Abstract
The Mediterranean diet (MD) has shown to reduce the occurrence of several chronic diseases. To evaluate its potential protective role on dementia incidence we studied 16,160 healthy participants from the European Prospective Investigation into Cancer and Nutrition (EPIC)-Spain Dementia Cohort study recruited between 1992–1996 and followed up for a mean (±SD) of 21.6 (±3.4) years. A total of 459 incident cases of dementia were ascertained through expert revision of medical records. Data on habitual diet was collected through a validated diet history method to assess adherence to the relative Mediterranean Diet (rMED) score. Hazard ratios (HR) of dementia by rMED levels (low, medium and high adherence levels: ≤6, 7–10 and ≥11 points, respectively) were estimated using multivariable Cox models, whereas time-dependent effects were evaluated using flexible parametric Royston-Parmar (RP) models. Results of the fully adjusted model showed that high versus low adherence to the categorical rMED score was associated with a 20% (HR = 0.80, 95%CI: 0.60–1.06) lower risk of dementia overall and HR of dementia was 8% (HR = 0.92, 0.85–0.99, p = 0.021) lower for each 2-point increment of the continuous rMED score. By sub-types, a favorable association was also found in women for non-AD (HR per 2-points = 0.74, 95%CI: 0.62–0.89), while not statistically significant in men for AD (HR per 2-points = 0.88, 0.76–1.01). The association was stronger in participants with lower education. In conclusion, in this large prospective cohort study MD was inversely associated with dementia incidence after accounting for major cardiovascular risk factors. The results differed by dementia sub-type, sex, and education but there was no significant evidence of effect modification.
1. Introduction
Dementia is one of the leading causes of disability and dependency among older people worldwide, with huge physical, social, and economic repercussions [1]. Over 50 million people suffer from dementia worldwide and this number is expected to rise by more than two-fold by 2050 [2], which has led the World Health Organization (WHO) to recognize dementia as a public health priority [3]. As an age-related disorder with no effective treatment to date [3], prevention through changes in modifiable risk factors and lifestyles such as diet remain paramount to tackle the health and social challenges imposed by the growing burden of dementia worldwide [4].
The Mediterranean diet (MD) pattern, described more than half a century ago, is characterized by a high intake of vegetables, fruits, legumes, unrefined cereals, nuts and olive oil, a moderate intake of fish and wine and low intake of dairy products, meat, poultry and saturated fat [5,6,7]. In the past few decades, the advantages of the MD have been extensively studied in relation to premature mortality and incidence of cancer or cardiovascular disease [8,9], while its health benefits have also led researchers to explore its potential role on cognition. Previous studies suggest that the MD may protect against age-related cognitive decline and mild cognitive impairment [10,11], but its potential role in reducing or delaying the onset of dementia is not well established [12,13,14,15,16]. Most studies conducted so far have focused on cognitive function as the end-point whereas only a few have assessed the potential beneficial role of the MD pattern on the occurrence of dementia or Alzheimer’s disease (AD), for which the level of the evidence is still regarded as moderate and warrants further research [2]. Moreover, many of the studies that have assessed the association between MD and dementia risk have taken place in non-Mediterranean countries, mostly the United States (US) [15,16,17]. These studies would support the external validity of the beneficial effect of the MD. However, there remains a need of supporting evidence from large-scale prospective studies carried out in Mediterranean countries in order to elucidate whether the MD could actually reduce the risk of cognitive decline or dementia. It is in these regions where the MD pattern remains closer to its original features. Moreover, the traditional lifestyles and healthy habits of Mediterranean countries could extend the benefits of the MD beyond its strictly nutritional effects. The EPIC (European Prospective Investigation into Cancer and nutrition)-Spain study is a large multicenter cohort study designed to evaluate the association between diet, lifestyles and incidence of cancer and other chronic and age-related diseases, such as dementia, accounting for wide exposure variability, long follow-up time and large number of cases. In this analysis we aimed to evaluate the relationship between adherence to a MD pattern and the subsequent risk of dementia and dementia sub-types in the EPIC-Spain Dementia Cohort.
2. Materials and Methods
2.1. Study Sample
The EPIC study is a multicenter prospective study carried out on over 500,000 volunteers from ten European countries [18,19]. The EPIC-Spain Dementia Cohort was established from a sample of 25,015 EPIC-Spain participants from three EPIC-Spain study centers which carried out the prospective ascertainment of dementia cases: Gipuzkoa, Navarra, and Murcia [20].
Participants were 30 to 70 years old at enrolment between 1992 and 1996, and were mostly blood donors, civil servants, and general population. Exclusion criteria were being pregnant or breastfeeding, or not being physically or mentally capable of participating. Baseline data on diet, lifestyles, and medical and reproductive history were collected during in-person interviews. In addition, participants underwent a physical examination to obtain anthropometric information, and provided a blood sample. Further details on study design and sample characteristics can be found elsewhere [19,20].
2.2. Dietary Assessment
Detailed information on habitual food consumption during the previous year was collected through a validated diet history method, administered face-to-face by trained dietitians [21].
Questionnaires were structured by meals according to occasions of food intake and subjects were asked about foods consumed in a typical week, accounting for food preparation, frequency of consumption and usual portion size. All foods consumed at least twice a month were considered (except liver, included when consumed at least once a month) and seasonal and weekly variations (working days or weekends) were taken into account. Total daily energy and nutrient intakes were calculated using country-specific food composition tables, further harmonized within the EPIC Nutrient DataBase project [22].
Plausibility of dietary intake was defined according to the predicted total energy expenditure (pTEE) method [23], identifying implausible reporters based on the ratio of estimated energy intake to predicted total energy expenditure (rEI:pTEE).
2.3. Relative Mediterranean Diet (rMED) Score
Adherence to the Mediterranean dietary pattern was assessed by means of the relative Mediterranean Diet score (rMED), a variation of the original Mediterranean diet score by Trichopoulou et al. [24] defined within the EPIC-Spain cohort.
The rMED score is based on 9 components: 6 considered as positive (fruit, vegetables, olive oil, legumes, fish, and cereals), 2 considered as negative or detrimental (meat and dairy products), and alcohol. Intake for each component was standardized as grams per 1000 kcal and divided by tertiles. Positive components score 0, 1, and 2 for the first, second, and third groups defined by tertiles of intake, respectively, while detrimental components score 2, 1, and 0 for the first, second, and third groups, respectively. For alcohol intake, 2 points are given to consumption within a sex-specific range (5–25 g/day for women, 10–50 g/day for men), and 0 points otherwise [24,25]. Thus, the rMED score ranged from 0 (minimum) to 18 (maximum). Levels of adherence to the MD were classified as low (0–6 points), medium (7–10 points), and high rMED scores (11–18 points) as previously defined [25].
Since associations of MD with health-related outcomes may vary depending on the operative definition of the MD pattern [6], three alternative MD indexes, i.e., the Mediterranean Diet Score (MDS) by Trichopoulou et al. [24], the adapted relative Mediterranean Diet score (arMED) by Buckland et al. [26], and the alternate Mediterranean Diet Index (aMED), by Fung et al. [27], were evaluated in a sensitivity analysis.
2.4. Ascertainment of Dementia Cases
A two-phase validation protocol was designed to ascertain incident dementia cases occurring in the cohort based on medical records, as detailed previously [28]. In brief, potential cases were identified by record linkage of the EPIC database with primary care records, hospital discharge databases, and regional mortality registries. Incident dementia cases were then validated after a careful and extensive examination of all medical records available for each potential case by a panel of neurologists, who determined the sub-type whenever possible.
Participants were followed up from recruitment until the date of diagnosis, death, loss to follow-up, or the last complete vital status check (31 December 2017 for Gipuzkoa, 31 December 2015 for Navarra, and 30 November 2016 for Murcia), whichever occurred first.
Participants in the EPIC study did not undergo a baseline cognitive assessment. However, all volunteers were considered to have normal cognition since they were required to be able to complete extensive and demanding questionnaires, including a dietary history interview which took 50–60 min on average. The case ascertainment process identified only one prevalent case, whereas four participants developed dementia within the first 5 years of the study (only one in the first 3 years).
From the 25,015 participants without evidence of prevalent dementia at baseline, we excluded a total of 1651 patients with at least one major chronic pathology (diabetes, ischemic heart disease, stroke, or cancer). We further excluded as energy mis-reporters a total of 7204 participants with a reported energy intake beyond 30% of the estimated energy requirement (corresponding to a cutoff of ±2 SD of the rEI:pTEE ratio), including 206 incident dementia cases. Thus, the final sample consisted of 16,160 participants, among which 308 incident AD cases and 151 incident non-AD (non-Alzheimer’s disease) cases were identified after a total observation time of 349,242 person-years, corresponding to a mean follow-up time (±SD) of 21.6 (±3.4) years).
2.5. Assessment of Anthropometric, Clinical and Lifestyle Data
Additional questionnaire information was collected at baseline on educational level, smoking status, and medical and reproductive history during in-person interviews. Data on prevalence of chronic diseases such as cancer, cardiovascular disease, or diabetes was self-reported. Height, weight, and waist and hip circumferences were measured by trained personnel during a physical examination following standard procedures [29]. Quetelet’s body mass index was estimated as weight (in kg) divided by squared height (in m).
2.6. Ethics
The EPIC study protocol was approved by the International Agency for Research on Cancer (IARC) Ethics Committee. All participants voluntarily agreed to take part and gave written informed consent. The current research has been conducted in accordance with the principles of the Declaration of Helsinki and the paper was written according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (https://www.equator-network.org/reporting-guidelines/strobe) (accessed on 22 November 2020).
2.7. Statistics
Baseline characteristics of cases and non-cases were described using absolute and relative frequencies for categorical variables and medians and inter-quartile ranges for continuous variables. Statistical differences by case status or rMED categories were assessed with χ2 (categorical variables), Mann-Whitney U (for comparisons between two groups) or Kruskal-Wallis (for comparisons across three categories) tests.
Hazard ratios (HR) of dementia and dementia sub-types were estimated using proportional hazards Cox models, stratified by age (in 5-year categories) and study setting (to account for heterogeneity in methods and population characteristics across centers). Entry time was defined as age at recruitment, and exit time was age at diagnosis for dementia cases, and age at death or censoring for non-cases. Adherence levels were defined based on the rMED, by categorizing the score into low (0–6 points), medium (7–10 points) and high (11–18 points) adherence groups. HR were estimated for ‘medium’ and ‘high’ rMED categories as compared with ‘low’ rMED scores, and for every 2-point increment of the score as a continuous variable under the linearity assumption. Besides, non-linear associations of dementia with the rMED score were evaluated after a restricted cubic spline transformation of the exposure (with internal knots at the 33rd and 67th centiles), by plotting the predicted HR against the rMED score. A basic model was fitted including sex, educational level, and energy intake as potential confounders, whereas final multivariable models were further adjusted by other variables which could partially account for confounding of the exposure–outcome association: smoking habit (never, former, current, unknown), BMI group (normal weight, overweight, obese), elevated waist circumference (≥102 cm in men, ≥88 cm in women), sum of household and recreational physical activity (in Metabolic equivalents [MET]∙h/week), self-reported hypertension (no, yes, unknown), self-reported hyperlipidemia (no, yes, unknown), combined coffee and tea consumption (in mL/day), and daily intake of potatoes, eggs, and cakes and biscuits, in (g/day). In women, final multivariable models also included menopausal status (pre-, peri-, post-menopausal), ever use of oral contraceptives (yes, no, unknown), and ever use of hormonal replacement therapy (yes, no, unknown). The proportional hazards assumption was checked in all models based on Schoenfeld residuals and visual inspection of log-log survival plots, and no significant deviations were found.
Main analyses were stratified by sex and dementia sub-types (AD and non-AD), whereas potential effect modification of the rMED and dementia association was evaluated for sex, educational level, smoking habit, and obesity. Likelihood ratio tests were used to assess heterogeneity.
Time-dependent effects were evaluated using flexible parametric Royston-Parmar (RP) multivariable models [30], by plotting the HR for high vs. low rMED scores over follow-up time. RP models implement the use of separate sets of spline terms to model baseline hazard rates and time-dependent effects of a covariate, thus allowing for a smooth representation of the change in HR over time.
All analyses were performed with Stata/SE v.14.2 (StataCorp LLC, College Station, TX, USA). Two-sided (when appropriate) p-values < 0.05 were considered statistically significant.
3. Results
459 incident cases of dementia (67% AD) were available for analysis after a mean of 21.6 (±3.4) years (see Figure S1, flowchart of study participants). Baseline characteristics of the sample, including dementia and AD cases, are summarized in Table 1.

Table 1.
Baseline characteristics of cases (overall dementia and Alzheimer’s disease, AD) and non-cases participating in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Spain Dementia Cohort study (N = 16,160).
Dementia cases were more likely to be older, obese, to have low educational level, higher intake of fruits and dairy products and lower intake of meat. On the contrary, non-cases were more likely to be smokers, and to consume more energy, alcohol, coffee and tea. High adherence to the rMED score was positively associated to age, male sex, obesity and intake of total energy and nuts (Supplementary Table S1). High adherence was also more frequent among post-menopausal women. By contrast, low adherence to the rMED was associated with smoking, leisure time physical activity, and higher intake of eggs, coffee and tea.
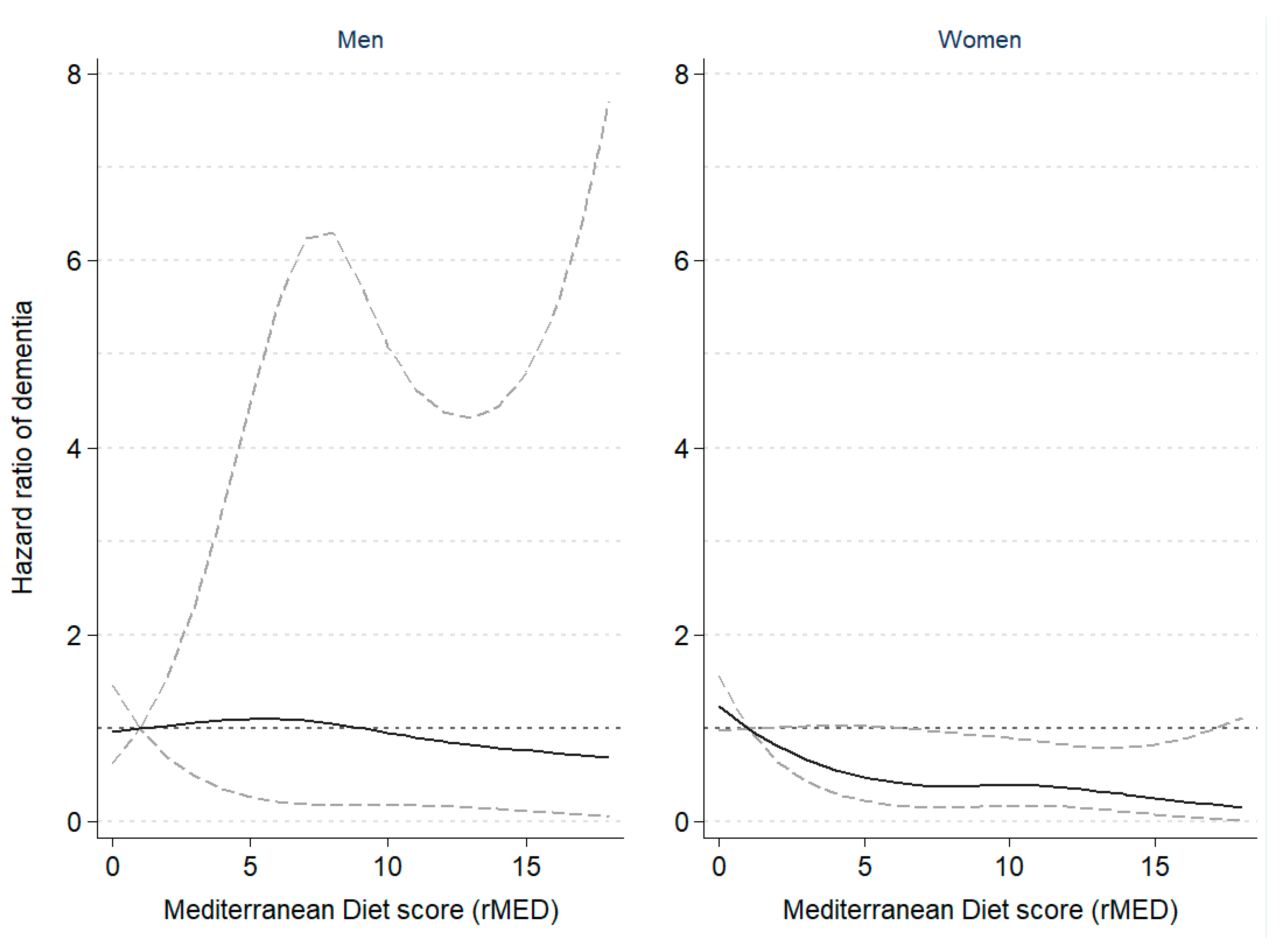
Table 2 shows the results of the multivariable survival analyses between rMED adherence and incidence of dementia, overall and by sex. A greater adherence to the MD pattern was associated with a 20% lower risk of dementia (HR = 0.80, 95%CI: 0.60–1.06), and an estimated 8% lower risk per 2-point increment in the continuous rMED score (p for linear trend on the continuous variable = 0.021). The negative trend was found to be statistically significant in women, but not men. The shape of the association is graphically illustrated in Figure 1, which shows the restricted cubic spline modelling of dementia risk by sex according to the rMED score, suggesting a non-linear relationship with a steeper slope at lower rMED scores. Although point estimates were similar in both sexes, results in men did not reach statistical significance.

Table 2.
Hazard ratio of dementia by levels of the Mediterranean Diet score (rMED) in participants from the EPIC-Spain Dementia Cohort study (N = 16,160).
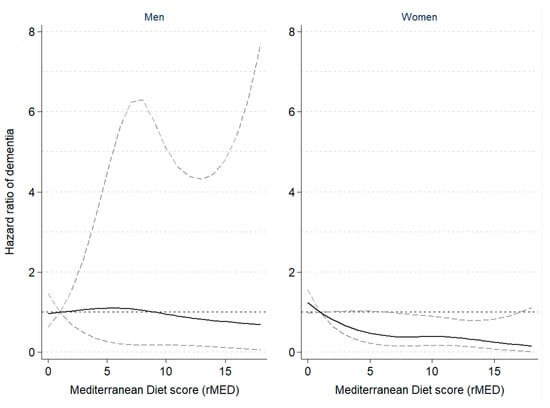
Figure 1.
Hazard ratio of dementia according to Mediterranean Diet scores in the EPIC-Spain Dementia Cohort study (N = 16,160), by sex. The dashed lines represent the upper and lower 95% confidence interval limits of the estimates. A non-linear inverse association between risk of dementia and rMED scores was observed among women, but not among men. Hazard ratios of dementia were estimated using Cox proportional hazards regression models, with age as the time scale, stratified by center and age (in 5-year categories), and adjusted by sex, education, energy intake, smoking, BMI category, elevated waist circumference, household and recreational physical activities, hypertension (self-reported), hyperlipidemia (self-reported), coffee and tea consumption (combined), and intake (in g/day per 2000 kcal) of potatoes, eggs, and cakes and biscuits. In women, models were further adjusted by menopausal status, oral contraceptive use, and hormone replacement therapy. Dementia risk was modelled following a restricted cubic spline transformation of the rMED variable with three degrees of freedom (knots were placed at the 33rd and 67th percentiles).
When further stratifying results by dementia sub-types, the negative association with MD was stronger among women for non-AD dementia, with up to 48% lower risk (95%CI: 0–73%) for those with high vs. low adherence to the MD, whereas rMED suggest a favorable while not statistically significant association with AD (HR high vs. low rMED = 0.57, 95%CI: 0.32–1.01) among men (Table 3). The decline in dementia risk was steeper for non-AD in the low-medium score range of the rMED, whereas the favorable association with AD risk suggested no evidence against a linear relationship (p for non-linearity = 0.353, Figure S5).

Table 3.
Hazard ratio of dementia sub-types (Alzheimer’s and non-Alzheimer’s disease) by levels of Mediterranean Diet score (rMED) in participants from the EPIC-Spain Dementia Cohort study (N = 16,160).
Table 4 evaluates selected variables as potential effect modifiers of the rMED and dementia association. The results, while not statistically significant, suggest a possible heterogeneity for education (p for interaction = 0.055). Among those with primary education or lower, participants with medium and high adherence to the MD had about 20% lower risk of dementia, a pattern that was not consistent in participants with higher education. The rMED score was significantly associated with dementia in women and non-smokers, but there was no formal evidence of heterogeneity by sex, smoking or ponderal status.

Table 4.
Hazard ratios of dementia by levels of adherence to the Mediterranean Diet score (rMED), stratified by selected variables in participants from the EPIC-Spain Dementia Cohort study (N = 16,160).
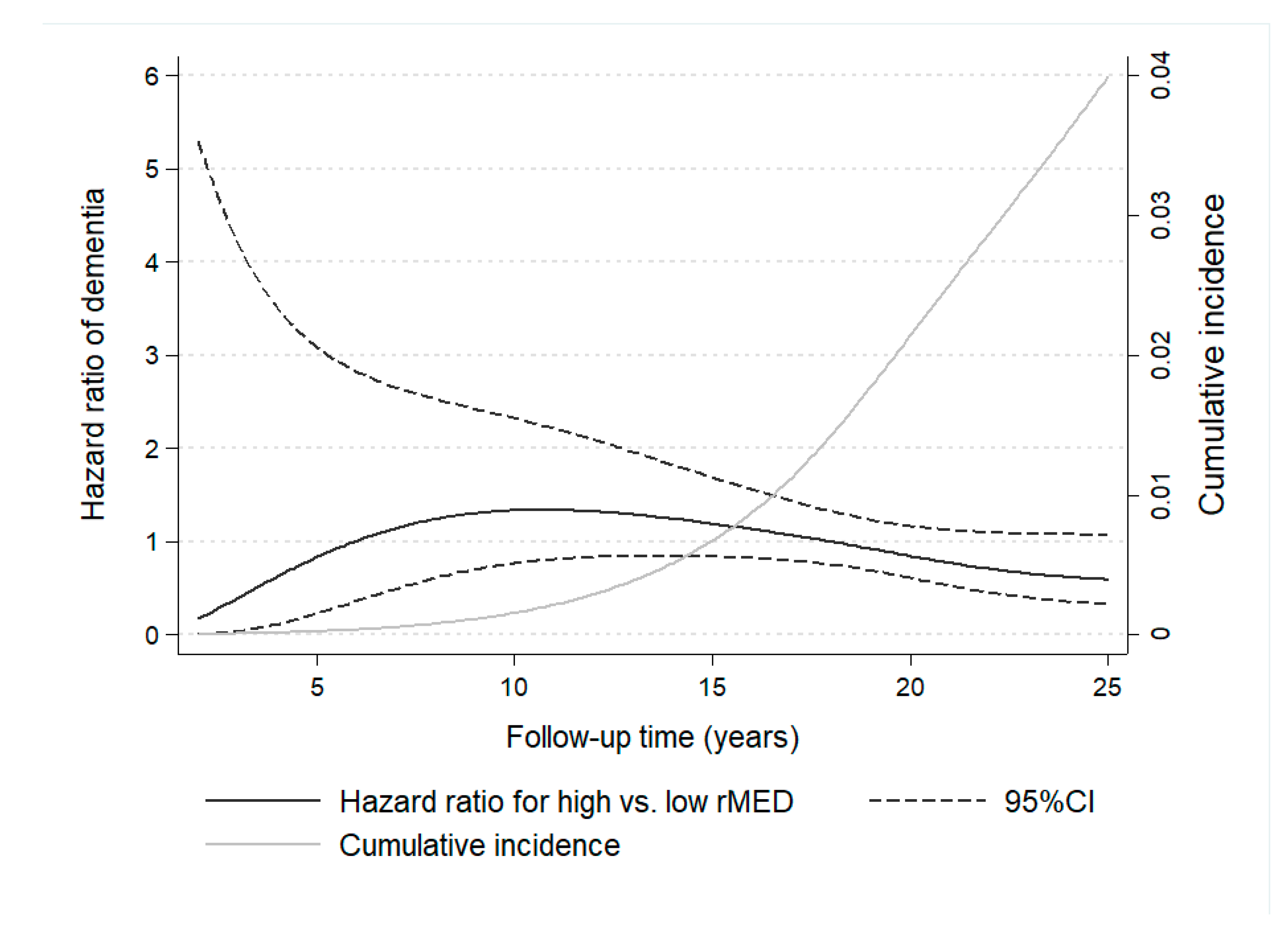
Time-varying effects for the association of rMED with dementia were studied by means of RP flexible survival models, by plotting HR as a function of follow-up time (Figure 2). Results show the change in HR estimates throughout study time, revealing that HR < 1 were detectable only after a long follow-up (~18 years), when the larger cumulative number of cases, which increased exponentially with the aging of the sample, allowed for more powerful estimations.
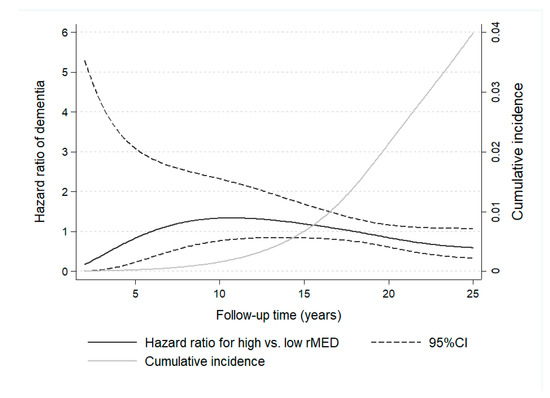
Figure 2.
Time-dependent variation in hazard ratio estimates of dementia for participants with high versus low Mediterranean Diet scores (rMED) throughout 25 years of follow-up in the EPIC-Spain Dementia Cohort study (N = 16,160). Hazard ratio estimates of dementia varied depending on follow-up time and cumulative number of cases. Time-varying hazard ratios were calculated using flexible parametric Royston-Parmar survival models, with time on study as the time scale. Models were adjusted by center, sex, education, energy intake, smoking, BMI category, elevated waist circumference, household and recreational physical activities, hypertension (self-reported), hyperlipidemia (self-reported), coffee and tea consumption (combined), and intake (in g/day per 2000 kcal) of potatoes, eggs, and cakes and biscuits.
The distribution of baseline characteristics and dietary variables according to rMED categories are shown in Table 5, separately for cases and non-cases. The rMED score was effective in ranking participants according to their dietary intake for almost all the diet groups included in the index (except for nuts and seeds, eggs, and coffee). Besides, higher adherence to the rMED score was associated with older age, female sex, lower prevalence of smoking, higher prevalence of obesity and less leisure time physical activity among non-cases. For cases, differences were only observed for sex and leisure time physical activity. Notably, there were no differences in educational level by rMED categories in any group.

Table 5.
Baseline characteristics of dementia cases and non-cases by rMED score group in the EPIC-Spain Dementia Cohort study (N = 16,160).
Further supplementary analyses showed the robustness of the results to exclusion of components of the rMED score on an item-by-item basis (Figure S2), to different multivariable models with increasing levels of adjustment, to other sensitivity analyses to account for potential reverse causation (Figure S3), and to alternative operational definitions of the MD (arMED, MDS, and aMED scores) (Figures S4 and S5). Stratification of the main analysis according to plausibility of energy reporting (Figure S6), which would eventually lead to misclassification of dietary exposure, resulted in wide confidence intervals and non-significant dementia risk estimates among mis-reporters (p for linear trend = 0.887).
4. Discussion
In this large prospective study involving 16,160 Spanish middle-aged and elderly participants followed up for over 20 years, participants with ‘high’ adherence compared to those with ‘low’ adherence to the MD (categorical rMED score) had a 20% lower risk of dementia overall. A negative linear trend was significant among women, those with lower educational level, and non-smokers. By dementia sub-types, associations were stronger for non-AD dementia in women and for AD in men. Of note, the association between rMED score and dementia incidence was revealed only after mis-reporters of energy intake were excluded from the analyses.
Mis-reporting (over- or under-reporting) of energy intake is a potential source of error in nutritional epidemiological studies, affecting the reliability and validity of nutritional assessment and confounding or attenuating diet–disease associations [31]. Different methods have been developed to minimize the impact of mis-reporting bias in epidemiological studies that rely on the ratio of reported intakes to predicted total energy expenditure, estimating a reference interval to account for individual variations in physical activity levels (PAL) [32,33]. Reporting of dietary energy can thus be regarded as plausible when the energy intake calculated from the dietary questionnaire falls within the defined cut-offs, or implausible (over- or under-reporting) otherwise. All EPIC-Spain participants have been assessed for reporting plausibility following the methodology described by Mendez et al. [23], and classified accordingly. Our data suggested that dietary mis-reporting had a non-negligible impact in the accuracy of estimates of dementia risk, increasing estimation errors and reducing the power to detect significant associations. Therefore, we have restricted the main analyses to plausible reporters.
There is suggestive evidence for a protective role of the MD with regards to dementia risk [2], and a considerable body of evidence suggests that MD may have a protective effect on cognition by decreasing the risk of cognitive impairment and delaying the onset of dementia [10,11,34]. In agreement with previous findings, our results suggest that adherence to MD could significantly decrease the risk of dementia [10,11]. Every 2-point increase of the rMED score was associated with an 8% and 6% lower risk of dementia and AD (although results for AD were not statistically significant), respectively, and results suggest a dose-response effect with decreasing risks of dementia at higher scores of the rMED score. However, a protective effect of the Mediterranean dietary pattern against cognitive decline or dementia has been reported in some [13,17,35,36,37,38] but not all [12,35] previous studies. Several cohort studies showed that the MD or its components were positively associated with a better cognition and contributed to delay the onset of mild cognitive impairment (MCI), both in Mediterranean [13,39,40] and non-Mediterranean countries [41,42]. Results from the PREDIMED (PREvención con DIeta MEDiterránea) clinical trial also support the role of MD and olive oil consumption on cognitive performance and the decreased risk of age-related cognitive decline [38]. However, only a few prospective cohort studies have examined the association between MD and the incidence of dementia or AD [12,13,17,37], and the available evidence in support of a protective role of MD remains inconclusive. In the WHICAP (Washington Heights-Inwood Columbia Aging Project) study, Scarmeas et al. [17] found that AD risk decreased by 9% for each additional point of the MeDi score (HR = 0.91; 95%CI: 0.83, 0.98), a MD scale ranging from 0 to 9 points, among 2258 US non-demented elderly followed up for a mean of 4 years. Furthermore, participants in the upper (vs. lower) scoring third had a 40% lower risk for AD (HR = 0.60, 95%CI: 0.42, 0.87). A further study by Morris et al. [16] in 923 US elderly participants from the Memory and Aging Project (MAP) project observed a 54% lower risk of AD when comparing participants in the high and low groups based on tertiles of the MIND (Mediterranean-Dietary Approach to Systolic Hypertension (DASH) diet intervention for neurodegenerative delay (MIND) diet) score (HR = 0.46; 95%CI: 0.27, 0.79), a hybrid pattern that combined the MD and the DASH diet. Our result of a negative trend in dementia risk for the continuous rMED score thus add to previous data in support of a beneficial role of MD against dementia. However, a recent large US prospective study conducted by Hu et al. in 13,630 participants from the Atherosclerosis Risk in Communities (ARIC) Study found no significant associations for the aMED, Healthy Eating Index (HEI)-2010 or DASH dietary scores with incident dementia after 27 years of follow-up [37]. This is in line with a previous French study by Feart et al. [12], who followed a cohort of 1410 French elderly over 5 years and could not find evidence that MD decreased dementia risk, despite a significant association with lower cognitive decline as defined by higher MMSE (Mini-Mental State Examination) scores. Of note, the authors acknowledged the limited power to detect significant effects given the scarce number of cases and short follow-up time. Nevertheless, in spite of the scarce prospective literature available on the association of MD with dementia or AD risk, our results add to previous evidence in support of a beneficial role for the MD against dementia [10,11,43,44].
The MD could be related to a lower risk of cognitive decline and dementia through several potential mechanisms, including anti-inflammatory, antioxidant and lipid-lowering actions [45] and a favorable effect on cardiovascular risk factors [44,46]. The MD has been associated with lower levels of C-reactive protein and interleukins, previously found to increase the risk of major forms of dementia such as AD and vascular dementia [47,48]. The MD has also been shown to modulate glucose and lipid metabolism, improving insulin sensitivity and blood lipid profile, and to reduce the risk for hypertension, diabetes, or cardiovascular disease [8,9,45], risk factors for cognitive decline and dementia [4]. Furthermore, several components of the MD, such as olive oil, fish, vegetables, or nuts are associated with reduced inflammation and dyslipidemia [46,49], and are rich in antioxidants, such as vitamin C, vitamin E, β-carotene, or flavonoids, and minerals such as selenium [45], that could ameliorate the age-related decline in cognitive function that precedes the onset of dementia. The pleiotropy of the advantageous effects of the MD supports the biological plausibility of its potential role in reduction of dementia risk.
We found no evidence that the association of rMED with dementia incidence varied by sex, smoking or obesity, which are potential effect modifiers as risk factors for dementia or cardio-metabolic risk that configure the patho-physiological environment (i.e., inflammation, oxidative stress, neurovascular dysfunction) through which diet would exert its effects on the prevention of dementia or dementia sub-types [44,45,46]. However, there was some indication of heterogeneity according to educational level (P interaction = 0.055), so that estimates were more robust among participants with lower education across categories of the rMED score. However, considering the similarity in HR estimates for the high vs. low rMED categories, as well as for the score as a continuous variable, the heterogeneity reported might be a consequence of the scarce number of events that occurred in the group with higher educational level, distorting the estimates in the medium score group. Our results were robust against a series of sensitivity analyses. The case-wise exclusion of single rMED components had little influence on overall estimates (Figure S2). We further excluded the first five years of follow-up and found no evidence of a potential reverse causation bias (Figure S3). We also considered alternative operational definitions of the MD, as the use of different scores might partly account for the heterogeneous results in the literature. We found a similar association in shape and magnitude for the rMED and arMED indices, which was slightly attenuated when using the MDS, whereas the aMED score showed a flat null association with dementia risk in our cohort. Of note, the aMED index was developed by Fung et al. as an attempt to adapt the MDS to the US population [27].
Our study has some limitations. The EPIC cohort was established to study the determinants of cancer and it was not designed to study dementia as a primary outcome. For this reason, participants did not undergo a baseline cognitive assessment to exclude prevalent cases of dementia or mild cognitive impairment. However, only one case was validated as prevalent after the revision of clinical records of potential cases, and the exclusion of participants with less than five years of follow-up had no impact on the estimates. Furthermore, diagnoses were based on clinical records available from the public health system, rather than prospective evaluations of the participants’ cognitive function. Nevertheless, given the universal coverage of the country health system, we are confident that virtually all incident cases from our cohort diagnosed with dementia have been ascertained. Other limitations are that diet and covariate data were only assessed at baseline and we could not evaluate potential exposure and lifestyle changes during follow-up, and that we had no data available on genetic traits such as apolipoprotein E (APOE) genotype (presence of ε4 alleles), although previous evidence suggest that APOE is not associated with MD and has no influence on the association between MD and AD [17]. As in any observational study, we cannot rule out the possibility of residual or unmeasured confounding. Finally, the lack of representativeness of the cohort could limit the generalizability of the results to other populations.
Major strengths of our study are the large sample size and long follow-up that were revealed as crucial for the ability to identify potential clinically relevant associations with sufficient precision. Nevertheless, statistical power could still be limited in some sub-group analyses with lower number of cases, as reflected by wider confidence intervals. Of note, this is the second largest study available evaluating the association of MD with a hard dementia or AD endpoint, and the largest ever conducted in a Mediterranean country. Lack of sufficient number of cases or an insufficient follow-up time could have prevented other studies from reaching significant conclusions. The Mediterranean setting of the cohort, its geographical variability within the country, and the availability of an extensive set of socio-demographic, lifestyle, and clinical variables to adjust for are important features. Finally, mis-reporting was shown to be an important factor to account for. Therefore, being able to control mis-reporters by identifying and excluding them from the analysis reinforced the accuracy of our estimates.
5. Conclusions
In conclusion, adherence to the Mediterranean diet was associated with a 20% lower risk of dementia overall in the EPIC-Spain Dementia Cohort, a Mediterranean study involving over 16,000 middle-aged and elderly participants followed up for over 20 years. Associations were stronger for non-AD dementia in women and for AD in men, and among participants with lower education. The significant associations were revealed after excluding energy mis-reporters and required a long follow-up time. Further cohort studies with sufficient number of cases and follow-up will contribute to reinforce the evidence for the role of MD in risk reduction of cognitive decline, dementia, and Alzheimer’s disease.
Supplementary Materials
The following are available online at https://www.mdpi.com/2072-6643/13/2/700/s1, Figure S1: Flow chart of study participants; Figure S2: Effect of an item-by-item exclusion of components of the Mediterranean Diet score (rMED) on the overall estimates of dementia risk; Figure S3: Sensitivity analyses of dementia risk under different multivariable models (M1–M6) in the EPIC-Spain Dementia Cohort study (N = 16,160); Figure S4: Hazard ratio of dementia in relation to alternative Mediterranean Diet scores in the EPIC-Spain Dementia Cohort study (N = 16,160); Figure S5: Hazard ratio of dementia, overall and by sub-types, according to Mediterranean Diet scores in the EPIC-Spain Dementia Cohort study (N = 16,160); Figure S6: Effect of dietary energy mis-reporting on hazard ratio estimates of dementia in the EPIC-Spain Dementia Cohort study (N =16,160). Table S1: Baseline characteristics of EPIC-Spain Dementia Cohort participants, by categories of the Mediterranean Diet score (N = 16,160).
Author Contributions
The authors’ responsibilities were as follows: M.E.A.-R. and J.M.H. performed the statistical analyses, drafted the manuscript, and had primary responsibility for the final content. M.D.C., C.N., D.G. and J.M.H. participated in the study conception and design. M.E.A.-R., J.M., M.T., R.L. conducted the case validation work. D.G., J.M.H., P.A., and E.A. coordinated the field work. S.M.C.-Y. and F.N.-M. revised the text for important intellectual content. All authors revised and approved the final manuscript. M.E.A.-R. and J.M.H. contributed equally to this work. All authors have read and agreed to the published version of the manuscript.
Funding
The EPIC study received financial support from the International Agency for Research on Cancer(AEP/93/06), the European Commission (SO-97-200302-05F02, SP23-CT-2005-006438), the Health Research Fund (FIS) of the Spanish Ministry of Health, the Red Temática de Investigación Cooperativa de Centros de Cáncer (RTICCC C03/10, RD06/0020), the CIBER de Epidemiología y Salud Pública (CIBERESP), the participating Regional Governments of Andalusia, Asturias, Basque Country, Murcia (no. 6236), and Navarra, and the Catalan Institute of Oncology (ICO). The present project received partial funding from the Fundación Séneca (19487/PI/14). The EPIC-Murcia study received partial funding from the Fundación Séneca (19487/PI/14), the Murcia Biomedical Research Institute (IMIB)-FFIS and the Spanish Biomedical Research Network Center (CIBER) (BOE-A-2020-6018).
Institutional Review Board Statement
The EPIC study protocol was approved by the IARC Ethics Committee. All participants voluntarily agreed to take part and gave written informed consent. The current research has been conducted according to the guidelines of the Declaration of Helsinki and the paper was written according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (https://www.equator-network.org/reporting-guidelines/strobe) (accessed on 22 November 2020).
Informed Consent Statement
All participants voluntarily agreed to take part and gave written informed consent.
Data Availability Statement
The data will be made available from the authors upon reasonable request.
Acknowledgments
We are grateful to Jone Alkorta for her assistance during the case validation phase of the study. We also thank all EPIC-Spain participants for their contribution to the study.
Conflicts of Interest
The authors declare no conflict of interest. Navarro-Mateu reports non-financial support from Otsuka outside the submitted work.
References
- Prince, M.; Wimo, A.; Guerchet, M.; Gemma-Claire, A.; Wu, Y.-T.; Prina, M. World Alzheimer Report 2015: The Global Impact of Dementia—An Analysis of Prevalence, Incidence, Cost and Trends; Alzheimer’s Disease International: London, UK, 2015. [Google Scholar]
- WHO. Risk reduction of cognitive decline and dementia: WHO guidelines. In Risk Reduction of Cognitive Decline and Dementia: WHO Guidelines; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- WHO. Towards a Dementia Plan: A WHO Guide; World Health Organization: Geneva, Switzerland, 2018; Licence: CC BY-NC-SA 3.0 IGO. [Google Scholar]
- Baumgart, M.; Snyder, H.M.; Carrillo, M.C.; Fazio, S.; Kim, H.; Johns, H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimers Dement. 2015, 11, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulos, D.; Lagiou, P. Editorial: Mediterranean diet and cardiovascular epidemiology. Eur. J. Epidemiol. 2003, 19, 7–8. [Google Scholar] [CrossRef]
- Davis, C.R.; Bryan, J.; Hodgson, J.M.; Murphy, K.J. Definition of the Mediterranean Diet; A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [CrossRef]
- Dinu, M.; Pagliai, G.; Casini, A.; Sofi, F. Mediterranean diet and multiple health outcomes: An umbrella review of meta-analyses of observational studies and randomised trials. Eur. J. Clin. Nutr. 2018, 72, 30–43. [Google Scholar] [CrossRef]
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2014, 17, 2769–2782. [Google Scholar] [CrossRef]
- Brink, A.C.V.D.; Brouwer-Brolsma, E.M.; Berendsen, A.A.M.; Van De Rest, O. The Mediterranean, Dietary Approaches to Stop Hypertension (DASH), and Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) Diets Are Associated with Less Cognitive Decline and a Lower Risk of Alzheimer’s Disease—A Review. Adv. Nutr. 2019, 10, 1040–1065. [Google Scholar] [CrossRef]
- Chen, X.; Maguire, B.; Brodaty, H.; O’Leary, F. Dietary Patterns and Cognitive Health in Older Adults: A Systematic Review. J. Alzheimers Dis. 2019, 67, 583–619. [Google Scholar] [CrossRef]
- Féart, C. Adherence to a Mediterranean Diet, Cognitive Decline, and Risk of Dementia. JAMA 2009, 302, 638–648. [Google Scholar] [CrossRef]
- Anastasiou, C.A.; Yannakoulia, M.; Kosmidis, M.H.; Dardiotis, E.; Hadjigeorgiou, G.M.; Sakka, P.; Arampatzi, X.; Bougea, A.; Labropoulos, I.; Scarmeas, N. Mediterranean diet and cognitive health: Initial results from the Hellenic Longitudinal Investigation of Ageing and Diet. PLoS ONE 2017, 12, e0182048. [Google Scholar] [CrossRef] [PubMed]
- Olsson, E.; Karlström, B.; Kilander, L.; Byberg, L.; Cederholm, T.; Sjögren, P. Dietary Patterns and Cognitive Dysfunction in a 12-Year Follow-up Study of 70 Year Old Men. J. Alzheimers Dis. 2014, 43, 109–119. [Google Scholar] [CrossRef]
- Haring, B.; Wu, C.; Mossavar-Rahmani, Y.; Snetselaar, L.; Brunner, R.; Wallace, R.B.; Neuhouser, M.L.; Wassertheil-Smoller, S. No Association between Dietary Patterns and Risk for Cognitive Decline in Older Women with 9-Year Follow-Up: Data from the Women’s Health Initiative Memory Study. J. Acad. Nutr. Diet. 2016, 116, 921–930.e1. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimers Dement. 2015, 11, 1007–1014. [Google Scholar] [CrossRef]
- Scarmeas, N.; Stern, Y.; Tang, M.-X.; Mayeux, R.; Luchsinger, J.A. Mediterranean diet and risk for Alzheimer’s disease. Ann. Neurol. 2006, 59, 912–921. [Google Scholar] [CrossRef]
- Riboli, E. The EPIC Project: Rationale and study design. European Prospective Investigation into Cancer and Nutrition. Int. J. Epidemiol. 1997, 26, S6–S14. [Google Scholar] [CrossRef]
- Riboli, E.; Hunt, K.J.; Slimani, N.; Ferraria, P.; Norata, T.; Faheya, M.; Charrondierea, U.R.; Hemona, B.; Casagrandea, C.; Vignata, J.; et al. European Prospective Investigation into Cancer and Nutrition (EPIC): Study populations and data collection. Public Health Nutr. 2002, 5, 1113–1124. [Google Scholar] [CrossRef]
- González, C.A.; Navarro, C.; Martínez, C.; Quirós, J.R.; Dorronsoro, M.; Barricarte, A.; Tormo, M.J.; Agudo, A.; Chirlaque, M.D.; Amiano, P.; et al. El estudio prospectivo europeo sobre cáncer y nutrición (EPIC). Rev. Española Salud Pública 2004, 78, 167–176. [Google Scholar] [CrossRef]
- Relative validity and reproducibility of a diet history questionnaire in Spain. I. Foods. EPIC Group of Spain. European Prospective Investigation into Cancer and Nutrition. Int. J. Epidemiol. 1997, 26, S91–S99. [CrossRef] [PubMed]
- Slimani, N.; Deharveng, G.; Unwin, I.; Southgate, D.A.T.; Vignat, J.; Skeie, G.; Salvini, S.; Parpinel, M.; Møller, A.; Ireland, J.; et al. The EPIC nutrient database project (ENDB): A first attempt to standardize nutrient databases across the 10 European countries participating in the EPIC study. Eur. J. Clin. Nutr. 2007, 61, 1037–1056. [Google Scholar] [CrossRef] [PubMed]
- Mendez, M.A.; Popkin, B.M.; Buckland, G.; Schroder, H.; Amiano, P.; Barricarte, A.; Huerta, J.-M.; Quirós, J.R.; Sánchez, M.-J.; A González, C. Alternative Methods of Accounting for Underreporting and Overreporting When Measuring Dietary Intake-Obesity Relations. Am. J. Epidemiol. 2011, 173, 448–458. [Google Scholar] [CrossRef]
- DeKoning, L.; Anand, S.S. Vascular viewpoint. Vasc. Med. 2004, 9, 145–146. [Google Scholar] [CrossRef]
- Buckland, G.; González, C.A.; Agudo, A.; Vilardell, M.; Berenguer, A.; Amiano, P.; Ardanaz, E.; Arriola, L.; Barricarte, A.; Basterretxea, M.; et al. Adherence to the Mediterranean Diet and Risk of Coronary Heart Disease in the Spanish EPIC Cohort Study. Am. J. Epidemiol. 2009, 170, 1518–1529. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; Travier, N.; Cottet, V.; González, C.; Luján-Barroso, L.; Agudo, A.; Trichopoulou, A.; Lagiou, P.; Trichopoulos, D.; Peeters, P.; et al. Adherence to the mediterranean diet and risk of breast cancer in the European prospective investigation into cancer and nutrition cohort study. Int. J. Cancer 2012, 132, 2918–2927. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; McCullough, M.L.; Newby, P.; E Manson, J.; Meigs, J.B.; Rifai, N.; Willett, W.C.; Hu, F.B. Diet-quality scores and plasma concentrations of markers of inflammation and endothelial dysfunction. Am. J. Clin. Nutr. 2005, 82, 163–173. [Google Scholar] [CrossRef]
- Andreu-Reinón, M.E.; Gavrila, D.; Chirlaque, M.D.; Colorado-Yohar, S.M.; Amiano, P.; Ardanaz, E.; Navarro-Mateu, F.; Navarro, C.; Huerta, J.M. Ascertainment of Dementia Cases in the Spanish European Prospective Investigation into Cancer and Nutrition-Murcia Cohort. Neuroepidemiology 2018, 52, 63–73. [Google Scholar] [CrossRef]
- Haftenberger, M.; Lahmann, P.; Panico, S.; Gonzalez-Martinez, A.; Seidell, J.C.; Boeing, H.; Giurdanella, M.C.; Krogh, V.; De Mesquita, H.B.B.; Peeters, P.H.M.; et al. Overweight, obesity and fat distribution in 50- to 64-year-old participants in the European Prospective Investigation into Cancer and Nutrition (EPIC). Public Health Nutr. 2002, 5, 1147–1162. [Google Scholar] [CrossRef]
- Royston, P.; Lambert, P.C. Flexible Parametric Survival Analysis Using Stata: Beyond the Cox Model, 1st ed.; STATA Press: College Station, TX, USA, 2011; p. 347. [Google Scholar]
- Livingstone, M.B.E.; Black, A.E. Markers of the Validity of Reported Energy Intake. J. Nutr. 2003, 133, 895S–920S. [Google Scholar] [CrossRef] [PubMed]
- E Black, A. Critical evaluation of energy intake using the Goldberg cut-off for energy intake:basal metabolic rate. A practical guide to its calculation, use and limitations. Int. J. Obes. 2000, 24, 1119–1130. [Google Scholar] [CrossRef]
- E Black, A.; Goldberg, G.R.; A Jebb, S.; Livingstone, M.B.; Cole, T.J.; Prentice, A.M. Critical evaluation of energy intake data using fundamental principles of energy physiology: 2. Evaluating the results of published surveys. Eur. J. Clin. Nutr. 1991, 45, 583–599. [Google Scholar]
- Aridi, Y.S.; Walker, J.L.; Wright, O.R.L. The Association between the Mediterranean Dietary Pattern and Cognitive Health: A Systematic Review. Nutrients 2017, 9, 674. [Google Scholar] [CrossRef]
- Psaltopoulou, T.; Kyrozis, A.; Stathopoulos, P.; Trichopoulos, D.; Vassilopoulos, D.; Trichopoulou, A. Diet, physical activity and cognitive impairment among elders: The EPIC–Greece cohort (European Prospective Investigation into Cancer and Nutrition). Public Health Nutr. 2008, 11, 1054–1062. [Google Scholar] [CrossRef]
- Shannon, O.M.; Stephan, B.C.M.; Granic, A.; Lentjes, M.; Hayat, S.; Mulligan, A.; Brayne, C.; Khaw, K.-T.; Bundy, R.; Aldred, S.; et al. Mediterranean diet adherence and cognitive function in older UK adults: The European Prospective Investigation into Cancer and Nutrition–Norfolk (EPIC-Norfolk) Study. Am. J. Clin. Nutr. 2019, 110, 938–948. [Google Scholar] [CrossRef]
- Hu, E.A.; Wu, A.; Dearborn, J.L.; Gottesman, R.F.; Sharrett, A.R.; Steffen, L.M.; Coresh, J.; Rebholz, C.M. Adherence to Dietary Patterns and Risk of Incident Dementia: Findings from the Atherosclerosis Risk in Communities Study. J. Alzheimers Dis. 2020, 78, 827–835. [Google Scholar] [CrossRef]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; De La Torre, R.; Martínez-González, M.Á.; Martínez-Lapiscina, E.H.; Fitó, M.; Pérez-Heras, A.; Salas-Salvadó, J.; et al. Mediterranean Diet and Age-Related Cognitive Decline: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 1094–1103. [Google Scholar] [CrossRef]
- Galbete, C.; Toledo, E.; Toledo, J.B.; Bes-Rastrollo, M.; Buil-Cosiales, P.; Marti, A.; Guillén-Grima, F.; Martínez-González, M.A. Mediterranean diet and cognitive function: The sun project. J. Nutr. Health Aging 2015, 19, 305–312. [Google Scholar] [CrossRef]
- Tanaka, T.; Talegawkar, S.A.; Jin, Y.; Colpo, M.; Ferrucci, L.; Bandinelli, S. Adherence to a Mediterranean Diet Protects from Cognitive Decline in the Invecchiare in Chianti Study of Aging. Nutrients 2018, 10, 2007. [Google Scholar] [CrossRef]
- Tangney, C.C.; Kwasny, M.J.; Li, H.; Wilson, R.S.; A Evans, D.; Morris, M.C. Adherence to a Mediterranean-type dietary pattern and cognitive decline in a community population. Am. J. Clin. Nutr. 2010, 93, 601–607. [Google Scholar] [CrossRef]
- Koyama, A.; Houston, D.K.; Simonsick, E.M.; Lee, J.S.; Ayonayon, H.N.; Shahar, D.R.; Rosano, C.; Satterfield, S.; Yaffe, K. Association Between the Mediterranean Diet and Cognitive Decline in a Biracial Population. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2015, 70, 354–359. [Google Scholar] [CrossRef]
- Singh, B.; Parsaik, A.K.; Mielke, M.M.; Erwin, P.J.; Knopman, D.S.; Petersen, R.C.; Roberts, R.O. Association of Mediterranean Diet with Mild Cognitive Impairment and Alzheimer’s Disease: A Systematic Review and Meta-Analysis. J. Alzheimers Dis. 2014, 39, 271–282. [Google Scholar] [CrossRef]
- Lourida, I.; Soni, M.; Thompson-Coon, J.; Purandare, N.; Lang, I.A.; Ukoumunne, O.C.; Llewellyn, D.J. Mediterranean Diet, Cognitive Function, and Dementia. Epidemiology 2013, 24, 479–489. [Google Scholar] [CrossRef]
- Tuttolomondo, A.; Simonetta, I.; Daidone, M.; Mogavero, A.; Ortello, A.; Pinto, A. Metabolic and Vascular Effect of the Mediterranean Diet. Int. J. Mol. Sci. 2019, 20, 4716. [Google Scholar] [CrossRef]
- Shen, J.; Wilmot, K.A.; Ghasemzadeh, N.; Molloy, D.L.; Burkman, G.; Mekonnen, G.; Gongora, M.C.; Quyyumi, A.A.; Sperling, L.S. Mediterranean Dietary Patterns and Cardiovascular Health. Annu. Rev. Nutr. 2015, 35, 425–449. [Google Scholar] [CrossRef]
- Lima, T.A.S.; Adler, A.L.; Minett, T.; Matthews, F.E.; Brayne, C.; Marioni, R.E.; on behalf of the Medical Research Council Cognitive Function and Ageing Study. C-reactive protein, APOE genotype and longitudinal cognitive change in an older population. Age Ageing 2014, 43, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Chrysohoou, C.; Panagiotakos, D.B.; Pitsavos, C.; Das, U.N.; Stefanadis, C. Adherence to the Mediterranean diet attenuates inflammation and coagulation process in healthy adults. J. Am. Coll. Cardiol. 2004, 44, 152–158. [Google Scholar] [CrossRef]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean Diet, its Components, and Cardiovascular Disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).