Relationships between Neonatal Nutrition and Growth to 36 Weeks’ Corrected Age in ELBW Babies–Secondary Cohort Analysis from the Provide Trial
Abstract
:1. Introduction
- Intakes of macronutrients in the first 4 weeks after birth are associated with changes in weight, length and head circumference z scores
- Relationships between nutritional intakes and growth are not different for girls and boys
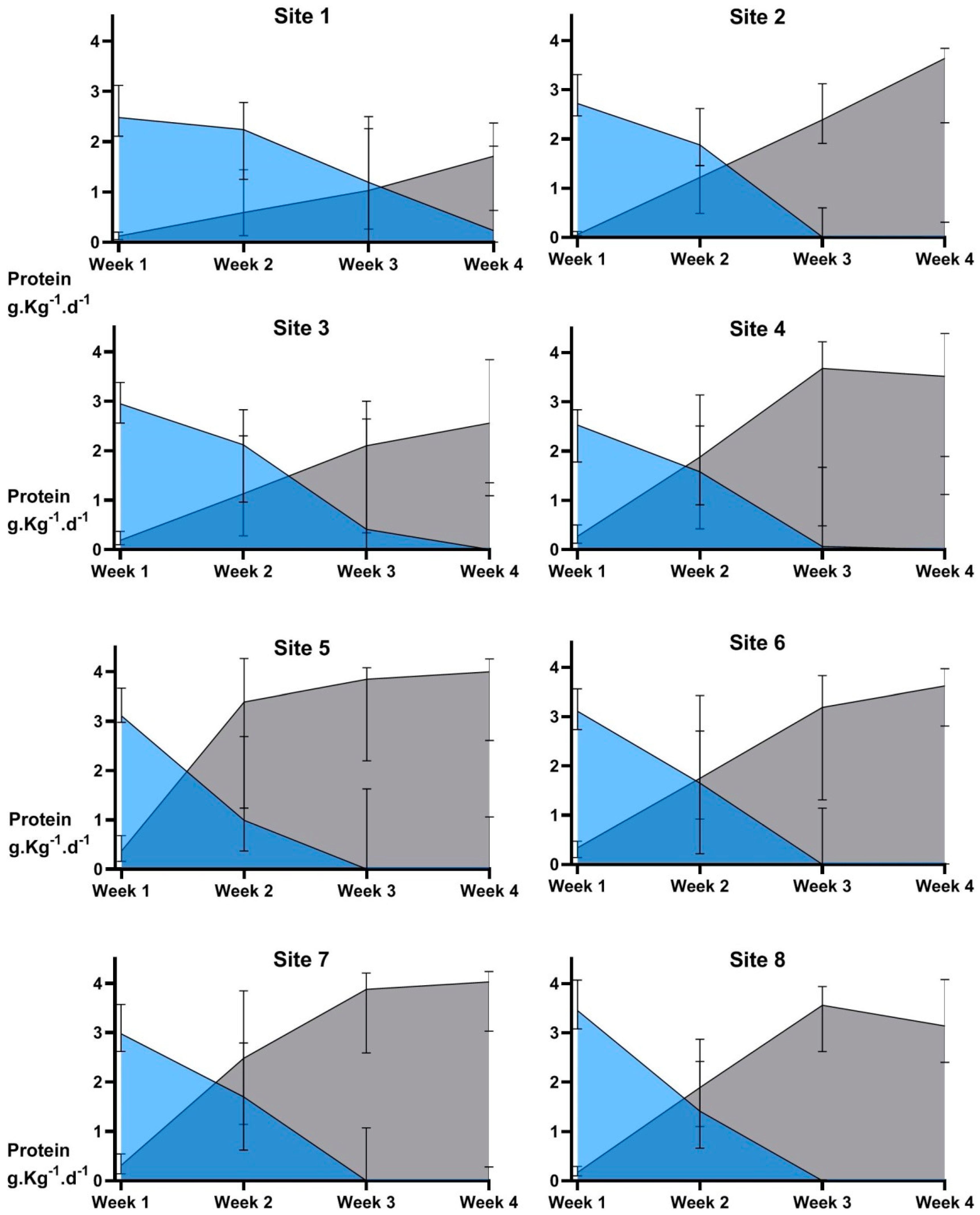
- Growth outcomes differ across hospital sites in accordance with nutrition practices
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.3. Statistical Analysis
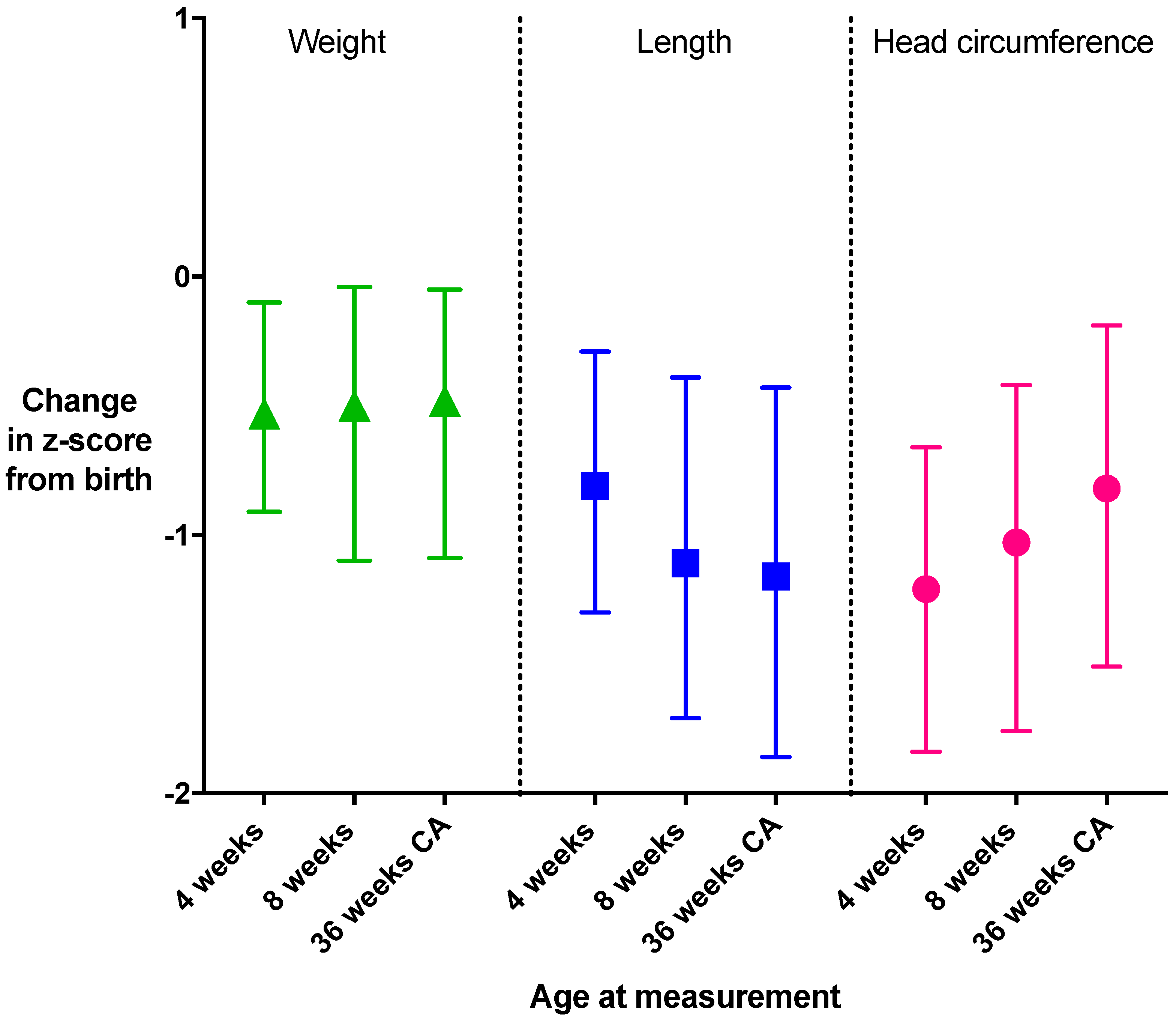
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ziegler, E.E.; O’Donnell, A.M.; Nelson, S.E.; Fomon, S.J. Body composition of the reference fetus. Growth 1976, 40, 329–341. [Google Scholar]
- Agostoni, C.; Buonocore, G.; Carnielli, V.P.; De Curtis, M.; Darmaun, D.; Decsi, T.; Domellof, M.; Embleton, N.D.; Fusch, C.; Genzel-Boroviczeny, O.; et al. Enteral nutrient supply for preterm infants: Commentary from the European Society of Paediatric Gastroenterology, Hepatology and Nutrition Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2010, 50, 85–91. [Google Scholar] [CrossRef]
- Joosten, K.; Embleton, N.; Yan, W.; Senterre, T. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Energy. Clin. Nutr. 2018, 37, 2309–2314. [Google Scholar] [CrossRef]
- van Goudoever, J.B.; Carnielli, V.; Darmaun, D.; Sainz de Pipaon, M.; ESPGHAN ESPEN ESPR CSPEN Working Group on Pediatric Parenteral Nutrition. Guidelines on pediatric parenteral nutrition: Amino acids. Clin. Nutr. 2018, 37, 2315–2323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horbar, J.D.; Ehrenkranz, R.A.; Badger, G.J.; Edwards, E.M.; Morrow, K.A.; Soll, R.F.; Buzas, J.S.; Bertino, E.; Gagliardi, L.; Bellù, R. Weight growth velocity and postnatal growth failure in infants 501 to 1500 g: 2000–2013. Pediatrics 2015, 136, e84–e92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clouchoux, C.; Guizard, N.; Evans, A.C.; du Plessis, A.J.; Limperopoulos, C. Normative fetal brain growth by quantitative in vivo magnetic resonance imaging. Am. J. Obstet. Gynecol. 2012, 206, 173.e1–173.e8. [Google Scholar] [CrossRef] [PubMed]
- Volpe, J.J. Brain injury in premature infants: A complex amalgam of destructive and developmental disturbances. Lancet Neurol. 2009, 8, 110–124. [Google Scholar] [CrossRef] [Green Version]
- Adams-Chapman, I.; Heyne, R.J.; DeMauro, S.B.; Duncan, A.F.; Hintz, S.R.; Pappas, A.; Vohr, B.R.; McDonald, S.A.; Das, A.; Newman, J.E.; et al. Neurodevelopmental impairment among extremely preterm infants in the neonatal research network. Pediatrics 2018, 141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- American Academy of Pediatrics; Committee on Nutrition. Nutritional needs of low-birth-weight infants. Pediatrics 1977, 60, 519–530. [Google Scholar]
- Osborn, D.A.; Schindler, T.; Jones, L.J.; Sinn, J.K.; Bolisetty, S. Higher versus lower amino acid intake in parenteral nutrition for newborn infants. Cochrane Database Syst. Rev. 2018, 3, CD005949. [Google Scholar] [CrossRef]
- Ottolini, K.M.; Andescavage, N.; Keller, S.; Limperopoulos, C. Nutrition and the developing brain: The road to optimizing early neurodevelopment: A systematic review. Pediatr. Res. 2020, 87, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Senterre, T.; Rigo, J. Reduction in postnatal cumulative nutritional deficit and improvement of growth in extremely preterm infants. Acta Paediatr. 2012, 101, e64–e70. [Google Scholar] [CrossRef] [PubMed]
- Cormack, B.E.; Bloomfield, F.H. Increased protein intake decreases postnatal growth faltering in ELBW babies. Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, F399–F404. [Google Scholar] [CrossRef] [PubMed]
- Olsen, I.E.; Harris, C.L.; Lawson, M.L.; Berseth, C.L. Higher protein intake improves length, not weight, z scores in preterm infants. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Andrews, E.T.; Ashton, J.J.; Pearson, F.; Beattie, R.M.; Johnson, M.J. Early postnatal growth failure in preterm infants is not inevitable. Arch. Dis. Child. Fetal Neonatal Ed. 2019, 104, F235–F241. [Google Scholar] [CrossRef]
- Cheong, J.L.; Hunt, R.W.; Anderson, P.J.; Howard, K.; Thompson, D.K.; Wang, H.X.; Bear, M.J.; Inder, T.E.; Doyle, L.W. Head growth in preterm infants: Correlation with magnetic resonance imaging and neurodevelopmental outcome. Pediatrics 2008, 121, e1534–e1540. [Google Scholar] [CrossRef]
- Kan, E.; Roberts, G.; Anderson, P.J.; Doyle, L.W.; Victorian Infant Collaborative Study Group. The association of growth impairment with neurodevelopmental outcome at eight years of age in very preterm children. Early Hum. Dev. 2008, 84, 409–416. [Google Scholar] [CrossRef]
- Leppanen, M.; Lapinleimu, H.; Lind, A.; Matomaki, J.; Lehtonen, L.; Haataja, L.; Rautava, P.; Pipari Study Group. Antenatal and postnatal growth and 5-year cognitive outcome in very preterm infants. Pediatrics 2014, 133, 63–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sammallahti, S.; Pyhala, R.; Lahti, M.; Lahti, J.; Pesonen, A.K.; Heinonen, K.; Hovi, P.; Eriksson, J.G.; Strang-Karlsson, S.; Andersson, S.; et al. Infant growth after preterm birth and neurocognitive abilities in young adulthood. J. Pediatr. 2014, 165, 1109–1115.e3. [Google Scholar] [CrossRef]
- Morgan, C.; McGowan, P.; Herwitker, S.; Hart, A.E.; Turner, M.A. Postnatal head growth in preterm infants: A randomized controlled parenteral nutrition study. Pediatrics 2014, 133, e120–e128. [Google Scholar] [CrossRef] [Green Version]
- Stephens, B.E.; Walden, R.V.; Gargus, R.A.; Tucker, R.; McKinley, L.; Mance, M.; Nye, J.; Vohr, B.R. First-week protein and energy intakes are associated with 18-month developmental outcomes in extremely low birth weight infants. Pediatrics 2009, 123, 1337–1343. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.J.; Cooke, R.W. Improving head growth in very preterm infants—A randomised controlled trial I: Neonatal outcomes. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F337–F341. [Google Scholar] [CrossRef] [PubMed]
- Stoltz Sjostrom, E.; Ohlund, I.; Ahlsson, F.; Engstrom, E.; Fellman, V.; Hellstrom, A.; Kallen, K.; Norman, M.; Olhager, E.; Serenius, F.; et al. Nutrient intakes independently affect growth in extremely preterm infants: Results from a population-based study. Acta Paediatr. 2013, 102, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.R.; Pohlandt, F.; Bode, H.; Mihatsch, W.A.; Sander, S.; Kron, M.; Steinmacher, J. Intrauterine, early neonatal, and postdischarge growth and neurodevelopmental outcome at 5.4 years in extremely preterm infants after intensive neonatal nutritional support. Pediatrics 2009, 123, e101–e109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isaacs, E.B.; Morley, R.; Lucas, A. Early diet and general cognitive outcome at adolescence in children born at or below 30 weeks gestation. J. Pediatr. 2009, 155, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Coviello, C.; Keunen, K.; Kersbergen, K.J.; Groenendaal, F.; Leemans, A.; Peels, B.; Isgum, I.; Viergever, M.A.; de Vries, L.S.; Buonocore, G.; et al. Effects of early nutrition and growth on brain volumes, white matter microstructure, and neurodevelopmental outcome in preterm newborns. Pediatr. Res. 2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leenders, E.; de Waard, M.; van Goudoever, J.B. Low- versus high-dose and early versus late parenteral amino-acid administration in very-low-birth-weight infants: A systematic review and meta-analysis. Neonatology 2018, 113, 187–205. [Google Scholar] [CrossRef]
- Bloomfield, F.H.; Crowther, C.A.; Harding, J.E.; Conlon, C.A.; Jiang, Y.; Cormack, B.E. The ProVIDe study: The impact of protein intravenous nutrition on development in extremely low birthweight babies. BMC Pediatr. 2015, 15, 100. [Google Scholar] [CrossRef]
- Cormack, B.E.; Embleton, N.D.; van Goudoever, J.B.; Hay, W.W., Jr.; Bloomfield, F.H. Comparing apples with apples: It is time for standardized reporting of neonatal nutrition and growth studies. Pediatr. Res. 2016, 79, 810–820. [Google Scholar] [CrossRef] [Green Version]
- Boyce, C.; Watson, M.; Lazidis, G.; Reeve, S.; Dods, K.; Simmer, K.; McLeod, G. Preterm human milk composition: A systematic literature review. Br. J. Nutr. 2016, 116, 1033–1045. [Google Scholar] [CrossRef]
- Cooper, A.R.; Barnett, D.; Gentles, E.; Cairns, L.; Simpson, J.H. Macronutrient content of donor human breast milk. Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, F539–F541. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.P.; Holding, R.E. Neonatometer: A new infant length measurer. Arch. Dis. Child. 1972, 47, 938–940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaw, V.; Lawson, M. Nutritional assessment, dietary requirements, feed supplementation. In Clinical Paediatric Dietetics, 3 ed.; Shaw, V., Lawson, M., Eds.; Blackwell Publishing: Oxford, UK, 2007; pp. 3–20. [Google Scholar]
- Fenton, T.R.; Kim, J.H. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013, 13, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papageorghiou, A.T.; Ohuma, E.O.; Altman, D.G.; Todros, T.; Cheikh Ismail, L.; Lambert, A.; Jaffer, Y.A.; Bertino, E.; Gravett, M.G.; Purwar, M.; et al. International standards for fetal growth based on serial ultrasound measurements: The Fetal Growth Longitudinal Study of the INTERGROWTH-21st Project. Lancet 2014, 384, 869–879. [Google Scholar] [CrossRef] [Green Version]
- Patel, A.L.; Engstrom, J.L.; Meier, P.P.; Kimura, R.E. Accuracy of methods for calculating postnatal growth velocity for extremely low birth weight infants. Pediatrics 2005, 116, 1466–1473. [Google Scholar] [CrossRef]
- Rochow, N.; Raja, P.; Liu, K.; Fenton, T.; Landau-Crangle, E.; Gottler, S.; Jahn, A.; Lee, S.; Seigel, S.; Campbell, D.; et al. Physiological adjustment to postnatal growth trajectories in healthy preterm infants. Pediatr. Res. 2016, 79, 870–879. [Google Scholar] [CrossRef]
- Cormack, B.E.; Jiang, Y.; Harding, J.E.; Crowther, C.A.; Lynn, A.; Nair, A.; Hewson, M.; Meyer, M.; Broadbent, R.; Webster, D.; et al. Plasma ammonia concentrations in extremely low birthweight infants in the first week after birth: Secondary analysis from the ProVIDe randomized clinical trial. Pediatr. Res. 2020. [Google Scholar] [CrossRef] [Green Version]
- Cormack, B.E.; Bloomfield, F.H. Early nutrition for preterm babies: Small changes can make a big difference. Nutr. Today 2015, 50, 230–239. [Google Scholar] [CrossRef]
- Balasubramanian, H.; Nanavati, R.N.; Kabra, N.S. Effect of two different doses of parenteral amino acid supplementation on postnatal growth of very low birth weight neonates—A randomized controlled trial. Indian Pediatr. 2013, 50, 1131–1136. [Google Scholar] [CrossRef]
- Uthaya, S.; Liu, X.; Babalis, D.; Dore, C.J.; Warwick, J.; Bell, J.; Thomas, L.; Ashby, D.; Durighel, G.; Ederies, A.; et al. Nutritional evaluation and optimisation in neonates: A randomized, double-blind controlled trial of amino acid regimen and intravenous lipid composition in preterm parenteral nutrition. Am. J. Clin. Nutr. 2016, 103, 1443–1452. [Google Scholar] [CrossRef] [Green Version]
- Balakrishnan, M.; Jennings, A.; Przystac, L.; Phornphutkul, C.; Tucker, R.; Vohr, B.; Stephens, B.E.; Bliss, J.M. Growth and neurodevelopmental outcomes of early, high-dose parenteral amino acid intake in very low birth weight infants: A randomized controlled trial. J. Parenter. Enter. Nutr. 2017. [Google Scholar] [CrossRef]
- Lapillonne, A.; Kermorvant-Duchemin, E. A systematic review of practice surveys on parenteral nutrition for preterm infants. J. Nutr. 2013, 143, 2061S–2065S. [Google Scholar] [CrossRef] [Green Version]
- Cormack, B.; Sinn, J.; Lui, K.; Tudehope, D. Australasian neonatal intensive care enteral nutrition survey: Implications for practice. J. Paediatr. Child Health 2013, 49, E340–E347. [Google Scholar] [CrossRef] [PubMed]
- Tottman, A.C.; Alsweiler, J.M.; Bloomfield, F.H.; Gamble, G.D.; Jiang, Y.; Leung, M.; Poppe, T.; Thompson, B.; Wouldes, T.A.; Harding, J.E.; et al. Relationships between early neonatal nutrition and neurodevelopment at school age in children born very preterm. J. Pediatr. Gastroenterol. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- McCarty, D.B.; Peat, J.R.; Malcolm, W.F.; Smith, P.B.; Fisher, K.; Goldstein, R.F. Dolichocephaly in preterm infants: Prevalence, risk factors, and early motor outcomes. Am. J. Perinatol. 2017, 34, 372–378. [Google Scholar] [CrossRef] [PubMed]





| Per 100 mL | Protein (g) | Carbohydrate (g) | Fat (g) | Energy (kcal) |
|---|---|---|---|---|
| Mother’s expressed breastmilk—day 1–4 | 1.83 | 5.98 | 3.22 | 67.35 |
| Mother’s expressed breastmilk—day 15–28 | 1.35 | 6.05 | 3.86 | 72.95 |
| Donor breastmilk | 0.90 | 6.60 | 4.00 | 66.00 |
| Characteristic | At birth |
|---|---|
| For mother: | |
| Age in years | 31 (16, 44) |
| Caesarean section | 234 (54) |
| Antenatal corticosteroids (any) | 410 (94) |
| Maternal diabetes | 24 (6) |
| For baby: | |
| Gestation (weeks) | 25.7 (22.7, 31.5) |
| Birthweight (g) | 777 (405, 998) |
| Birthweight z score | 0.06 (−2.46, 2.64) |
| Birth length (cm) | 33.0 (26.0, 39.0) |
| Birth length z score | −0.09 (−4.08, 2.40) |
| Birth head circumference (cm) | 23.4 (18.3, 27.0) |
| Birth head circumference z score | 0.05 (−3.34, 2.34) |
| Small-for-gestational-age | 48 (11%) |
| Male | 212 (49%) |
| Singleton | 340 (78%) |
| Nutrition | Week 1 | Fortnight 1 | Month 1 |
|---|---|---|---|
| Fluid volumes (ml·Kg−1·d−1) | |||
| Total | 139 (125, 151) | 151 (143, 161) | 158 (152, 167) |
| Intravenous | 123 (106, 149) | 105 (77, 133) | 66 (44, 110) |
| Enteral | 11 (5, 23) | 44 (16, 73) | 93 (46, 117) |
| Breastmilk | 11 (5, 23) | 42 (15, 72) | 87 (38, 115) |
| Macronutrients | |||
| Total energy (kcal·Kg−1·d−1) | 76 (70, 83) | 92 (83, 102) | 109 (95, 122) |
| Intravenous | 66 (58, 73) | 57 (42, 70) | 36 (24, 57) |
| Enteral | 8 (4, 16) | 31 (11, 56) | 73 (36, 96) |
| Total protein (g·Kg−1·d−1) | 3.34 (2.86, 3.76) | 3.48 (3.01, 3.90) | 3.50 (2.95, 3.92) |
| Intravenous | 2.99 (2.50, 3.45) | 2.40 (1.85, 2.40) | 1.46 (1.04, 2.24) |
| Enteral | 0.20 (0.01, 0.43) | 0.88 (0.27, 1.66) | 1.82 (0.75, 2.63) |
| Total carbohydrate (g·Kg−1·d−1) | 10.33 (9.13, 11.79) | 11.70 (10.88, 12.58) | 12.62 (11.72, 13.56) |
| Intravenous | 9.43 (7.97, 11.11) | 8.13 (5.95, 10.51) | 5.33 (3.51, 8.44) |
| Enteral | 0.67 (0.32, 1.41) | 2.90 (0.95, 5.34) | 6.61 (3.10, 9.34) |
| Total fat (g·Kg−1·d−1) | 2.57 (2.21, 3.01) | 3.45 (2.84, 3.95) | 4.56 (3.67, 5.16) |
| Intravenous | 2.06 (1.73, 2.43) | 1.76 (1.30, 1.76) | 1.07 (0.73, 1.73) |
| Enteral | 0.36 (0.17, 0.75) | 1.46 (0.54, 2.46) | 3.50 (1.70, 4.40) |
| Total energy: protein ratio (kcals/g) | 23 (20, 28) | 27 (24, 31) | 32 (29, 36) |
| Intravenous | 22 (19, 27) | 24 (20, 29) | 24 (21, 30) |
| Enteral | 37 (37, 37) | 37 (35, 38) | 39 (36, 44) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cormack, B.E.; Jiang, Y.; Harding, J.E.; Crowther, C.A.; Bloomfield, F.H., for the ProVIDe study group. Relationships between Neonatal Nutrition and Growth to 36 Weeks’ Corrected Age in ELBW Babies–Secondary Cohort Analysis from the Provide Trial. Nutrients 2020, 12, 760. https://doi.org/10.3390/nu12030760
Cormack BE, Jiang Y, Harding JE, Crowther CA, Bloomfield FH for the ProVIDe study group. Relationships between Neonatal Nutrition and Growth to 36 Weeks’ Corrected Age in ELBW Babies–Secondary Cohort Analysis from the Provide Trial. Nutrients. 2020; 12(3):760. https://doi.org/10.3390/nu12030760
Chicago/Turabian StyleCormack, Barbara E., Yannan Jiang, Jane E. Harding, Caroline A. Crowther, and Frank H. Bloomfield for the ProVIDe study group. 2020. "Relationships between Neonatal Nutrition and Growth to 36 Weeks’ Corrected Age in ELBW Babies–Secondary Cohort Analysis from the Provide Trial" Nutrients 12, no. 3: 760. https://doi.org/10.3390/nu12030760
APA StyleCormack, B. E., Jiang, Y., Harding, J. E., Crowther, C. A., & Bloomfield, F. H., for the ProVIDe study group. (2020). Relationships between Neonatal Nutrition and Growth to 36 Weeks’ Corrected Age in ELBW Babies–Secondary Cohort Analysis from the Provide Trial. Nutrients, 12(3), 760. https://doi.org/10.3390/nu12030760






