Can the FUT 2 Gene Variant Have an Effect on the Body Weight of Patients Undergoing Bariatric Surgery?—Preliminary, Exploratory Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
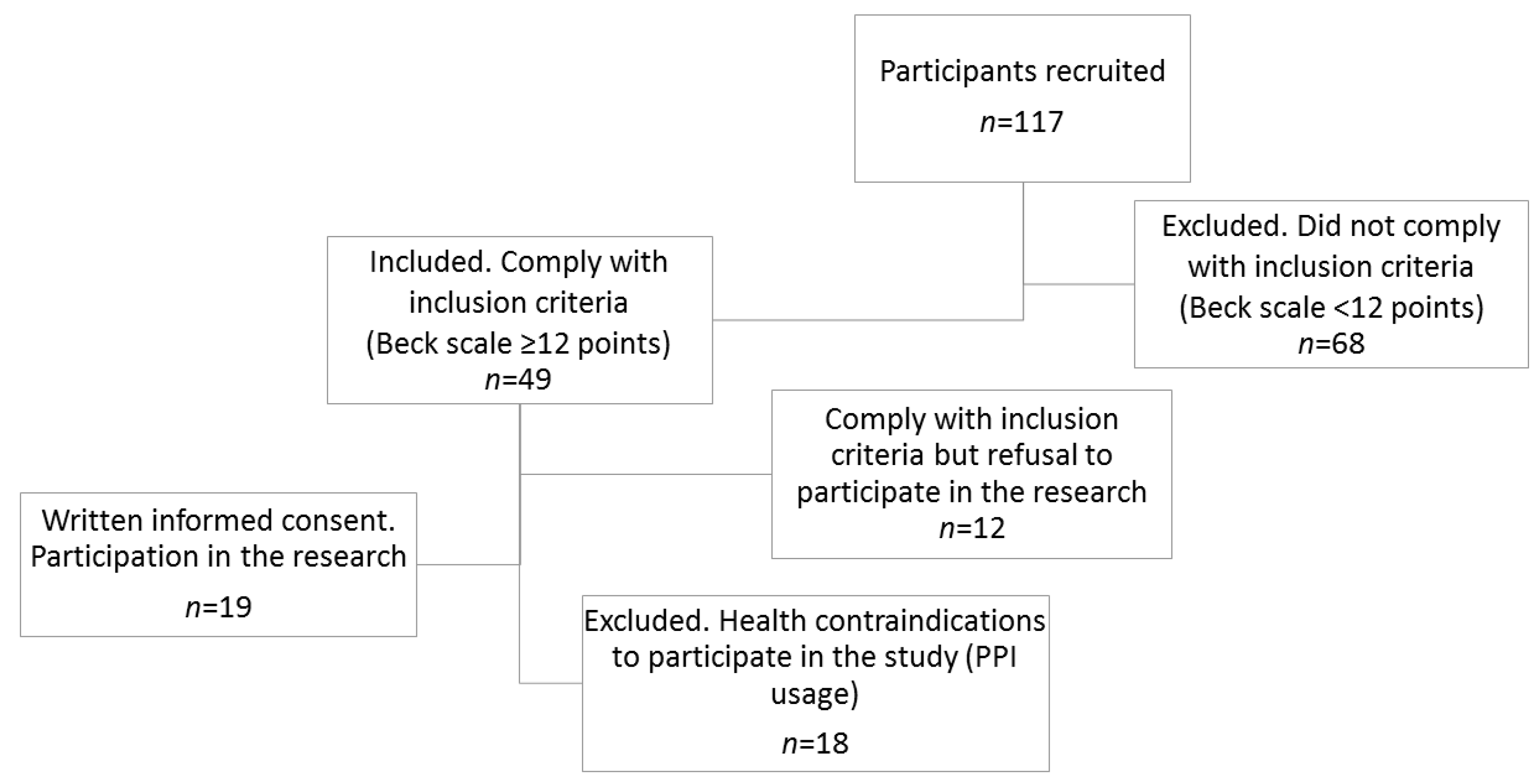
2.2. Study Protocol, the Anthropometric Data
2.3. DNA Isolation and Real-Time PCR SNP Analysis
2.4. DNA Extraction and the Analysis of Bacterial 16S RNA Gene Sequencing
2.5. Sequence Statistical Analysis
3. Results
3.1. Study Group Characteristics
3.2. Characteristics of the Stool Microbial Composition
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Conflicts of Interest
References
- Lo, T.; Tavakkoli, A. Bariatric surgery and its role in obesity pandemic. Curr. Opin. Physiol. 2019, 12, 51–56. [Google Scholar] [CrossRef]
- English, W.J.; DeMaria, E.J.; Hutter, M.M.; Kothari, S.N.; Mattar, S.G.; Brethauer, S.A.; Morton, J.M. American Society for Metabolic and Bariatric Surgery 2018 estimate of metabolic and bariatric procedures performed in the United States. Surg. Obes. Relat. Dis. 2020, 16, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; West, N.P.; Cripps, A.W. Obesity, inflammation, and the gut microbiota. Lancet Diabetes Endocrinol. 2015, 3, 207–215. [Google Scholar] [CrossRef]
- Luijten, J.C.H.B.M.; Vugts, G.; Nieuwenhuijzen, G.A.P.; Luyer, M.D.P. The Importance of the Microbiome in Bariatric Surgery: A Systematic Review. Obes. Surg. 2019, 29, 2338–2349. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, P.; Mäkivuokko, H.; Alakulppi, N.; Nikkilä, J.; Tenkanen, H.; Räbinä, J.; Partanen, J.; Aranko, K.; Mättö, J. Secretor genotype (FUT2 gene) is strongly associated with the composition of Bifidobacteria in the human intestine. PLoS ONE 2011, 6, e20113. [Google Scholar] [CrossRef] [Green Version]
- Czerwiński, M. Grupy krwi—minusy i plusy Czy antygeny grupowe krwi chronią nas przed chorobami zakaźnymi? Postepy Hig. Med. Dosw. 2015, 69, 703–722. [Google Scholar] [CrossRef]
- Ferrer-Admetlla, A.; Sikora, M.; Laayouni, H.; Esteve, A.; Roubinet, F.; Blancher, A.; Calafell, F.; Bertranpetit, J.; Casals, F. A natural history of FUT2 polymorphism in humans. Mol. Biol. Evol. 2009, 26, 1993–2003. [Google Scholar] [CrossRef] [Green Version]
- Parmar, A.S.; Alakulppi, N.; Paavola-Sakki, P.; Kurppa, K.; Halme, L.; Färkkilä, M.; Turunen, U.; Lappalainen, M.; Kontula, K.; Kaukinen, K.; et al. Association study of FUT2 (rs601338) with celiac disease and inflammatory bowel disease in the Finnish population. Tissue Antigens 2012, 80, 488–493. [Google Scholar] [CrossRef]
- Azad, M.B.; Wade, K.H.; Timpson, N.J. FUT2 secretor genotype and susceptibility to infections and chronic conditions in the ALSPAC cohort. Wellcome Open Res. 2018, 3. [Google Scholar] [CrossRef]
- Fujitani, N.; Liu, Y.; Toda, S.; Shirouzu, K.; Okamura, T.; Kimura, H. Expression of H type 1 antigen of ABO histo-blood group in normal colon and aberrant expressions of H type 2 and H type 3/4 antigens in colon cancer. Glycoconj. J. 2000, 17, 331–338. [Google Scholar] [CrossRef]
- Thorven, M.; Grahn, A.; Hedlund, K.-O.; Johansson, H.; Wahlfrid, C.; Larson, G.; Svensson, L. A homozygous nonsense mutation (428G→A) in the human secretor (FUT2) gene provides resistance to symptomatic norovirus (GGII) infections. J. Virol. 2005, 79, 15351–15355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindesmith, L.; Moe, C.; Marionneau, S.; Ruvoen, N.; Jiang, X.; Lindblad, L.; Stewart, P.; LePendu, J.; Baric, R. Human susceptibility and resistance to Norwalk virus infection. Nat. Med. 2003, 9, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Tong, M.; McHardy, I.; Ruegger, P.; Goudarzi, M.; Kashyap, P.C.; Haritunians, T.; Li, X.; Graeber, T.G.; Schwager, E.; Huttenhower, C.; et al. Reprograming of gut microbiome energy metabolism by the FUT2 Crohn’s disease risk polymorphism. ISME J. 2014, 8, 2193–2206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imhann, F.; Vila, A.V.; Bonder, M.J.; Fu, J.; Gevers, D.; Visschedijk, M.C.; Spekhorst, L.M.; Alberts, R.; Franke, L.; van Dullemen, H.M.; et al. Interplay of host genetics and gut microbiota underlying the onset and clinical presentation of inflammatory bowel disease. Gut 2018, 67, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, P.; Tuimala, J.; Nikkilä, J.; Tims, S.; Mäkivuokko, H.; Alakulppi, N.; Laine, P.; Rajilic-Stojanovic, M.; Paulin, L.; de Vos, W.M.; et al. Faecal microbiota composition in adults is associated with the FUT2 gene determining the secretor status. PLoS ONE 2014, 9, e94863. [Google Scholar] [CrossRef] [Green Version]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain fatty acids in health and disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar] [CrossRef]
- Xiong, Y.; Miyamoto, N.; Shibata, K.; Valasek, M.A.; Motoike, T.; Kedzierski, R.M.; Yanagisawa, M. Short-chain fatty acids stimulate leptin production in adipocytes through the G protein-coupled receptor GPR41. Proc. Natl. Acad. Sci. USA 2004, 101, 1045–1050. [Google Scholar] [CrossRef] [Green Version]
- De la Cuesta-Zuluaga, J.; Mueller, N.T.; Álvarez-Quintero, R.; Velásquez-Mejía, E.P.; Sierra, J.A.; Corrales-Agudelo, V.; Carmona, J.A.; Abad, J.M.; Escobar, J.S. Higher Fecal Short-Chain Fatty Acid Levels Are Associated with Gut Microbiome Dysbiosis, Obesity, Hypertension and Cardiometabolic Disease Risk Factors. Nutrients 2018, 11, 51. [Google Scholar] [CrossRef] [Green Version]
- Canetti, L.; Bachar, E.; Bonne, O. Deterioration of mental health in bariatric surgery after 10 years despite successful weight loss. Eur. J. Clin. Nutr. 2016, 70, 17–22. [Google Scholar] [CrossRef]
- Waters, G.S.; Pories, W.J.; Swanson, M.S.; Meelheim, H.D.; Flickinger, E.G.; May, H.J. Long-term studies of mental health after the Greenville gastric bypass operation for morbid obesity. Am. J. Surg. 1991, 161, 154–157. [Google Scholar] [CrossRef]
- Beck, A.T.; Ward, C.H.; Mendelson, M.; Mock, J.; Erbaugh, J. An Inventory for Measuring Depression. Arch. Gen. Psychiatry 1961, 4, 561–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parnowski, T.; Jernajczyk, W. Beck’s depression inventory in the rating of mood in normal subjects and in patients with affective disturbances. Psychiatr. Pol. 1977, 11, 417–421. [Google Scholar] [PubMed]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [Green Version]
- Masella, A.P.; Bartram, A.K.; Truszkowski, J.M.; Brown, D.G.; Neufeld, J.D. PANDAseq: Paired-end assembler for illumina sequences. BMC Bioinform. 2012, 13, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [Green Version]
- R: The R Project for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 26 July 2020).
- McNeil, N.I. The contribution of the large intestine to energy supplies in man. Am. J. Clin. Nutr. 1984, 39, 338–342. [Google Scholar] [CrossRef]
- Schwiertz, A.; Taras, D.; Schäfer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef]
- Rahat-Rozenbloom, S.; Fernandes, J.; Gloor, G.B.; Wolever, T.M.S. Evidence for greater production of colonic short-chain fatty acids in overweight than lean humans. Int. J. Obes. 2014, 38, 1525–1531. [Google Scholar] [CrossRef] [Green Version]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Jumpertz, R.; Le, D.S.; Turnbaugh, P.J.; Trinidad, C.; Bogardus, C.; Gordon, J.I.; Krakoff, J. Energy-balance studies reveal associations between gut microbes, caloric load, and nutrient absorption in humans. Am. J. Clin. Nutr. 2011, 94, 58–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liou, A.P.; Paziuk, M.; Luevano, J.-M.; Machineni, S.; Turnbaugh, P.J.; Kaplan, L.M. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci. Transl. Med. 2013, 5, 178ra41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meek, C.L.; Lewis, H.B.; Reimann, F.; Gribble, F.M.; Park, A.J. The effect of bariatric surgery on gastrointestinal and pancreatic peptide hormones. Peptides 2016, 77, 28–37. [Google Scholar] [CrossRef]
- Chambers, E.S.; Viardot, A.; Psichas, A.; Morrison, D.J.; Murphy, K.G.; Zac-Varghese, S.E.K.; MacDougall, K.; Preston, T.; Tedford, C.; Finlayson, G.S.; et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 2015, 64, 1744–1754. [Google Scholar] [CrossRef] [Green Version]
- Adab, P.; Pallan, M.; Whincup, P.H. Is BMI the best measure of obesity? BMJ 2018, 360, k1274. [Google Scholar] [CrossRef] [Green Version]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [Green Version]
- Furet, J.-P.; Kong, L.-C.; Tap, J.; Poitou, C.; Basdevant, A.; Bouillot, J.-L.; Mariat, D.; Corthier, G.; Doré, J.; Henegar, C.; et al. Differential adaptation of human gut microbiota to bariatric surgery-induced weight loss: Links with metabolic and low-grade inflammation markers. Diabetes 2010, 59, 3049–3057. [Google Scholar] [CrossRef] [Green Version]
- Kong, L.-C.; Tap, J.; Aron-Wisnewsky, J.; Pelloux, V.; Basdevant, A.; Bouillot, J.-L.; Zucker, J.-D.; Doré, J.; Clément, K. Gut microbiota after gastric bypass in human obesity: Increased richness and associations of bacterial genera with adipose tissue genes. Am. J. Clin. Nutr. 2013, 98, 16–24. [Google Scholar] [CrossRef] [Green Version]
- Arboleya, S.; Watkins, C.; Stanton, C.; Ross, R.P. Gut Bifidobacteria Populations in Human Health and Aging. Front. Microbiol. 2016, 7, 1204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobyliak, N.; Conte, C.; Cammarota, G.; Haley, A.P.; Styriak, I.; Gaspar, L.; Fusek, J.; Rodrigo, L.; Kruzliak, P. Probiotics in prevention and treatment of obesity: A critical view. Nutr. Metab. 2016, 13, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, H.M.; Park, S.Y.; Lee, D.K.; Kim, J.R.; Cha, M.K.; Lee, S.W.; Lim, H.T.; Kim, K.J.; Ha, N.J. Antiobesity and lipid-lowering effects of Bifidobacterium spp. in high fat diet-induced obese rats. Lipids Health Dis. 2011, 10, 116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanauchi, O.; Fujiyama, Y.; Mitsuyama, K.; Araki, Y.; Ishii, T.; Nakamura, T.; Hitomi, Y.; Agata, K.; Saiki, T.; Andoh, A.; et al. Increased growth of Bifidobacterium and Eubacterium by germinated barley foodstuff, accompanied by enhanced butyrate production in healthy volunteers. Int. J. Mol. Med. 1999, 3, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.I.; Sellin, J.H. Review article: Short chain fatty acids in health and disease. Aliment. Pharmacol. Ther. 1998, 12, 499–507. [Google Scholar] [CrossRef]
- Delzenne, N.M.; Neyrinck, A.M.; Cani, P.D. Modulation of the gut microbiota by nutrients with prebiotic properties: Consequences for host health in the context of obesity and metabolic syndrome. Microb. Cell Fact. 2011, 10, S10. [Google Scholar] [CrossRef] [Green Version]
- Martinez, K.B.; Leone, V.; Chang, E.B. Western diets, gut dysbiosis, and metabolic diseases: Are they linked? Gut Microbes 2017, 8, 130–142. [Google Scholar] [CrossRef] [Green Version]
- Alcaraz, F.; Frey, S.; Iannelli, A. Surgical Management of Small Intestinal Bacterial Overgrowth After Roux-en-Y Gastric Bypass. Obes. Surg. 2020. [Google Scholar] [CrossRef]
- Harris, L.A.; Baffy, N. Modulation of the gut microbiota: A focus on treatments for irritable bowel syndrome. Postgrad. Med. 2017, 129, 872–888. [Google Scholar] [CrossRef]







| Feature | |
|---|---|
| Age (years) | 46.3 (±11.7) |
| Type of surgery | 10 patients Roux-en-Y gastric bypass (RYGB) 9 patients sleeve gastrectomy (SG) |
| Weight before surgery (kg) | 122.3 (±22.1) |
| BMI before surgery (kg/m2) | 43.2 (±5.9) |
| Post-operative period (years) | 3.2 (±3.4) |
| Actual weight (kg) | 95.5 (±23.5) |
| Actual BMI (kg/m2) | 30.6 (±5.3) |
| Actual waist circumference (cm) | 94.2(±12.2) |
| Actual WHR | 0.82 (±0.07) |
| Fut2 | n | Delta-BMI | SD |
|---|---|---|---|
| AA | 2 | −13.4000 | 3.1113 |
| GA | 9 | −12.1571 | 4.9784 |
| GG | 8 | −13.8023 | 4.5286 |
| Significance level | p = 0.77 | ||
| Energy (kcal) | Total Protein (g) | Animal Protein (g) | Plant Protein (g) | Fat (g) | Carbohydrates (g) |
| 1138 ± 366 | 65 ± 16.2 | 44.89 ± 14 | 14.87 ± 5.68 g | 45.2 ± 18.8 | 117.6 ± 44.9 |
| Fiber (g) | Sucrose (g) | Sodium (mg) | Potassium (mg) | Calcium (mg) | Magnesium (mg) |
| 15.5 ± 6.4 | 19.7 ± 12.4 | 1144 ± 544.5 | 2622 ± 774.4 | 497.2 ± 152.4 | 256.2 ± 88.5 |
| Iron (mg) | Phosphorus (mg) | Zinc (mg) | Copper (mg) | Vitamin A (µg) | Vitamin D (µg) |
| 8.9 ± 2.6 | 1008.3 ± 231.3 | 7.1 ± 1.8 | 1.03 ± 0.3 | 1311.9 ± 1671 | 3.2 ± 3.3 |
| Vitamin E (mg) | Vitamin B1 (mg) | Vitamin B12 (µg) | Folate (µg) | Vitamin B6 (mg) | Vitamin C (mg) |
| 7.7 ± 4 | 0.8 ± 0.3 | 5.7 ± 6.5 | 284.8 ± 148.1 | 1.6 ± 0.6 | 131.1 ± 92.4 |
| Genus | Relative % Abundance (Cutoff > 1) |
|---|---|
| Haemophilus | 1.10 |
| Lachnospiracea_incertae_sedis | 1.26 |
| Phascolarctobacterium | 1.35 |
| Veillonella | 1.42 |
| Lactobacillus | 1.64 |
| Roseburia | 1.87 |
| Faecalibacterium | 1.88 |
| Blautia | 1.95 |
| Clostridium sensu stricto | 2.05 |
| Streptococcus | 2.19 |
| Oscillibacter | 2.62 |
| Barnesiella | 2.68 |
| Escherichia/Shigella | 4.31 |
| Parabacteroides | 4.69 |
| Alistipes | 9.71 |
| Prevotella | 12.29 |
| Bacteroides | 26.40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komorniak, N.; Martynova-Van Kley, A.; Nalian, A.; Wardziukiewicz, W.; Skonieczna-Żydecka, K.; Styburski, D.; Palma, J.; Kowalewski, B.; Kaseja, K.; Stachowska, E. Can the FUT 2 Gene Variant Have an Effect on the Body Weight of Patients Undergoing Bariatric Surgery?—Preliminary, Exploratory Study. Nutrients 2020, 12, 2621. https://doi.org/10.3390/nu12092621
Komorniak N, Martynova-Van Kley A, Nalian A, Wardziukiewicz W, Skonieczna-Żydecka K, Styburski D, Palma J, Kowalewski B, Kaseja K, Stachowska E. Can the FUT 2 Gene Variant Have an Effect on the Body Weight of Patients Undergoing Bariatric Surgery?—Preliminary, Exploratory Study. Nutrients. 2020; 12(9):2621. https://doi.org/10.3390/nu12092621
Chicago/Turabian StyleKomorniak, Natalia, Alexandra Martynova-Van Kley, Armen Nalian, Wiktoria Wardziukiewicz, Karolina Skonieczna-Żydecka, Daniel Styburski, Joanna Palma, Bartosz Kowalewski, Krzysztof Kaseja, and Ewa Stachowska. 2020. "Can the FUT 2 Gene Variant Have an Effect on the Body Weight of Patients Undergoing Bariatric Surgery?—Preliminary, Exploratory Study" Nutrients 12, no. 9: 2621. https://doi.org/10.3390/nu12092621





