1. Introduction
Metabolic syndrome is widely known as a multi-factorial disorder with symptoms such as hypertension, hyperglycemia, insulin resistance, and visceral fat obesity. This syndrome is promoted by irregular eating, lack of exercise, excessive drinking of alcohol and stress, which may cause risk factors such as cardiovascular disease, coronary disease, and type 2 diabetes [
1]. In particular, quality of diet is closely associated with the development of metabolic syndrome. In recent years, the beneficial effects of whole-grains have become apparent: a meta-analysis of cohort studies showed that a high consumption of whole-grains is associated with reduced incidence of type 2 diabetes [
2]. Another report showed that whole-grain consumption (48–80 g/3–5 serving/day) reduced the incidence of obesity, type 2 diabetes, and concentrations of total and LDL-cholesterol in serum [
3]. A systematic review also reported that intake of a whole-grain and fiber diet reduced the risk of type 2 diabetes, the incidence of being overweight and obesity in Japanese men and women [
4]. Whole-grain and grains are good sources of fiber, and the effect of dietary fiber on lipid metabolism has been studied over a long period [
5,
6,
7].
Barley, a cereal grain, contains higher amounts of β-glucan compared with other grains such as oats, rye, and wheat. Since barley β-glucan is distributed throughout the kernel, the amount of β-glucan in refined barley is unchanged. Barley β-glucan is a polysaccharide polymer in which glucose is linked by β-glycoside bonds (β-1,3–1,4), and is almost soluble in water [
8]. β-Glucan increases water solubilization in the stomach and small intestine and delays the absorption of other nutrients [
9]. These effects suppress increases in postprandial blood glucose, and maintains satiety, thereby improving insulin resistance and suppressing visceral fat accumulation by reduced excessive insulin secretion [
10]. It has also been reported that blood cholesterol levels were reduced by barley intake in vivo and human studies [
11,
12,
13]. Studies have indicated that the mechanism of barley β-glucan involves inhibition of cholesterol synthesis in the liver, inhibition of bile acid reabsorption and alteration of cholesterol metabolism due to decreased insulin secretion and promotion of cholesterol excretion [
14,
15].
Water-soluble dietary fibers such as barley β-glucan are fermented by bacteria in the cecum to the distal colon. Fermentation produces short-chain fatty acids (SCFAs) such as acetic acid, propionic acid, and butyric acid as metabolites which serve as energy sources for the host. It was recently reported that SCFAs affect lipid metabolism in several organs such as liver, muscle, and brown adipose tissue [
16]. Acetic acid is mainly metabolized in the liver and is involved in fat and cholesterol synthesis in peripheral tissues [
17]. Propionic acid is mainly metabolized in the liver and intestinal tract, and butyric acid is involved in cell differentiation and maintenance of intestinal barrier formation in the intestinal tract [
18]. These SCFAs also contribute to reducing intestinal pH, which inhibits the absorption of toxic products such as
p-cresol and phenols [
19]. Moreover, recent studies have indicated the SCFAs act in the host metabolism as signal molecules, regulating intestinal hormone secretion and insulin signaling via SCFA receptors [
20].
The findings of the above studies support the beneficial effects of barley β-glucan in normalizing cholesterol concentration, improving glucose tolerance, and reducing visceral fat. These effects are considered to be related to the physiological functions of delaying digestion and absorption of nutrients in the diet from the digestive tract, and physiological functions of host energy by gut microbiota-derived SCFAs. However, the mechanism of barley β-glucan affecting lipid metabolism is still not understood at the gene level. There are very few reports which confirm the effects of barley β-glucan on lipid metabolism in several organs, such as intestinal tract, adipose, and liver tissues; therefore, it is still debatable whether the intake of β-glucan-rich barley flour is involved in lipid metabolism at the gene level.
To investigate the effect of β-glucan-rich barley on glucose and lipid metabolism, we measured gene expression levels using DNA microarrays, which are a tool to comprehensively detect total gene expression. In recent years, several studies have clarified the physiological effects of dietary fiber materials using DNA microarrays. Drew et al. reported that genes involved in the intestinal barrier function and metabolism of the intestinal epithelium were upregulated in the cecum of mice fed inulin or β-glucan extract of barley [
21]. Other studies have reported that the expression of genes involved in fatty acid oxidation and lipid transport in skeletal muscle are upregulated in mice fed psyllium [
22]. The purpose of this study was to investigate whether the intake of β-glucan-rich barley flour affects the expression levels of genes related to glucose and lipid metabolism in the ileum, liver, and adipose tissues of mice fed a high-fat diet using a DNA microarray and subsequent analysis by q-PCR. We also investigated the relationship between gene expression levels in each organ and SCFAs in the cecum.
4. Discussion
We investigated whether β-glucan-rich barley flour affected glucose and lipid metabolism at the gene expression level in high-fat obesity model mice by comparing them with a control group. Firstly, we performed OGTTs and found that barley flour intake reduced postprandial blood glucose levels in BF mice compared with the control mice. Barley β-glucan is known to moderate the digestion and absorption of carbohydrates in the intestine, which results in suppressing excessive elevation of postprandial blood glucose. Previous studies have shown that long-term barley intake suppressed elevated blood glucose levels in mice [
29]. Brockman et al. reported that consumption of high concentrations of barley β-glucan improved glucose control during glucose tolerance tests [
30]. Our results were consistent with these previous studies and suggested that barley intake improves glucose tolerance. Moreover, this effect may be regulated by SCFAs. A previous study speculated that barley suppresses food intake and improves insulin sensitivity through SCFAs, which promote intestinal hormone secretion, such as glucagon-like peptide 1 (GLP-1) from intestinal enteroendocrine cells [
31]. GLP-1 has the ability to decrease blood glucose levels by modulating glucose-dependent insulin secretion [
32]. We confirmed that barley intake increased SCFAs in the cecum in this study (
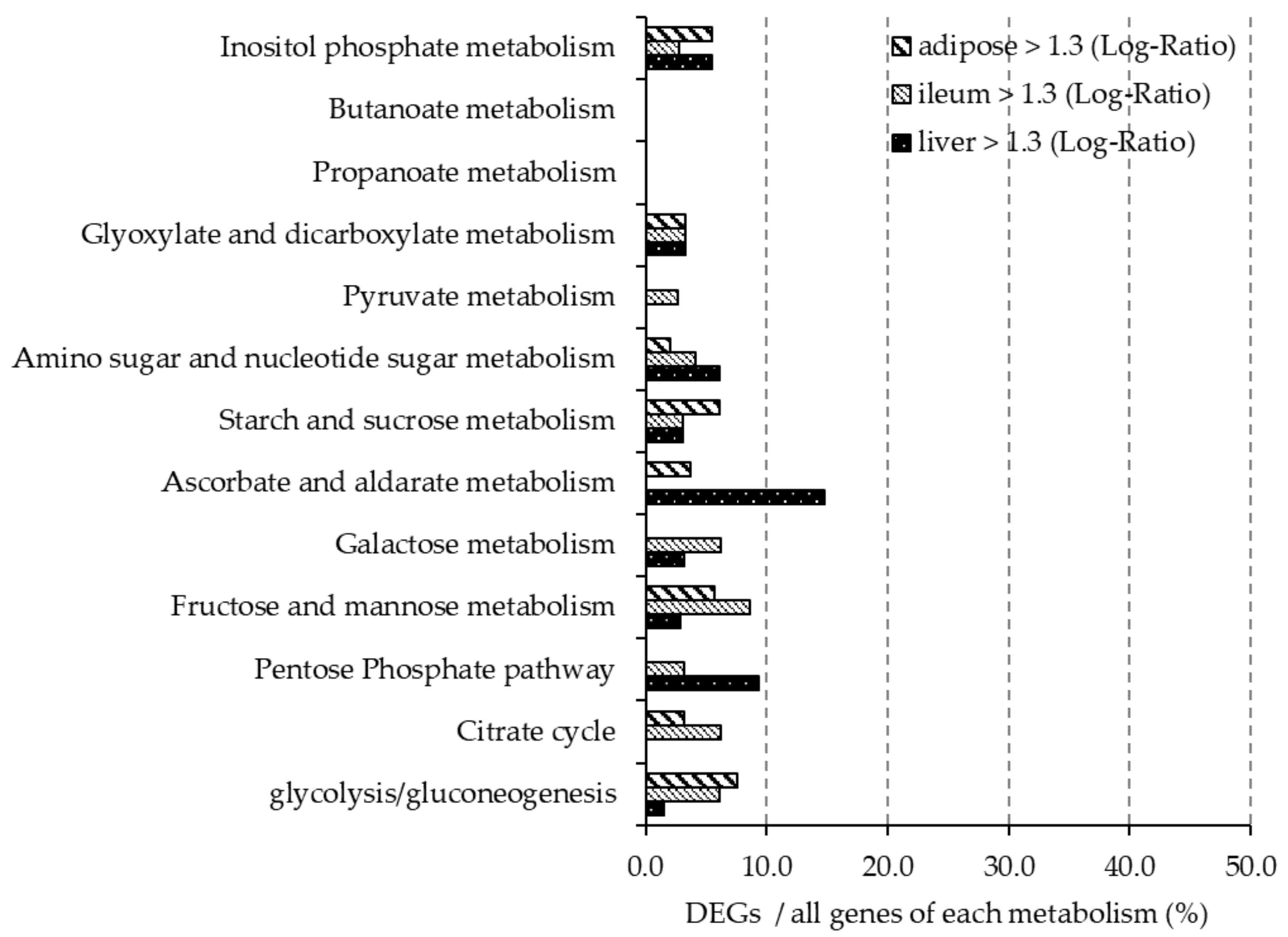
Table 4); however, further studies are needed to elucidate the effect of gut hormone secretion on glucose metabolism. DNA microarray analysis showed that there was only a slight change in the expression level of genes related to glucose metabolism in ileum, liver and adipose tissues after barley intake. Therefore, we speculated that barley flour intake reduced postprandial blood glucose levels and was mainly influenced by retardation of glucose absorption and secretion of intestinal hormones, but not through changes in the expression of genes related to glucose metabolism in ileum, liver, and adipose tissues.
Intake of a diet rich in β-glucan barley improved lipid metabolism in mice. DNA microarray analysis showed that the expression of 50% of the genes related to steroid biosynthesis (mmu00100) pathways were greatly downregulated in the BF group compared with the control group in liver and adipose tissues. In particular, the mRNA expression level of HMG-CoA reductase, the rate-limiting enzyme in cholesterol synthesis, was lower in the BF group compared with the control group in the liver. Xia et al. also reported that intake of whole-grain significantly lowered the expression of HMG-CoA reductase in the liver and plasma and liver lipid levels when compared with the no barley group in high-fat diet model rats [
33]. These reports are similar to the results obtained in our study. We found that intake of barley flour lowered liver and serum TC concentrations by consistently reducing the expression of genes involved in cholesterol synthesis. Moreover, it was previously suggested that improvements in lipid metabolism after barley intake is due to the promotion of cholesterol excretion in the intestine and inhibition of bile acid re-absorption. It is known that CYP7a1 is the rate-limiting enzyme for bile acid synthesis in the liver. A previous study reported that intake of barley β-glucan and waxy barley promotes excretion of bile acids in the feces; concomitantly increasing CYP7a1 expression and bile acid synthesis, thereby suppressing cholesterol synthesis [
34]. However, in our study, the mRNA expression levels of CYP7a1 were significantly lower in the BF group compared with the control group. We also found that the fecal lipid levels did not change between the experimental and control groups (data not shown). The differing results may be due to differences in the source of barley, the experimental period and/or the animal model. Further studies are needed to elucidate how changes in the intake of barley affects bile acid metabolism.
The metabolic syndrome indices (abdominal fat organs, liver lipids, serum TC and LDL) is improved in mice fed barley flour: we previously reported that barley intake reduced fat accumulation in mice [
35] and Choi et al. reported that consumption of barley β-glucan extract is involved in preventing obesity in mice fed a high-fat diet [
36]. Another study reported that barley intake has an improving effect, both histologically and biochemically, on the liver of diabetic-model rats [
37]. Similar effects have been shown in human studies; Japanese patients with hyperlipidemia had decreased abdominal fat and serum LDL after barley intake [
38] and abdominal fat was decreased by whole-grain cereal intake in healthy humans [
39]. Our results support previous studies. A DNA microarray showed that more than 10% of genes involved in glycerolipid metabolism (mmu00561) and the fatty acid elongation pathway (mmu00062) were downregulated in the BF group compared with the control group in the liver. q-PCR analyses also showed the expression of genes involved in lipid synthesis, such as DGAT1, SCD-1, FAS, and degradation, ACOX, CPT-1, were lowered after β-glucan-rich barley flour intake. The data suggest that gene expression levels related to lipid metabolism were lower overall in the liver, and affected an improvement in the obesity index.
Intake of β-glucan-rich barley flour affected the expression levels of mRNA related to lipid metabolism in adipocytes in a different way to the control group. Adipocytes are not only energy storage organs but also secrete some adipocytokines to regulate energy metabolism. In the present study, intake of barley flour downregulated genes related to several lipid metabolism pathways, such as steroid biosynthesis (mmu00100), and glycerolipid metabolism (mmu00561), in adipose tissue. It is considered that lipids flowing into adipose tissue were lower in the BF group compared with the control group due to lower lipid synthesis in the liver. The mRNA expression levels of HSL were significantly higher in the BF group compared with the control group. It is known that HSL catalyzes the degradation of triacylglycerol and decreases in the expression of HSL are associated with insulin concentration and obesity [
40,
41]. In this study, serum insulin concentration was reduced by barley flour intake (BF: 3.9 ± 1.5 ng/mL, control 6.0 ± 3.3 ng/mL); therefore, it was suggested that increased HSL expression occurs by the lower concentration of insulin accelerating fat degradation. Positive correlations between several SCFAs and HSL expression in adipose tissue were detected; however, further studies are needed to evaluate the relationship between HSL and SCFAs.
The expression levels of genes related to glucose and lipid metabolism were unchanged in the ileum. A previous study using fructo-oligosaccharides reported that succinic acid induced intestinal gluconeogenesis in vivo [
42]; however, our study produced different results and this may be due to the differences in soluble fiber material. Therefore, our results suggest that the main organs in which barley flour is involved in lipid metabolism at the gene expression level are the liver and adipose tissues, but not the ileum.
Some inflammatory markers (MCP-1, p67
phox, F4/80) were significantly decreased in the adipose tissue of barley-supplemented mice. It has been reported that adipocytes induce early inflammation by producing active oxygen via activation of NADPH oxidase in mice on a high-fat diet [
43]. These results suggest that a decrease in adipocyte hypertrophy reduces fat synthesis in the liver, thus lowering visceral fat weight and inflammation in adipocytes.
Previous studies have described SCFAs as energy sources for the intestine and liver [
44,
45]. We found that several SCFAs negatively correlated with the expression levels of mRNA involved in hepatic lipid synthesis and degradation. Our results are supported by previous studies in which SCFAs suppressed lipid synthesis in the liver [
46] and the expression levels of FAS and carbohydrate-response element-binding protein (Chrebp) were lower in mice fed SCFAs compared with mice fed cellulose [
47]. Based on these results, we suggest that SCFAs alter the expression of genes related to lipid metabolism in the cecum. A recent study revealed that SCFAs influence the metabolism of lipids and glucose to regulate GLP-1 and transcription factor activity involved in lipid metabolism via intestinal SCFA receptors [
48]. It is known that G protein-coupled receptors (GPCRs) are activated by SCFAs; notably, GPR43 and GPR41 regulate gut hormone secretion [
49]. Further studies using mice deficient in these SCFA receptors will clarify the relationship between barley intake, SCFAs and the expression of genes related to lipid metabolism.