Dietary Supplements for Male Infertility: A Critical Evaluation of Their Composition
Abstract
1. Introduction
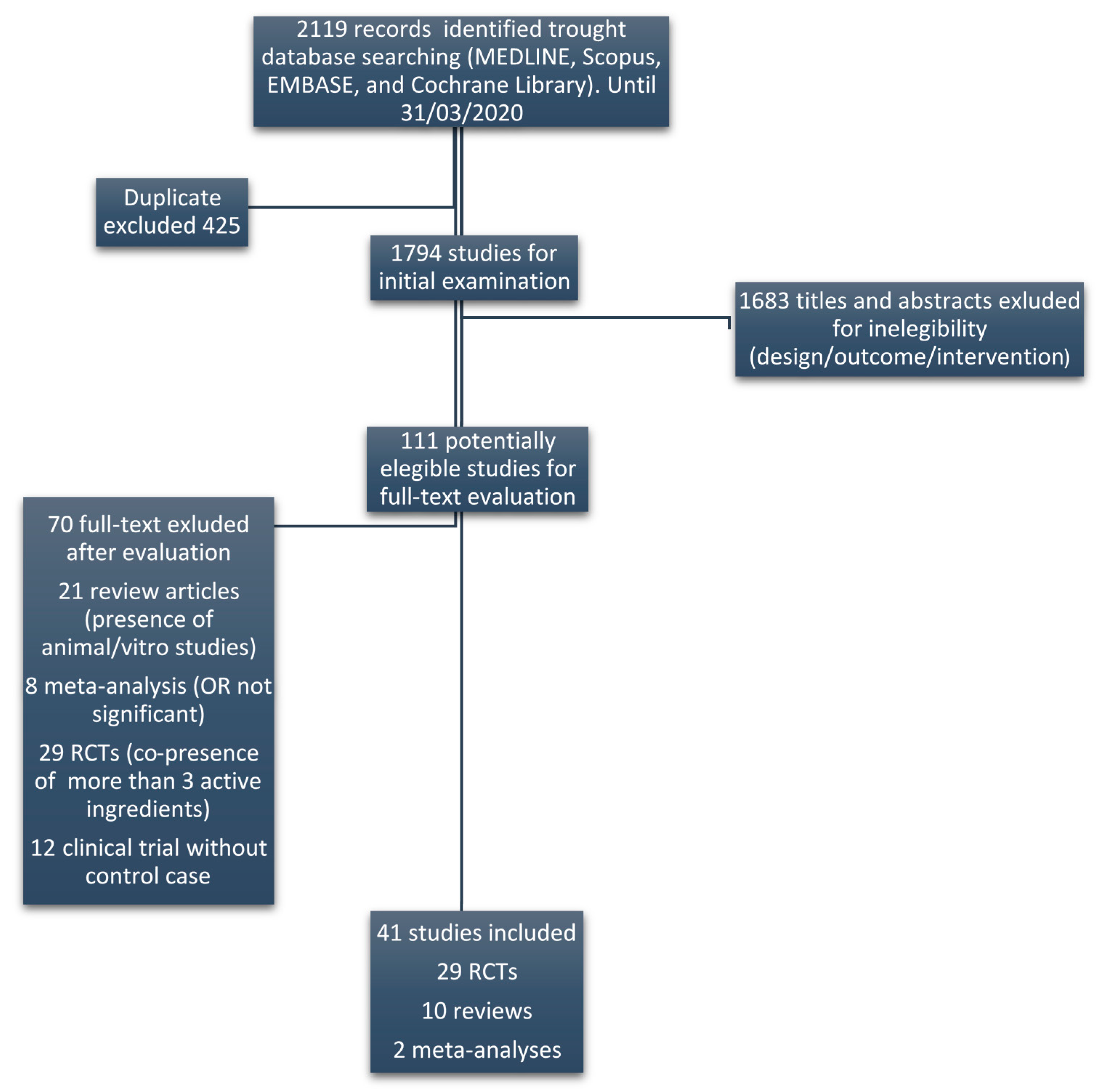
2. Materials and Methods
- (1)
- Each class of ingredients was given an arbitrary value: A = +2, B = +1 and C = −1;
- (2)
- These values were multiplied for the respective number of ingredients within each supplement (A, B and C respectively), obtaining a total score given by the sum of each category (2A + B − C);
- (3)
- As the number of ingredients highly differed between supplements, we standardized the above total score by dividing it for the maximum possible score for that supplement, by assuming that each ingredient was of class A (=2N, where N is the total number of ingredients in each supplement);
- (4)
- In order to correct this value for the number of ingredients of only categories A and B, the relative score was multiplied for the sum of high efficacy ingredients plus half (as a proxy of their lower efficacy) the number of moderate efficacy ingredients (A + B/2), finally obtaining a corrected score for each supplement.
- (5)
- Given the distribution of the scores resulted in three main clusters, we classified DS into three categories, resembling the efficacy of the ingredients: higher expected efficacy (corrected score ≥ 4), lower expected efficacy (4 < corrected score > 1) and no expected efficacy (corrected score ≤ 1).
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Task Force on Methods for the Regulation of Male Fertility. Contraceptive efficacy of testosterone-induced azoospermia in normal men. Lancet 1990, 336, 955–959. [Google Scholar] [CrossRef]
- Istituto Superiore di Sanità. Available online: http://old.iss.it/binary/rpma/cont/7_2017_Report.pdf (accessed on 20 March 2020).
- Winters, B.R.; Walsh, T.J. The Epidemiology of Male Infertility. Urol. Clin. N. Am. 2014, 41, 195–204. [Google Scholar] [CrossRef]
- Lotti, F.; Maggi, M. Ultrasound of the male genital tract in relation to male reproductive health. Hum. Reprod. Updat. 2014, 21, 56–83. [Google Scholar] [CrossRef] [PubMed]
- Ferlin, A.; Foresta, C. New genetic markers for male infertility. Curr. Opin. Obstet. Gynecol. 2014, 26, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Falsig, A.L.; Gleerup, C.S.; Knudsen, U.B. The influence of omega-3 fatty acids on semen quality markers: A systematic PRISMA review. Andrology 2019, 7, 794–803. [Google Scholar] [CrossRef] [PubMed]
- EUR-Lex. Available online: http://data.europa.eu/eli/reg/2006/1924/2012-11-29 (accessed on 20 March 2020).
- Cui, T.; Kovell, R.C.; Brooks, D.C.; Terlecki, R.P. A Urologist’s Guide to Ingredients Found in Top-Selling Nutraceuticals for Men’s Sexual Health. J. Sex. Med. 2015, 12, 2105–2117. [Google Scholar] [CrossRef]
- Henkel, R.; Sandhu, I.S.; Agarwal, A. The excessive use of antioxidant therapy: A possible cause of male inferility? Andrologia 2019, 51, 303–4569. [Google Scholar] [CrossRef]
- Calogero, A.E.; Aversa, A.; La Vignera, S.; Corona, G.; Ferlin, A. The use of nutraceuticals in male sexual and reproductive disturbances: Position statement from the Italian Society of Andrology and Sexual Medicine (SIAMS). J. Endocrinol. Investig. 2017, 40, 1389–1397. [Google Scholar] [CrossRef]
- Otten, J.J.; Hellwig, J.P.; Meyers, D.L. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; The National Academies Press: Washington, DC, USA, 2006; pp. 529–542. [Google Scholar]
- Bardelli, P.; Bardelli, M. Commento alle linee guida emanate dal Ministero della Salute sulle disfunzioni posturali. Med. Chir. Caviglia Piede 2019, 43. [Google Scholar] [CrossRef]
- Agarwal, A.; Majzoub, A. Antioxidant therapy in idiopathic oligoasthenoteratozoospermia. Indian J. Urol. 2017, 33, 207–214. [Google Scholar] [CrossRef]
- Ebisch, I.M.W.; Pierik, F.H.; De Jong, F.H.; Thomas, C.M.G.; Steegers-Theunissen, R.P.M. Does folic acid and zinc sulphate intervention affect endocrine parameters and sperm characteristics in men? Int. J. Androl. 2006, 29, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Azizollahi, G.; Azizollahi, S.; Babaei, H.; Kianinejad, M.; Baneshi, M.M.; Nematollahi-Mahani, S.N. Effects of supplement therapy on sperm parameters, protamine content and acrosomal integrity of varicocelectomized subjects. J. Assist. Reprod. Genet. 2013, 30, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Smits, R.M.; Mackenzie-Proctor, R.; Yazdani, A.; Stankiewicz, M.T.; Jordan, V.; Showell, M.G. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 2019, 3, CD007411. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Safarinejad, S. Efficacy of Selenium and/or N-Acetyl-Cysteine for Improving Semen Parameters in Infertile Men: A Double-Blind, Placebo Controlled, Randomized Study. J. Urol. 2009, 181, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A. Vitamin B12 and Semen Quality. Biomology 2017, 7, 42. [Google Scholar] [CrossRef] [PubMed]
- Kumamoto, Y.; Maruta, H.; Ishigami, J.; Kamidono, S.; Orikasa, S.; Kimura, M.; Yamanaka, H.; Kurihara, H.; Koiso, K.; Okada, K. Clinical efficacy of mecobalamin in the treatment of oligozoospermia—Results of double-blind comparative clinical study. Hinyokika kiyo. Acta Urol. Jpn. 1988, 34, 1109–1132. [Google Scholar]
- Moriyama, H.; Nakamura, K.; Sanda, N.; Fujiwara, E.; Seko, S.; Yamazaki, A.; Mizutani, M.; Sagami, K.; Kitano, T. Studies on the usefulness of a long-term, high-dose treatment of methylcobalamin in patients with oligozoospermia. Hinyokika kiyo. Acta Urol. Jpn. 1987, 33, 151–156. [Google Scholar]
- Calogero, A.E.; Gullo, G.; La Vignera, S.; Condorelli, R.A.; Vaiarelli, A. Myoinositol improves sperm parameters and serum reproductive hormones in patients with idiopathic infertility: A prospective double-blind randomized placebo-controlled study. Andrology 2015, 3, 491–495. [Google Scholar] [CrossRef]
- Wong, W.Y.; Merkus, H.M.W.M.; Thomas, C.M.G.; Menkveld, R.; Zielhuis, G.A.; Steegers-Theunissen, R.P. Effects of folic acid and zinc sulfate on male factor subfertility: A double-blind, randomized, placebo-controlled trial. Fertil. Steril. 2002, 77, 491–498. [Google Scholar] [CrossRef]
- Showell, M.G.; Brown, J.; Yazdani, A.; Stankiewicz, M.T.; Hart, R.J. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef]
- Stanislavov, R.; Nikolova, V.; Rohdewald, P. Improvement of seminal parameters with Prelox®: A randomized, double-blind, placebo-controlled, cross-over trial. Phytother. Res. 2009, 23, 297–302. [Google Scholar] [CrossRef]
- Nikolova, V.; Stanislavov, R.; Vatev, I.; Nalbanski, B.; Pŭnevska, M. Sperm parameters in male idiopathic infertility after treatment with prelox. Akush Ginekol (Sofiia) 2007, 46, 7–12. [Google Scholar] [PubMed]
- Stanislavov, R.; Rohdewald, P. Sperm quality in men is improved by supplementation with a combination of L-arginine, L-citrullin, roburins and Pycnogenol®. Minerva Urol. Nefrol. 2014, 66, 217–223. [Google Scholar]
- Haghighian, H.K.; Haidari, F.; Mohammadi-Asl, J.; Dadfar, M. Randomized, triple-blind, placebo-controlled clinical trial examining the effects of alpha-lipoic acid supplement on the spermatogram and seminal oxidative stress in infertile men. Fertil. Steril. 2015, 104, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Zhang, X.; Yang, F.; Li, J.; Yu, X.; Li, Y. Effect of oral alpha-lipoic acid (ALA) on the treatment of male infertility. Medicine (Baltimore) 2019, 98, e18453. [Google Scholar] [CrossRef] [PubMed]
- Mehni, N.M.; Ketabchi, A.A.; Hosseini, E. Combination effect of Pentoxifylline and L-carnitine on idiopathic oligoasthenoteratozoospermia. Iran. J. Reprod. Med. 2014, 12, 817–824. [Google Scholar]
- Peivandi, S.; Abasali, K.; Narges, M. Effects of L-carnitine on infertile men’s spermogram; a randomised clinical trial. J. Reprod. Infertil. 2010, 10, 331. [Google Scholar]
- Lenzi, A.; Lombardo, F.; Sgrò, P.; Salacone, P.; Caponecchia, L.; Dondero, F.; Gandini, L. Use of carnitine therapy in selected cases of male factor infertility: A double-blind crossover trial. Fertil. Steril. 2003, 79, 292–300. [Google Scholar] [CrossRef]
- Ahmadi, S.; Bashiri, R.; Ghadiri-Anari, A.; Nadjarzadeh, A. Antioxidant supplements and semen parameters: An evidence based review. Int. J. Reprod. BioMed. 2016, 14, 729–736. [Google Scholar] [CrossRef]
- Walczak-Jedrzejowska, R.; Wolski, J.K.; Slowikowska-Hilczer, J. The role of oxidative stress and antioxidants in male fertility. Central Eur. J. Urol. 2013, 66, 60–67. [Google Scholar] [CrossRef]
- Çiftçi, H.; Verit, A.; Savas, M.; Yeni, E.; Erel, O. Effects of N-acetylcysteine on Semen Parameters and Oxidative/Antioxidant Status. Urology 2009, 74, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Nadjarzadeh, A.; Sadeghi, M.R.; Amirjannati, N.; Vafa, M.; Motevalian, S.A.; Gohari, M.R.; Akhondi, M.A.; Yavari, P.; Shidfar, F. Coenzyme Q10 improves seminal oxidative defense but does not affect on semen parameters in idiopathic oligoasthenoteratozoospermia: A randomized double-blind, placebo controlled trial. J. Endocrinol. Investig. 2011, 34, 224–228. [Google Scholar]
- Safarinejad, M.R.; Safarinejad, S.; Shafiei, N.; Safarinejad, S. Effects of the Reduced Form of Coenzyme Q10 (Ubiquinol) on Semen Parameters in Men with Idiopathic Infertility: A Double-Blind, Placebo Controlled, Randomized Study. J. Urol. 2012, 188, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Safarinejad, M.R. Efficacy of Coenzyme Q10 on Semen Parameters, Sperm Function and Reproductive Hormones in Infertile Men. J. Urol. 2009, 182, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Comhaire, F.H.; El Garem, Y.; Mahmoud, A.; Eertmans, F.; Schoonjans, F. Combined conventional/antioxidant “Astaxanthin” treatment for male infertility: A double blind, randomized trial. Asian J. Androl. 2005, 7, 257–262. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, A.; Ronsini, S.; Notari, T.; Grieco, N.; Infante, V.; D’Angel, N.; Mascia, F.; Fiore, M.; Fisher, G. D-Aspartate, a Key Element for the Improvement of Sperm Quality. Adv. Sex. Med. 2012, 2, 45–53. [Google Scholar] [CrossRef]
- Ismail, S.B.; Bakar, M.B.; Nik Hussain, N.H.; Norhayati, M.N.; Sulaiman, S.A.; Jaafar, H.; Draman, S.; Ramli, R.; Wan Yusoff, W.Z. Comparison on the effects and safety of tualang honey and tribestaan in sperm parameters, erectile function and hormonal profile among oligospermia males. Evid.-Based Compliment. Altern. Med. 2014, 2014, 126138. [Google Scholar]
- Setiawan, L. Tribulus terrestris L. extract improves spermatozoa motility and increases the efficiency of acrosome reaction in subjects diagnosed with oligoastheno-teratozoospermia. Adv. Male. Reprod. Physiol. 1996, 2, 105–114. [Google Scholar]
- Sanagoo, S.; Oskouei, B.S.; Abdollahi, N.G.; Salehi-Pourmehr, H.; Hazhir, N.; Farshbaf-Khalili, A. Effect of Tribulus terrestris L. on sperm parameters in men with idiopathic infertility: A systematic review. Complement. Ther. Med. 2018, 42, 95–103. [Google Scholar] [CrossRef]
- Vazquez-Levin, M.H.; Verón, G.L. Myo-inositol in health and disease: Its impact on semen parameters and male fertility. Andrology 2019, 8, 277–298. [Google Scholar] [CrossRef]
- Condorelli, R.A.; La Vignera, S.; Mongioì, L.M.; Vitale, S.G.; Laganà, A.S.; Cimino, L.; Calogero, A.E. Myo-inositol as a male fertility molecule: Speed them up! Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 30–35. [Google Scholar] [PubMed]
- Omu, A.E.; Al-Azemi, M.; Kehinde, E.; Anim, J.; Oriowo, M.; Mathew, T. Indications of the Mechanisms Involved in Improved Sperm Parameters by Zinc Therapy. Med Princ. Pr. 2008, 17, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Suleiman, S.A.; Ali, M.E.; Zaki, Z.M.; El-Malik, E.M.; Nasr, M.A. Lipid peroxidation and human sperm motility: Protective role of vitamin E. J. Androl. 1996, 17, 530–537. [Google Scholar] [PubMed]
- Greco, E.; Iacobelli, M.; Ferrero, S.; Tesarik, J.; Rienzi, L.; Ubaldi, F. Reduction of the Incidence of Sperm DNA Fragmentation by Oral Antioxidant Treatment. J. Androl. 2005, 26, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Cyrus, A.; Kabir, A.; Goodarzi, D.; Moghimi, M. The effect of adjuvant vitamin C after varicocele surgery on sperm quality and quantity in infertile men: A double blind placebo controlled clinical trial. Int. Braz. J. Urol. 2015, 41, 230–238. [Google Scholar] [CrossRef]
- Dawson, E.B.; Harris, W.A.; Powell, L.C. Relationship between ascorbic acid and male fertility. World Rev. Nutr. Diet. 1990, 62, 1–26. [Google Scholar]
- Safarinejad, M.R. MP-11.10: Effect of Omega-3 Polyunsaturated Fatty Acid Supplementation on Semen Profile and Enzymatic Anti-Oxidant Capacity of Seminal Plasma in Infertile Men with Idiopathic Oligoasthenoteratospermia: A Double Blind, Placebo-Controlled, Randomized Study. Urology 2009, 74, S96. [Google Scholar] [CrossRef]
- Martinez-Soto, J.; Domingo, J.; Cordobilla, B.; Palbero, L.; Pellicer, A.; Landeras, J. Effect of dietary DHA supplementation on sperm DNA integrity. Fertil. Steril. 2010, 94, S235–S236. [Google Scholar] [CrossRef]
- Gupta, N.P.; Kumar, R. Lycopene therapy in idiopathic male infertility–A preliminary report. Int. Urol. Nephrol. 2002, 34, 369–372. [Google Scholar] [CrossRef]
- Agarwal, A.; Durairajanayagam, D.; Ong, C.; Prashast, P. Lycopene and male infertility. Asian J. Androl. 2014, 16, 420–425. [Google Scholar] [CrossRef]
- Nassan, F.L.; Chavarro, J.E.; Tanrikut, C. Diet and men’s fertility: Does diet affect sperm quality? Fertil. Steril. 2018, 110, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Terai, K.; Horie, S.; Fukuhara, S.; Miyagawa, Y.; Kobayashi, K.; Tsujimura, A. Combination therapy with antioxidants improves total motile sperm counts: A Preliminary Study. Reprod. Med. Boil. 2019, 19, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Kumalic, S.I.; Pinter, B. Review of Clinical Trials on Effects of Oral Antioxidants on Basic Semen and Other Parameters in Idiopathic Oligoasthenoteratozoospermia. BioMed Res. Int. 2014, 2014, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Keszthelyi, M.; Sofikitis, N. Administration of Antioxidants in the Infertile Male: When it may have a Beneficial Effect? Curr. Pharm. Des. 2020, 26, 1. [Google Scholar] [CrossRef]
- Federsalus. Available online: https://www.federsalus.it/il-mercato-degli-integratori-dinamiche-aggiornate-a-marzo-2019/ (accessed on 20 March 2020).
- Ronis, M.J.; Pedersen, K.B.; Watt, J. Adverse Effects of Nutraceuticals and Dietary Supplements. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 583–601. [Google Scholar] [CrossRef]
- Szewczyk, K.; Zidorn, C. Ethnobotany, phytochemistry, and bioactivity of the genus Turnera (Passifloraceae) with a focus on damiana—Turnera diffusa. J. Ethnopharmacol. 2014, 152, 424–443. [Google Scholar] [CrossRef]
- Jung, H.A.; Roy, A.; Jung, J.H.; Choi, J.S. Evaluation of the inhibitory effects of eckol and dieckol isolated from edible brown alga Eisenia bicyclis on human monoamine oxidases A and B. Arch. Pharmacal Res. 2017, 40, 480–491. [Google Scholar] [CrossRef]
- Naderi, N.; House, J.D. Recent Developments in Folate Nutrition. Adv. Food Nutr. Res. 2018, 83, 195–213. [Google Scholar] [CrossRef]
- Najafipour, R.; Moghbelinejad, S.; Aleyasin, A.; Jalilvand, A. Effect of B9 and B12 vitamin intake on semen parameters and fertility of men with MTHFR polymorphisms. Andrology 2017, 5, 704–710. [Google Scholar] [CrossRef]
- Qi, Y.-N.; Ma, J.; Han, R.-Y.; Liu, W.-J.; Wang, S.-S. Correlation of the levels of seminal plasma homocysteine, folate and cobalamin with semen parameters in obese men. Zhonghua Nan Ke Xue 2018, 24, 883–886. [Google Scholar]
- Duncan, A.; Yacoubian, C.; Watson, N.; Morrison, I. The risk of copper deficiency in patients prescribed zinc supplements. J. Clin. Pathol. 2015, 68, 723–725. [Google Scholar] [CrossRef] [PubMed]
- Foresta, C.; Flohé, L.; Garolla, A.; Roveri, A.; Ursini, F.; Maiorino, M. Male fertility is linked to the selenoprotein phospholipid hydroperoxide glutathione peroxidase. Boil. Reprod. 2002, 67, 967–971. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M. Do micronutrient deficiencies contribute to mitochondrial failure in critical illness? Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Garolla, A.; Maiorino, M.; Roverato, A.; Roveri, A.; Ursini, F.; Foresta, C. Oral carnitine supplementation increases sperm motility in asthenozoospermic men with normal sperm phospholipid hydroperoxide glutathione peroxidase levels. Fertil. Steril. 2005, 83, 355–361. [Google Scholar] [CrossRef]
- Zhang, X.; Cui, Y.; Dong, L.; Sun, M.; Zhang, Y. The efficacy of combined l-carnitine and l-acetyl carnitine in men with idiopathic oligoasthenoteratozoospermia: A systematic review and meta-analysis. Andrology 2019, 52, e13470. [Google Scholar] [CrossRef]
- Foresta, C.; Garolla, A.; Cosci, I.; Menegazzo, M.; Ferigo, M.; Gandin, V.; De Toni, L. Role of zinc trafficking in male fertility: From germ to sperm. Hum. Reprod. 2014, 29, 1134–1145. [Google Scholar] [CrossRef]
- Fallah, A.; Mohammad-Hasani, A.; Colagar, A.H. Zinc is an Essential Element for Male Fertility: A Review of Zn Roles in Men’s Health, Germination, Sperm Quality, and Fertilization. J. Reprod. Infertil. 2018, 19, 69–81. [Google Scholar]
- Manish, K.; Yash, S.; Premal, P.; Neel, P.; Ranjith, R. A Systematic Review and Evidence-based Analysis of Ingredients in Popular Male Fertility Supplements. Urology 2020, 136, 133–141. [Google Scholar]
- Budoff, M.J.; Achenbach, S.; Blumenthal, R.S.; Carr, J.J.; Goldin, J.; Greenland, P.; Guerci, A.D.; Lima, J.A.; Rader, D.J.; Rubin, G.D.; et al. Assessment of Coronary Artery Disease by Cardiac Computed Tomography. Circulation 2006, 114, 1761–1791. [Google Scholar] [CrossRef]

| Active Ingredients | References | Evaluated Sperm Parameters | Employed Daily Dose | Minimal Effective Dose (mED) |
|---|---|---|---|---|
| Zinc | [13] * (Rev) | concentration | 50 mg | 50 mg |
| [14] * (RCT) | concentration | 66 mg | ||
| [15] * (RCT) | morphology | 66 mg | ||
| Selenium | [13] (Rev) | linear progression | 50 µg | 50 µg |
| [16] (Met) | concentration | 100 µg | ||
| [17] (RCT) | concentration/motility | 200 µg | ||
| Vitamin B12 | [18] (Rev) | count | 25 µg | 25 µg |
| [19] (RCT) | count | 1500 µg | ||
| [20] (RCT) | count | 6000 µg | ||
| Folic Acid | [21] (RCT) | count/motility | 400 µg | 400 µg |
| [22] (RCT) | volume/motility | 500 µg | ||
| [23] (Met) | DNA damage | 500 µg | ||
| L-Arginine | [24] (RCT) | progressive motility | 1.4 g | 1.4 g |
| [25] (RCT) | concentration/motility | 1.4 g | ||
| L-Citrulline | [26] (RCT) | volume/concentration motility/vitality | 1.2 g | 1.2 g |
| α-Lipoic Acid | [27] (RCT) | concentration/motility | 600 mg | 600 mg |
| [28] (Rev) | motility/morphology | 600 mg | ||
| L-Carnitine (LC/LAC) | [29] (RCT) | motility | 1 g | 1 g |
| [30] (RCT) | count/motility | 2 g | ||
| [31] (RCT) | concentration/motility | 2 g | ||
| [32] (Rev) | motility | 3 g | ||
| N-Acetyl Cysteine (NAC) | [33] (Rev) | motility/DNA damage | 600 mg | 600 mg |
| [34] (RCT) | motility/DNA damage | 600 mg | ||
| [17] (RCT) | motility/morphology | 600 mg | ||
| Coenzyme Q10 | [35] (RCT) | motility | 200 mg | 200 mg |
| [36] (RCT) | count/motility | 200 mg | ||
| [37] (RCT) | concentration/morphology | 300 mg | ||
| Astaxanthin | [38] (RCT) | motility | 16 mg | 16 mg |
| D-Aspartic Acid (DAA) | [39] (RCT) | count/motility | 2.7 g | 2.7 g |
| Tribulus Terrestris DE | [40] (RCT) | count/motility | 250 mg | 250 mg |
| [41] (RCT) | morphology/motility | 500 mg | ||
| [42] (Rev) | count/morphology | 6000 mg | ||
| Myoinositol | [43] (Rev) | motility | 2 g | 2 g |
| [44] (Rev) | concentration | 4 g | ||
| α-Tocopherol | [45] (RCT) | motility/DNA damage | 20 mg | 20 mg |
| [32] (Rev) | motility/morphology | 268.46 mg | ||
| [46] (RCT) | motility/lipid oxidation | 300 mg | ||
| [47] * (RCT) | DNA damage | 1000 mg | ||
| Vitamin C | [48] (RCT) | concentration/motility DNA damage | 0.5 g | 0.5 g |
| [49] (Rev) | 1 g | |||
| [47] (RCT) | 1 g | |||
| EPA + DHA EPA + DHA | [50] (RCT) | concentration/motility | 0.72 g + 0.48 g | DHA 0.48 g |
| [51] (RCT) | DNA damage | 0.14 g + 1 g | ||
| Lycopene | [52] (RCT) | concentration/motility | 4 mg | 4 mg |
| [53] (Rev) | count/morphology | 8 mg |
| Astragalus DE |
| Damiana DE |
| Nettle DE |
| Catuba DE |
| Ecklonia bicyclis DE |
| L-Taurine |
| Glutathione |
| Glucosamine |
| SOD |
| Vitamin D3 |
| Vitamin B1 |
| Riboflavin |
| Niacin |
| Vitamin B5 |
| Vitamin B6 |
| Biotin |
| Manganese |
| Active Ingredients | DS 1 | DS 2 | DS 3 | DS 4 | DS 5 | DS 6 | DS 7 | |||||||
| S = 3.12 | S = 2.08 | S = 3.66 | S = 0.16 | S = 2.1 | S = 2.25 | S = 3.37 | ||||||||
| Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | |
| Zinc | 7.5 mg | B | 10 mg | B | 12.5 mg | B | 1.5 mg | B | 13 mg | B | ||||
| Selenium | 60 µg | A | 83 µg | A | 30 µg | B | 55 µg | A | ||||||
| Vitamin B12 | 33 mg | A | ||||||||||||
| Folic Acid | 200 µg | B | 400 µg | A | 400 µg | A | 200 µg | B | ||||||
| L-Arginine | 100 mg | B | 1000 mg | B | 2500 mg | A | 125 mg | B | 30 mg | B | ||||
| L-Citrulline | ||||||||||||||
| α-Lipoic Acid | 50 mg | B | ||||||||||||
| L-Carnitine | 1000 mg | A | 200 mg | B | 30 mg | B | ||||||||
| N-Acetyl Cysteine (NAC) | ||||||||||||||
| Coenzyme Q10 | 10 mg | B | 200 mg | A | 10 mg | B | 200 mg | A | 7.5 mg | B | ||||
| Astaxanthin | 15 mg | B | ||||||||||||
| D-Aspartic Acid (DAA) | 2660 mg | A | ||||||||||||
| Tribulus terrestris DE | 800 mg | A | ||||||||||||
| Myoinositol | 1000 mg | A | ||||||||||||
| α-Tocopherol | 30 mg | A | 12 mg | B | 30 mg | A | 36 mg | A | 30 mg | A | ||||
| Vitamin C | 60 mg | B | 180 mg | B | ||||||||||
| DHA | ||||||||||||||
| Lycopene | 15 mg | A | ||||||||||||
| Astragalus DE | 300 mg | C | ||||||||||||
| Damiana DE | ||||||||||||||
| Nettle DE | ||||||||||||||
| Catuba DE | ||||||||||||||
| Ecklonia bicyclis DE | ||||||||||||||
| L-Taurine | 500 mg | C | ||||||||||||
| Glutathione | 30 mg | C | 40 mg | C | ||||||||||
| Glucosamine | ||||||||||||||
| SOD | 154 UI | C | ||||||||||||
| Vitamin D3 | ||||||||||||||
| Vitamin B1 | ||||||||||||||
| Vitamin B2 | 25 mg | C | ||||||||||||
| Vitamin B3 | 36 mg | C | ||||||||||||
| Vitamin B5 | ||||||||||||||
| Vitamin B6 | 9.5 mg | C | ||||||||||||
| Biotin | ||||||||||||||
| Manganese | ||||||||||||||
| Active Ingredients | DS 8 | DS 9 | DS 10 | DS 11 | DS 12 | DS 13 | DS 14 | |||||||
| S = 1 | S = 2.81 | S = 2.66 | S = 0.5 | S = 1.14 | S = 4.32 | S = 2.5 | ||||||||
| Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | |
| Zinc | 40 mg | B | 12.5 mg | B | 10 mg | B | 15 mg | B | ||||||
| Selenium | 60 µg | A | 55 µg | A | 55 µg | A | 83 µg | A | ||||||
| Vitamin B12 | 2.5 µg | B | 5 µg | B | ||||||||||
| Folic Acid | 800 µg | A | 400 µg | A | 200 µg | B | ||||||||
| L-Arginine | 200 mg | B | 250 mg | B | 100 mg | B | 30 mg | B | ||||||
| L-Citrulline | 800 mg | B | ||||||||||||
| α-Lipoic Acid | 300 mg | B | 800 mg | A | ||||||||||
| L-Carnitine | 200 mg | B | 400 mg | B | 500 mg | B | 30 mg | B | ||||||
| N-Acetyl Cysteine (NAC) | 300 mg | B | 600 mg | A | ||||||||||
| Coenzyme Q10 | 15 mg | B | 15 mg | B | 100 mg | B | 20 mg | B | 200 mg | A | ||||
| Astaxanthin | ||||||||||||||
| D-Aspartic Acid (DAA) | 80 mg | B | ||||||||||||
| Tribulus terrestris DE | 300 mg | A | ||||||||||||
| Myoinositol | 1000 mg | A | 100 mg | B | 1000 mg | A | 1000 mg | A | ||||||
| α-Tocopherol | 30 mg | A | 120 mg | A | 30 mg | A | 30 mg | A | ||||||
| Vitamin C | ||||||||||||||
| DHA | ||||||||||||||
| Lycopene | 4 mg | A | ||||||||||||
| Astragalus DE | ||||||||||||||
| Damiana DE | ||||||||||||||
| Nettle DE | ||||||||||||||
| Catuba DE | 50 mg | C | ||||||||||||
| Ecklonia bicyclis DE | 200 mg | C | ||||||||||||
| L-Taurine | ||||||||||||||
| Glutathione | 80 mg | C | ||||||||||||
| Glucosamine | 150 mg | C | ||||||||||||
| SOD | ||||||||||||||
| Vitamin D3 | ||||||||||||||
| Vitamin B1 | 1.1 mg | C | ||||||||||||
| Vitamin B2 | 1.4 mg | C | 2.8 mg | C | ||||||||||
| Vitamin B3 | 16 mg | C | ||||||||||||
| Vitamin B5 | 6 mg | C | ||||||||||||
| Vitamin B6 | 1.4 mg | C | 2.8 mg | C | ||||||||||
| Biotin | 100 µg | C | ||||||||||||
| Manganese | 2 mg | C | ||||||||||||
| Active Ingredients | DS 15 | DS 16 | DS 17 | DS 18 | DS 19 | DS 20 | DS 21 | |||||||
| S = 4.33 | S = 2.45 | S = 2.06 | S = 2.06 | S = 1 | S = 1.05 | S = 2 | ||||||||
| Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | Daily Dose | EV | |
| Zinc | 15 mg | B | 22.5 mg | B | 10 mg | B | 10 mg | B | 6.5 mg | B | 10 mg | B | ||
| Selenium | 50 µg | A | 80 µg | A | 50 µg | A | 55 µg | A | ||||||
| Vitamin B12 | 2.5 µg | B | 1.5 µg | B | ||||||||||
| Folic Acid | 400 µg | A | 300 µg | B | 200 µg | B | 200 µg | B | 400 µg | A | ||||
| L-Arginine | 2500 mg | A | 200 mg | B | 30 mg | B | ||||||||
| L-Citrulline | 3000 mg | A | 200 mg | B | ||||||||||
| α-Lipoic Acid | ||||||||||||||
| L-Carnitine | 1000 mg | A | 200 mg | B | 400 mg | B | 44,7 mg | B | ||||||
| N-Acetyl Cysteine (NAC) | ||||||||||||||
| Coenzyme Q10 | 200 mg | A | 100 mg | B | 90 mg | B | ||||||||
| Astaxanthin | 16 mg | A | 10 mg | B | ||||||||||
| D-Aspartic Acid (DAA) | 1000 mg | B | ||||||||||||
| Tribulus terrestris DE | ||||||||||||||
| Myoinositol | 50 mg | B | 500 mg | B | 4000 mg | A | ||||||||
| α-Tocopherol | 40 mg | A | 30 mg | A | 12 mg | B | ||||||||
| Vitamin C | 80 mg | B | 100 mg | B | 180 mg | B | ||||||||
| DHA | 100 mg | B | ||||||||||||
| Lycopene | 10 mg | A | ||||||||||||
| Astragalus DE | ||||||||||||||
| Damiana DE | 400 mg | C | ||||||||||||
| Nettle DE | 300 mg | C | ||||||||||||
| Catuba DE | ||||||||||||||
| Ecklonia bicyclis DE | ||||||||||||||
| L-Taurine | 300 mg | B | ||||||||||||
| Glutathione | 40 mg | C | 40 mg | C | ||||||||||
| Glucosamine | ||||||||||||||
| SOD | 150 mg | C | ||||||||||||
| Vitamin D3 | 3.75 µg | C | ||||||||||||
| Vitamin B1 | ||||||||||||||
| Vitamin B2 | ||||||||||||||
| Vitamin B3 | ||||||||||||||
| Vitamin B5 | ||||||||||||||
| Vitamin B6 | ||||||||||||||
| Biotin | ||||||||||||||
| Manganese | ||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garolla, A.; Petre, G.C.; Francini-Pesenti, F.; De Toni, L.; Vitagliano, A.; Di Nisio, A.; Foresta, C. Dietary Supplements for Male Infertility: A Critical Evaluation of Their Composition. Nutrients 2020, 12, 1472. https://doi.org/10.3390/nu12051472
Garolla A, Petre GC, Francini-Pesenti F, De Toni L, Vitagliano A, Di Nisio A, Foresta C. Dietary Supplements for Male Infertility: A Critical Evaluation of Their Composition. Nutrients. 2020; 12(5):1472. https://doi.org/10.3390/nu12051472
Chicago/Turabian StyleGarolla, Andrea, Gabriel Cosmin Petre, Francesco Francini-Pesenti, Luca De Toni, Amerigo Vitagliano, Andrea Di Nisio, and Carlo Foresta. 2020. "Dietary Supplements for Male Infertility: A Critical Evaluation of Their Composition" Nutrients 12, no. 5: 1472. https://doi.org/10.3390/nu12051472
APA StyleGarolla, A., Petre, G. C., Francini-Pesenti, F., De Toni, L., Vitagliano, A., Di Nisio, A., & Foresta, C. (2020). Dietary Supplements for Male Infertility: A Critical Evaluation of Their Composition. Nutrients, 12(5), 1472. https://doi.org/10.3390/nu12051472







