Chronic Alcohol Consumption is Inversely Associated with Insulin Resistance and Fatty Liver in Japanese Males
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Clinical and Laboratory Assessments
2.3. Evaluation of Alcohol Consumption
2.4. Statistical Analysis
3. Results
3.1. Clinical Characteristics
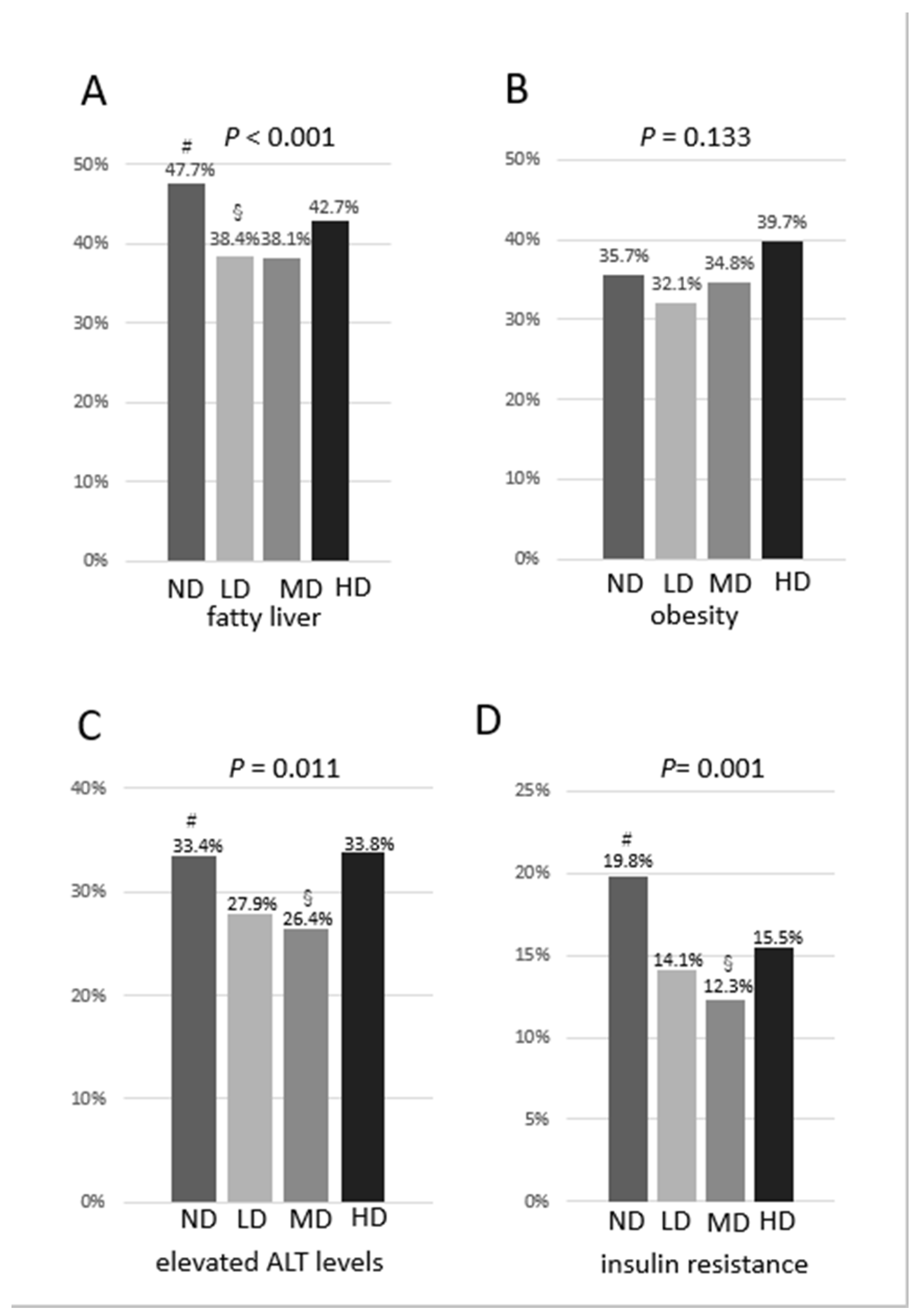
3.2. Comparisons of Prevalence of Fatty Liver, Obesity, Elevated Levels of ALT and Resistance to Insulin among the 4 Groups
3.3. Predictors of Fatty Liver and Resistance to Insulin
3.4. Comparisons of Biological Features among Non-Drinkers and Various Types of Drinkers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Shea, R.S.; Dasarathy, S.; McCullough, A.J. Alcoholic Liver Disease. Am. J. Gastroenterol. 2010, 105, 14–32. [Google Scholar] [CrossRef]
- Neuschwander-Tetri, B.A. Nonalcoholic Steatohepatitis and the Metabolic Syndrome. Am. J. Med. Sci. 2005, 330, 326–335. [Google Scholar] [CrossRef] [Green Version]
- Marchesini, G.; Brizi, M.; Bianchi, G.; Tomassetti, S.; Bugianesi, E.; Lenzi, M.; McCullough, A.J.; Natale, S.; Forlani, G.; Melchionda, N. Nonalcoholic Fatty Liver Disease: A Feature of the Metabolic Syndrome. Diabetes 2001, 50, 1844–1850. [Google Scholar] [CrossRef] [Green Version]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The Metabolic Syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Kitade, H.; Chen, G.; Ni, Y.; Ota, T. Nonalcoholic Fatty Liver Disease and Insulin Resistance: New Insights and Potential New Treatments. Nutrients 2017, 9, 387. [Google Scholar] [CrossRef] [Green Version]
- Stampfer, M.J.; Colditz, G.A.; Willett, W.C.; Speizer, F.E.; Hennekens, C.H. A Prospective Study of Moderate Alcohol Consumption and the Risk of Coronary Disease and Stroke in Women. N. Engl. J. Med. 1988, 319, 267–273. [Google Scholar] [CrossRef]
- Fuchs, C.S.; Stampfer, M.J.; Colditz, G.A.; Giovannucci, E.L.; Manson, J.E.; Kawachi, I.; Hunter, D.J.; Hankinson, S.E.; Hennekens, C.H.; Rosner, B.; et al. Alcohol Consumption and Mortality among Women. N. Engl. J. Med. 1995, 332, 1245–1250. [Google Scholar] [CrossRef]
- Gaziano, J.M.; Buring, J.E.; Breslow, J.L.; Goldhaber, S.Z.; Rosner, B.; VanDenburgh, M.; Willett, W.; Hennekens, C.H. Moderate Alcohol Intake, Increased Levels of High-Density Lipoprotein and Its Subfractions, and Decreased Risk of Myocardial Infarction. N. Engl. J. Med. 1993, 329, 1829–1834. [Google Scholar] [CrossRef]
- Tsumura, K.; Hayashi, T.; Suematsu, C.; Endo, G.; Fujii, S.; Okada, K. Daily Alcohol Consumption and the Risk of Type 2 Diabetes in Japanese Men: The Osaka Health Survey. Diabetes Care 1999, 22, 1432–1437. [Google Scholar] [CrossRef]
- Wei, M.; Gibbons, L.W.; Mitchell, T.L.; Kampert, J.B.; Blair, S.N. Alcohol Intake and Incidence of Type 2 Diabetes in Men. Diabetes Care 2000, 23, 18–22. [Google Scholar] [CrossRef] [Green Version]
- Davies, M.J.; Baer, D.J.; Judd, J.T.; Brown, E.D.; Campbell, W.S.; Taylor, P.R. Effects of Moderate Alcohol Intake on Fasting Insulin and Glucose Concentrations and Insulin Sensitivity in Postmenopausal Women: A Randomized Controlled Trial. JAMA 2002, 287, 2559–2562. [Google Scholar] [CrossRef] [Green Version]
- Gunji, T.; Matsuhashi, N.; Sato, H.; Fujibayashi, K.; Okumura, M.; Sasabe, N.; Urabe, A. Light and Moderate Alcohol Consumption Significantly Reduces the Prevalence of Fatty Liver in the Japanese Male Population. Am. J. Gastroenterol. 2009, 104, 2189–2195. [Google Scholar] [CrossRef]
- Hiramine, Y.; Imamura, Y.; Uto, H.; Koriyama, C.; Horiuchi, M.; Oketani, M.; Hosoyamada, K.; Kusano, K.; Ido, A.; Tsubouchi, H. Alcohol Drinking Patterns and the Risk of Fatty Liver in Japanese Men. J. Gastroenterol. 2011, 46, 519–528. [Google Scholar] [CrossRef]
- Chang, Y.; Ryu, S.; Kim, Y.; Cho, Y.K.; Sung, E.; Kim, H.N.; Ahn, J.; Jung, H.S.; Yun, K.E.; Kim, S.; et al. Low Levels of Alcohol Consumption, Obesity, and Development of Fatty Liver with and without Evidence of Advanced Fibrosis. Hepatology 2020, 71, 861–873. [Google Scholar] [CrossRef]
- Liu, P.; Xu, Y.; Tang, Y.; Du, M.; Yu, X.; Sun, J.; Xiao, L.; He, M.; Wei, S.; Yuan, J.; et al. Independent and Joint Effects of Moderate Alcohol Consumption and Smoking on the Risks of Non-Alcoholic Fatty Liver Disease in Elderly Chinese Men. PLoS ONE 2017, 12, e0181497. [Google Scholar] [CrossRef] [Green Version]
- Ajmera, V.H.; Terrault, N.A.; Harrison, S.A. Is Moderate Alcohol Use in Nonalcoholic Fatty Liver Disease Good or Bad? A Critical Review. Hepatology 2017, 65, 2090–2099. [Google Scholar] [CrossRef] [Green Version]
- Corrao, G.; Bagnardi, V.; Zambon, A.; La Vecchia, C. A Meta-Analysis of Alcohol Consumption and the Risk of 15 Diseases. Prev. Med. 2004, 38, 613–619. [Google Scholar] [CrossRef]
- Ronksley, P.E.; Brien, S.E.; Turner, B.J.; Mukamal, K.J.; Ghali, W.A. Association of Alcohol Consumption with Selected Cardiovascular Disease Outcomes: A Systematic Review and Meta-Analysis. BMJ 2011, 342, d671. [Google Scholar] [CrossRef] [Green Version]
- Rimm, E.B.; Williams, P.; Fosher, K.; Criqui, M.; Stampfer, M.J. Moderate Alcohol Intake and Lower Risk of Coronary Heart Disease: Meta-Analysis of Effects on Lipids and Haemostatic Factors. BMJ 1999, 319, 1523–1528. [Google Scholar] [CrossRef] [Green Version]
- Rehm, J.; Room, R.; Graham, K.; Monteiro, M.; Gmel, G.; Sempos, C.T. The Relationship of Average Volume of Alcohol Consumption and Patterns of Drinking to Burden of Disease: An Overview. Addiction 2003, 98, 1209–1228. [Google Scholar] [CrossRef]
- Koppes, L.L.; Dekker, J.M.; Hendriks, H.F.; Bouter, L.M.; Heine, R.J. Moderate Alcohol Consumption Lowers the Risk of Type 2 Diabetes: A Meta-Analysis of Prospective Observational Studies. Diabetes Care 2005, 28, 719–725. [Google Scholar] [CrossRef] [Green Version]
- Corrao, G.; Rubbiati, L.; Bagnardi, V.; Zambon, A.; Poikolainen, K. Alcohol and Coronary Heart Disease: A Meta-Analysis. Addiction 2000, 95, 1505–1523. [Google Scholar] [CrossRef]
- Knott, C.; Bell, S.; Britton, A. Alcohol Consumption and the Risk of Type 2 Diabetes: A Systematic Review and Dose-Response Meta-Analysis of More Than 1.9 Million Individuals from 38 Observational Studies. Diabetes Care 2015, 38, 1804–1812. [Google Scholar] [CrossRef] [Green Version]
- Sookoian, S.; Castano, G.O.; Pirola, C.J. Modest Alcohol Consumption Decreases the Risk of Non-Alcoholic Fatty Liver Disease: A Meta-Analysis of 43 175 Individuals. Gut 2014, 63, 530–532. [Google Scholar] [CrossRef]
- Cao, G.; Yi, T.; Liu, Q.; Wang, M.; Tang, S. Alcohol Consumption and Risk of Fatty Liver Disease: A Meta-Analysis. PeerJ 2016, 4, e2633. [Google Scholar] [CrossRef]
- Bell, R.A.; Mayer-Davis, E.J.; Martin, M.A.; D’Agostino, R.B., Jr.; Haffner, S.M. Associations between Alcohol Consumption and Insulin Sensitivity and Cardiovascular Disease Risk Factors: The Insulin Resistance and Atherosclerosis Study. Diabetes Care 2000, 23, 1630–1636. [Google Scholar] [CrossRef] [Green Version]
- Greenfield, J.R.; Samaras, K.; Jenkins, A.B.; Kelly, P.J.; Spector, T.D.; Campbell, L.V. Moderate Alcohol Consumption, Estrogen Replacement Therapy, and Physical Activity Are Associated with Increased Insulin Sensitivity: Is Abdominal Adiposity the Mediator? Diabetes Care 2003, 26, 2734–2740. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.M.; Carlson, G.P. Effects of Ethanol on Glutathione Conjugation in Rat Liver and Lung. Biochem. Pharmacol. 1991, 41, 923–929. [Google Scholar] [CrossRef]
- Guarino, M.P.; Afonso, R.A.; Raimundo, N.; Raposo, J.F.; Macedo, M.P. Hepatic Glutathione and Nitric Oxide Are Critical for Hepatic Insulin-Sensitizing Substance Action. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G588–G594. [Google Scholar] [CrossRef] [Green Version]
- Kanuri, G.; Landmann, M.; Priebs, J.; Spruss, A.; Löscher, M.; Ziegenhardt, D.; Röhl, C.; Degen, C.; Bergheim, I. Moderate Alcohol Consumption Diminishes the Development of Non-Alcoholic Fatty Liver Disease (Nafld) in Ob/Ob Mice. Eur. J. Nutr. 2016, 55, 1153–1164. [Google Scholar] [CrossRef]
- Lindtner, C.; Scherer, T.; Zielinski, E.; Filatova, N.; Fasshauer, M.; Tonks, N.K.; Puchowicz, M.; Buettner, C. Binge Drinking Induces Whole-Body Insulin Resistance by Impairing Hypothalamic Insulin Action. Sci. Transl. Med. 2013, 5, 170ra114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pareek, M.; Olsen, M.H. Alcohol and Blood Pressure. Lancet Public Health 2017, 2, e63–e64. [Google Scholar] [CrossRef] [Green Version]
- European Association for the Study of the Liver. Electronic address, e.e.e., European Association for the Study of the, L. Easl Clinical Practice Guidelines: Management of Alcohol-Related Liver Disease. J. Hepatol. 2018, 69, 154–181. [Google Scholar] [CrossRef] [Green Version]
- Dunn, W.; Xu, R.; Schwimmer, J.B. Modest Wine Drinking and Decreased Prevalence of Suspected Non-alcoholic Fatty Liver Disease. Hepatology 2008, 47, 1947–1954. [Google Scholar] [CrossRef]
| ND | LD | MD | HD | p-Value | |
|---|---|---|---|---|---|
| N (%) | 894 (36.8) | 857 (35.3) | 444 (18.3) | 234 (9.6) | |
| Alcohol consumption (95% CI) (g/d) | 0 (0, 0) | 15.8 ± 5.9 (15.4, 16.2) | 41.6 ± 5.8 (41.0, 42.1) | 77.4 ± 18.8 (75.0, 79.8) | <0.001 |
| Age (y) | 53.4 ± 9.2 | 54.7 ± 9.8 | 54.7 ± 8.5 | 54.3 ± 8.5 | 0.016 |
| BMI (kg/m2) | 24.2 ± 3.4 | 23.9 ± 3.1 | 24.0 ± 3.0 | 24.4 ± 3.0 | 0.074 |
| Habit of drinking sweet beverages (%) | 322 (36) # | 248 (28.9) | 96 (21.6) § | 39 (16.7) § | <0.001 |
| Habit of eating greasy meat (%) | 322 (36) | 315 (36.8) | 184 (41.4) | 96 (41) | 0.162 |
| Physical activity (%) | 316 (35.3) § | 366 (42.7) # | 187 (42.1) | 84 (35.9) | 0.005 |
| Systolic BP | 127 ± 15 | 129 ± 14 | 131 ± 15 *** | 134 ± 14 *** | <0.001 |
| Diastolic BP | 78 ± 11 | 79 ± 11 | 82 ± 10 *** | 84 ± 11 *** | <0.001 |
| Hypertension (%) | 277 (31) § | 304 (35.5) | 191 (43) # | 137 (58.5) # | <0.001 |
| Use of antihypertensive medications | 165 (18.5) § | 173 (20.2) | 122 (27.5) # | 72 (30.8) # | <0.001 |
| AST (IU/L) | 24 ± 19 | 24 ± 7 | 26 ± 22 ** | 28 ± 14 *** | <0.001 |
| ALT (IU/L) | 30 ± 19 | 27 ± 15 ** | 27 ± 20 | 30 ± 20 | 0.001 |
| GGT (IU/L) | 34 ± 30 | 42 ± 36 * | 62 ± 53 *** | 96 ± 119 *** | <0.001 |
| Triglycerides (mg/dL) | 125 ± 86 | 123 ± 76 | 139 ± 103 * | 169 ± 148 *** | <0.001 |
| Total cholesterol (mg/dL) | 205 ± 33 | 207 ± 32 | 206 ± 32 | 207 ± 34 | 0.524 |
| HDL cholesterol (mg/dL) | 53 ± 13 | 56 ± 13 *** | 60 ± 15 *** | 61 ± 15 *** | <0.001 |
| LDL cholesterol (mg/dL) | 129 ± 29 | 127 ± 29 | 120 ± 31 *** | 116 ± 33 *** | <0.001 |
| Use of lipid-lowering medications (%) | 129 (14.4) | 94 (11.0) | 53 (11.9) | 21 (9.0) | 0.054 |
| Fasting glucose (mg/dL) | 103 ± 20 | 103 ± 20 | 105 ± 20 | 107 ± 21 | 0.24 |
| Use of antidiabetic medications (%) | 62 (6.9) | 47 (5.5) | 27 (6.1) | 15 (6.4) | 0.657 |
| HOMA-IR | 1.9 ± 1.7 | 1.6 ± 1.4 ** | 1.5 ± 1.0 *** | 1.6 ± 1.2 * | <0.001 |
| Metabolic syndrome (%) | 202 (22.6) | 172 (20.1) § | 111 (25.1) | 80 (34.2) # | <0.001 |
| Odds Ratio (95% CI) | p-Value | |
|---|---|---|
| ND | 1 | |
| LD | 0.682 (0.549–0.846) | 0.001 |
| MD | 0.771 (0.675–0.880) | <0.001 |
| HD | 0.840 (0.749–0.941) | 0.003 |
| Odds Ratio (95% CI) | p-Value | |
|---|---|---|
| ND | 1 | |
| LD | 0.724 (0.539–0.974) | 0.033 |
| MD | 0.701 (0.577–0.852) | <0.001 |
| HD | 0.800 (0.686–0.933) | 0.005 |
| ND | Mixed Drinkers | Beer Drinkers | Sake Drinkers | Shochu Drinkers | Whiskey Drinkers | Wine Drinkers | p-Value | |
|---|---|---|---|---|---|---|---|---|
| N (%) | 894 (36.8) | 349 (14.4) | 813 (33.5) | 103 (4.2) | 236 (9.7) | 17 (0.7) | 17 (0.7) | |
| Alcohol consumption (95% CI) (g/d) | 0 | 54.3 ± 23.9 (51.8, 56.8) | 20.4 ± 13.8 (19.4, 21.3) | 32.9 ± 16.9 (29.6, 36.2) | 43.7 ± 26.2 (40.3, 47.1) | 33.4 ± 18.4 (23.9, 42.9) | 16.4 ± 18.2 (7.0, 25.7) | <0.001 |
| LD (%), MD (%), HD (%) | 0 | 29 (8.3), 187 (53.6), 133 (38.1) | 653 (80.3), 131 (16.1), 29 (3.6) | 49 (47.6), 43 (41.7), 11 (10.7) | 105 (44.5), 74 (31.4), 57 (24.2) | 7 (41.2), 7 (41.2), 3 (17.6) | 14 (82.4), 2 (11.8), 1 (5.9) | <0.001 |
| Age (y) | 53.4 ± 9.2 | 54.6 ± 8.3 | 53.2 ± 9.4 | 60.8 ± 8.3 | 56.9 ± 8.6 | 52.4 ± 9.2 | 56.8 ± 10.3 | <0.001 |
| BMI (kg/m2) | 24.2 ± 3.4 | 24.2 ± 3.0 | 23.9 ± 3.1 | 23.4 ± 2.5 | 24.1 ± 3.2 | 24.0 ± 3.2 | 23.8 ± 2.8 | 0.243 |
| AST (IU/L) | 24 ± 9 | 26 ± 26 * | 24 ± 9 | 27 ± 9 | 26 ± 9 | 22 ± 3 | 25 ± 8 | 0.004 |
| ALT (IU/L) | 30 ± 19 | 27 ± 22 | 28 ± 17 | 28 ± 15 | 27 ± 13 | 23 ± 8 | 24 ± 12 | 0.058 |
| GGT (IU/L) | 34 ± 30 | 66 ± 85 *** | 47 ± 49 *** | 59 ± 70 *** | 67 ± 67 *** | 42 ± 27 | 55 ± 51 | <0.001 |
| Triglycerides (mg/dL) | 125 ± 86 | 142 ± 102 | 129 ± 80 | 121 ± 79 | 152 ± 149** | 148 ± 114 | 119 ± 81 | 0.001 |
| Use of lipid-lowering medications (%) | 129 (14.4) | 34 (9.7) | 89 (10.9) | 11 (10.7) | 30 (12.7) | 1 (5.9) | 3 (17.9) | 0.193 |
| Fasting glucose (mg/dL) | 103 ± 20 | 105 ± 20 | 104 ± 21 | 102 ± 14 | 107 ± 18 | 100 ± 10 | 102 ± 10 | 0.156 |
| Use of antidiabetic medications (%) | 62 (6.9) | 18 (5.2) | 43 (5.3) | 3 (2.9) | 24 (10.4) | 0 (0) | 1 (5.9) | 0.061 |
| HOMA-IR | 1.9 ± 1.7 | 1.5 ± 1.1 ** | 1.6 ± 1.4 * | 1.3 ± 0.9 * | 1.5 ± 1.0* | 1.7 ± 1.0 | 1.7 ± 2.0 | <0.001 |
| Fatty liver (%) | 426 (47.7) | 139 (39.8) | 312 (38.4) | 37 (35.9) | 97 (41.1) | 6 (35.3) | 7 (41.2) | 0.005 |
| LD | MD | HD | |
|---|---|---|---|
| Mixed drinkers (fatty liver/total number, (%)) | 13/29 (44.8) | 76/187 (40.6) | 50/133 (37.6) |
| Beer drinkers (fatty liver/total number, (%)) | 254/653 (38.9) | 42/131 (32.1) | 16/29 (55.2) |
| Sake drinkers (fatty liver/total number, (%)) | 14/49 (28.6) | 15/43 (34.9) | 8/11 (72.7) |
| Shochu drinkers (fatty liver/total number, (%)) | 41/105 (39.0) | 32/74 (43.2) | 24/57 (42.1) |
| Whiskey drinkers (fatty liver/total number, (%)) | 3/7 (42.9) | 2/7 (28.6) | 1/3 (33.3) |
| Wine drinkers (fatty liver/total number, (%)) | 4/14 (28.6) | 2/2 (100) | 1/1 (100) |
| p-Value | 0.665 | 0.215 | 0.119 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akahane, T.; Namisaki, T.; Kaji, K.; Moriya, K.; Kawaratani, H.; Takaya, H.; Sawada, Y.; Shimozato, N.; Fujinaga, Y.; Furukawa, M.; et al. Chronic Alcohol Consumption is Inversely Associated with Insulin Resistance and Fatty Liver in Japanese Males. Nutrients 2020, 12, 1036. https://doi.org/10.3390/nu12041036
Akahane T, Namisaki T, Kaji K, Moriya K, Kawaratani H, Takaya H, Sawada Y, Shimozato N, Fujinaga Y, Furukawa M, et al. Chronic Alcohol Consumption is Inversely Associated with Insulin Resistance and Fatty Liver in Japanese Males. Nutrients. 2020; 12(4):1036. https://doi.org/10.3390/nu12041036
Chicago/Turabian StyleAkahane, Takemi, Tadashi Namisaki, Kosuke Kaji, Kei Moriya, Hideto Kawaratani, Hiroaki Takaya, Yasuhiko Sawada, Naotaka Shimozato, Yukihisa Fujinaga, Masanori Furukawa, and et al. 2020. "Chronic Alcohol Consumption is Inversely Associated with Insulin Resistance and Fatty Liver in Japanese Males" Nutrients 12, no. 4: 1036. https://doi.org/10.3390/nu12041036
APA StyleAkahane, T., Namisaki, T., Kaji, K., Moriya, K., Kawaratani, H., Takaya, H., Sawada, Y., Shimozato, N., Fujinaga, Y., Furukawa, M., Kitagawa, K., Ozutsumi, T., Tsuji, Y., Kaya, D., Ogawa, H., Takagi, H., Ishida, K., & Yoshiji, H. (2020). Chronic Alcohol Consumption is Inversely Associated with Insulin Resistance and Fatty Liver in Japanese Males. Nutrients, 12(4), 1036. https://doi.org/10.3390/nu12041036






