Hesperidin Effects on Gut Microbiota and Gut-Associated Lymphoid Tissue in Healthy Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Sample Collection and Processing
2.3. Lymphocyte Isolation from Mesenteric Lymph Nodes
2.4. Quantification of Urine Total Phenolic Content
2.5. Fluorescence In Situ Hybridization of Gut Microbiota
2.6. Quantification of Bacteria Coated to IgA
2.7. MLN Lymphocyte Composition
2.8. MLN Lymphocyte Proliferative Capacity
2.9. Cytokine Quantification
2.10. Immunoglobulin Quantification by ELISA
2.11. Gene Expression by Real-Time Polymerase Chain Reaction (RT-PCR)
2.12. Statistical Analysis
3. Results
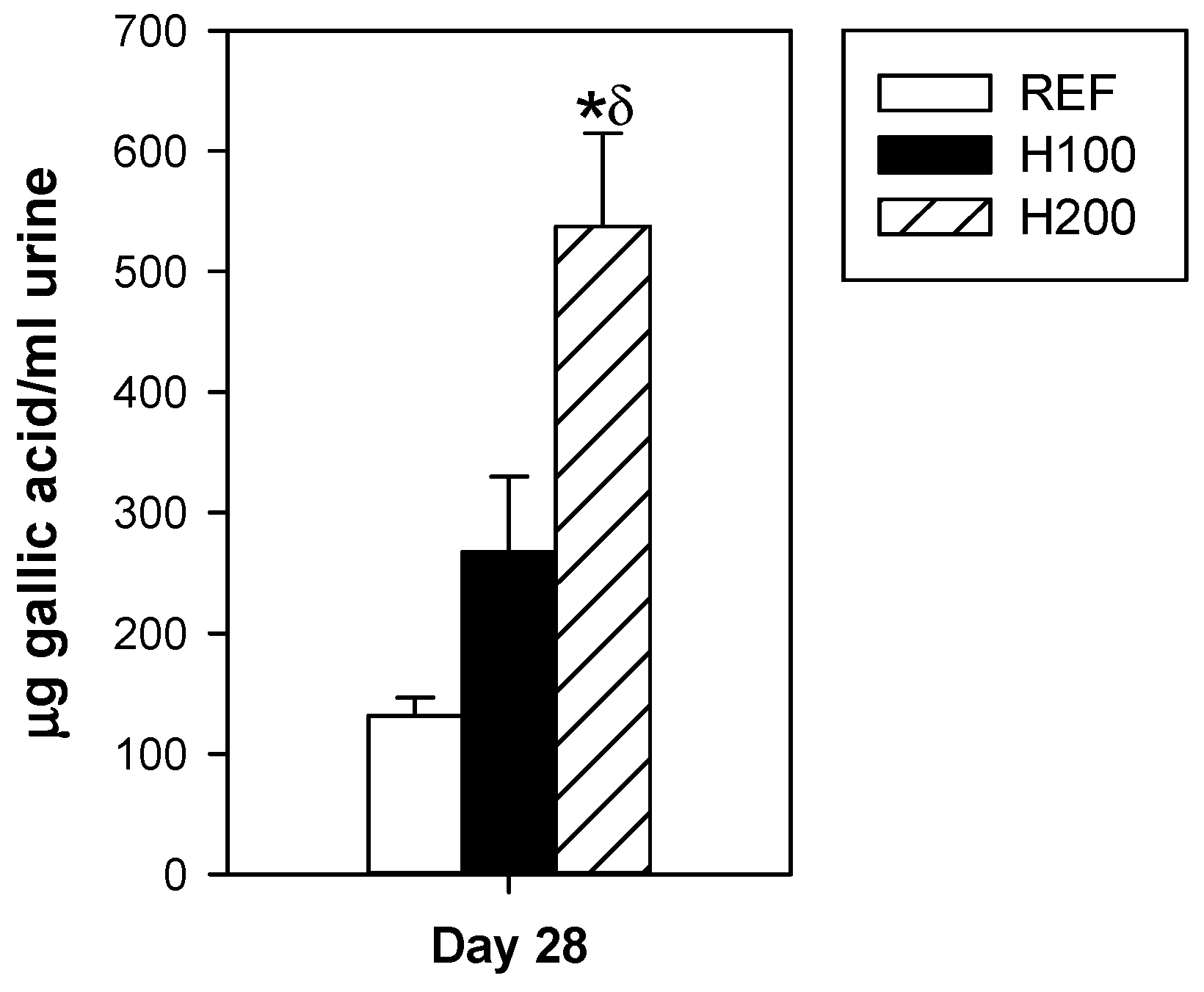
3.1. Influence of Hesperidin on Body Weight, Food Intake and Urine Flavonoid Content
3.2. Influence of Hesperidin on Caecal Microbiota
3.3. Influence of Hesperidin on MLNL Composition and Functionality
3.4. Effect of Hesperidin on Small Intestine Cytokines and Gene Expression
3.5. Effect of Hesperidin on Intestinal Immunoglobulins
3.6. Influence of Hesperidin on Serum IgG, IgM and IgA Concentrations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Romier, B.; Schneider, Y.J.; Larondelle, Y.; During, A. Dietary polyphenols can modulate the intestinal inflammatory response. Nutr. Rev. 2009, 67, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Kawser Hossain, M.; Abdal Dayem, A.; Han, J.; Yin, Y.; Kim, K.; Kumar Saha, S.; Yang, G.-M.; Choi, H.; Cho, S.-G. Molecular mechanisms of the anti-obesity and anti-diabetic properties of flavonoids. Int. J. Mol. Sci. 2016, 17, 569. [Google Scholar] [CrossRef] [PubMed]
- González-Gallego, J.; García-Mediavilla, M.V.; Sánchez-Campos, S.; Tuñón, M.J. Fruit polyphenols, immunity and inflammation. Br. J. Nutr. 2010, 104, S15–S27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williamson, G. The role of polyphenols in modern nutrition. Nutr. Bull. 2017, 42, 226–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mkhize, N.V.P.; Qulu, L.; Mabandla, M.V. The effect of quercetin on pro- and anti-inflammatory cytokines in a prenatally stressed rat model of febrile seizures. J. Exp. Neurosci. 2017, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ota, A.; Ulrih, N.P. An overview of herbal products and secondary metabolites used for management of type two diabetes. Front. Pharmacol. 2017, 8, 436. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cano, F.J.; Massot-Cladera, M.; Rodríguez-Lagunas, M.; Castell, M. Flavonoids affect host-microbiota crosstalk through TLR modulation. Antioxidants 2014, 3, 649–670. [Google Scholar] [CrossRef] [PubMed]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: An updated review of their molecular mechanisms and experimental models. Phyther. Res. 2015, 29, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, F.; Larrosa, M.; Escudero, E.; Zafrilla, M.P.; Cerdá, B.; Boza, J.; García-Conesa, M.T.; Espín, J.C.; Tomás-Barberán, F.A. Concentration and solubility of flavanones in orange beverages affect their bioavailability in humans. J. Agric. Food Chem. 2010, 58, 6516–6524. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Konishi, Y. Transepithelial transport of flavanone in intestinal Caco-2 cell monolayers. Biochem. Biophys. Res. Commun. 2008, 368, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Garg, S.; Zaneveld, L.J.; Singla, A.K. Chemistry and pharmacology of the citrus bioflavonoid hesperidin. Phyther. Res. 2001, 15, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Rupasinghe, H.P.V. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxid. Med. Cell. Longev. 2013, 2013, 891748. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.S.; Khan, M.M.; Ahmad, A.; Ashafaq, M.; Khuwaja, G.; Tabassum, R.; Javed, H.; Siddiqui, M.S.; Safhi, M.M.; Islam, F. Hesperidin ameliorates functional and histological outcome and reduces neuroinflammation in experimental stroke. Brain Res. 2011, 1420, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Tanaka, T.; Tanaka, M.; Kuno, T. Cancer chemoprevention by citrus pulp and juices containing high amounts of β-cryptoxanthin and hesperidin. J. Biomed. Biotechnol. 2012, 2012, 516981. [Google Scholar] [CrossRef] [PubMed]
- Bentli, R.; Ciftci, O.; Cetin, A.; Unlu, M.; Basak, N.; Çay, M. Oral administration of hesperidin, a citrus flavonone, in rats counteracts the oxidative stress, the inflammatory cytokine production, and the hepatotoxicity induced by the ingestion of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). Eur. Cytokine Netw. 2013, 24, 91–96. [Google Scholar]
- Antunes, M.S.; Jesse, C.R.; Ruff, J.R.; de Oliveira Espinosa, D.; Gomes, N.S.; Altvater, E.E.T.; Donato, F.; Giacomeli, R.; Boeira, S.P. Hesperidin reverses cognitive and depressive disturbances induced by olfactory bulbectomy in mice by modulating hippocampal neurotrophins and cytokine levels and acetylcholinesterase activity. Eur. J. Pharmacol. 2016, 789, 411–420. [Google Scholar] [CrossRef]
- Ahmadi, A.; Shadboorestan, A. Oxidative stress and cancer; the role of hesperidin, a citrus natural bioflavonoid, as a cancer chemoprotective agent. Nutr. Cancer 2015, 68, 29–39. [Google Scholar] [CrossRef]
- Allam, G.; Abuelsaad, A.S.A. Differential effect of hesperidin on Th1, Th2, Th17, and proinflammatory cytokines production from splenocyte of Schistosoma mansoni-infected mice. Cent. Eur. J. Immunol. 2013, 38, 29–36. [Google Scholar] [CrossRef]
- Wei, D.; Ci, X.; Chu, X.; Wei, M.; Hua, S.; Deng, X. Hesperidin suppresses ovalbumin-induced airway inflammation in a mouse allergic asthma model. Inflammation 2012, 35, 114–121. [Google Scholar] [CrossRef]
- Kim, S.-H.; Kim, B.-K.; Lee, Y.-C. Antiasthmatic effects of hesperidin, a potential Th2 cytokine antagonist, in a mouse model of allergic asthma. Mediators Inflamm. 2011, 2011, 485402. [Google Scholar] [CrossRef] [PubMed]
- Abuelsaad, A.S.A.; Allam, G.; Al-Solumani, A.A.A. Hesperidin inhibits inflammatory response induced by Aeromonas hydrophila infection and alters CD4+/CD8+ T cell ratio. Mediators Inflamm. 2014, 2014, 393217. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Influence of hesperidin on the systemic and intestinal rat immune response. Nutrients 2017, 9, 580. [Google Scholar] [CrossRef] [PubMed]
- Unno, T.; Hisada, T.; Takahashi, S. Hesperetin modifies the composition of fecal microbiota and increases cecal levels of short-chain fatty acids in rats. J. Agric. Food Chem. 2015, 63, 7952–7957. [Google Scholar] [CrossRef] [PubMed]
- Cianci, R.; Pagliari, D.; Piccirillo, C.A.; Fritz, J.H.; Gambassi, G. The microbiota and immune system crosstalk in health and disease. Mediators Inflamm. 2018, 2018, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Forchielli, M.L.; Walker, W.A. The role of gut-associated lymphoid tissues and mucosal defence. Br. J. Nutr. 2005, 93, S41. [Google Scholar] [CrossRef] [PubMed]
- Ramiro-Puig, E.; Pérez-Cano, F.J.; Ramos-Romero, S.; Pérez-Berezo, T.; Castellote, C.; Permanyer, J.; Franch, A.; Izquierdo-Pulido, M.; Castell, M. Intestinal immune system of young rats influenced by cocoa-enriched diet. J. Nutr. Biochem. 2008, 19, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Abril-Gil, M.; Saldaña-Ruiz, S.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Cocoa diet prevents antibody synthesis and modifies lymph node composition and functionality in a rat oral sensitization model. Nutrients 2016, 8, 242. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Camps-Bossacoma, M.; Massot-Cladera, M.; Rigo-Adrover, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Effect of cocoa’s theobromine on intestinal microbiota of rats. Mol. Nutr. Food Res. 2017, 201700238, 1700238. [Google Scholar] [CrossRef]
- Massot-Cladera, M.; Abril-Gil, M.; Torres, S.; Franch, À.; Castell, M.; Pérez-Cano, F.J. Impact of cocoa polyphenol extracts on the immune system and microbiota in two strains of young rats. Br. J. Nutr. 2014, 112, 1944–1954. [Google Scholar] [CrossRef]
- Massot-Cladera, M.; Pérez-Berezo, T.; Franch, A.; Castell, M.; Pérez-Cano, F.J. Cocoa modulatory effect on rat faecal microbiota and colonic crosstalk. Arch. Biochem. Biophys. 2012, 527, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Lay, C.; Sutren, M.; Rochet, V.; Saunier, K.; Doré, J.; Rigottier-Gois, L. Design and validation of 16S rRNA probes to enumerate members of the Clostridium leptum subgroup in human faecal microbiota. Environ. Microbiol. 2005, 7, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, H.J.M.; Gibson, G.R.; Elfferich, P.; Raangs, G.C.; Wildeboer-Veloo, A.C.; Argaiz, A.; Roberfroid, M.B.; Welling, G.W. Comparison of viable cell counts and fluorescence in situ hybridization using specific rRNA-based probes for the quantification of human fecal bacteria. FEMS Microbiol. Lett. 2000, 183, 125–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harmsen, H.J.M.; Elfferich, P.; Schut, F.; Welling, G.W. A 16S rRNA-targeted probe for detection of lactobacilli and enterococci in faecal samples by fluorescent in situ hybridization. Microb. Ecol. Health Dis. 1999, 11, 3–12. [Google Scholar]
- Trebesius, K.; Leitritz, L.; Adler, K.; Schubert, S.; Autenrieth, I.B.; Heesemann, J. Culture independent and rapid identification of bacterial pathogens in necrotising fasciitis and streptococcal toxic shock syndrome by fluorescence in situ hybridisation. Med. Microbiol. Immunol. 2000, 188, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Manz, W.; Amann, R.; Ludwig, W.; Vancanneyt, M.; Schleifer, K.H. Application of a suite of 16S rRNA-specific oligonucleotide probes designed to investigate bacteria of the phylum cytophaga-flavobacter-bacteroides in the natural environment. Microbiology 1996, 142, 1097–1106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langendijk, P.S.; Schut, F.; Jansen, G.J.; Raangs, G.C.; Kamphuis, G.R.; Wilkinson, M.H.; Welling, G.W. Quantitative fluorescence in situ hybridization of Bifidobacterium spp. with genus-specific 16S rRNA-targeted probes and its application in fecal samples. Appl. Environ. Microbiol. 1995, 61, 3069–3075. [Google Scholar] [PubMed]
- Poulsen, L.K.; Lan, F.; Kristensen, C.S.; Hobolth, P.; Molin, S.; Krogfelt, K.A. Spatial distribution of Escherichia coli in the mouse large intestine inferred from rRNA in situ hybridization. Infect. Immun. 1994, 62, 5191–5194. [Google Scholar]
- Grases-Pintó, B.; Abril-Gil, M.; Rodríguez-Lagunas, M.J.; Castell, M.; Pérez-Cano, F.J.; Franch, À. Leptin and adiponectin supplementation modifies mesenteric lymph node lymphocyte composition and functionality in suckling rats. Br. J. Nutr. 2018, 119, 486–495. [Google Scholar] [CrossRef]
- Camps-Bossacoma, M.; Abril-Gil, M.; Franch, A.; Perez-Cano, F.; Castell, M. Induction of an oral sensitization model in rats. Clin. Immunol. Endocr. Metab. Drugs 2014, 1, 89–101. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef] [PubMed]
- Perche, O.; Vergnaud-Gauduchon, J.; Morand, C.; Dubray, C.; Mazur, A.; Vasson, M.P. Orange juice and its major polyphenol hesperidin consumption do not induce immunomodulation in healthy well-nourished humans. Clin. Nutr. 2014, 33, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Alard, J.; Peucelle, V.; Boutillier, D.; Breton, J.; Kuylle, S.; Pot, B.; Holowacz, S.; Grangette, C. New probiotic strains for inflammatory bowel disease management identified by combining in vitro and in vivo approaches. Benef. Microbes 2018, 9, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Tankou, S.K.; Regev, K.; Healy, B.C.; Cox, L.M.; Tjon, E.; Kivisakk, P.; Vanande, I.P.; Cook, S.; Gandhi, R.; Glanz, B.; et al. Investigation of probiotics in multiple sclerosis. Mult. Scler. J. 2018, 24, 58–63. [Google Scholar] [CrossRef] [PubMed]
- An, H.M.; Lee, D.K.; Kim, J.R.; Lee, S.W.; Cha, M.K.; Lee, K.O.; Ha, N.J. Antiviral activity of Bifidobacterium adolescentis SPM 0214 against herpes simplex virus type 1. Arch. Pharm. Res. 2012, 35, 1665–1671. [Google Scholar] [CrossRef] [PubMed]
- Sabico, S.; Al-Mashharawi, A.; Al-Daghri, N.M.; Yakout, S.; Alnaami, A.M.; Alokail, M.S.; McTernan, P.G. Effects of a multi-strain probiotic supplement for 12 weeks in circulating endotoxin levels and cardiometabolic profiles of medication naïve T2DM patients: A randomized clinical trial. J. Transl. Med. 2017, 15, 249. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Fernández-Quirós, B.; Ludwig, I.A.; Pradas, I.; Crozier, A.; Moreno-Rojas, J.M. Catabolism of citrus flavanones by the probiotics Bifidobacterium longum and Lactobacillus rhamnosus. Eur. J. Nutr. 2018, 57, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar] [PubMed]
- Daly, K.; Darby, A.C.; Hall, N.; Nau, A.; Bravo, D.; Shirazi-Beechey, S.P. Dietary supplementation with lactose or artificial sweetener enhances swine gut Lactobacillus population abundance. Br. J. Nutr. 2014, 111, S30–S35. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimiä, R.; Nohynek, L.; Hartmann-Schmidlin, S.; Kähkönen, M.; Heinonen, M.; Määttä-Riihinen, K.; Oksman-Caldentey, K.M. Berry phenolics selectively inhibit the growth of intestinal pathogens. J. Appl. Microbiol. 2005, 98, 991–1000. [Google Scholar] [CrossRef]
- Camps-Bossacoma, M.; Pérez-Cano, F.J.; Franch, À.; Castell, M. Gut microbiota in a rat oral sensitization model: Effect of a cocoa-enriched diet. Oxid. Med. Cell. Longev. 2017, 2017, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Massot-Cladera, M.; Costabile, A.; Childs, C.E.; Yaqoob, P.; Franch, À.; Castell, M.; Pérez-Cano, F.J. Prebiotic effects of cocoa fibre on rats. J. Funct. Foods 2015, 19, 341–352. [Google Scholar] [CrossRef] [Green Version]
- Yeom, M.; Sur, B.J.; Park, J.; Cho, S.G.; Lee, B.; Kim, S.T.; Kim, K.S.; Lee, H.; Hahm, D.H. Oral administration of Lactobacillus casei variety rhamnosus partially alleviates TMA-induced atopic dermatitis in mice through improving intestinal microbiota. J. Appl. Microbiol. 2015, 119, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, A.J.; Yilmaz, B.; Limenitakis, J.P.; Ganal-Vonarburg, S.C. IgA function in relation to the intestinal microbiota. Annu. Rev. Immunol. 2018, 36, 359–381. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y.; Han, Y.; Kayahara, M.; Watanabe, T.; Arishige, H.; Kato, N. Consumption of curcumin elevates fecal immunoglobulin A, an index of intestinal immune function, in rats fed a high-fat diet. J. Nutr. Sci. Vitaminol. (Tokyo) 2010, 56, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Taira, T.; Yamaguchi, S.; Takahashi, A.; Okazaki, Y.; Yamaguchi, A.; Sakaguchi, H.; Chiji, H. Dietary polyphenols increase fecal mucin and immunoglobulin A and ameliorate the disturbance in gut microbiota caused by a high fat diet. J. Clin. Biochem. Nutr. 2015, 57, 212–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, S.; Liu, W.; Bi, J.; Liu, S.; Zhao, H.; Gong, N.; Xing, D. Anti-inflammatory effect of hesperidin enhances chondrogenesis of human mesenchymal stem cells for cartilage tissue repair. J. Inflamm. 2018, 15, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.H. Anti-inflammatory effects and its mechanisms of hesperidin in an asthmatic mouse model induced by ovalbumin. J. Exp. Biomed. Sci. 2010, 16, 83–90. [Google Scholar]






| Phylum | Bacterial Group Targeted | Sequence (5′–3′) | Hybridization Temperature (°C) | Reference |
|---|---|---|---|---|
| Firmicutes | Clostridium coccoides/Eubacterium rectale | GCTTCTTAGTCARGRACCG | 50 | [32] |
| Clostridium histolyticum/C. perfringens | TTATGCGGTATTAATCT(C/T)CCTTT | 50 | [33] | |
| Lactobacillus/Enterococcus | GGTATTAGCAYCTGTTTCCA | 50 (lysozyme) | [34] | |
| Staphylococcus spp. | TCCTCCATATCTCTGCGC | 46 | [35] | |
| Streptococcus spp. | CACTCTCCCCTTCTGCAC | 46 | [35] | |
| Bacteroidetes | Bacteroides/Prevotella | CCAATGTGGGGGACCTT | 46 | [36] |
| Actinobacteria | Bifidobacterium spp. | CATCCGGCATTACCACCC | 50 | [37] |
| Proteobacteria | Escherichia coli | CACCGTAGTGCCTCGTCATCA | 37 | [38] |
| Day | Body Weight (g) | Food Intake (g/100 g rat/day) | Water Intake (g/100 g rat/day) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| REF | H100 | H200 | REF | H100 | H200 | REF | H100 | H200 | |
| 0 | 48.8 ± 1.6 | 48.5 ± 0.8 | 48.4 ± 0.9 | - | - | - | - | - | - |
| 7 | 77.2 ± 2.6 | 75.3 ± 1.4 | 74.8 ± 1.4 | 14.3 ± 0.5 | 14.7 ± 0.1 | 14.7 ± 0.2 | 10.8 ± 0.1 | 11.1 ± 0.2 | 13.8 ± 2.5 |
| 14 | 117.0 ± 3.8 | 117.6 ± 1.9 | 119.2 ± 2.2 | 11.80± 2.4 | 9.2 ± 0.0 | 10.0 ± 0.8 | 10.9 ± 0.0 | 12.0 ± 0.2 | 11.6 ± 0.3 |
| 21 | 158.0 ± 3.8 | 162.6 ± 1.8 | 161.1 ± 3.0 | 12.6 ± 0.2 | 12.7 ± 0.4 | 12.3 ± 0.2 | 10.1 ± 0.3 | 11.6 ± 0.3 | 10.7 ± 0.1 |
| 28 | 190.5 ± 4.7 | 189.9 ± 1.9 | 189.9 ± 3.3 | 9.8 ± 0.3 | 9.8 ± 0.0 | 9.4 ± 0.2 | 11.5 ± 0.0 | 11.7 ± 0.2 | 11.0 ± 0.8 |
| Gene | Relative Gene Expression (%) | ||
|---|---|---|---|
| REF | H100 | H200 | |
| IgA | 100.00 ± 13.24 | 148.46 ± 82.70 | 151.91 ± 37.14 |
| TGF-β | 100.00 ± 11.88 | 63.00 ± 14.39 | 85.96 ± 23.61 |
| CCR9 | 100.00 ± 12.94 | 69.05 ± 14.85 | 113.66 ± 31.96 |
| CD40 | 100.00 ± 36.80 | 82.97 ± 30.91 | 62.36 ± 12.78 |
| CCL25 | 100.00 ± 13.80 | 66.57 ± 22.26 | 102.75 ± 17.79 |
| CCL28 | 100.00 ± 15.62 | 104.00 ± 14.08 | 100.00 ± 14.14 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estruel-Amades, S.; Massot-Cladera, M.; Pérez-Cano, F.J.; Franch, À.; Castell, M.; Camps-Bossacoma, M. Hesperidin Effects on Gut Microbiota and Gut-Associated Lymphoid Tissue in Healthy Rats. Nutrients 2019, 11, 324. https://doi.org/10.3390/nu11020324
Estruel-Amades S, Massot-Cladera M, Pérez-Cano FJ, Franch À, Castell M, Camps-Bossacoma M. Hesperidin Effects on Gut Microbiota and Gut-Associated Lymphoid Tissue in Healthy Rats. Nutrients. 2019; 11(2):324. https://doi.org/10.3390/nu11020324
Chicago/Turabian StyleEstruel-Amades, Sheila, Malén Massot-Cladera, Francisco J. Pérez-Cano, Àngels Franch, Margarida Castell, and Mariona Camps-Bossacoma. 2019. "Hesperidin Effects on Gut Microbiota and Gut-Associated Lymphoid Tissue in Healthy Rats" Nutrients 11, no. 2: 324. https://doi.org/10.3390/nu11020324







