Cereal Processing By-Products as Rich Sources of Phenolic Compounds and Their Potential Bioactivities
Abstract
:1. Introduction
2. Cereal Processing
2.1. Corn Processing
2.2. Wheat Processing
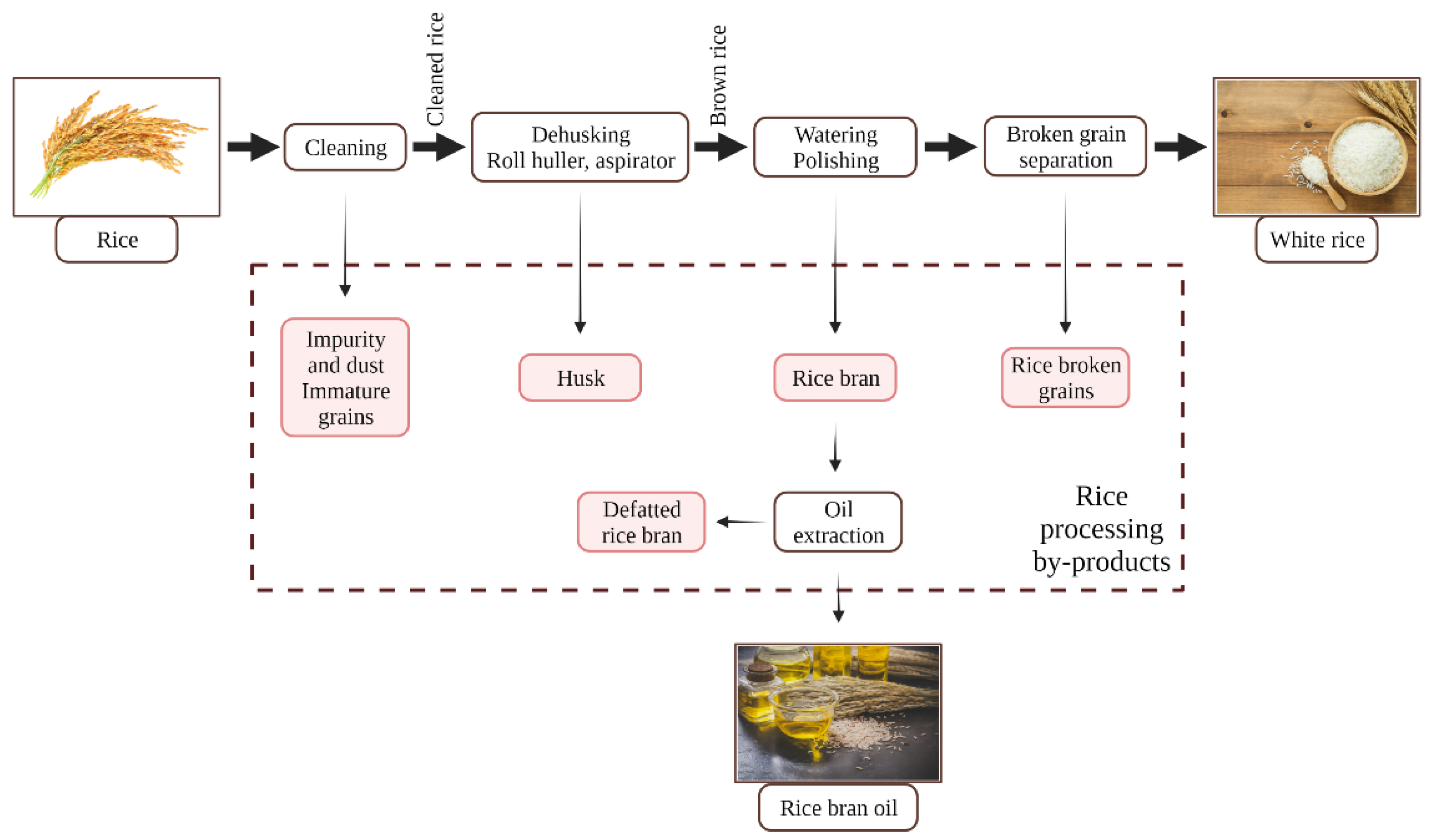
2.3. Rice Processing
2.4. Barley Processing in Brewing Industry
3. Polyphenols in Cereal By-Products
4. Extraction Methods
5. The Potential Health Effects of Extracted Polyphenols
5.1. Antioxidant Activity
5.2. Anti-Carcinogenic Effects
5.2.1. Cell Viability
5.2.2. Cell Proliferation and Apoptosis
5.2.3. DNA Damaging
5.2.4. Inflammation
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| EU | European Union |
| FAO | Food and Agriculture Organization |
| dCGM | Dry Corn Germ Meal |
| CE | Catechin Equivalent |
| dWB | Dry Wheat Bran |
| dWG | Dry Wheat Germ |
| dRB | Dry Rice Bran |
| GAE | Gallic Acid Equivalent |
| dRG | Dry Rice Germ |
| RDA | Recommended Dietary Allowance |
| BSG | Brewer’s spent Grain |
| SC-CO2 | Supercritical Carbon Dioxide |
| UAE | Ultrasound Assisted Extraction |
| MAE | Microwave Assisted Extraction |
| ROS | Reactive Oxygen Species |
| RNS | Reactive Nitrogen Species |
| CAT | Catalase |
| NO | Nitric Oxide |
| iNOS | Nitric Oxide Synthase |
| LPS | Lipopolysaccharide |
| AP | Apurinic/Apirimidinic |
| NF-kB | Nuclear Factor Kappa-light-chain-enhancer of activated B cells |
| TNFα | Tumor Necrosis Factor alpha |
| COX-2 | Cyclooxygenase-isoform 2 |
References
- Cattaneo, A.; Sánchez, M.V.; Torero, M.; Vos, R. Reducing Food Loss and Waste: Five Challenges for Policy and Research. Food Policy 2021, 98, 101974. [Google Scholar] [CrossRef] [PubMed]
- Skendi, A.; Zinoviadou, K.G.; Papageorgiou, M.; Rocha, J.M. Advances on the Valorisation and Functionalization of By-Products and Wastes from Cereal-Based Processing Industry. Foods 2020, 9, 1243. [Google Scholar] [CrossRef]
- Sharma, P.; Gaur, V.K.; Sirohi, R.; Varjani, S.; Hyoun Kim, S.; Wong, J.W.C. Sustainable Processing of Food Waste for Production of Bio-Based Products for Circular Bioeconomy. Bioresour. Technol. 2021, 325, 124684. [Google Scholar] [CrossRef]
- Ubando, A.; Felix, C.; Chen, W. Biorefineries in Circular Bioeconomy: A Comprehensive Review. Bioresour. Technol. 2020, 299, 122585. [Google Scholar] [CrossRef] [PubMed]
- Venkata Mohan, S.; Varjani, S.; Pant, D.; Sauer, M.; Chang, J.S. Circular Bioeconomy Approaches for Sustainability. Bioresour. Technol. 2020, 318, 124084. [Google Scholar] [CrossRef]
- Belc, N.; Mustatea, G.; Apostol, L.; Iorga, S.; Vlǎduţ, V.N.; Mosoiu, C. Cereal Supply Chain Waste in the Context of Circular Economy. E3S Web Conf. 2019, 112, 03031. [Google Scholar] [CrossRef]
- FAO. Cereal Supply and Demand Brief|World Food Situation|Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/worldfoodsituation/csdb/en/ (accessed on 9 August 2021).
- Grain Production Worldwide by Type, 2020/21|Statista. Available online: https://www.statista.com/statistics/263977/world-grain-production-by-type/ (accessed on 2 September 2021).
- Singh, A.; Karmakar, S.; Jacob, B.S.; Bhattacharya, P.; Kumar, S.P.J.; Banerjee, R. Enzymatic Polishing of Cereal Grains for Improved Nutrient Retainment. J. Food Sci. Technol. 2015, 52, 3147. [Google Scholar] [CrossRef] [Green Version]
- Saleh, A.S.M.; Wang, P.; Wang, N.; Yang, S.; Xiao, Z. Technologies for Enhancement of Bioactive Components and Potential Health Benefits of Cereal and Cereal-Based Foods: Research Advances and Application Challenges. Crit. Rev. Food Sci. Nutr. 2019, 59, 207–227. [Google Scholar] [CrossRef] [PubMed]
- Dapčević-Hadnađev, T.; Hadnađev, M.; Pojić, M. The Healthy Components of Cereal By-Products and Their Functional Properties. In Sustainable Recovery and Reutilization of Cereal Processing By-Products; Elsevier: Amsterdam, The Netherlands, 2018; pp. 27–61. [Google Scholar] [CrossRef]
- Tan, J.P.; Jahim, J.M.; Wu, T.Y.; Harun, S.; Mumtaz, T. Use of Corn Steep Liquor as an Economical Nitrogen Source for Biosuccinic Acid Production by Actinobacillus Succinogenes. IOP Conf. Ser. Earth Environ. Sci. 2016, 36, 012058. [Google Scholar] [CrossRef]
- Albuquerque, C.; Rabello, C.; Santos, M.; Lima, M.; Silva, E.; Lima, T.; Ventura, D.; Dutra, W., Jr. Chemical Composition and Metabolizable Energy Values of Corn Germ Meal Obtained by Wet Milling for Layers. Brazilian J. Poult. Sci. 2014, 16, 107–112. [Google Scholar] [CrossRef]
- Smuda, S.S.; Mohsen, S.M.; Olsen, K.; Aly, M.H. Bioactive Compounds and Antioxidant Activities of Some Cereal Milling By-Products. J. Food Sci. Technol. 2018, 55, 1134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rose, D.; Inglett, G.; Liu, S. Utilisation of Corn (Zea Mays) Bran and Corn Fiber in the Production of Food Components. J. Sci. Food Agric. 2010, 90, 915–924. [Google Scholar] [CrossRef]
- Hu, R.; Dunmire, K.M.; Truelock, C.N.; Paulk, C.B.; Aldrich, G.; Li, Y. Antioxidant Performances of Corn Gluten Meal and DDGS Protein Hydrolysates in Food, Pet Food, and Feed Systems. J. Agric. Food Res. 2020, 2, 100030. [Google Scholar] [CrossRef]
- Najera, G.; Patricia, S. A Compositional Breakage Equation for First Break Roller Milling of Wheat; University of Manchester—CEAS: Manchester, UK, 2014. [Google Scholar]
- Rosenfelder, P.; Eklund, M.; Mosenthin, R. Nutritive Value of Wheat and Wheat By-Products in Pig Nutrition: A Review. Anim. Feed Sci. Technol. 2013, 185, 107–125. [Google Scholar] [CrossRef]
- Brandolini, A.; Hidalgo, A. Wheat Germ: Not Only a by-Product. Int. J. Food Sci. Nutr. 2012, 63, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Prückler, M.; Siebenhandl-Ehn, S.; Apprich, S.; Höltinger, S.; Haas, C.; Schmid, E.; Kneifel, W. Wheat Bran-Based Biorefinery 1: Composition of Wheat Bran and Strategies of Functionalization. LWT Food Sci. Technol. 2014, 56, 211–221. [Google Scholar] [CrossRef]
- Gul, K.; Yousuf, B.; Singh, A.K.; Singh, P.; Wani, A.A. Rice Bran: Nutritional Values and Its Emerging Potential for Development of Functional Food—A Review. Bioact. Carbohydrates Diet. Fibre 2015, 6, 24–30. [Google Scholar] [CrossRef]
- Moongngarm, A.; Daomukda, N.; Khumpika, S. Chemical Compositions, Phytochemicals, and Antioxidant Capacity of Rice Bran, Rice Bran Layer, and Rice Germ. APCBEE Procedia 2012, 2, 73–79. [Google Scholar] [CrossRef] [Green Version]
- Bodie, A.R.; Micciche, A.C.; Atungulu, G.G.; Rothrock, M.J.J.; Ricke, S.C. Current Trends of Rice Milling Byproducts for Agricultural Applications and Alternative Food Production Systems. Front. Sustain. Food Syst. 2019, 3, 47. [Google Scholar] [CrossRef]
- Choi, O.-K.; Yun, S.-K.; Hwang, S.-Y. The Chemical Components of Korean Rice Germ. J. Korean Soc. Food Cult. 2000, 15, 253–258. [Google Scholar]
- Rondanelli, M.; Miccono, A.; Peroni, G.; Nichetti, M.; Infantino, V.; Spadaccini, D.; Alalwan, T.A.; Faliva, M.A.; Perna, S. Rice Germ Macro- and Micronutrients: A New Opportunity for the Nutraceutics. Nat. Prod. Res. 2021, 35, 1532–1536. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Dragone, G.; Roberto, I.C. Brewers’ Spent Grain: Generation, Characteristics and Potential Applications. J. Cereal Sci. 2006, 43, 1–14. [Google Scholar] [CrossRef]
- Mussatto, S.I. Brewer’s Spent Grain: A Valuable Feedstock for Industrial Applications. J. Sci. Food Agric. 2014, 94, 1264–1275. [Google Scholar] [CrossRef] [Green Version]
- Almeida, A.d.R.; Geraldo, M.R.F.; Ribeiro, L.F.; Silva, M.V.; Maciel, M.V.d.O.B.; Haminiuk, C.W.I. Compostos Bioativos Do Bagaço de Malte: Compostos Fenólicos, Ácidos Graxos e Capacidade Antioxidante in Vitro. Acta Sci. Technol. 2017, 39, 269–277. [Google Scholar] [CrossRef] [Green Version]
- Codina-Torrella, I.; Rodero, L.; Almajano, M.P. Brewing By-Products as a Source of Natural Antioxidants for Food Preservation. Antioxidants 2021, 10, 1512. [Google Scholar] [CrossRef]
- Stefanello, F.S.; dos Santos, C.O.; Bochi, V.C.; Fruet, A.P.B.; Soquetta, M.B.; Dörr, A.C.; Nörnberg, J.L. Analysis of Polyphenols in Brewer’s Spent Grain and Its Comparison with Corn Silage and Cereal Brans Commonly Used for Animal Nutrition. Food Chem. 2018, 239, 385–401. [Google Scholar] [CrossRef]
- Socaci, S.A.; Fărcaş, A.C.; Diaconeasa, Z.M.; Vodnar, D.C.; Rusu, B.; Tofană, M. Influence of the Extraction Solvent on Phenolic Content, Antioxidant, Antimicrobial and Antimutagenic Activities of Brewers’ Spent Grain. J. Cereal Sci. 2018, 80, 180–187. [Google Scholar] [CrossRef]
- Rachwał, K.; Waśko, A.; Gustaw, K.; Polak-Berecka, M. Utilization of Brewery Wastes in Food Industry. PeerJ 2020, 8, e9427. [Google Scholar] [CrossRef]
- Gupta, M.; Abu-Ghannam, N.; Gallaghar, E. Barley for Brewing: Characteristic Changes during Malting, Brewing and Applications of Its by-Products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 318–328. [Google Scholar] [CrossRef] [Green Version]
- Kao, T.-H. Health Potential for Beer Brewing Byproducts. In Current Topics on Superfoods; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef] [Green Version]
- Tlais, A.Z.A.; Fiorino, G.M.; Polo, A.; Filannino, P.; Di Cagno, R. High-Value Compounds in Fruit, Vegetable and Cereal Byproducts: An Overview of Potential Sustainable Reuse and Exploitation. Molecules 2020, 25, 2987. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Sun, Y.; Chen, Z.; Yang, Y.; Wang, Y.; Trabelsi, N. Functional Properties of Polyphenols in Grains and Effects of Physicochemical Processing on Polyphenols. J. Food Qual. 2019, 2019. [Google Scholar] [CrossRef]
- Van Hung, P. Phenolic Compounds of Cereals and Their Antioxidant Capacity. Crit. Rev. Food Sci. Nutr. 2016, 56, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Luithui, Y.; Nisha, R.B.; Meera, M.S. Cereal By-Products as an Important Functional Ingredient: Effect of Processing. J. Food Sci. Technol. 2019, 56, 1. [Google Scholar] [CrossRef]
- Bonifácio-Lopes, T.; Teixeira, J.A.; Pintado, M. Current Extraction Techniques towards Bioactive Compounds from Brewer’s Spent Grain–A Review. Crit. Rev. Food Sci. Nutr. 2020, 60, 2730–2741. [Google Scholar] [CrossRef] [PubMed]
- Macias-Garbett, R.; Serna-Hernández, S.O.; Sosa-Hernández, J.E.; Parra-Saldívar, R. Phenolic Compounds From Brewer’s Spent Grains: Toward Green Recovery Methods and Applications in the Cosmetic Industry. Front. Sustain. Food Syst. 2021, 5, 196. [Google Scholar] [CrossRef]
- Alonso-Riaño, P.; Diez, M.T.S.; Blanco, B.; Beltrán, S.; Trigueros, E.; Benito-Román, O. Water Ultrasound-Assisted Extraction of Polyphenol Compounds from Brewer’s Spent Grain: Kinetic Study, Extract Characterization, and Concentration. Antioxidants 2020, 9, 265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guido, L.F.; Moreira, M.M. Techniques for Extraction of Brewer’s Spent Grain Polyphenols: A Review. Food Bioprocess Technol. 2017, 10, 1192–1209. [Google Scholar] [CrossRef] [Green Version]
- Fărcaş, A.; Tofană, M.; Socaci, S.; Scrob, S. Preliminary Study on Antioxidant Activity and Polyphenols Content in Discharged Waste from Beer Production. J. Agroaliment. Process. Technol. 2013, 19, 319–324. [Google Scholar]
- Bonifácio-Lopes, T.; Vilas Boas, A.A.; Coscueta, E.R.; Costa, E.M.; Silva, S.; Campos, D.; Teixeira, J.A.; Pintado, M. Bioactive Extracts from Brewer’s Spent Grain. Food Funct. 2020, 11, 8963–8977. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, A.L.; O’Callaghan, Y.C.; Connolly, A.; Piggott, C.O.; Fitzgerald, R.J.; O’Brien, N.M. Phenolic Extracts of Brewers’ Spent Grain (BSG) as Functional Ingredients—Assessment of Their DNA Protective Effect against Oxidant-Induced DNA Single Strand Breaks in U937 Cells. Food Chem. 2012, 134, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.W.; Khong, N.M.H.; Iqbal, S.; Ismail, M. Isolation and Antioxidative Properties of Phenolics-Saponins Rich Fraction from Defatted Rice Bran. J. Cereal Sci. 2013, 57, 480–485. [Google Scholar] [CrossRef]
- Ciulu, M.; Cádiz-Gurrea, M.d.l.L.; Segura-Carretero, A. Extraction and Analysis of Phenolic Compounds in Rice: A Review. Molecules 2018, 23, 2890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buranov, A.U.; Mazza, G. Extraction and Purification of Ferulic Acid from Flax Shives, Wheat and Corn Bran by Alkaline Hydrolysis and Pressurised Solvents. Food Chem. 2009, 115, 1542–1548. [Google Scholar] [CrossRef]
- Inglett, G.E.; Chen, D. Antioxidant Activity and Phenolic Content of Air-Classified Corn Bran. Cereal Chem. 2011, 88, 36–40. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, Y.; Sun, X.; Raddatz, J.; Zhou, Z.; Chen, G. Effect of Microfluidisation on Antioxidant Properties of Corn Bran. Food Chem. 2013, 152, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Rebolleda, S.; José, M.L.G.S.; Sanz, M.T.; Beltrán, S.; Solaesa, Á.G. Bioactive Compounds of a Wheat Bran Oily Extract Obtained with Supercritical Carbon Dioxide. Foods 2020, 9, 625. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sun, B.; Cao, Y.; Tian, Y.; Li, X. Optimisation of Ultrasound-Assisted Extraction of Phenolic Compounds from Wheat Bran. Food Chem. 2008, 106, 804–810. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, R.; Liu, C. Extraction and Antioxidant Activity of Phenolic Compounds from Wheat Bran Treated by Steam Explosion. Trop. J. Pharm. Res. 2015, 14, 1857–1863. [Google Scholar] [CrossRef] [Green Version]
- Radenkovs, V.; Juhnevica-Radenkova, K.; Górnaś, P.; Seglina, D. Non-Waste Technology through the Enzymatic Hydrolysis of Agro-Industrial by-Products. Trends Food Sci. Technol. 2018, 77, 64–76. [Google Scholar] [CrossRef]
- Bian, Y.Y.; Guo, J.; Zhu, K.X.; Guo, X.N.; Peng, W.; Zhou, H.M. Resistance Investigation of Wheat Bran Polyphenols Extracts on HEK293 Cells against Oxidative Damage. RSC Adv. 2015, 5, 16116–16124. [Google Scholar] [CrossRef]
- Vieira, E.F.; da Silva, D.D.; Carmo, H.; Ferreira, I.M. Protective Ability against Oxidative Stress of Brewers’ Spent Grain Protein Hydrolysates. Food Chem. 2017, 228, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Zhang, H.; Cheng, L.; Wang, L.; Qian, H.; Qi, X. In Vitro and in Vivo Antioxidant Activity of Polyphenols Extracted from Black Highland Barley. Food Chem. 2016, 194, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and Biological Activities of Natural Polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Henderson, A.J.; Ollila, C.A.; Kumar, A.; Borresen, E.C.; Raina, K.; Agarwal, R.; Ryan, E.P. Chemopreventive Properties of Dietary Rice Bran: Current Status and Future Prospects. Adv. Nutr. 2012, 3, 643–653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polonskiy, V.; Loskutov, I.; Sumina, A. Biological Role and Health Benefits of Antioxidant Compounds in Cereals. Biol. Commun. 2020, 65, 53–67. [Google Scholar] [CrossRef]
- Davatgaran-Taghipour, Y.; Masoomzadeh, S.; Farzaei, M.H.; Bahramsoltani, R.; Karimi-Soureh, Z.; Rahimi, R.; Abdollahi, M. Polyphenol Nanoformulations for Cancer Therapy: Experimental Evidence and Clinical Perspective. Int. J. Nanomed. 2017, 12, 2689. [Google Scholar] [CrossRef] [Green Version]
- Makris, D.P.; Şahin, S. Polyphenolic Antioxidants from Agri-Food Waste Biomass. Antioxidants 2019, 8, 624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martín-Diana, A.B.; García-Casas, M.J.; Martínez-Villaluenga, C.; Frías, J.; Peñas, E.; Rico, D. Wheat and Oat Brans as Sources of Polyphenol Compounds for Development of Antioxidant Nutraceutical Ingredients. Foods 2021, 10, 115. [Google Scholar] [CrossRef] [PubMed]
- Mccarthy, A.L.; O’Callaghan, Y.C.; Connolly, A.; Piggott, C.O.; Fitzgerald, R.J.; O’Brien, N.M. Phenolic-Enriched Fractions from Brewers’ Spent Grain Possess Cellular Antioxidant and Immunomodulatory Effects in Cell Culture Model Systems. J. Sci. Food Agric. 2014, 94, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, A.; Burlini, I.; Huerta Lorenzo, B.; Grandini, A.; Vertuani, S.; Tacchini, M.; Sacchetti, G. Antioxidant and Antimicrobial Extracts Obtained from Agricultural By-Products: Strategies for a Sustainable Recovery and Future Perspectives. Food Bioprod. Process. 2020, 124, 397–407. [Google Scholar] [CrossRef]
- Masisi, K.; Beta, T.; Moghadasian, M.H. Antioxidant Properties of Diverse Cereal Grains: A Review on in Vitro and in Vivo Studies. Food Chem. 2016, 196, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Link, A.; Balaguer, F.; Goel, A. Cancer Chemoprevention by Dietary Polyphenols: Promising Role for Epigenetics. Biochem. Pharmacol. 2010, 80, 1771–1792. [Google Scholar] [CrossRef] [Green Version]
- Niedzwiecki, A.; Roomi, M.W.; Kalinovsky, T.; Rath, M. Anticancer Efficacy of Polyphenols and Their Combinations. Nutrients 2016, 8, 552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Son, T.G.; Camandola, S.; Mattson, M.P. Hormetic Dietary Phytochemicals. NeuroMolecular Med. 2008, 10, 236–246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crowley, D.; O’Callaghan, Y.; McCarthy, A.L.; Connolly, A.; Fitzgerald, R.J.; O’Brien, N.M. Aqueous and Enzyme-Extracted Phenolic Compounds from Brewers’ Spent Grain (BSG): Assessment of Their Antioxidant Potential. J. Food Biochem. 2017, 41, 1–11. [Google Scholar] [CrossRef]
- Donato, M.T.; Tolosa, L.; Gómez-Lechón, M.J. Culture and Functional Characterization of Human Hepatoma HepG2 Cells. Methods Mol. Biol. 2015, 1250, 77–93. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Santhakumar, A.B.; Chinkwo, K.A.; Vanniasinkam, T.; Luo, J.; Blanchard, C.L. Chemopreventive Potential of Cereal Polyphenols. Nutr. Cancer 2018, 70, 913–927. [Google Scholar] [CrossRef] [PubMed]
- Feitelson, M.A.; Arzumanyan, A.; Kulathinal, R.J.; Blain, S.W.; Holcombe, R.F.; Mahajna, J.; Marino, M.; Martinez-Chantar, M.L.; Nawroth, R.; Sanchez-Garcia, I.; et al. Sustained Proliferation in Cancer: Mechanisms and Novel Therapeutic Targets. Semin. Cancer Biol. 2015, 35, S25–S54. [Google Scholar] [CrossRef] [PubMed]
- Kong, C.K.L.; Lam, W.S.; Chiu, L.C.M.; Ooi, V.E.C.; Sun, S.S.M.; Wong, Y.S. A Rice Bran Polyphenol, Cycloartenyl Ferulate, Elicits Apoptosis in Human Colorectal Adenocarcinoma SW480 and Sensitizes Metastatic SW620 Cells to TRAIL-Induced Apoptosis. Biochem. Pharmacol. 2009, 77, 1487–1496. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Norhaizan, M.E.; Huynh, K.; Heshu, S.R.; Yeap, S.K.; Hazilawati, H.; Roselina, K. Water Extract of Brewers’ Rice Induces Apoptosis in Human Colorectal Cancer Cells via Activation of Caspase-3 and Caspase-8 and Downregulates the Wnt/β-Catenin Downstream Signaling Pathway in Brewers’ Rice-Treated Rats with Azoxymethane-Induced Colon Carc. BMC Complement. Altern. Med. 2015, 15, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-Maqueda, D.; Miralles, B.; Recio, I. HT29 Cell Line. In The Impact of Food Bioactives on Health; In Vitro and Ex Vivo Models; Springer: Berlin/Heidelberg, Germany, 2015; pp. 113–124. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.J.; Yoon, W.J.; Kim, S.S. Phytochemical Compositions of Immature Wheat Bran, and Its Antioxidant Capacity, Cell Growth Inhibition, and Apoptosis Induction through Tumor Suppressor Gene. Molecules 2016, 21, 1292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carusillo, A.; Mussolino, C. DNA Damage: From Threat to Treatment. Cells 2020, 9, 1665. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.; Ahmad, A.; Khan, H.; Zubair, H.; Sarkar, F.; Hadi, S. The Prooxidant Action of Dietary Antioxidants Leading to Cellular DNA Breakage and Anticancer Effects: Implications for Chemotherapeutic Action against Cancer. Cell Biochem. Biophys. 2013, 67, 431–438. [Google Scholar] [CrossRef] [PubMed]
- León-González, A.J.; Auger, C.; Schini-Kerth, V.B. Pro-Oxidant Activity of Polyphenols and Its Implication on Cancer Chemoprevention and Chemotherapy. Biochem. Pharmacol. 2015, 98, 371–380. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, A.L.; O’Callaghan, Y.C.; Piggott, C.O.; FitzGerald, R.J.; O’Brien, N.M. Brewers’ Spent Grain; Bioactivity of Phenolic Component, Its Role in Animal Nutrition and Potential for Incorporation in Functional Foods: A Review. Proc. Nutr. Soc. 2013, 72, 117–125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murakami, A.; Nakamura, Y.; Koshimizu, K.; Takahashi, D.; Matsumoto, K.; Hagihara, K.; Taniguchi, H.; Nomura, E.; Hosoda, A.; Tsuno, T.; et al. FA15, a Hydrophobic Derivative of Ferulic Acid, Suppresses Inflammatory Responses and Skin Tumor Promotion: Comparison with Ferulic Acid. Cancer Lett. 2002, 180, 121–129. [Google Scholar] [CrossRef]




| Cereal By-Product | Extraction Method | Conditions | Polyphenols | References |
|---|---|---|---|---|
| Brewer’s spent grain | Supercritical carbon dioxide (SC-CO2) | - CO2 + Ethanol (0–60%, v/v) - p = 15–35 MPa - t° = 40–60 °C | Very low quantities of polyphenols | [2,40] |
| Ultrasound Assisted Extraction (UAE) | - Acetone/Water (60/40%, v/v) or NaOH/Water (0.75/99.25%, v/v) - Ultrasound frequency = 20 kHz - t° = 39 → 47 °C | p-Hydroxybenzoic acid, Ferulic acid, Sinapic acid | [40,41] | |
| Microwave Assisted Extraction (MAE) | - NaOH (0.75%)/Choline chloride:glycerol - t° = 100 °C | p-Coumaric acid, Ferulic acid, Syringic acid | [40,42] | |
| Methanol/Methanol-Water | - VMethanol = 10 mL - t° = 4 °C | Very low quantities of polyphenols | [40,43] | |
| Water | - VH2O = 50 mL - t° = 25 °C; 80 °C | 4-Hydroxybenzoic acid, p-Coumaric acid, Protocatechuic acid, Vanillin, Catechin, Vanillic acid | [44] | |
| Ethanol/ Ethanol-Water | - VEthanol (60–100%, v/v) = 50 mL - t° = 25 °C; 80 °C | 4-Hydroxybenzoic, p-Coumaric acid, Protocatechuic acid, Vanillin, Catechin, Syringic acid | [44] | |
| Acetone/ Acetone-Water | - Acetone (60%, v/v) - t° = 60 °C | p-Hydroxybenzoic acid, Protocatechuic acid, Chlorogenic acid, 8-8′-DiFA, 5-5′-DiFA, 5-5′,8′-O-4′-TriFA, p-Coumaric acid, Ferulic acid, Sinapic acid | [41] | |
| H2SO4 and NaOH | - H2SO4 + NaOH - t° = 120 °C | Ferulic acid | [40] | |
| Saponification | - CM = 1–4 M NaOH | Ferulic acid, p-Coumaric | [45] | |
| Rice bran | Supercritical carbon dioxide (SC-CO2) | - CO2 + Ethanol (0, 5 and 10%) - t° = 40 °C, 60 °C - p = 30, 40 MPa | (+)-Catechin, Chlorogenic acid, Caffeic acid, p-Coumaric acid, Protocatechuic acid, Cyanidin-3-glucoside | [2,46] |
| Ultrasound Assisted Extraction (UAE) for black and purple rice bran | - Solvent (20–60%) - t° = 30–60 °C - pH = 2–4 | [2,46] | ||
| Microwave Assisted Extraction (MAE) | - Methanol (100%) - t° = 185 °C - Microwave power = 1000 W | [46,47] | ||
| Green method | - Glycerol (10–70%, v/v) - t° = 40–70 °C - Liquid-to-solid ratio = 10–40 mL/g | [2,46] | ||
| Corn bran | Pressurized alkaline hydrolysis | - CM = 0.5 M NaOH—30% Ethanol - 15% Ammonia/Water - t° = 180 °C | Ferulic acid, p-Coumaric acid, Vanillin (derived) | [48] |
| Single alkaline and acid extraction | - VNaOH = 5 mL (2 N NaOH) - VHCl = 5 mL (2 N HCl) | Vanillic acid, Cis-ferulic acid, p-Coumaric acid, Caffeic acid, Syringic acid, Sinapic acid | [49] | |
| Single neutral extraction | - VEthanol (50%, v/v) = 10 mL | [49] | ||
| Acetone | - VAcetone (50%, v/v) = 20 mL - t° = 25 °C | p-Coumaric acid, Syringic acid, Ferulic acid | [50] | |
| Wheat bran | Supercritical carbon dioxide (SC-CO2) | - CO2 (8 ± 1 kg CO2/h) - t° = 40 ± 2 °C - p = 25.0 ± 1.0 MPa | p-Hydroxybenzaldehyde, Ferulic acid, Syringic acid, Syringic aldehyde, Vanillic acid, Vanillin | [51] |
| Ultrasound Assisted Extraction (UAE) | - Methanol/Ethanol/Acetone (70/70/70%, v/v) - Ultrasound frequency = 40 kHz - t° = 50 °C | [52] | ||
| Steam Explosion | -Ethanol -t° = 224 °C -p = 2.5 MPa | Ferulic acid predominantly | [53] | |
| Enzymatic Hydrolysis | - Multi-enzyme complex Viscozyme L/Xylanase/Feruloyl esterase | Ferulic acid | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fărcaș, A.; Drețcanu, G.; Pop, T.D.; Enaru, B.; Socaci, S.; Diaconeasa, Z. Cereal Processing By-Products as Rich Sources of Phenolic Compounds and Their Potential Bioactivities. Nutrients 2021, 13, 3934. https://doi.org/10.3390/nu13113934
Fărcaș A, Drețcanu G, Pop TD, Enaru B, Socaci S, Diaconeasa Z. Cereal Processing By-Products as Rich Sources of Phenolic Compounds and Their Potential Bioactivities. Nutrients. 2021; 13(11):3934. https://doi.org/10.3390/nu13113934
Chicago/Turabian StyleFărcaș, Anca, Georgiana Drețcanu, Teodora Daria Pop, Bianca Enaru, Sonia Socaci, and Zorița Diaconeasa. 2021. "Cereal Processing By-Products as Rich Sources of Phenolic Compounds and Their Potential Bioactivities" Nutrients 13, no. 11: 3934. https://doi.org/10.3390/nu13113934
APA StyleFărcaș, A., Drețcanu, G., Pop, T. D., Enaru, B., Socaci, S., & Diaconeasa, Z. (2021). Cereal Processing By-Products as Rich Sources of Phenolic Compounds and Their Potential Bioactivities. Nutrients, 13(11), 3934. https://doi.org/10.3390/nu13113934













