Food Liking-Based Diet Quality Indexes (DQI) Generated by Conceptual and Machine Learning Explained Variability in Cardiometabolic Risk Factors in Young Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Procedure and Measures
2.3. Diet Quality Index Methods
2.4. Statistical Analyses
3. Results
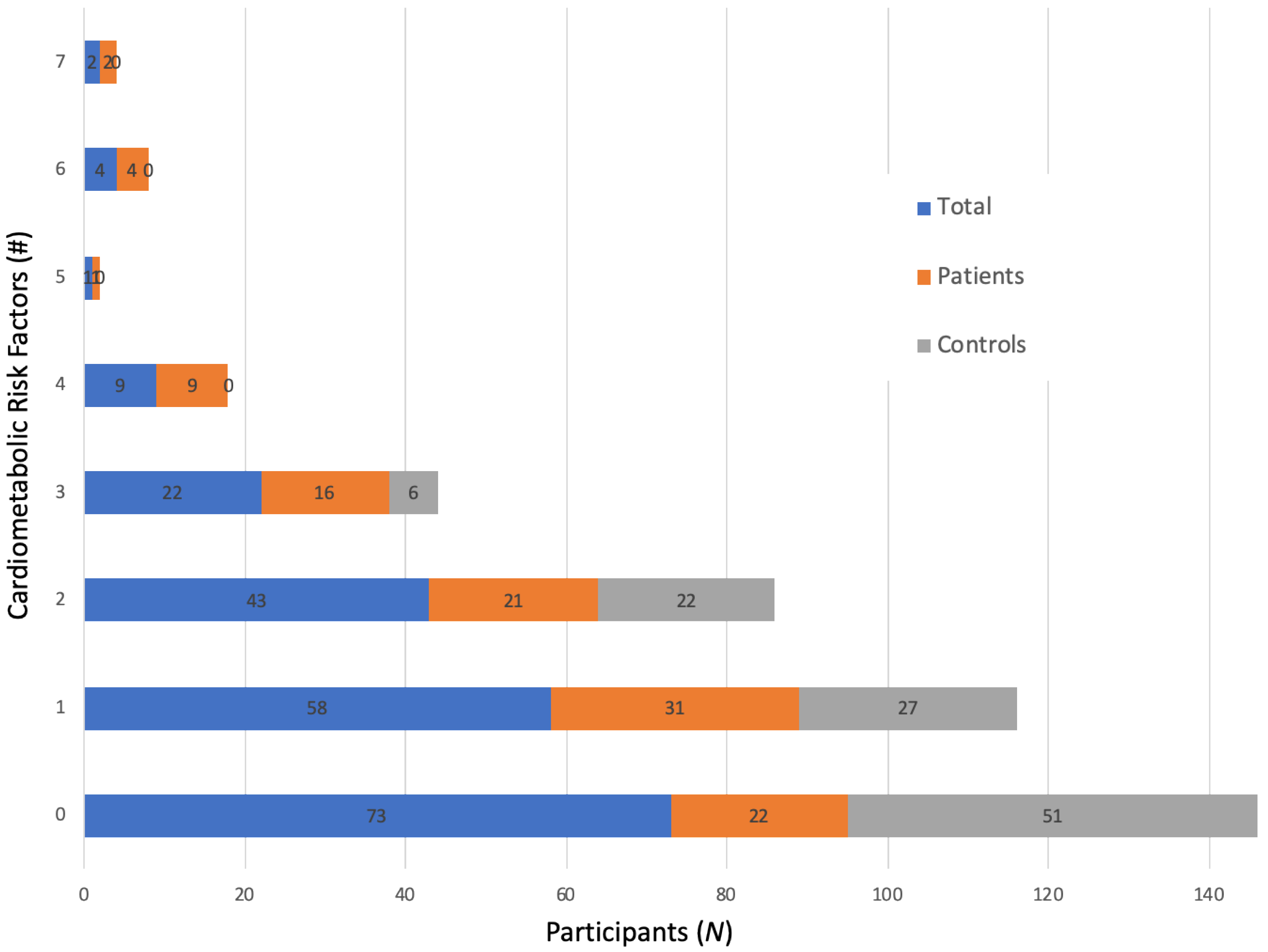
3.1. Descriptive Findings—Cardiometabolic Risk Factors and Food Liking
3.2. Descriptive Findings—Diet Quality Indexes
3.3. Predicting CFRS: Comparisons between the DQIs
3.4. Standardized Hybrid DQI Components and Scoring Standards
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Andes, L.J.; Cheng, Y.J.; Rolka, D.B.; Gregg, E.W.; Imperatore, G. Prevalence of Prediabetes among Adolescents and Young Adults in the United States, 2005–2016. JAMA Pediatr. 2019, 174, e194498. [Google Scholar] [CrossRef]
- Moore, J.X.; Chaudhary, N.; Akinyemiju, T. Metabolic Syndrome Prevalence by Race/Ethnicity and Sex in the United States, National Health and Nutrition Examination Survey, 1988–2012. Prev. Chronic Dis. 2017, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, B.J.; Todaro, J.F.; Niaura, R.; McCaffery, J.M.; Zhang, J.; Spiro, A., 3rd; Ward, K.D. Are metabolic risk factors one unified syndrome? Modeling the structure of the metabolic syndrome X. Am. J. Epidemiol. 2003, 157, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Magge, S.N.; Goodman, E.; Armstrong, S.C.; Committee On Nutrition; Section On Endocrinology; Section On Obesity. The Metabolic Syndrome in Children and Adolescents: Shifting the Focus to Cardiometabolic Risk Factor Clustering. Pediatrics 2017, 140, e20171063. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodman, E.; Dolan, L.M.; Morrison, J.A.; Daniels, S.R. Factor analysis of clustered cardiovascular risks in adolescence: Obesity is the predominant correlate of risk among youth. Circulation 2005, 111, 1970–1977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, A.S.; Steinberger, J.; Jacobs, D.R.; Hong, C.P.; Moran, A.; Sinaiko, A.R. Predicting cardiovascular risk in young adulthood from the metabolic syndrome, its component risk factors, and a cluster score in childhood. Int. J. Pediatr. Obes. 2011, 6, e283–e289. [Google Scholar] [CrossRef] [Green Version]
- McCaffery, J.M.; Shen, B.J.; Muldoon, M.F.; Manuck, S.B. Ambulatory blood pressure and the metabolic syndrome in normotensive and untreated hypertensive men. Metab. Syndr. Relat. Disord. 2007, 5, 34–44. [Google Scholar] [CrossRef]
- Barbour-Tuck, E.N.; Erlandson, M.C.; Sherar, L.B.; Eisenmann, J.C.; Muhajarine, N.; Foulds, H.; Vatanparast, H.; Nisbet, C.; Kontulainen, S.; Baxter-Jones, A.D.G. Relationship Between Trajectories of Trunk Fat Development in Emerging Adulthood and Cardiometabolic Risk at 36 Years of Age. Obesity 2019, 27, 1652–1660. [Google Scholar] [CrossRef]
- Callo Quinte, G.; Barros, F.; Gigante, D.P.; de Oliveira, I.O.; Dos Santos Motta, J.V.; Horta, B.L. Overweight trajectory and cardio metabolic risk factors in young adults. BMC Pediatr. 2019, 19, 75. [Google Scholar] [CrossRef] [Green Version]
- DiPietro, L.; Zhang, Y.; Mavredes, M.; Simmens, S.J.; Whiteley, J.A.; Hayman, L.L.; Faro, J.; Malin, S.K.; Winston, G.; Napolitano, M.A. Physical Activity and Cardiometabolic Risk Factor Clustering in Young Adults with Obesity. Med. Sci. Sports Exerc. 2019. [Google Scholar] [CrossRef]
- Kant, A.K. Indexes of overall diet quality: A review. J. Am. Diet Assoc. 1996, 96, 785–791. [Google Scholar] [CrossRef]
- Wrobleski, M.M.; Parker, E.A.; Hurley, K.M.; Oberlander, S.; Merry, B.C.; Black, M.M. Comparison of the HEI and HEI-2010 Diet Quality Measures in Association with Chronic Disease Risk among Low-Income, African American Urban Youth in Baltimore, Maryland. J. Am. Coll. Nutr. 2018, 37, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Mayra, S.; Ugarte, N.; Johnston, C.S. Health Biomarkers in Adults Are More Closely Linked to Diet Quality Attributes Than to Plant-Based Diet Categorization. Nutrients 2019, 11, 1427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, R.A.; Rose, A.M.; Bruno, R.S.; Hanks, A.S.; Kennel, J.A.; McDonald, J.D.; Labyk, A.N.; Gunther, C. Examination of the relationship of diet quality with cardiometabolic risk factors in apparently healthy college students. J. Educ. Health Promot. 2019, 8, 148. [Google Scholar] [CrossRef] [PubMed]
- Aljuraiban, G.S.; Gibson, R.; Oude Griep, L.M.; Okuda, N.; Steffen, L.M.; Van Horn, L.; Chan, Q. Perspective: The Application of A Priori Diet Quality Scores to Cardiovascular Disease Risk-A Critical Evaluation of Current Scoring Systems. Adv. Nutr. 2020, 11, 10–24. [Google Scholar] [CrossRef]
- Krebs-Smith, S.M.; Pannucci, T.E.; Subar, A.F.; Kirkpatrick, S.I.; Lerman, J.L.; Tooze, J.A.; Wilson, M.M.; Reedy, J. Update of the Healthy Eating Index: HEI-2015. J. Acad. Nutr. Diet. 2018, 118, 1591–1602. [Google Scholar] [CrossRef] [Green Version]
- Shivappa, N.; Steck, S.E.; Hurley, T.G.; Hussey, J.R.; Hebert, J.R. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 2014, 17, 1689–1696. [Google Scholar] [CrossRef] [Green Version]
- Kirkpatrick, S.I.; Baranowski, T.; Subar, A.F.; Tooze, J.A.; Frongillo, E.A. Best Practices for Conducting and Interpreting Studies to Validate Self-Report Dietary Assessment Methods. J. Acad. Nutr. Diet. 2019, 119, 1801–1816. [Google Scholar] [CrossRef]
- National Cancer Institute. Dietary Assessment Primer. Available online: Dietassessmentprimer.cancer.gov (accessed on 28 February 2020).
- Duffy, V.B.; Hayes, J.E.; Sullivan, B.S.; Faghri, P. Surveying food and beverage liking: A tool for epidemiological studies to connect chemosensation with health outcomes. Ann. N. Y. Acad. Sci. 2009, 1170, 558–568. [Google Scholar] [CrossRef]
- Duffy, V.B.; Lanier, S.A.; Hutchins, H.L.; Pescatello, L.S.; Johnson, M.K.; Bartoshuk, L.M. Food preference questionnaire as a screening tool for assessing dietary risk of cardiovascular disease within health risk appraisals. J. Am. Diet Assoc. 2007, 107, 237–245. [Google Scholar] [CrossRef]
- Sharafi, M.; Hayes, J.E.; Duffy, V.B. Masking vegetable bitterness to improve palatability depends on vegetable type and taste phenotype. Chemosens. Percept. 2013, 6, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Takachi, R.; Ishihara, J.; Iwasaki, M.; Ishii, Y.; Tsugane, S. Self-reported taste preference can be a proxy for daily sodium intake in middle-aged Japanese adults. J. Acad. Nutr. Diet. 2014, 114, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Shelley, M.; Liu, C.T.; Chang, Y.C. Assessing the Association of Food Preferences and Self-Reported Psychological Well-Being among Middle-Aged and Older Adults in Contemporary China-Results from the China Health and Nutrition Survey. Int. J. Environ. Res. Public Health 2018, 15, 463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beheshti, R.; Jones-Smith, J.C.; Igusa, T. Taking dietary habits into account: A computational method for modeling food choices that goes beyond price. PLoS ONE 2017, 12, e0178348. [Google Scholar] [CrossRef] [Green Version]
- Garnier, S.; Vallee, K.; Lemoine-Morel, S.; Joffroy, S.; Drapeau, V.; Tremblay, A.; Auneau, G.; Mauriege, P. Food group preferences and energy balance in moderately obese postmenopausal women subjected to brisk walking program. Appl. Physiol. Nutr. Metab. 2015, 40, 741–748. [Google Scholar] [CrossRef]
- Charlot, K.; Malgoyre, A.; Bourrilhon, C. Proposition for a shortened version of the Leeds Food Preference Questionnaire (LFPQ). Physiol. Behav. 2019, 199, 244–251. [Google Scholar] [CrossRef]
- Park, H.; Shin, Y.; Kwon, O.; Kim, Y. Association of Sensory Liking for Fat with Dietary Intake and Metabolic Syndrome in Korean Adults. Nutrients 2018, 10, 877. [Google Scholar] [CrossRef] [Green Version]
- Ramsay, S.A.; Rudley, M.; Tonnemaker, L.E.; Price, W.J. A Comparison of College Students’ Reported Fruit and Vegetable Liking and Intake from Childhood to Adulthood. J. Am. Coll. Nutr. 2017, 36, 28–37. [Google Scholar] [CrossRef]
- Sharafi, M.; Rawal, S.; Fernandez, M.L.; Huedo-Medina, T.B.; Duffy, V.B. Taste phenotype associates with cardiovascular disease risk factors via diet quality in multivariate modeling. Physiol. Behav. 2018, 194, 103–112. [Google Scholar] [CrossRef]
- Tuorila, H.; Huotilainen, A.; Lähteenmäki, L.; Ollila, S.; Tuomi-Nurmi, S.; Urala, N. Comparison of affective rating scales and their relationship to variables reflecting food consumption. Food Qual. Pref. 2008, 19, 51–61. [Google Scholar] [CrossRef]
- Pallister, T.; Sharafi, M.; Lachance, G.; Pirastu, N.; Mohney, R.P.; MacGregor, A.; Feskens, E.J.; Duffy, V.; Spector, T.D.; Menni, C. Food Preference Patterns in a UK Twin Cohort. Twin Res. Hum. Genet. 2015, 18, 793–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharafi, M.; Duffy, V.B.; Miller, R.J.; Winchester, S.B.; Sullivan, M.C. Dietary Behaviors of Adults Born Prematurely May Explain Future Risk for Cardiovascular Disease. Appetite 2016, 99, 157–167. [Google Scholar] [CrossRef] [Green Version]
- Low, J.Y.Q.; Lacy, K.E.; McBride, R.L.; Keast, R.S.J. The Associations Between Oral Complex Carbohydrate Sensitivity, BMI, Liking, and Consumption of Complex Carbohydrate Based Foods. J. Food Sci. 2018, 83, 2227–2236. [Google Scholar] [CrossRef] [PubMed]
- Ledikwe, J.H.; Ello-Martin, J.; Pelkman, C.L.; Birch, L.L.; Mannino, M.L.; Rolls, B.J. A reliable, valid questionnaire indicates that preference for dietary fat declines when following a reduced-fat diet. Appetite 2007, 49, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, S.I.; Reedy, J.; Krebs-Smith, S.M.; Pannucci, T.E.; Subar, A.F.; Wilson, M.M.; Lerman, J.L.; Tooze, J.A. Applications of the Healthy Eating Index for Surveillance, Epidemiology, and Intervention Research: Considerations and Caveats. J. Acad. Nutr. Diet. 2018, 118, 1603–1621. [Google Scholar] [CrossRef]
- Ocke, M.C. Evaluation of methodologies for assessing the overall diet: Dietary quality scores and dietary pattern analysis. Proc. Nutr. Soc. 2013, 72, 191–199. [Google Scholar] [CrossRef] [Green Version]
- Vyncke, K.; Cruz Fernandez, E.; Fajo-Pascual, M.; Cuenca-Garcia, M.; De Keyzer, W.; Gonzalez-Gross, M.; Moreno, L.A.; Beghin, L.; Breidenassel, C.; Kersting, M.; et al. Validation of the Diet Quality Index for Adolescents by comparison with biomarkers, nutrient and food intakes: The HELENA study. Br. J. Nutr. 2013, 109, 2067–2078. [Google Scholar] [CrossRef]
- Sharafi, M.; Perrachio, H.; Scarmo, S.; Huedo-Medina, T.B.; Mayne, S.T.; Cartmel, B.; Duffy, V.B. Preschool-Adapted Liking Survey (PALS): A brief and valid method to assess dietary quality of preschoolers. Child. Obes. 2015, 11, 530–540. [Google Scholar] [CrossRef]
- Manios, Y.; Kourlaba, G.; Grammatikaki, E.; Androutsos, O.; Ioannou, E.; Roma-Giannikou, E. Comparison of two methods for identifying dietary patterns associated with obesity in preschool children: The GENESIS study. Eur. J. Clin. Nutr. 2010, 64, 1407–1414. [Google Scholar] [CrossRef] [Green Version]
- Pereira, D.L.; Juvanhol, L.L.; Silva, D.C.; Longo, G.Z. Dietary patterns and metabolic phenotypes in Brazilian adults: A population-based cross-sectional study. Public Health Nutr. 2019, 22, 3377–3383. [Google Scholar] [CrossRef]
- Biesbroek, S.; van der, A.D.; Brosens, M.C.; Beulens, J.W.; Verschuren, W.M.; van der Schouw, Y.T.; Boer, J.M. Identifying cardiovascular risk factor-related dietary patterns with reduced rank regression and random forest in the EPIC-NL cohort. Am. J. Clin. Nutr. 2015, 102, 146–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jessri, M.; Wolfinger, R.D.; Lou, W.Y.; L’Abbe, M.R. Identification of dietary patterns associated with obesity in a nationally representative survey of Canadian adults: Application of a priori, hybrid, and simplified dietary pattern techniques. Am. J. Clin. Nutr. 2017, 105, 669–684. [Google Scholar] [CrossRef] [PubMed]
- Pilleron, S.; Peres, K.; Jutand, M.A.; Helmer, C.; Dartigues, J.F.; Samieri, C.; Feart, C. Dietary patterns and risk of self-reported activity limitation in older adults from the Three-City Bordeaux Study. Br. J. Nutr. 2018, 120, 549–556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cesari, M.; Penninx, B.W.; Newman, A.B.; Kritchevsky, S.B.; Nicklas, B.J.; Sutton-Tyrrell, K.; Rubin, S.M.; Ding, J.; Simonsick, E.M.; Harris, T.B.; et al. Inflammatory markers and onset of cardiovascular events: Results from the Health ABC study. Circulation 2003, 108, 2317–2322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flahault, A.; Paquette, K.; Fernandes, R.O.; Delfrate, J.; Cloutier, A.; Henderson, M.; Lavoie, J.C.; Masse, B.; Nuyt, A.M.; Luu, T.M.; et al. Increased Incidence but Lack of Association Between Cardiovascular Risk Factors in Adults Born Preterm. Hypertension 2020, 75, 796–805. [Google Scholar] [CrossRef]
- Sun, Y.; Magnussen, C.G.; Dwyer, T.; Oddy, W.H.; Venn, A.J.; Smith, K.J. Cross-Sectional Associations between Dietary Fat-Related Behaviors and Continuous Metabolic Syndrome Score among Young Australian Adults. Nutrients 2018, 10, 972. [Google Scholar] [CrossRef] [Green Version]
- Wijndaele, K.; Beunen, G.; Duvigneaud, N.; Matton, L.; Duquet, W.; Thomis, M.; Lefevre, J.; Philippaerts, R.M. A continuous metabolic syndrome risk score: Utility for epidemiological analyses. Diabetes Care 2006, 29, 2329. [Google Scholar] [CrossRef] [Green Version]
- DHHS/USDA. Dietary Guidelines 2015–2020. Available online: https://health.gov/sites/default/files/2019-09/2015-2020_Dietary_Guidelines.pdf (accessed on 24 March 2020).
- Ivanescu, A.E.; Li, P.; George, B.; Brown, A.W.; Keith, S.W.; Raju, D.; Allison, D.B. The importance of prediction model validation and assessment in obesity and nutrition research. Int. J. Obes. 2016, 40, 887–894. [Google Scholar] [CrossRef] [Green Version]
- Marquardt, D.W.; Snee, R.D. Ridge regression in practice. Am. Stat. 1975, 29, 3–20. [Google Scholar]
- Hayes, J.E.; Bartoshuk, L.M.; Kidd, J.R.; Duffy, V.B. Supertasting and PROP bitterness depends on more than the TAS2R38 gene. Chem. Senses 2008, 33, 255–265. [Google Scholar] [CrossRef] [Green Version]
- Hubert, P.; Papasavas, P.; Stone, A.; Swede, H.; Huedo-Medina, T.; Tishler, D.; Duffy, V. Associations between weight loss, food likes, dietary behaviors and chemosensory function in bariatric surgery: A case-control analysis in women. Nutrients 2019, 11, 804. [Google Scholar] [CrossRef] [Green Version]
- Johnson, M. A multiple-entry, modular memory system. In The Psychology of Learning and Motivation: Advances in Research and Theory; Bower, G., Ed.; Academic Press: New York, NY, USA, 1983; Volume 17, pp. 81–123. [Google Scholar]
- Johnson, M.; Multhaup, K. Emotion and MEM. In The Handbook of Emotion and Memory: Current Research and Theory; Christianson, S.-A., Ed.; Erlbaum: Hillsdale, NJ, USA, 1992; Volume 33–66. [Google Scholar]
- Lähteenmäki, L.; Tuorila, H. Three-factor eating questionnaire and the use and liking of sweet and fat among dieters. Physiol. Behav. 1995, 57, 81–88. [Google Scholar] [CrossRef]
- Dinehart, M.E.; Hayes, J.E.; Bartoshuk, L.M.; Lanier, S.L.; Duffy, V.B. Bitter taste markers explain variability in vegetable sweetness, bitterness, and intake. Physiol. Behav. 2006, 87, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.E.; Sullivan, B.S.; Duffy, V.B. Explaining variability in sodium intake through oral sensory phenotype, salt sensation and liking. Physiol. Behav. 2010, 100, 369–380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guenther, P.M.; Casavale, K.O.; Reedy, J.; Kirkpatrick, S.I.; Hiza, H.A.; Kuczynski, K.J.; Kahle, L.L.; Krebs-Smith, S.M. Update of the Healthy Eating Index: HEI-2010. J. Acad. Nutr. Diet. 2013, 113, 569–580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vosburgh, K.; Smith, S.R.; Oldman, S.; Huedo-Medina, T.; Duffy, V.B. Pediatric-Adapted Liking Survey (PALS): A Diet and Activity Screener in Pediatric Care. Nutrients 2019, 11, 1641. [Google Scholar] [CrossRef] [Green Version]
- Zoghbi, M.; Stone, A.; Pavlos, P.; Swede, H.; Hubert, P.; Tishler, D.; Duffy, V. Evaluating taste preferences and dietary quality with a simple liking survey: Application in bariatric treatment settings. Bariatr. Surg. Pract. Patient Care 2019, 14, 41–48. [Google Scholar] [CrossRef]
- Avalos, L.A.; Caan, B.; Nance, N.; Zhu, Y.; Li, D.K.; Quesenberry, C.; Hyde, R.J.; Hedderson, M.M. Prenatal Depression and Diet Quality During Pregnancy. J. Acad. Nutr. Diet. 2020. [Google Scholar] [CrossRef]
- Burrows, K.; Stewart, J.L.; Antonacci, C.; Kuplicki, R.; Thompson, K.; Taylor, A.; Teague, T.K.; Paulus, M.P. Association of poorer dietary quality and higher dietary inflammation with greater symptom severity in depressed individuals with appetite loss. J. Affect. Disord. 2020, 263, 99–106. [Google Scholar] [CrossRef]
- Teasdale, S.B.; Burrows, T.L.; Hayes, T.; Hsia, C.Y.; Watkins, A.; Curtis, J.; Ward, P.B. Dietary intake, food addiction and nutrition knowledge in young people with mental illness. Nutr. Diet. 2019. [Google Scholar] [CrossRef]
- Ahluwalia, N.; Andreeva, V.A.; Kesse-Guyot, E.; Hercberg, S. Dietary patterns, inflammation and the metabolic syndrome. Diabetes Metab. 2013, 39, 99–110. [Google Scholar] [CrossRef]
- Hosseini, Z.; Whiting, S.J.; Vatanparast, H. Current evidence on the association of the metabolic syndrome and dietary patterns in a global perspective. Nutr. Res. Rev. 2016, 29, 152–162. [Google Scholar] [CrossRef]
- Pan, Y.; Pratt, C.A. Metabolic syndrome and its association with diet and physical activity in US adolescents. J. Am. Diet Assoc. 2008, 108, 276–286, discussion 286. [Google Scholar] [CrossRef]
- Fogli-Cawley, J.J.; Dwyer, J.T.; Saltzman, E.; McCullough, M.L.; Troy, L.M.; Meigs, J.B.; Jacques, P.F. The 2005 Dietary Guidelines for Americans and risk of the metabolic syndrome. Am. J. Clin. Nutr. 2007, 86, 1193–1201. [Google Scholar] [CrossRef] [Green Version]
- Osadnik, K.; Osadnik, T.; Lonnie, M.; Lejawa, M.; Regula, R.; Fronczek, M.; Gawlita, M.; Wadolowska, L.; Gasior, M.; Pawlas, N. Metabolically healthy obese and metabolic syndrome of the lean: The importance of diet quality. Analysis of MAGNETIC cohort. Nutr. J. 2020, 19, 1–13. [Google Scholar] [CrossRef]
- Lafreniere, J.; Carbonneau, E.; Laramee, C.; Corneau, L.; Robitaille, J.; Labonte, M.E.; Lamarche, B.; Lemieux, S. Is the Canadian Healthy Eating Index 2007 an Appropriate Diet Indicator of Metabolic Health? Insights from Dietary Pattern Analysis in the PREDISE Study. Nutrients 2019, 11, 1597. [Google Scholar] [CrossRef] [Green Version]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Khan, T.A.; Tayyiba, M.; Agarwal, A.; Mejia, S.B.; de Souza, R.J.; Wolever, T.M.S.; Leiter, L.A.; Kendall, C.W.C.; Jenkins, D.J.A.; Sievenpiper, J.L. Relation of Total Sugars, Sucrose, Fructose, and Added Sugars with the Risk of Cardiovascular Disease: A Systematic Review and Dose-Response Meta-analysis of Prospective Cohort Studies. Mayo Clin. Proc. 2019, 94, 2399–2414. [Google Scholar] [CrossRef] [Green Version]
- Schwingshackl, L.; Neuenschwander, M.; Hoffmann, G.; Buyken, A.E.; Schlesinger, S. Dietary sugars and cardiometabolic risk factors: A network meta-analysis on isocaloric substitution interventions. Am. J. Clin. Nutr. 2020, 111, 187–196. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): A prospective cohort study. Lancet 2017, 390, 2050–2062. [Google Scholar] [CrossRef] [Green Version]
- Grundy, S.M. Metabolic syndrome update. Trends Cardiovasc. Med. 2016, 26, 364–373. [Google Scholar] [CrossRef]
- Murthy, V.L.; Abbasi, S.A.; Siddique, J.; Colangelo, L.A.; Reis, J.; Venkatesh, B.A.; Carr, J.J.; Terry, J.G.; Camhi, S.M.; Jerosch-Herold, M.; et al. Transitions in Metabolic Risk and Long-Term Cardiovascular Health: Coronary Artery Risk Development in Young Adults (CARDIA) Study. J. Am. Heart Assoc. 2016, 5, e003934. [Google Scholar] [CrossRef]
- Pyorala, M.; Miettinen, H.; Halonen, P.; Laakso, M.; Pyorala, K. Insulin resistance syndrome predicts the risk of coronary heart disease and stroke in healthy middle-aged men: The 22-year follow-up results of the Helsinki Policemen Study. Arter. Thromb. Vsasc. Biol. 2000, 20, 538–544. [Google Scholar] [CrossRef] [Green Version]
- Lakka, H.M.; Laaksonen, D.E.; Lakka, T.A.; Niskanen, L.K.; Kumpusalo, E.; Tuomilehto, J.; Salonen, J.T. The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA 2002, 288, 2709–2716. [Google Scholar] [CrossRef]
- Hanley, A.J.; Karter, A.J.; Festa, A.; D’Agostino, R., Jr.; Wagenknecht, L.E.; Savage, P.; Tracy, R.P.; Saad, M.F.; Haffner, S.; Insulin Resistance Atherosclerosis Study. Factor analysis of metabolic syndrome using directly measured insulin sensitivity: The Insulin Resistance Atherosclerosis Study. Diabetes 2002, 51, 2642–2647. [Google Scholar] [CrossRef] [Green Version]
- Fryar, C.D.; Ostchega, Y.; Hales, C.M.; Zhang, G.; Kruszon-Moran, D. Hypertension Prevalence and Control Among Adults: United States, 2015–2016. Available online: https://www.cdc.gov/nchs/products/databriefs/db289.htm. (accessed on 24 March 2020).
- Leyva, F.; Godsland, I.F.; Worthington, M.; Walton, C.; Stevenson, J.C. Factors of the metabolic syndrome: Baseline interrelationships in the first follow-up cohort of the HDDRISC Study (HDDRISC-1). Heart Disease and Diabetes Risk Indicators in a Screened Cohort. Arter. Thromb. Vasc. Biol. 1998, 18, 208–214. [Google Scholar] [CrossRef] [Green Version]
- Anderson, P.J.; Critchley, J.A.; Chan, J.C.; Cockram, C.S.; Lee, Z.S.; Thomas, G.N.; Tomlinson, B. Factor analysis of the metabolic syndrome: Obesity vs insulin resistance as the central abnormality. Int. J. Obes. 2001, 25, 1782–1788. [Google Scholar] [CrossRef] [Green Version]
- Timar, O.; Sestier, F.; Levy, E. Metabolic syndrome X: A review. Can. J. Cardiol. 2000, 16, 779–789. [Google Scholar]
- Lemieux, S.; Despres, J.P. Metabolic complications of visceral obesity: Contribution to the aetiology of type 2 diabetes and implications for prevention and treatment. Diabete Metab. 1994, 20, 375–393. [Google Scholar]
- O’Neill, S.; O’Driscoll, L. Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies. Obes. Rev. 2015, 16, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Moller, D.E.; Kaufman, K.D. Metabolic syndrome: A clinical and molecular perspective. Annu. Rev. Med. 2005, 56, 45–62. [Google Scholar] [CrossRef]
- National Center for Chronic Disease Prevention and Health Promotion, Division for Heart Disease and Stroke Prevention. Heart Disease Facts. Available online: https://www.cdc.gov/heartdisease/facts.htm (accessed on 28 February 2020).
- Vaccarino, V.; McClure, C.; Johnson, B.D.; Sheps, D.S.; Bittner, V.; Rutledge, T.; Shaw, L.J.; Sopko, G.; Olson, M.B.; Krantz, D.S.; et al. Depression, the metabolic syndrome and cardiovascular risk. Psychosom. Med. 2008, 70, 40–48. [Google Scholar] [CrossRef]
- Mistler, B.J.; Reetz, D.R.; Krylowicz, B.; Barr, V. The Association for University and College Counseling Center Directors Annual Survey. Aurora 2014, 51, 60506. [Google Scholar]
- National Institutes of Mental Health. Major Depression, Statistics. Available online: https://www.nimh.nih.gov/health/statistics/major-depression.shtml (accessed on 28 February 2020).



| GROUP | FOOD GROUP ITEMS | Alpha |
|---|---|---|
| High-Fat Protein | Sausage, hotdog, beef steak, fried chicken, bologna, bacon | 0.79 |
| Refined Carbohydrate | Rice, bagels, pasta, cracker, pizza | 0.742 |
| Sweets/sugary beverages | Ice cream, cookies/cakes/pastries, cake icing, cheesecake, chocolate milk, soda, sweetened coffee drink | 0.723 |
| Healthy Fat/seafood | Tuna, salmon, baked fish, shrimp/other shellfish, olive oil | 0.716 |
| Fruit | Blueberry, melon, strawberry, mango, pineapple | 0.713 |
| Alcoholic beverages | Wine, scotch, dark beer | 0.689 |
| Vegetable | Broccoli, carrots, greens, sweet potato, mushrooms, tomatoes, tomato juice | 0.687 |
| Spicy/flavorful | Horseradish/wasabi, burn of a spicy meal, tabasco sauce, soy sauce, grapefruit juice, black coffee, dark chocolate | 0.663 |
| Saturated fat | Mayonnaise, whole milk, full fat dressing, cheddar cheese | 0.654 |
| Salty | Soup, lean ham, baked chicken, chips, salty pretzels, French fries | 0.608 |
| Complex carbohydrate | Whole wheat bread, oatmeal, shredded wheat cereal | 0.509 |
| Low-Fat Dairy | Low-fat cottage cheese, skim milk, plain yogurt | 0.435 |
| Patients (n = 106) | Students (n = 106) | T-Value, Chi Squared, or Mann-Whitney U Z | |
|---|---|---|---|
| Gender | χ2(1) = 0.321 | ||
| male (n = 80) | 39.6% | 35.8% | |
| female (n = 132) | 60.4% | 64.2% | |
| Age (years, mean ± SEM) | 21.5 ± 0.21 | 20.3 ± 0.13 | Z = 4.029 ** |
| Body mass index (kg/m2) (mean ± SEM) | 27.4 ± 0.9 | 23.2 ± 0.7 | Z = 3.388 ** |
| underweight (<18.5) (n = 9) | 4.7% | 3.8% | χ2(1) = 8.732 ** |
| normal (18.5–24.9) (n = 120) | 46.2% | 67% | |
| overweight (25–29.9) (n = 54) | 23.6% | 27.4% | |
| obese (>30) (n = 29) | 25.5% | 1.9% | |
| Waist: Hip Ratio (mean ± Sem) | 0.79 ± 0.6 | 0.86 ± 0.7 | Z = 7.60 *** |
| Blood pressure (BP) | |||
| diastolic mmHG (mean ± SEM) | 72.16 ± 1.07 | 71.20 ± 0.79 | Z = 0.75 |
| systolic mmHG (mean ± SEM) | 112.81± 1.58 | 110.56 ± 1.21 | Z = 0.98 |
| Total cholesterol mg/dL | 169.4 ± 3.47 | 166.3 ± 2.74 | T = −0.563 |
| LDL-C mg/dL † | 93.3 ± 3.01 | 88.8 ± 2.41 | T = −1.009 |
| HDL-C mg/DL † | 54.4 ± 1.49 | 61.2 ± 1.43 | T = 3.425 ** |
| cholesterol:HDL ratio † | 3.33 ± 0.11 | 2.85 ± 0.07 | T = −3.549 ** |
| Triglycerides mg/dL | 108.9 ± 5.93 | 82 ± 3.24 | T = −3.783 ** |
| Fasting blood glucose mg/dL | 91.4 ± 0.87 | 87.2 ± 0.77 | Z = 3.156 ** |
| Insulin mIU/L | 17.8 ± 2.27 | 9.6 ± 0.46 | Z = 3.723 ** |
| Variable | Fruit | Refined Carbohydrate | Sweet | Salty | Healthy Fat | Saturated Fat | Vegetable | High-Fat Protein | Complex Carbohydrate | Spicy/Flavorful | Alcohol | Low-Fat Dairy |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | 46.29 | 40.45 | 33.37 | 26.01 | 11.01 | 10.02 | 5.44 | 5.08 | 2.51 | −5.59 | −10.28 | −11.54 |
| Std. Dev. | 33.13 | 29.02 | 30.09 | 28.31 | 40.89 | 39.58 | 34.19 | 41.36 | 36.23 | 34.75 | 51.63 | 39.20 |
| Min | −96.5 | −61.2 | −94.25 | −79.66 | −96.8 | −97.5 | −96.16 | −100 | −100 | −87.28 | −100 | −100 |
| Max | 100 | 100 | 100 | 100 | 91.4 | 100 | 91 | 93.33 | 100 | 72.85 | 97.66 | 100 |
| Fruit | 1 | 0.06 | −0.06 | −0.01 | 0.13 | 0.03 | 0.43 *** | −0.13 | 0.34 *** | 0.21 ** | 0.18 * | 0.32 *** |
| Refined Carbohydrate | 1 | 0.67 *** | 0.55 *** | −0.002 | 0.55 *** | 0.003 | 0.28 *** | 0.048 | 0.036 | 0.10 | −0.004 | |
| Sweet | 1 | 0.467 *** | 0.041 | 0.590 *** | −0.144 * | 0.342 *** | −0.065 | 0.067 | 0.097 | 0.006 | ||
| Salty | 1 | 0.245 *** | 0.369 *** | −0.063 | 0.664 *** | −0.066 | 0.133 | 0.021 | 0.0015 | |||
| Healthy Fat | 1 | 0.133 | 0.276 *** | 0.326 *** | 0.164 * | 0.309 *** | 0.124 | 0.213 ** | ||||
| Saturated Fat | 1 | 0.041 | 0.375 *** | 0.0005 | 0.168 * | 0.135 | 0.185 ** | |||||
| Vegetable | 1 | −0.148 * | 0.447 *** | 0.393 *** | 0.125 | 0.335 *** | ||||||
| High-Fat Protein | 1 | −0.125 | 0.161 * | 0.086 | −0.057 | |||||||
| Complex Carbohydrate | 1 | 0.26 *** | 0.165 * | 0.417 *** | ||||||||
| Spicy/Flavorful | 1 | 0.305 *** | 0.392 *** | |||||||||
| Alcohol | 1 | 0.223 ** | ||||||||||
| Low-Fat Diary | 1 |
| Component | Maximum Points | Liking Score for Maximum Score | Liking Score for Minimum Score of Zero |
|---|---|---|---|
| Adequacy | |||
| Vegetables | 18 | 100 | −100 |
| Alcohol | 3 | 100 | −100 |
| Fruit | 2 | 100 | −100 |
| Moderation | |||
| Sweet, Fat, and Refined Carbohydrates | 52 | −100 | 100 |
| Complex Carbohydrates | 15 | −100 | 100 |
| Spicy/Flavorful | 4 | −100 | 100 |
| Low-Fat Diary | 3 | −100 | 100 |
| Salty and High Fat Protein | 2 | −100 | 100 |
| Healthy Fat | 1 | −100 | 100 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, R.; Blanchard, B.E.; McCaffrey, J.M.; Woolley, S.; Corso, L.M.L.; Duffy, V.B. Food Liking-Based Diet Quality Indexes (DQI) Generated by Conceptual and Machine Learning Explained Variability in Cardiometabolic Risk Factors in Young Adults. Nutrients 2020, 12, 882. https://doi.org/10.3390/nu12040882
Xu R, Blanchard BE, McCaffrey JM, Woolley S, Corso LML, Duffy VB. Food Liking-Based Diet Quality Indexes (DQI) Generated by Conceptual and Machine Learning Explained Variability in Cardiometabolic Risk Factors in Young Adults. Nutrients. 2020; 12(4):882. https://doi.org/10.3390/nu12040882
Chicago/Turabian StyleXu, Ran, Bruce E. Blanchard, Jeanne M. McCaffrey, Stephen Woolley, Lauren M. L. Corso, and Valerie B. Duffy. 2020. "Food Liking-Based Diet Quality Indexes (DQI) Generated by Conceptual and Machine Learning Explained Variability in Cardiometabolic Risk Factors in Young Adults" Nutrients 12, no. 4: 882. https://doi.org/10.3390/nu12040882
APA StyleXu, R., Blanchard, B. E., McCaffrey, J. M., Woolley, S., Corso, L. M. L., & Duffy, V. B. (2020). Food Liking-Based Diet Quality Indexes (DQI) Generated by Conceptual and Machine Learning Explained Variability in Cardiometabolic Risk Factors in Young Adults. Nutrients, 12(4), 882. https://doi.org/10.3390/nu12040882






