Abstract
Attention deficit hyperactivity disorder (ADHD) is a common neurodevelopmental disorder in children and adolescents, with environmental and biological causal influences. Pharmacological medication is the first choice in ADHD treatment; recently, many studies have concentrated on dietary supplementation approaches to address nutritional deficiencies, to which part of non-responses to medications have been imputed. This review aims to evaluate the efficacy of non-pharmacological supplementations in children or adolescents with ADHD. We reviewed 42 randomized controlled trials comprised of the following supplementation categories: polyunsaturated fatty acids (PUFAs), peptides and amino acids derivatives, single micronutrients, micronutrients mix, plant extracts and herbal supplementations, and probiotics. The reviewed studies applied heterogeneous methodologies, thus making it arduous to depict a systematic overview. No clear effect on single cognitive, affective, or behavioral domain was found for any supplementation category. Studies on PUFAs and micronutrients found symptomatology improvements. Peptides and amino acids derivatives, plant extracts, herbal supplementation, and probiotics represent innovative research fields and preliminary results may be promising. In conclusion, such findings, if confirmed through future research, should represent evidence for the efficacy of dietary supplementation as a support to standard pharmacological and psychological therapies in children and adolescents with ADHD.
1. Introduction
Attention deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder affecting about 5% of children and 2.5% of adults worldwide. It is characterized by dysregulated cognition and behaviors, resulting in inattention, excessive motor activity, and impulsivity [1].
Drug treatments for ADHD mainly act to potentiate the action of catecholamines, which are neurotransmitters involved in the prefrontal cortex responsible for the maintenance of attention and cognitive control [2].
Stimulants, the first choice for ADHD drug treatment, include methylphenidate that inhibits dopamine reuptake and amphetamines that inhibit dopamine and noradrenaline reuptake and increase dopamine release. Atomoxetine is the most common non-stimulant medication for ADHD and acts by inhibiting norepinephrine reuptake [3].
The efficacy of stimulants and non-stimulants in reducing ADHD symptoms is well documented in children and adults [4,5]. However, the tolerability of these drugs can be scarce in some patients, warranting treatment interruption. At least 10–30% of children with ADHD may not benefit from these medications due to non-response or adverse effects [6,7] such as decreased appetite, insomnia, stomachache, headache, weight loss (potentially leading to growth retardation), tics, increases in blood pressure, and potential abuse or misuse [8]. Although less effective than stimulants, non-stimulant medication are first choice treatments for individuals with co-diagnoses including tic, mood, and anxiety disorders [3]. However, atomoxetine use has been associated with increased risk of suicidal behavior in youths [9].
ADHD is a multifactorial disorder, in which genetic and biological factors have important roles; ADHD symptomatology expression is also influenced by environmental factors, like nutrition [10]. To explain the role of nutritional deficiencies, to which part of non-responses to medications have been imputed, several studies focused on supplementation approaches, as alternative or additional therapies.
Within this research area, many studies have focused on macronutrient supplementations, mainly polyunsaturated fatty acids (PUFAs), that are precursors of eicosanoids and are relevant components of cell membranes and of phospholipids. PUFAs influence the quality of growth and development [11]. Research about PUFAs adjunctive treatments for ADHD did not produce convincing evidence, probably due to the heterogeneity of methods and doses administered [12,13].
Few studies have also examined the role of other macronutrients such as peptides and amino acids derivatives, which can act as metabolic adjuvants. In particular, there are experimental trials using Acetyl-L-carnitine (ALC) to optimize mitochondrial fatty acid oxidation. It has been reported that ALC treatment increased sodium levels in the cingulate cortex and the 5HIAA/5HT ratio in both cingulate cortex and medial frontal cortex in adolescent impulsive rats [14] and it could have a link with hyperactivity and impulsivity symptoms in children with ADHD [15]. L-carnosine (a dipeptide composed of β-alanine and l-histidine) is known as an antiaging antioxidant and neuroprotective compound, and it is found highly concentrated in brain tissues [16]. L-carnosine has also been associated to ADHD symptoms [17]: it accumulates in the subfrontal cortex and may enhance frontal lobe functions [18], interesting for ADHD research. Lastly, Phosphatidylserine (PS), a naturally occurring phospholipid, modulates the activity of receptors, enzymes, ion channels and signaling molecules, and contributes to regulate membrane fluidity [19]. PS has been shown to counteract the stress-induced activation of the hypothalamic-pituitary-adrenal axis [20]; it has been involved in ADHD symptomatology and short-term auditory memory [21].
Other studies have investigated the role of various micronutrients (e.g., oligoelements, vitamins, minerals) as alternative or supporting non-pharmacological treatment for ADHD.
Vitamin D deficiency is relevant in the pathogenesis of psychiatric disorders including autism, depression, schizophrenia, and ADHD [22,23,24]. Vitamin D can act on central dopamine levels via different mechanisms, including calcium transition, antioxidant properties and gene expression [25].
Another micronutrient linked to dopamine metabolism is zinc, a cofactor of many metalloenzymes and metal–enzyme complexes [26]. Studies conducted on animals and humans associated zinc deficiency and hyperactivity [27,28]. Researchers also focused on broad-spectrum supplementation approaches, highlighting the efficacy of minerals and vitamins combinations [29].
Complementary or alternative medicine approaches propose herbal extracts for children with ADHD. However, these studies have not yet produced structured evidence [30].
Lastly, an interesting hypothesis is that gut microbiota and probiotics could influence brain activity and behaviors and psychiatric symptoms [31]. In rodent studies, modifications of gut microbiota are associated with cerebral signals modification, in cortical and subcortical regions, linked to affective and sensory functions [32]. Evidence in humans is preliminary; however, chronic intake of probiotics is associated in healthy adults with altered brain connectivity during affective and attentive tasks [32].
Given the number and heterogeneity of studies conducted on nutritional supplementation in ADHD, the present review aims at offering a systematization for the results of studies conducted from January 2010 to March 2020 and evaluating the efficacy of non-pharmacological dietary supplementations in ADHD.
We defined the present review as “critical” to highlight our aim to offer clinicians an analytical and up-to-date point of view in the clinical management of children and adolescents with ADHD, specifically in a nutritional supplementation framework alone or in combination with pharmacological treatment. Moreover, this review did not have a systematic or quantitative objective.
2. Materials and Methods
The present review is reported according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) [33]. We searched PubMed from January 2010 to March 2020 using the following string: (probiotic OR prebiotic OR vitamin OR mineral OR phytonutrient OR amino acid OR supplementation OR non-pharmacological) AND (ADHD OR attention deficit hyperactivity disorder) NOT review [All Fields] NOT adult NOT infant. The search manually completed with relevant articles. We included randomized controlled trials involving children and adolescents with ADHD receiving non-pharmacological supplementation. We focused on studies addressing cognitive or behavioral outcomes; we excluded papers not written in English, researches addressing other diagnoses than ADHD, studies with no administration of non-pharmacological supplementations, studies that did not report behavioral/cognitive outcomes, and studies on preschool or adult subjects. Specifically, our search did not include infants (<5 years) given ADHD onset which typically corresponds to school age [1]. Moreover, since this review aim was to offer evidences regarding clinical management of children and adolescents with ADHD, we decided to exclude adults in the PubMed search.
3. Results
Our search found 414 articles. After title and abstract screening, articles retained were 44; we excluded two articles after full text review. In total, 42 articles are included in the present review. The PRISMA flow chart is shown in Figure 1.
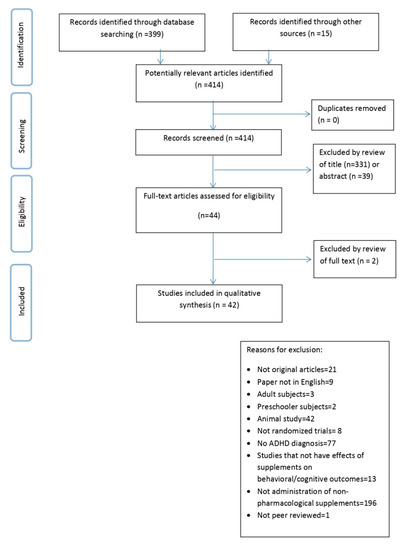
Figure 1.
Preferred reporting items for systematic reviews and meta-analyses (PRISMA).Flow diagram of the study selection process.
In the following text and tables, results are presented according to supplementation categories as follows:
- PUFAs;
- peptides and amino acids derivatives;
- single micronutrient (Zinc or Vitamin D);
- micronutrients mix;
- plant extracts or herbal supplementations;
- probiotics.
3.1. PUFAs
3.1.1. Methodologies
In total, 20 studies focused on supplementation with PUFAs [34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53] (see Table 1a): fourteen were on patients without comorbidities [34,36,37,38,40,41,42,43,44,45,46,47,50,53], two with oppositional defiant disorder (ODD) [35,39], and four with various co-diagnoses, such as specific learning disorder, ODD, conduct disorder, tic disorder, anxiety, Tourette syndrome, or psychomotor difficulties [48,49,51,52]. Two researches [42,43] include very small samples.

Table 1.
(a) Studies comprising supplementation with polyunsaturated fatty acids (PUFAs). (b) Studies comprising supplementation with peptides and amino acids derivatives. (c) Studies comprising supplementation with a single micronutrient. (d) Study comprising supplementation with micronutrients mix. (e) Studies comprising supplementation with plant or herbal extracts. (f) Study comprising supplementation with probiotics.
These studies supplemented with various omega-3 fatty acids. In total, 18 studies provided docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) [34,35,36,37,38,39,40,41,42,44,45,46,47,48,49,50,52,53], one study provided alpha linolenic acid (ALA) [43], and one study did not specify which omega-3 was used. Three studies supplemented omega-6: one used gamma-linolenic acid (GLA) [36], while two did not specify which omega-6 was used [35,51]. One study provided, together with EPA and DHA, conjugated linoleic acid (CLA), a precursor of a distinct family of PUFAs (both omega-3 and omega-6) [47]. In two linked studies, authors prescribed linoleic acid (LA) omega-6 as a control condition compared to two omega-3 groups [48,49]. Seven studies added vitamins to EPA and DHA [40,44,47,48,49,52,53]; each of the seven provided vitamin E, whereas in Cornu et al. added vitamins A and D [40]. Six studies allowed pharmacotherapy concomitant with PUFA supplementation [34,35,36,37,50,52], while two added to EPA and DHA supplementation with PS [46] or docosapentaenoic acid [52]. Although PUFAs represent now the most used supplementation in this field, there is too much heterogeneity around the specific PUFAs used. It remains unclear if the efficacy of results depend on individual type of omega-3/6 or the whole of macronutrients.
Randomization between treatment and placebo groups is equal, except for one study [46], but there is still too much heterogeneity around the number of treatment groups.
Concerning outcomes, several studies used both multiple questionnaires/ratings [36,37,41,44,46] and cognitive tasks [38,39,40,42,43,48,49,52,53] and only one includes fMRI assessment [38]. However, other many studies kept in consideration only one questionnaire [34,35,45,47,50,51], as a result future researches could integrate better outcome measures.
3.1.2. Results
Regarding results, 13 studies highlighted various improvements related to PUFAs supplementation [35,36,38,39,41,44,45,46,48,49,51,52,53]. Six of those used EPA and DHA combinations [38,39,41,45,48,49], three used unspecified omega3 + omega6 combinations [35,36,51], three added vitamin E to the omega3 formula [44,52,53], and one study prescribed PS together with omega3 [46]. In these studies, ameliorations were not related to specific cognitive or behavioral domains. The remaining seven studies [34,40,42,43,47,49,50] did not find positive results linked to PUFAs supplementation. Future investigations with homogeneous methodologies are needed to clarify the reason of non-significant results or could better specify the role of PUFAs supplementation on ADHD symptoms.
3.2. Peptides and Amino Acids Derivatives
3.2.1. Methodologies
We identified three studies [15,17,21] using Acetyl-L-carnitine (ALC), l-carnosine, and PS (Table 1b). It is not possible to depict a systematic comparison of those studies, given the heterogeneous supplementation approaches. Specifically, two works [15,17] compared subjects who tok a placebo to a group that took supplementation as an add-on to MPH. On the contrary, one study [21] used PS alone in the active treatment group and placebo in control group. All studies comprised uniform samples, except Ghajar et al. [17], who also included children with ODD and applied equal randomization in treatment and placebo groups. Two studies [15,17] considered similar outcome measures, including parent and teacher questionnaires regarding children’s behavioral and cognitive symptoms. Hirayama et al. [21] used a go/no-go task and ADHD diagnostic criteria. Although these studies focused on different supplementations, they applied similar methodologies, especially regarding sample, randomization, and outcomes. The use of neuropsychological and physiological parameters could be improved and integrated to better understand response to treatment mechanisms. Moreover, future researches could address PS efficacy as an add-on to MPH.
3.2.2. Results
Regarding these studies results, ALC alone did not improve ADHD symptoms [15], but it reduced adverse effects when taken in addition to MPH. Moreover, l-carnosine seemed to improve behavioral problems according to parents [17]. PS had effects on ADHD symptoms, short-term auditory memory, and cognitive tasks [21]. In conclusion, l-carnosine and PS seem to have some effects on ADHD symptoms or cognitive domains but it remains unclear their specific role and mechanisms of action.
3.3. Single Micronutrient (Zinc or Vitamin D)
3.3.1. Methodologies
Some studies tested zinc [54,55,56] or vitamin D [25,57,58] (Table 1c). Zinc was used as sulfate [55,56] or glycinate [54] and vitamin D as generic [25] or D3 [57,58]. All studies provided micronutrients in addition to MPH [25,55,56,57,58] or dextroamphetamine [54]. Moreover, only Arnold et al. [54] compared the effects of micronutrients (and no pharmacological treatment) with placebo. All studies except two [54,58] included patients without comorbidity. Each study applied equal randomization between groups, even if the number of subjects was low [e.g., 58]. These works used similar methodologies, but several studies [25,55,57,58] used only parent-ratings as outcome measure.
3.3.2. Results
A link between zinc consumption and attention improvement was found in two studies [55,56]: zinc treatment improved inattention scores on parent questionnaires [55] and had effects on attention deficit disorder subtype of ADHD [56]. One study [54] did not find significant differences between zinc supplementation and placebo in the outcome measures; however, children taking zinc supplementation in addition to dextroamphetamine showed better drug dose optimization. All studies regarding vitamin D supplementation highlighted positive outcomes on parent-rated behavioral indexes or ADHD symptoms [25,57,58].
3.4. Micronutrients Mix
3.4.1. Methodologies
Several studies focused on broad combinations of vitamins and minerals [59,60,61,62,63] (Table 1d). One used a mixture of vitamin D and magnesium [60], two provided “daily essential nutrient formula”, which contained 13 vitamins, 17 minerals, and 4 amino acids [59,61]. The remaining studies represent further analyses [63] and follow-up research [62] of the Rucklidge et al. paper [61]. All studies were comprised of children with several comorbidities and applied equal randomization between groups. All studies used multiple ratings, except for Hemamy et al. [60], and one work using magnetic resonance imaging (MRI) data as an outcome (however, this last did not find any significant effect) [59].
3.4.2. Results
These studies highlighted improvements in several behavioral, emotional, and cognitive ADHD symptoms. Furthermore, the follow-up study [62] offers additional evidences regarding possible efficacy of micronutrients mix. However, these studies are characterized by low numerosity and high heterogeneity of samples and treatments.
3.5. Plant Extracts or Herbal Supplementations
3.5.1. Methodologies
Seven studies focused on plant or herbal extracts containing a mix of micronutrients, vitamins, and macronutrients (Table 1e). It is not possible to depict a systematic comparison of those studies, given the heterogeneous supplementation approaches. Specifically, one work used Ginkgo biloba plant extract as an add-on to MPH [69]. Others examined the efficacy of an herbal compound [64], Korean red ginseng (KRG) [65] or tocotrienol-rich fractions [70], as a single treatment compared to placebo, ningdong granule [66], sweet almond syrup [67], or Ginkgo biloba [68], compared to MPH. These studies focused on patients with ADHD symptoms without comorbidities and they applied an equal randomization, except for one work [64]. Each study used multiple ratings as outcomes, one of which [65] considered neurophysiological assessment and another cognitive tasks measures [64].
3.5.2. Results
All but two studies [68,70] found beneficial effects related to supplementation, in terms of improved symptomatology and/or less adverse effects as compared to MPH. Improvement in attention indexes was found in studies using a blend of herbs [64], Korean red ginseng [65], and Ginkgo biloba as an add-on to MPH [69]. Parent-, teacher-, or clinician-rated behavioral improvement was reported with KRG [65], ningdong granule [66], sweet almond syrup [67], and Ginkgo biloba as an add-on to MPH [69]. No adverse effects of supplementations were highlighted except for one study that reported increased appetite with sweet almond syrup [67], whereas half of the studies reported adverse effects of MPH [66,67,68]. One study [65] showed that KRG reduced the electroencephalography theta/beta ratio, a marker of cognitive processing capacity, significantly more than placebo. These results are heterogeneous and preliminary, and thus future homogeneous investigations that consider physiological parameters could offer more systematic evidences regarding herbal or extract supplementations. Furthermore, conflicting results [e.g., 68–69] between identical supplementation may be due to the different objectives: in one case authors aimed at comparing effects of supplementation with MPH [68], in the other case effects of MPH and non-pharmacological treatment together were compared to placebo [69].
3.6. Probiotics: Methodologies and Results
Only one paper focused on probiotics supplementation, through a different study design compared to the other reviewed studies [71] (Table 1f). A six-months-lasting probiotic supplementation was administered soon after childbirth and a follow-up assessment was conducted after 13 years. The quantity of Bifidobacterium species bacteria in the feces of children later diagnosed with ADHD or Asperger syndrome was found to be lower as compared to healthy children. ADHD or Asperger syndrome was diagnosed in 6/35 (17.1%) children in the placebo and none in the probiotic group (p = 0.008). This last study offers preliminary suggestions regarding probiotics supplementation as a preventive treatment, however further randomized clinical trials are needed to offer more systematic evidence regarding this treatment efficacy.
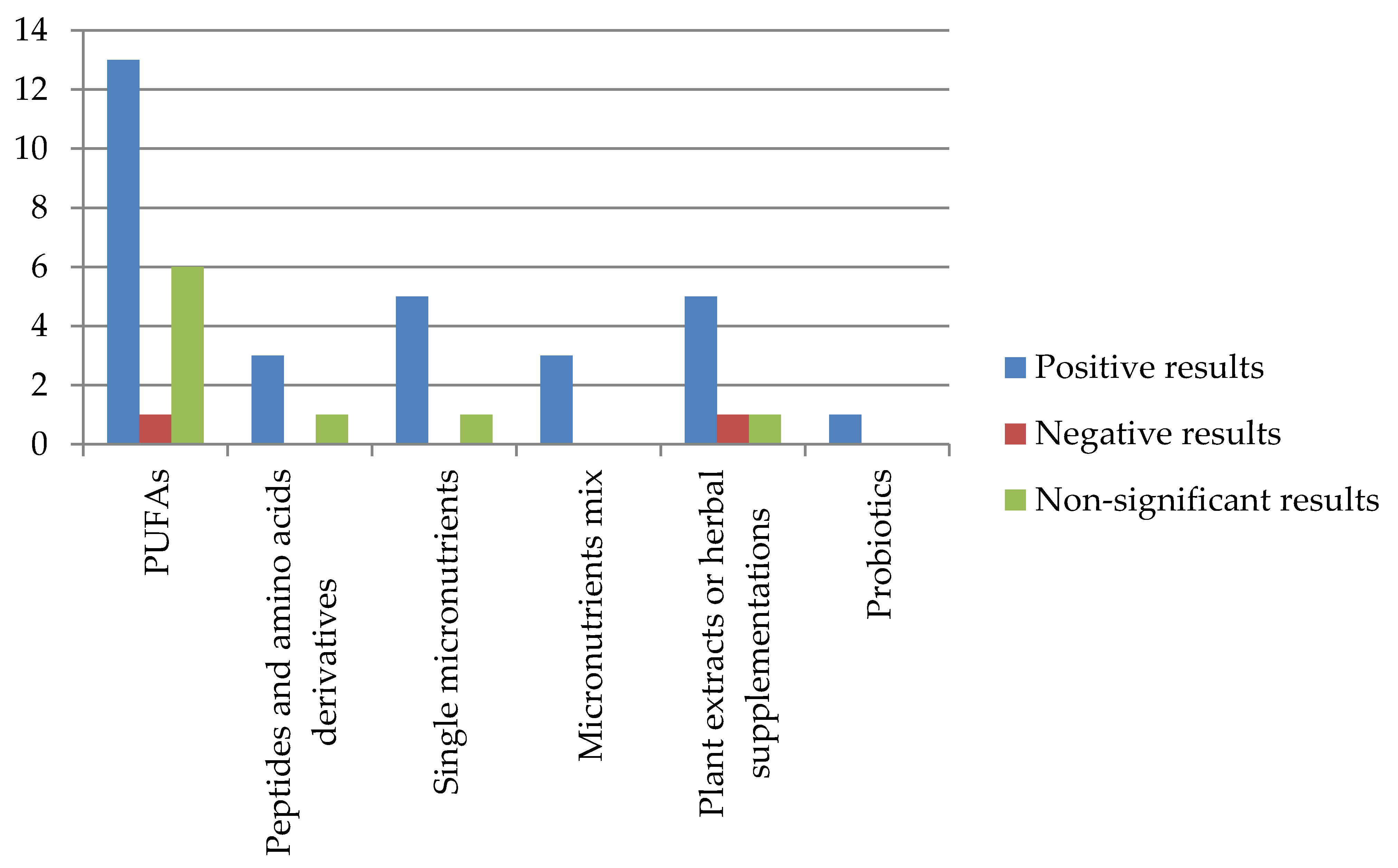
Figure 2 summarizes the main findings reported by the included works regarding nutritional supplementations on ADHD behavioral or cognitive symptoms; the vertical axis indicates the number of studies.
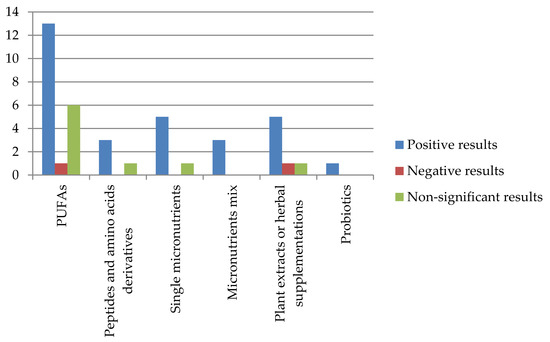
Figure 2.
Effect of supplementations in ADHD symptoms.
4. Discussion
We investigated the recent literature about the efficacy of non-pharmacological treatments for ADHD in children and adolescents, alone or in combination with pharmacological treatment.
4.1. Discussion of Methodologies
It is relevant that nearly half of the reviewed studies used supplementation with PUFA (mostly EPA and DHA as omega3 PUFAs and, for some, omega6; see Table 1a). This supplementation approach could be linked to previous evidence suggesting the involvement of lower blood levels of DHA in children and adolescents with ADHD [12].
The other reviewed studies addressed the efficacy of peptides and amino acids derivatives (Table 1b), micronutrients (alone or in combination; see Table 1c,d), and plant or herbal extracts (Table 1e); one study investigated the association between early-life probiotics supplementation and ADHD or Asperger syndrome diagnoses at puberty (Table 1f). These supplementation approaches are less frequently reported in the scientific literature as compared to PUFA supplementation.
Each reviewed study used different combinations and doses of drugs and/or non-pharmacological supplementations. Therefore, it is not possible to draw systematic conclusions on optimal type or dose of compound that could be useful in the treatment of ADHD symptomatology.
Regarding outcome measures, Table 2 depicts a summary of sources of information that were considered in the reviewed studies. Table 2 highlights the need to consider homogeneous outcome variables in future research to obtain more systematic evidence related to the same outcomes. Moreover, objective neurophysiological outcomes should be more consistently evaluated together with clinical evidence.

Table 2.
Summary of sources of information that were considered in the reviewed studies.
4.2. Discussion of Results
The majority of reviewed papers reported improvements but no specific effect of different supplements was found, thus suggesting a non-specific beneficial influence of micro- and macro-nutrients on a broad spectrum of functions and symptoms. A possible explanation of this result could be ascribed to general environmental and dietary influences that have been previously associated to the severity of ADHD symptoms in children and adolescents, such as low socioeconomic status, parents’ education, and unhealthy diet [72,73]. In this framework, it is still unclear whether ADHD onset and persistence over time represent the cause or the effect of unhealthy dietary patterns that could lead to nutritional deficits [72,73]. In any case, this review suggests that non-pharmacological supplementation, prescribed on the basis of individual nutritional deficiencies, could constitute a valid clinical path. It is not clear whether supplementation has a role for patients with no dietary imbalance. Moreover, the substances that are contained in various supplementations could benefit brain functioning but may also influence overall physiological functioning in children and adolescents, given a non-specific effect of these compounds. Clinicians should support alternative or additional treatment options only after appropriate blood tests and medical examinations.
In any case, the supplementation approach seems to be valid in combination with pharmacological treatment, as highlighted by positive results of MPH combination with PUFAs [35,36,37,52,56], peptides or amino acid derivatives [15,17], zinc [55,56], vitamin D [25,57,58], vitamin D and magnesium [60], and sweet almond syrup [67]. In these studies, ameliorations were found in behavioral symptoms as reported by parents and clinicians, together with less adverse events compared to pharmacotherapy alone. Hence, drugs and ad hoc nutritional supplementation could represent a valid therapeutic approach.
Other studies focused on children and adolescents who were not under pharmacological treatment for reasons including low compliance, adverse effects or non-response. This second group of studies found mixed results, in terms of finding beneficial effects of supplementation alone and of finding no effect at all. However, the majority of these studies reported a beneficial effect of supplements over placebo.
Specifically, 8 out of 14 studies regarding PUFA supplementation alone found symptoms amelioration over placebo in attention, psychosocial functioning, emotional problems, behavior as reported by parents and teachers, and working memory [38,39,41,44,45,46,51,53]. The only study addressing phosphatidylserine supplementation found positive effects of treatment over placebo in behavioral and cognitive symptomatology as reported by clinicians and through a go/no-go computerized task [21]. However, one study prescribed zinc supplementation against placebo and found no improvement in behavior, memory, or attention [54]. Five studies used plant or herbal extracts versus placebo. Two found significant beneficial effects of a patented blend of herbs (compound herbal preparation) and Korean red ginseng on attention and symptomatology as reported by clinicians [64,65]. Two studies found similar effects of ningdong granule or sweet almond syrup as compared to MPH treatment in behavioral measures reported by parents and teachers, with fewer side effects related to herbal supplementation than MPH [66,67]. Lastly, one study reported greater parent- and teacher-rated behavioral amelioration effects of MPH as compared to Ginkgo biloba supplementation alone [68] and another [70] did not find efficacy using tocotrienol-rich fractions compared to placebo. The only study concerning early-life probiotic supplementation revealed positive effects compared to placebo preventing ADHD onset later in life [71]. Studies regarding micronutrients mix supplementation found beneficial effects over placebo in general functioning, emotional dysregulation, aggression, and attention [59,61,62]. Importantly, a follow-up work by Rucklidge et al. identified various factors related to response to treatment with micronutrients mix, such as lower pre-treatment folate and B12 levels, being female, greater severity of symptoms and co-occurring disorders in pre-treatment condition, more pregnancy complications, and fewer birth problems [63]. This work highlighted the role of biological and environmental variables related to response to non-pharmacological treatment. This last area of research needs further research, given the high heterogeneity of results due to confounding biological and environmental variables.
5. Limitations
There were limitations within the articles described in this review. Results should be interpreted in the light of high heterogeneity related to various methodological factors. Indeed, the included works considered heterogeneous treatments, trial durations, methodologies (e.g., supplementation used as unique or combined treatment), and outcomes, even within the same category of supplements. Hence, it was not possible to carry out a meta-analysis of research results, which instead would be auspicable to provide clinicians with more systematic evidence. Moreover, samples were not uniformly involving only children with ADHD diagnoses; other comorbidities or typically developing children were included in some samples. Studies in this research field are also susceptible of cultural influences such as local dietary habits, thus making results difficult to generalize.
Although the majority of studies used similar parent and/or teacher assessment measures as primary outcomes, in many cases clinicians’ evaluation or neurophysiological / neuropsychological assessments were lacking. Only three studies [38,59,65] used neurophysiological data, like magnetic resonance imaging or electroencephalography. These kinds of assessments should be included in future research.
Lastly, the majority of studies found beneficial effects, but this may be due to the fact that only studies that found effects were published. However, a formal evaluation of bias was not conducted due to the non-systematic nature of this review.
6. Conclusions
This review suggest that supplementation approaches may be effective in add-on to pharmacotherapy in improving some behavioral and neuropsychological indicators in children and adolescents with ADHD. The heterogeneity of results suggests that supplementation should be personalized based on each patient’s dietary issues. Several supplementation components that are still poorly investigated and may be effective. Moreover, some nutritional supplementations could represent an alternative treatment or rehabilitation in situations of non-response or poor compliance or lack of tolerability of drug treatments, a field that must still be investigated further.
Author Contributions
E.R., S.G., M.P., F.M.V., and E.G. conducted the electronic search and study selection; E.R., S.G, M.M. (Maddalena Mauri), and M.N. wrote the first version of the manuscript; E.R., S.G., M.N., M.M. (Maddalena Mauri), F.M.V., M.P. and M.M. (Massimo Molteni) contributed to the discussion of the results; All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Italian Ministry of Health (RC 2018-2020 MIMOSA and Ricerca Corrente 2018-2020 to M.P.).
Conflicts of Interest
The authors declare no conflict of interest.
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®); American Psychiatric Pub: Washington, DC, USA, 2013. [Google Scholar]
- Arnsten, A.F.; Pliszka, S.R. Catecholamine influences on prefrontal cortical function: Relevance to treatment of attention deficit/hyperactivity disorder and related disorders. Pharmacol. Biochem. Behav. 2011, 99, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Stahl, S.M. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications, 3rd ed.; Cambridge University Press: New York, NY, USA, 2013. [Google Scholar]
- Storebø, O.J.; Krogh, H.B.; Ramstad, E.; Moreira-Maia, C.R.; Holmskov, M.; Skoog, M.; Nilausen, T.D.; Magnusson, F.L.; Zwi, M.; Gillies, D.; et al. Methylphenidate for attention-deficit/hyperactivity disorder in children and adolescents: Cochrane systematic review with meta-analyses and trial sequential analyses of randomised clinical trials. BMJ 2015, 351, h5203. [Google Scholar] [CrossRef] [PubMed]
- Punja, S.; Shamseer, L.; Hartling, L.; Urichuk, L.; Vandermeer, B.; Nikles, J.; Vohra, S. Amphetamines for attention deficit hyperactivity disorder (ADHD) in children and adolescents. Cochrane Database Syst. Rev. 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.R.; Akhondzadeh, S. Pharmacotherapy of attention-deficit/hyperactivity disorder: Nonstimulant medication approaches. Exp. Rev. Neurother. 2007, 7, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Steele, M.; Jensen, P.S.; Quinn, D.M. Remission versus response as the goal of therapy in ADHD: A new standard for the field? Clin. Ther. 2006, 28, 1892–1908. [Google Scholar] [CrossRef]
- Groenman, A.P.; Schweren, L.J.; Dietrich, A.; Hoekstra, P.J. An update on the safety of psychostimulants for the treatment of attention-deficit/hyperactivity disorder. Expert Opin. Drug Saf. 2017, 16, 455–464. [Google Scholar] [CrossRef]
- Bangs, M.E.; Wietecha, L.A.; Wang, S.; Buchanan, A.S.; Kelsey, D.K. Meta-analysis of suicide-related behavior or ideation in child, adolescent, and adult patients treated with atomoxetine. J. Child Adolesc. Psychopharmacol. 2014, 24, 426–434. [Google Scholar] [CrossRef]
- Thapar, A.; Cooper, M.; Jefferies, R.; Stergiakouli, E. What causes attention deficit hyperactivity disorder? Arch. Dis. Child. 2012, 97, 260–265. [Google Scholar] [CrossRef]
- Janssen, C.I.; Kiliaan, A.J. Long-chain polyunsaturated fatty acids (LCPUFA) from genesis to senescence: The influence of LCPUFA on neural development, aging, and neurodegeneration. Progr. Lipid Res. 2014, 53, 1–17. [Google Scholar] [CrossRef]
- Agostoni, C.; Nobile, M.; Ciappolino, V.; Delvecchio, G.; Tesei, A.; Turolo, S.; Crippa, A.; Mazzocchi, A.; Altamura, C.A.; Brambilla, P. The role of omega-3 fatty acids in developmental psychopathology: A systematic review on early psychosis, autism, and ADHD. Int. J. Mol. Sci. 2017, 18, 2608. [Google Scholar] [CrossRef]
- Tesei, A.; Crippa, A.; Busti Ceccarelli, S.; Mauri, M.; Molteni, M.; Agostoni, C.; Nobile, M. The potential relevance of docosahexaenoic acid and eicosapentaenoic acid to the etiopathogenesis of childhood neuropsychiatric disorders. Eur. Child Adolesc. Psychiatry 2017, 26, 1011–1030. [Google Scholar] [CrossRef] [PubMed]
- Adriani, W.; Rea, M.; Baviera, M.; Invernizzi, W.; Carli, M.; Ghirardi, O.; Caprioli, A.; Laviola, G. Acetyl-L-carnitine reduces impulsive behaviour in adolescent rats. Psychopharmacology 2004, 176, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, S.-H.; Heidari, S.; Mohammadi, M.-R.; Tabrizi, M.; Ghaleiha, A.; Akhondzadeh, S. Acetyl-L-Carnitine as an Adjunctive Therapy in the Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents: A Placebo-Controlled Trial. Child Psychiatry Hum. Dev. 2011, 42, 367–375. [Google Scholar] [CrossRef]
- Prokopieva, V.; Yarygina, E.; Bokhan, N.; Ivanova, S. Use of carnosine for oxidative stress reduction in different pathologies. Oxid. Med. Cell. Longev. 2016, 2939087. [Google Scholar] [CrossRef] [PubMed]
- Ghajar, A.; Aghajan-Nashtaei, F.; Afarideh, M.; Mohammadi, M.-R.; Akhondzadeh, S. L-Carnosine as Adjunctive Therapy in Children and Adolescents with Attention-Deficit/Hyperactivity Disorder: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Child Adolesc. Psychopharmacol. 2018, 5, 331–337. [Google Scholar] [CrossRef]
- Chez, M.G.; Buchanan, C.P.; Aimonovitch, M.C.; Becker, M.; Schaefer, K.; Black, C.; Komen, J. Double-blind, placebo-controlled study of l-carnosine supplementation in children with autistic spectrum disorders. J. Child Neurol. 2002, 17, 833–837. [Google Scholar] [CrossRef]
- Pepeu, G.; Pepeu, I.M.; Amaducci, L. A review of phosphatidylserine pharmacological and clinical effects. Is phosphatidylserine a drug for the ageing brain? Pharmacol. Res. 1996, 33, 73–80. [Google Scholar] [CrossRef]
- Starks, M.A.; Starks, S.L.; Kingsley, M.; Purpura, M.; Jäger, R. The effects of phosphatidylserine on endocrine response to moderate intensity exercise. J. Int. Soc. Sports Nutr. 2008, 5, 11. [Google Scholar] [CrossRef]
- Hirayama, S.; Terasawa, K.; Rabeler, R.; Hirayama, T.; Inoue, T.; Tatsumi, Y.; Purpura, M.; Jager, R. The effect of phosphatidylserine administration on memory and symptoms of attention-deficit hyperactivity disorder: A randomised, double-blind, placebo-controlled clinical. J. Hum. Nutr. Diet. 2013, 27, 284–291. [Google Scholar] [CrossRef]
- Bener, A.; Khattab, A.O.; Al-Dabbagh, M.M. Is high prevalence of Vitamin D deficiency evidence for autism disorder? In a highly endogamous population. J. Pediatr. Neurosci. 2014, 9, 227–233. [Google Scholar] [CrossRef]
- Chiang, M.; Natarajan, R.; Fan, X. Vitamin D in schizophrenia: A clinical review. Evid. Based Ment. Health 2016, 19, 6–9. [Google Scholar] [CrossRef]
- Meyer, T.; Becker, A.; Sundermann, J.; Rothenberger, A.; Herrmann-Lingen, C. Attention deficit-hyperactivity disorder is associated with reduced blood pressure and serum vitamin D levels: Results from the nationwide German Health Interview and Examination Survey for Children and Adolescents (KiGGS). Eur. Child Adolesc. Psychiatry 2017, 26, 165–175. [Google Scholar] [CrossRef]
- Mohammadpour, N.; Jazayeri, S.; Tehrani-Doost, M.; Djalali, M.; Hosseini, M.; Effatpanah, M.; Davari-Ashtiani, R.; Karami, E. Effect of vitamin D supplementation as adjunctive therapy to methylphenidate on ADHD symptoms: A randomized, double blind, placebo-controlled trial. Nutr. Neurosci. 2018, 21, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Toren, P.; Eldar, S.; Sela, B.A.; Wolmer, L.; Weitz, R.; Inbar, D.; Koren, S.; Reiss, A.; Weizman, R.; Laor, N. Zinc deficiency in attention-deficit hyperactivity disorder. Biol. Psychiatry 1996, 40, 1308–1310. [Google Scholar] [CrossRef]
- Sandstead, H.H.; Fosmire, G.J.; Halas, E.S.; Jacob, R.A.; Strobel, D.A.; Marks, E.O. Zinc deficiency: Effects on brain and behavior of rats and rhesus monkeys. Teratology 1977, 16, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Aggett, P.J.; Harries, J.T. Current status of zinc in health and disease states. Arch. Dis. Child. 1979, 54, 909–917. [Google Scholar] [CrossRef]
- Rucklidge, J.J.; Kaplan, B.J. Broad-spectrum micronutrient treatment for attention-deficit/hyperactivity disorder: Rationale and evidence to date. CNS Drugs 2014, 28, 775–785. [Google Scholar] [CrossRef]
- Sinha, D.; Efron, D. Complementary and alternative medicine use in children with attention deficit hyperactivity disorder. J. Paediatr. Child Health 2005, 41, 23–26. [Google Scholar] [CrossRef]
- Tillisch, K.; Labus, J.; Kilpatrick, L.; Jiang, Z.; Stains, J.; Ebrat, B.; Guyonnet, D.; Legrain-Raspaud, S.; Trotin, B.; Naliboff, B.; et al. Consumption of fermented milk product with probiotic modulates brain activity. Gastroenterology 2013, 144, 1394–1401. [Google Scholar] [CrossRef]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA Statement. Phys. Ther. 2009, 89, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Sachdeva, A. Effect of Poly Unsaturated Fatty Acids Administration on Children with Attention Deficit Hyperactivity Disorder: A Randomized Controlled Trial. J. Clin. Diagn. Res. 2016, 10, 1–5. [Google Scholar] [CrossRef]
- Assareh, M.; Ashtiani, R.D.; Khademi, M.; Jazayeri, S.; Rai, A.; Nikoo, M. Efficacy of Polyunsaturated Fatty Acids (PUFA) in the Treatment of Attention Deficit Hyperactivity Disorder: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Atten. Disord. 2017, 21, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Barragán, E.; Breuer, D.; Döpfner, M. Efficacy and Safety of Omega-3/6 Fatty Acids, Methylphenidate, and a Combined Treatment in Children with ADHD. J. Atten. Disord. 2017, 21, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Behdani, F.; Hebrani, P.; Naseraee, A.; Haghighi, M.B.; Akhavanrezayat, A. Does omega-3 supplement enhance the therapeutic results of methylphenidate in attention deficit hyperactivity disorder patients? J. Res. Med. Sci. 2013, 18, 653–658. [Google Scholar] [PubMed]
- Bos, D.J.; Oranje, B.; Veerhoek, E.S.; Van Diepen, R.M.; Weusten, J.M.; Demmelmair, H.; Koletzko, B.; GM de Sain-van der Velden, M.; Eilander, A.; Hoeksma, M.; et al. Reduced symptoms of inattention after dietary omega-3 fatty acid supplementation in boys with and without attention deficit/hyperactivity disorder. Neuropsychopharmacology 2015, 40, 2298–2306. [Google Scholar] [CrossRef]
- Pei-Chen Chang, J.; Su, K.P.; Mondelli, V.; Satyanarayanan, S.K.; Yang, H.-T.; Chiang, Y.-J.; Chen, H.-T.; Pariante, C.M. High-dose eicosapentaenoic acid (EPA) improves attention and vigilance in children and adolescents with attention deficit hyperactivity disorder (ADHD) and low endogenous EPA levels. Transl. Psychiatry 2019, 9, 303–312. [Google Scholar] [CrossRef]
- Cornu, C.; Mercier, C.; Ginhoux, T.; Masson, S.; Mouchet, J.; Nony, P.; Kassai, B.; Laudy, V.; Berquin, P.; Franc, N.; et al. A double-blind placebo-controlled randomised trial of omega-3 supplementation in children with moderate ADHD symptoms. Eur. Child Adolesc. Psychiatry 2018, 27, 377–384. [Google Scholar] [CrossRef]
- Crippa, A.; Tesei, A.; Sangiorgio, F.; Salandi, A.; Trabattoni, S.; Grazioli, S.; Agostoni, C.; Molteni, M.; Nobile, M. Behavioral and cognitive effects of docosahexaenoic acid in drug-naïve children with attention-deficit/hyperactivity disorder: A randomized, placebo-controlled clinical trial. Eur. Child Adolesc.Psychiatry 2019, 28, 571–583. [Google Scholar] [CrossRef]
- Dean, A.J.; Bor, W.; Adam, K.; Bowling, F.G.; Bellgrove, M.A. A Randomized, Controlled, Crossover Trial of Fish Oil Treatment for Impulsive Aggression in Children and Adolescents with Disruptive Behavior Disorders. J. Child Adolesc. Psychopharmacol. 2014, 24, 140–148. [Google Scholar] [CrossRef]
- Dubnov-Raz, G.; Khoury, Z.; Wright, I.; Raz, R.; Berger, I. The effect of alpha-linolenic acid supplementation on ADHD symptoms in children: A randomized controlled double-blind study. Front. Hum. Neurosci. 2014, 8, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, P.A.; Birberg-Thornberg, U.; Duchén, K.; Landgren, M.; Malmberg, K.; Pelling, H.; Strandvik, B.; Karlsson, T. EPA supplementation improves teacher-rated behaviour and oppositional symptoms in children with ADHD. Acta Paediatr. 2010, 99, 1540–1549. [Google Scholar] [CrossRef] [PubMed]
- Hariri, M.; Djazayery, A.; Djalali, M.; Saedisomeolia, A.; Rahimi, A.; Abdolahian, E. Effect of n-3 supplementation on hyperactivity, oxidative stress and inflammatory mediators in children with attention-deficit-hyperactivity disorder. Malays J. Nutr. 2012, 18, 329–335. [Google Scholar] [PubMed]
- Manor, I.; Magen, A.; Keidar, D.; Rosen, S.; Tasker, H.; Cohen, T.; Richter, Y.; Zaaroor-Regev, D.; Manor, Y.; Weizman, A. The effect of phosphatidylserine containing Omega3 fatty-acids on attention-deficit hyperactivity disorder symptoms in children: A double-blind placebo-controlled trial, followed by an open-label extension. Eur. Psychiatry 2012, 27, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Matsudaira, T.; Gow, R.V.; Kelly, J.; Murphy, C.; Potts, L.; Sumich, A.; Ghebremeskel, K.; Crawford, M.A.; Taylor, E. Biochemical and Psychological Effects of Omega-3/6 Supplements in Male Adolescents with Attention-Deficit/Hyperactivity Disorder: A Randomized, Placebo-Controlled, Clinical Trial. J. Child Adolesc. Psychopharmacol. 2015, 25, 775–782. [Google Scholar] [CrossRef]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R.C. Eicosapentaenoic and docosahexaenoic acids, cognition, and behavior in children with attention-deficit/hyperactivity disorder: A randomized controlled trial. Nutrition 2012, 28, 670–677. [Google Scholar] [CrossRef]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R.C. Increased Erythrocyte Eicosapentaenoic Acid and Docosahexaenoic Acid Are Associated With Improved Attention and Behavior in Children With ADHD in a Randomized Controlled Three-Way Crossover Trial. J. Atten. Disord. 2015, 19, 954–964. [Google Scholar] [CrossRef]
- Mohammadzadeh, S.; Baghi, N.; Yousefi, F.; Yousefzamani, B. Effect of omega-3 plus methylphenidate as an alternative therapy to reduce attention deficit-hyperactivity disorder in children. Korean J. Pediatr. 2019, 62, 360–366. [Google Scholar] [CrossRef]
- Perera, H.; Jeewandara, K.C.; Seneviratne, S.; Guruge, C. Combined ω3 and ω6 supplementation in children with attention-deficit hyperactivity disorder (ADHD) refractory to methylphenidate treatment: A double-blind, placebo-controlled study. J. Child Neurol. 2012, 27, 747–753. [Google Scholar] [CrossRef]
- Rodríguez, C.; García, T.; Areces, D.; Fernández, E.; García-Noriega, M.; Domingo, J.C. Supplementation with high-content docosahexaenoic acid triglyceride in attention-deficit hyperactivity disorder: A randomized double-blind placebo-controlled trial. Neuropsychiatr. Dis. Treat. 2019, 15, 1193–1209. [Google Scholar] [CrossRef]
- Widenhorn-Müller, K.; Schwanda, S.; Scholz, E.; Spitzer, M.; Bode, H. Effect of supplementation with long-chain ω-3 polyunsaturated fatty acids on behavior and cognition in children with attention deficit/hyperactivity disorder (ADHD): A randomized placebo-controlled intervention trial. Prostaglandins Leukot Essent Fatty Acids 2014, 91, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Arnold, L.E.; DiSilvestro, R.A.; Bozzolo, D.; Bozzolo, H.; Crowl, L.; Fernandez, S.; Ramadan, Y.; Thompson, S.; Mo, X.; Abdel-Rasoul, M.; et al. Zinc for attention-deficit/hyperactivity disorder: Placebo-controlled double-blind pilot trial alone and combined with amphetamine. J. Child Adolesc. Psychopharmacol. 2011, 21, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Noorazar, S.G.; Malek, A.; Aghaei, S.M.; Yasamineh, N.; Kalejahi, P. The efficacy of zinc augmentation in children with attention deficit hyperactivity disorder under treatment with methylphenidate: A randomized controlled trial. Asian J. Psychiatry 2020, 48, 101868. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Mohammadbeigi, A.; Sheykholeslam, H.; Moshiri, E.; Dorreh, F. Omega-3 and Zinc supplementation as complementary therapies in children with attention-deficit/hyperactivity disorder. J. Res. Pharm. Pract. 2016, 5, 22–26. [Google Scholar]
- Dehbokri, N.; Noorazar, G.; Ghaffari, A.; Mehdizadeh, G.; Sarbakhsh, P.; Ghaffary, S. Effect of vitamin D treatment in children with attention-deficit hyperactivity disorder. World J. Pediatr. 2019, 15, 78–84. [Google Scholar] [CrossRef]
- Elshorbagy, H.H.; Barseem, N.F.; Abdelghani, W.E.; Suliman, H.A.I.; Al-shokary, A.H.; Abdulsamea, S.E.; Elsadek, A.E.; Abdel Maksoud, Y.H.; Nour El Din, D.M.A.E.H. Impact of Vitamin D Supplementation on Attention-Deficit Hyperactivity Disorder in Children. Ann. Pharmacother. 2018, 52, 623–631. [Google Scholar] [CrossRef]
- Borlase, N.; Melzer, T.; Darling, K.; Eggleston, M.J.F.; Rucklidge, J. Resting-state networks and neurometabolites in children with ADHD after 10weeks of treatment with micronutrients: Results of a randomised placebo-controlled trial. Nutr. Neurosci. 2019, 1, 1–11. [Google Scholar] [CrossRef]
- Hemamy, M.; Heidari-Beni, M.; Askari, G.; Karahmadi, M.; Maracy, M. Effect of Vitamin D and Magnesium Supplementation on Behavior Problems in Children with Attention-Deficit Hyperactivity Disorder. Int. J. Prev. Med. 2020, 24, 11–14. [Google Scholar]
- Rucklidge, J.J.; Eggleston, M.J.F.; Johnstone, J.M.; Darling, K.; Frampton, C.M. Vitamin-mineral treatment improves aggression and emotional regulation in children with ADHD: A fully blinded, randomized, placebo-controlled trial. J. Child Psychol. Psychiatry 2018, 59, 232–246. [Google Scholar] [CrossRef]
- Darling, K.A.; Eggleston, M.J.F.; Retallick-Brown, H.; Rucklidge, J.J. Mineral-Vitamin Treatment Associated with Remission in Attention-Deficit/Hyperactivity Disorder Symptoms and Related Problems: 1-Year Naturalistic Outcomes of a 10-Week Randomized Placebo-Controlled Trial. J. Child Adolesc. Psychopharmacol. 2019, 29, 688–704. [Google Scholar] [CrossRef]
- Rucklidge, J.J.; Eggleston, M.J.F.; Darling, K.A.; Stevens, A.J.; Kennedy, M.A.; Frampton, C.M. Can we predict treatment response in children with ADHD to a vitamin-mineral supplement? An investigation into pre-treatment nutrient serum levels, MTHFR status, clinical correlates and demographic variables. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 89, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Katz, M.; Adar Levine, A.; Kol-Degani, H.; Kav-Venaki, L. A compound herbal preparation (CHP) in the treatment of children with ADHD: A randomized controlled trial. J. Attent. Disord. 2010, 14, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.J.; Kim, I.; Kim, J.B.; Moon, Y.; Whang, M.C.; Lee, K.M.; Jung, S.P. Effects of Korean red ginseng extract on behavior in children with symptoms of inattention and hyperactivity/impulsivity: A double-blind randomized placebo-controlled trial. J. Child Adolesc. Psychopharmacol. 2014, 24, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J.; Li, Z.W.; Wang, S.Z.; Qi, F.H.; Zhao, L.; Lv, H.; Li, A.Y. Ningdong granule: A complementary and alternative therapy in the treatment of attention deficit/hyperactivity disorder. Psychopharmacology 2011, 216, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Motaharifard, M.S.; Effatpanah, M.; Akhondzadeh, S.; Rahimi, H.; Yasrebi, S.A.; Nejatbakhsh, F. Effect of sweet almond syrup versus methylphenidate in children with ADHD: A randomized triple-blind clinical trial. Complement. Ther. Clin. Pract. 2019, 36, 170–175. [Google Scholar] [CrossRef]
- Salehi, B.; Imani, R.; Mohammadi, M.R.; Fallah, J.; Mohammadi, M.; Ghanizadeh, A.; Akhondzadeh, S. Ginkgo biloba for attention-deficit/hyperactivity disorder in children and adolescents: A double blind, randomized controlled trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 76–80. [Google Scholar] [CrossRef]
- Shakibaei, F.; Radmanesh, M.; Salari, E.; Mahaki, B. Ginkgo biloba in the treatment of attention-deficit/hyperactivity disorder in children and adolescents. A randomized, placebo-controlled, trial. Complement. Ther. Clin. Pract. 2015, 21, 61–67. [Google Scholar] [CrossRef]
- Tan, M.L.; Foong, S.C.; Foong, W.C.; Yusuff, Y.; Chettiar, S.M. Tocotrienol-rich fractions (TRF) supplementation in school-going children with Attention Deficit/Hyperactive Disorder (ADHD): A randomized controlled trial. BMC Nutr. 2016, 2, 14–23. [Google Scholar] [CrossRef]
- Pärtty, A.; Kalliomäki, M.; Wacklin, P.; Salminen, S.; Isolauri, E. A possible link between early probiotic intervention and the risk of neuropsychiatric disorders later in childhood: A randomized trial. Pediatr. Res. 2015, 77, 823–828. [Google Scholar] [CrossRef]
- Howard, A.L.; Robinson, M.; Smith, G.J.; Ambrosini, G.L.; Piek, J.P.; Oddy, W.H. ADHD is associated with a “Western” dietary pattern in adolescents. J. Atten. Disord. 2011, 15, 403–411. [Google Scholar] [CrossRef]
- Woo, H.D.; Kim, D.W.; Hong, Y.S.; Kim, Y.M.; Seo, J.H.; Choe, B.M.; Park, J.H.; Kang, J.-W.; Yoo, J.-H.; Chueh, H.W.; et al. Dietary patterns in children with attention deficit/hyperactivity disorder (ADHD). Nutrients 2014, 6, 1539–1553. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).