metformin and N-Acetyl Cysteine against Hyperglycaemia-Induced Cardiac Damage" Nutrients 11, no. 12: 2850. https://doi.org/10.3390/nu11122850




