Exercise-Induced Hyperhomocysteinemia Is Not Related to Oxidative Damage or Impaired Vascular Function in Amateur Middle-Aged Runners under Controlled Nutritional Intake
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Experimental Design
2.3. Baseline Evaluation
2.4. Dietary Control
2.5. Blood Sampling
2.6. Biochemical Determinations
2.6.1. Homocysteine and Vitamins
2.6.2. Oxidative Stress
2.6.3. Endothelial Function
2.7. Statistical Analysis
3. Results
3.1. Physical Characteristics and Training Profile
3.2. tHcy Kinetics
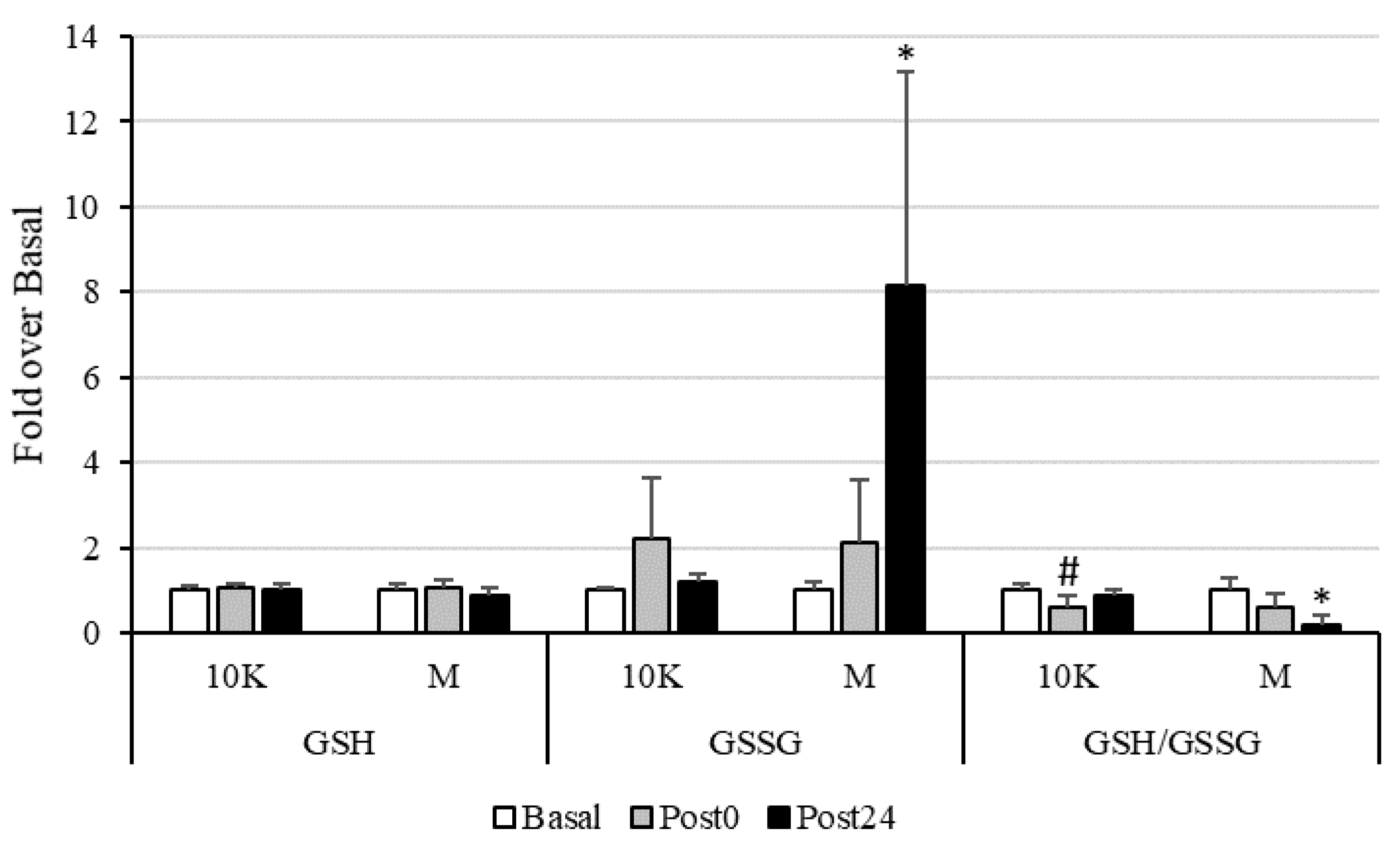
3.3. Oxidative Status
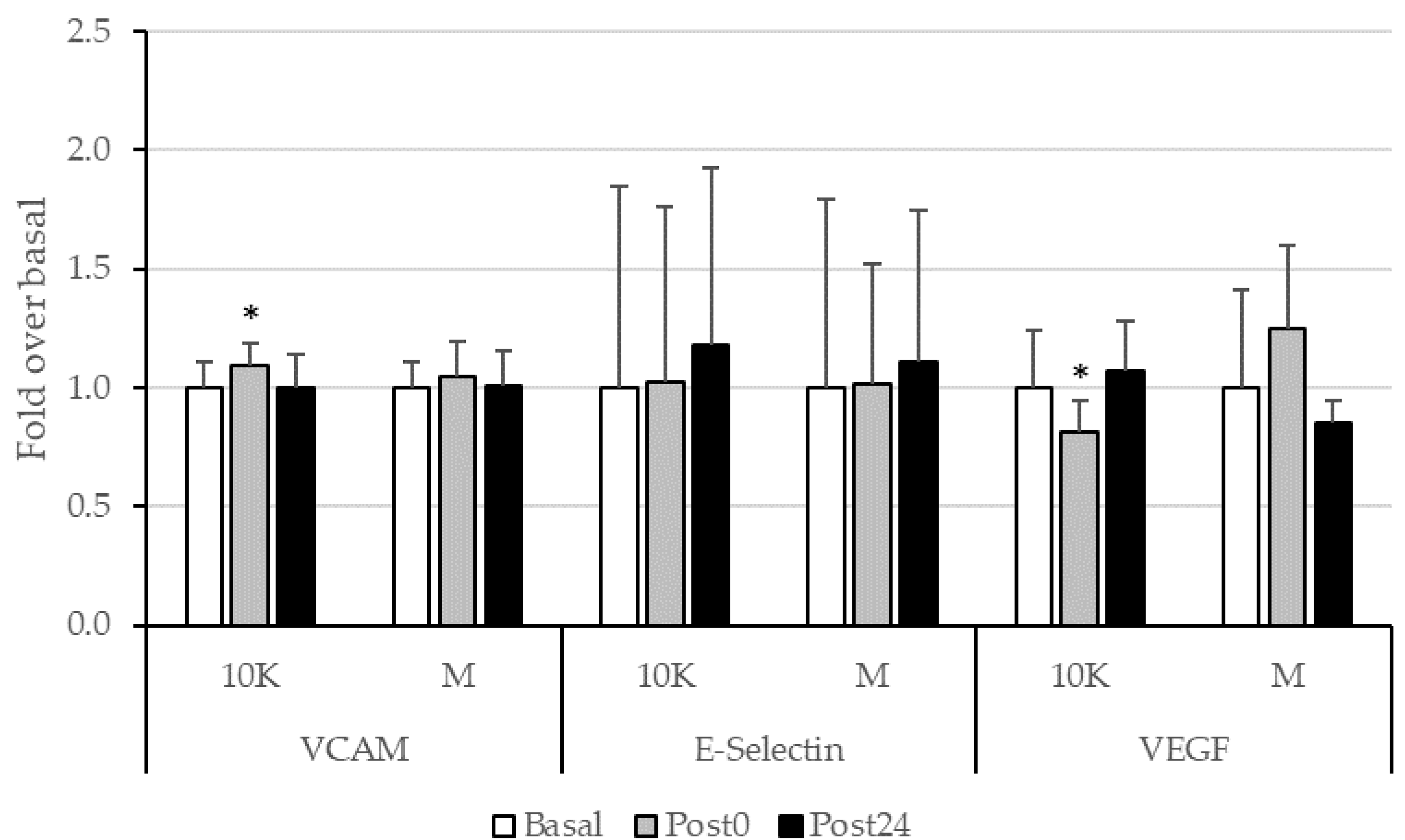
3.4. Endothelial Function
3.5. Vitamin Levels and Dietary Intake
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Myers, J. Exercise and cardiovascular health. Circulation 2003, 107, e2–e5. [Google Scholar] [CrossRef]
- Stewart, J.; Manmathan, G.; Wilkinson, P. Primary prevention of cardiovascular disease: A review of contemporary guidance and literature. JRSM Cardiovasc. Dis. 2017, 6, 2048004016687211. [Google Scholar] [CrossRef]
- Haskell, W.L.; Lee, I.M.; Pate, R.R.; Powell, K.E.; Blair, S.N.; Franklin, B.A.; Macera, C.A.; Heath, G.W.; Thompson, P.D.; Bauman, A. Physical activity and public health: Updated recommendation for adults from the American College of Sports Medicine and the American Heart Association. Med. Sci. Sport. Exerc. 2007, 39, 1423–1434. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sport. Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- WHO. Global Recommendations on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Prescribing exercise as preventive therapy. CMAJ 2006, 174, 961–974. [Google Scholar] [CrossRef] [PubMed]
- White, G.P. Clinical significance of cardiac damage and changes in function after exercise. Med. Sci. Sport. Exerc. 2008, 40, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, K.; Rohde, T.; Asp, S.; Schjerling, P.; Pedersen, B.K. Pro- and anti-inflammatory cytokine balance in strenuous exercise in humans. J. Physiol. 1999, 515, 287–291. [Google Scholar] [CrossRef]
- Jeukendrup, A.E.; Vet-Joop, K.; Sturk, A.; Stegen, J.H.; Senden, J.; Saris, W.H.; Wagenmakers, A.J. Relationship between gastro-intestinal complaints and endotoxaemia, cytokine release and the acute-phase reaction during and after a long-distance triathlon in highly trained men. Clin. Sci. (Lond) 2000, 98, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Comassi, M.; Vitolo, E.; Pratali, L.; Del Turco, S.; Dellanoce, C.; Rossi, C.; Santini, E.; Solini, A. Acute effects of different degrees of ultra-endurance exercise on systemic inflammatory responses. Intern. Med. J. 2015, 45, 74–79. [Google Scholar] [CrossRef]
- De Gonzalo-Calvo, D.; Dávalos, A.; Montero, A.; García-González, Á.; Tyshkovska, I.; González-Medina, A.; Soares, S.M.; Martínez-Camblor, P.; Casas-Agustench, P.; Rabadán, M.; et al. Circulating inflammatory miRNA signature in response to different doses of aerobic exercise. J. Appl. Physiol. (1985) 2015, 119, 124–134. [Google Scholar] [CrossRef]
- Rubio-Arias, J.Á.; Ávila-Gandía, V.; López-Román, F.J.; Soto-Méndez, F.; Alcaraz, P.E.; Ramos-Campo, D.J. Muscle damage and inflammation biomarkers after two ultra-endurance mountain races of different distances: 54 km vs. 111 km. Physiol. Behav. 2019, 205, 51–57. [Google Scholar] [CrossRef]
- Smith, K.A.; Kisiolek, J.N.; Willingham, B.D.; Morrissey, M.C.; Leyh, S.M.; Saracino, P.G.; Baur, D.A.; Cook, M.D.; Ormsbee, M.J. Ultra-endurance triathlon performance and markers of whole-body and gut-specific inflammation. Eur. J. Appl. Physiol. 2020, 120, 349–357. [Google Scholar] [CrossRef]
- Mrakic-Sposta, S.; Gussoni, M.; Vezzoli, A.; Dellanoce, C.; Comassi, M.; Giardini, G.; Bruno, R.M.; Montorsi, M.; Corciu, A.; Greco, F.; et al. Acute effects of triathlon race on oxidative stress biomarkers. Oxid. Med. Cell. Longev. 2020, 2020, 3062807. [Google Scholar] [CrossRef] [PubMed]
- Knez, W.L.; Coombes, J.S.; Jenkins, D.G. Ultra-endurance exercise and oxidative damage: Implications for cardiovascular health. Sport. Med. 2006, 36, 429–441. [Google Scholar] [CrossRef]
- Turner, J.E.; Hodges, N.J.; Bosch, J.A.; Aldred, S. Prolonged depletion of antioxidant capacity after ultraendurance exercise. Med. Sci. Sport. Exerc. 2011, 43, 1770–1776. [Google Scholar] [CrossRef] [PubMed]
- De Lucas, R.D.; Caputo, F.; Mendes de Souza, K.; Sigwalt, A.R.; Ghisoni, K.; Lock Silveira, P.C.; Remor, A.P.; da Luz Scheffer, D.; Guglielmo, L.G.; Latini, A. Increased platelet oxidative metabolism, blood oxidative stress and neopterin levels after ultra-endurance exercise. J. Sport. Sci. 2014, 32, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Vezzoli, A.; Dellanoce, C.; Mrakic-Sposta, S.; Montorsi, M.; Moretti, S.; Tonini, A.; Pratali, L.; Accinni, R. Oxidative stress assessment in response to ultraendurance exercise: Thiols redox status and ROS production according to duration of a competitive race. Oxid. Med. Cell. Longev. 2016, 2016, 6439037. [Google Scholar] [CrossRef] [PubMed]
- Trivax, J.E.; Franklin, B.A.; Goldstein, J.A.; Chinnaiyan, K.M.; Gallagher, M.J.; deJong, A.T.; Colar, J.M.; Haines, D.E.; McCullough, P.A. Acute cardiac effects of marathon running. J. Appl. Physiol. (1985) 2010, 108, 1148–1153. [Google Scholar] [CrossRef]
- Wilson, M.; O’Hanlon, R.; Prasad, S.; Oxborough, D.; Godfrey, R.; Alpendurada, F.; Smith, G.; Wong, J.; Basavarajaiah, S.; Sharma, S.; et al. Biological markers of cardiac damage are not related to measures of cardiac systolic and diastolic function using cardiovascular magnetic resonance and echocardiography after an acute bout of prolonged endurance exercise. Br. J. Sport. Med. 2011, 45, 780–784. [Google Scholar] [CrossRef]
- Scherr, J.; Braun, S.; Schuster, T.; Hartmann, C.; Moehlenkamp, S.; Wolfarth, B.; Pressler, A.; Halle, M. 72-h kinetics of high-sensitive troponin T and inflammatory markers after marathon. Med. Sci. Sport. Exerc. 2011, 43, 1819–1827. [Google Scholar] [CrossRef]
- Scott, J.M.; Esch, B.T.; Shave, R.; Warburton, D.E.; Gaze, D.; George, K. Cardiovascular consequences of completing a 160-km ultramarathon. Med. Sci. Sport. Exerc. 2009, 41, 26–34. [Google Scholar] [CrossRef]
- Seidl, J.; Asplund, C.A. Effects of excessive endurance activity on the heart. Curr. Sport. Med. Rep. 2014, 13, 361–364. [Google Scholar] [CrossRef]
- De Gonzalo-Calvo, D.; Dávalos, A.; Fernández-Sanjurjo, M.; Amado-Rodríguez, L.; Díaz-Coto, S.; Tomás-Zapico, C.; Montero, A.; García-González, Á.; Llorente-Cortés, V.; Heras, M.E.; et al. Circulating microRNAs as emerging cardiac biomarkers responsive to acute exercise. Int. J. Cardiol. 2018, 264, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.; Schorr, H.; Obeid, R.; Scharhag, J.; Urhausen, A.; Kindermann, W.; Herrmann, W. Homocysteine increases during endurance exercise. Clin. Chem. Lab. Med. 2003, 41, 1518–1524. [Google Scholar] [CrossRef] [PubMed]
- Real, J.T.; Merchante, A.; Gómez, J.L.; Chaves, F.J.; Ascaso, J.F.; Carmena, R. Effects of marathon running on plasma total homocysteine concentrations. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 134–139. [Google Scholar] [CrossRef]
- Refsum, H.; Ueland, P.M.; Nygard, O.; Vollset, S.E. Homocysteine and cardiovascular disease. Annu. Rev. Med. 1998, 49, 31–62. [Google Scholar] [CrossRef] [PubMed]
- Moat, S.J. Plasma total homocysteine: Instigator or indicator of cardiovascular disease? Ann. Clin. Biochem. 2008, 45, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.K.; Kan, M.Y. Homocysteine-induced endothelial dysfunction. Ann. Nutr. Metab. 2015, 67, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, P.; Alam, S.F. Role of homocysteine in the development of cardiovascular disease. Nutr. J. 2015, 14, 6. [Google Scholar] [CrossRef]
- Pushpakumar, S.; Kundu, S.; Sen, U. Endothelial dysfunction: The link between homocysteine and hydrogen sulfide. Curr. Med. Chem. 2014, 21, 3662–3672. [Google Scholar] [CrossRef]
- Škovierová, H.; Vidomanová, E.; Mahmood, S.; Sopková, J.; Drgová, A.; Červeňová, T.; Halašová, E.; Lehotský, J. The molecular and cellular effect of homocysteine metabolism imbalance on human health. Int. J. Mol. Sci. 2016, 17, 1733. [Google Scholar] [CrossRef] [PubMed]
- Esse, R.; Barroso, M.; Tavares de Almeida, I.; Castro, R. The contribution of homocysteine metabolism disruption to endothelial dysfunction: State-of-the-Art. Int. J. Mol. Sci. 2019, 20, 867. [Google Scholar] [CrossRef]
- Chrysohoou, C.; Panagiotakos, D.B.; Pitsavos, C.; Zeimbekis, A.; Zampelas, A.; Papademetriou, L.; Masoura, C.; Stefanadis, C. The associations between smoking, physical activity, dietary habits and plasma homocysteine levels in cardiovascular disease-free people: The ‘ATTICA’ study. Vasc. Med. 2004, 9, 117–123. [Google Scholar] [CrossRef]
- Maroto-Sánchez, B.; Lopez-Torres, O.; Palacios, G.; González-Gross, M. What do we know about homocysteine and exercise? A review from the literature. Clin. Chem. Lab. Med. 2016, 54, 1561–1577. [Google Scholar] [CrossRef]
- Deminice, R.; Ribeiro, D.F.; Frajacomo, F.T. The effects of acute exercise and exercise training on plasma homocysteine: A meta-analysis. PLoS ONE 2016, 11, e0151653. [Google Scholar] [CrossRef]
- Buckner, S.L.; Loenneke, J.P.; Loprinzi, P.D. Single and combined associations of accelerometer-assessed physical activity and muscle-strengthening activities on plasma homocysteine in a national sample. Clin. Physiol. Funct. Imaging 2017, 37, 669–674. [Google Scholar] [CrossRef]
- Marston, K.J.; Brown, B.M.; Rainey-Smith, S.R.; Bird, S.; Wijaya, L.; Teo, S.Y.M.; Laws, S.M.; Martins, R.N.; Peiffer, J.J. Twelve weeks of resistance training does not influence peripheral levels of neurotrophic growth factors or homocysteine in healthy adults: A randomized-controlled trial. Eur. J. Appl. Physiol. 2019, 119, 2167–2176. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-Gutiérrez, E.; Egan, B.; Díaz-Martínez, A.E.; Peñalvo, J.L.; González-Medina, A.; Martínez-Camblor, P.; O’Gorman, D.J.; Úbeda, N. Transient increase in homocysteine but not hyperhomocysteinemia during acute exercise at different intensities in sedentary individuals. PLoS ONE 2012, 7, e51185. [Google Scholar] [CrossRef] [PubMed]
- Úbeda, N.; Carson, B.P.; García-González, Á.; Aguilar-Ros, A.; Díaz-Martínez, Á.E.; Venta, R.; Terrados, N.; O’Gorman, D.J.; Iglesias-Gutiérrez, E. Muscular contraction frequency does not affect plasma homocysteine concentration in response to energy expenditure- and intensity-matched acute exercise in sedentary males. Appl. Physiol. Nutr. Metab. 2018, 43, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Sotgia, S.; Carru, C.; Caria, M.A.; Tadolini, B.; Deiana, L.; Zinellu, A. Acute variations in homocysteine levels are related to creatine changes induced by physical activity. Clin. Nutr. 2007, 26, 444–449. [Google Scholar] [CrossRef]
- Zinellu, A.; Sotgia, S.; Caria, M.A.; Tangianu, F.; Casu, G.; Deiana, L.; Carru, C. Effect of acute exercise on low molecular weight thiols in plasma. Scand. J. Med. Sci. Sport. 2007, 17, 452–456. [Google Scholar] [CrossRef]
- E Silva Ade, S.; da Mota, M.P. Effects of physical activity and training programs on plasma homocysteine levels: A systematic review. Amino Acids 2014, 46, 1795–1804. [Google Scholar] [CrossRef] [PubMed]
- Joubert, L.M.; Manore, M.M. The role of physical activity level and B-vitamin status on blood homocysteine levels. Med. Sci. Sport. Exerc. 2008, 40, 1923–1931. [Google Scholar] [CrossRef] [PubMed]
- Borrione, P.; Rizzo, M.; Spaccamiglio, A.; Salvo, R.A.; Dovio, A.; Termine, A.; Parisi, A.; Fagnani, F.; Angeli, A.; Pigozzi, F. Sport related hyperhomocysteinaemia: A putative marker of muscular demand to be noted for cardiovascular risk. Br. J. Sport. Med. 2008, 42, 594–600. [Google Scholar] [CrossRef]
- Kyle, U.G.; Genton, L.; Karsegard, L.; Slosman, D.O.; Pichard, C. Single prediction equation for bioelectrical impedance analysis in adults aged 20–94 years. Nutrition 2001, 17, 248–253. [Google Scholar] [CrossRef]
- Alvero Cruz, J.R.; Cabañas Armesilla, M.D.; Herrero de Lucas, A.; Martínez Riaza, L.; Moreno Pascual, C.; Porta Manzañido, J.; Sillero Quintana, M.; Sirvent Belando, J.E. Protocolo de valoración de la composición corporal para el reconocimiento médico-deportivo. Documento de consenso del Grupo Español de Cineantropometría de la Federación Española de Medicina del Deporte. Arch Med Deporte 2009, XXVI, 166–179. [Google Scholar]
- Moreiras, O.; Carbajal, A.; Cabrera, M.L.; Cuadrado, C. Tablas de Composición de Alimentos, 19th ed.; Pirámide: Madrid, Spain, 2018. [Google Scholar]
- SENC (Sociedad Española de Nutrición Comunitaria). Guías alimentarias para la población española. La nueva pirámide de la alimentación saludable. Nutr Hosp 2016, 33, 1–48. [Google Scholar]
- Lee, I.M. Dose-response relation between physical activity and fitness: Even a little is good; more is better. JAMA 2007, 297, 2137–2139. [Google Scholar] [CrossRef] [PubMed]
- Maslov, P.Z.; Schulman, A.; Lavie, C.J.; Narula, J. Personalized exercise dose prescription. Eur. Heart J. 2018, 39, 2346–2355. [Google Scholar] [CrossRef]
- Running USA. Available online: https://runningusa.org/RUSA/News/2018/U.S._Road_Race_Participation_Numbers_Hold_Steady_for_2017.aspx (accessed on 13 July 2002).
- Kim, J.H.; Malhotra, R.; Chiampas, G.; d’Hemecourt, P.; Troyanos, C.; Cianca, J.; Smith, R.N.; Wang, T.J.; Roberts, W.O.; Thompson, P.D.; et al. Race Associated Cardiac Arrest Event Registry (RACER) study group. Cardiac arrest during long-distance running races. N. Engl. J. Med. 2012, 366, 130–140. [Google Scholar] [CrossRef]
- Ueland, P.M.; Refsum, H.; Stabler, S.P.; Malinow, M.R.; Andersson, A.; Allen, R.H. Total homocysteine in plasma or serum: Methods and clinical applications. Clin. Chem. 1993, 39, 1764–1779. [Google Scholar] [CrossRef] [PubMed]
- Welch, G.N.; Loscalzo, J. Homocysteine and atherothrombosis. N. Engl. J. Med. 1998, 338, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.T.; Schalinske, K.L. Homocysteine metabolism and its relation to health and disease. Biofactors 2010, 36, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Manolescu, B.N.; Oprea, E.; Farcasanu, I.C.; Berteanu, M.; Cercasov, C. Homocysteine and vitamin therapy in stroke prevention and treatment: A review. Acta Biochim. Pol. 2010, 57, 467–477. [Google Scholar] [CrossRef]
- Schalinske, K.L.; Smazal, A.L. Homocysteine imbalance: A pathological metabolic marker. Adv. Nutr. 2012, 3, 755–762. [Google Scholar] [CrossRef]
- Benedetti, S.; Catalani, S.; Peda, F.; Luchetti, F.; Citarella, R.; Battistelli, S. Impact of the 24-h ultramarathon race on homocysteine, oxidized low-density lipoprotein, and paraoxonase 1 levels in professional runners. PLoS ONE 2018, 13, e0192392. [Google Scholar] [CrossRef]
- Knez, W.L.; Jenkins, D.G.; Coombes, J.S. Oxidative stress in half and full Ironman triathletes. Med. Sci. Sport. Exerc. 2007, 39, 283–288. [Google Scholar] [CrossRef]
- Skenderi, K.P.; Tsironi, M.; Lazaropoulou, C.; Anastasiou, C.A.; Matalas, A.L.; Kanavaki, I.; Thalmann, M.; Goussetis, E.; Papassotiriou, I.; Chrousos, G.P. Changes in free radical generation and antioxidant capacity during ultramarathon foot race. Eur. J. Clin. Investig. 2008, 38, 159–165. [Google Scholar] [CrossRef]
- Neubauer, O.; König, D.; Kern, N.; Nics, L.; Wagner, K.H. No indications of persistent oxidative stress in response to an ironman triathlon. Med. Sci. Sport. Exerc. 2008, 40, 2119–2128. [Google Scholar] [CrossRef]
- Pinho, R.A.; Silva, L.A.; Pinho, C.A.; Scheffer, D.L.; Souza, C.T.; Benetti, M.; Carvalho, T.; Dal-Pizzol, F. Oxidative stress and inflammatory parameters after an Ironman race. Clin. J. Sport. Med. 2010, 20, 306–311. [Google Scholar] [CrossRef]
- Pingitore, A.; Pereira Lima, G.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef]
- Pillon Barcelos, R.; Freire Royes, L.F.; Gonzalez-Gallego, J.; Bresciani, G. Oxidative stress and inflammation: Liver responses and adaptations to acute and regular exercise. Free Radic. Res. 2017, 51, 222–236. [Google Scholar] [CrossRef]
- De Sousa, C.V.; Magalhães Sales, M.; Santos Rosa, T.; Lewis, J.E.; Vieira de Andrade, R.; Simões, H.G. The antioxidant effect of exercise: A systematic review and meta-analysis. Sport. Med. 2017, 47, 277–293. [Google Scholar] [CrossRef] [PubMed]
- Margaritelis, N.V.; Theodorou, A.A.; Paschalis, V.; Veskoukis, A.S.; Dipla, K.; Zafeiridis, A.; Panayiotou, G.; Vrabas, I.S.; Kyparos, A.; Nikolaidis, M.G. Adaptations to endurance training depend on exercise-induced oxidative stress: Exploiting redox interindividual variability. Acta Physiol (Oxf). 2018, 222, e12898. [Google Scholar] [CrossRef]
- Farney, T.M.; McCarthy, C.G.; Canale, R.E.; Schilling, B.K.; Whitehead, P.N.; Bloomer, R.J. Absence of blood oxidative stress in trained men after strenuous exercise. Med. Sci. Sport. Exerc. 2012, 44, 1855–1863. [Google Scholar] [CrossRef]
- Ginsburg, G.S.; Agil, A.; O’Toole, M.; Rimm, E.; Douglas, P.S.; Rifai, N. Effects of a single bout of ultraendurance exercise on lipid levels and susceptibility of lipids to peroxidation in triathletes. JAMA 1996, 276, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Margaritis, I.; Tessier, F.; Richard, M.J.; Marconnet, P. No evidence of oxidative stress after a triathlon race in highly trained competitors. Int. J. Sport. Med. 1997, 18, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Jee, H.; Jin, Y. Effects of prolonged endurance exercise on vascular endothelial and inflammation markers. J. Sport. Sci. Med. 2012, 11, 719–726. [Google Scholar]
- Kargarfard, M.; Lam, E.T.C.; Shariat, A.; Mohammadi, M.A.; Afrasiabi, S.; Shaw, I.; Shaw, B.S. Effects of endurance and high intensity training on ICAM-1 and VCAM-1 levels and arterial pressure in obese and normal weight adolescents. Phys. Sportsmed. 2016, 44, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Reihmane, D.; Jurka, A.; Tretjakovs, P.; Dela, F. Increase in IL-6, TNF-α, and MMP-9, but not sICAM-1, concentrations depends on exercise duration. Eur. J. Appl. Physiol. 2013, 113, 851–858. [Google Scholar] [CrossRef]
- Akimoto, T.; Furudate, M.; Saitoh, M.; Sugiura, K.; Waku, T.; Akama, T.; Kono, I. Increased plasma concentrations of intercellular adhesion molecule-1 after strenuous exercise associated with muscle damage. Eur. J. Appl. Physiol. 2002, 86, 185–190. [Google Scholar] [CrossRef]
- Many, G.M.; Jenkins, N.T.; Witkowski, S.; Damsker, J.M.; Hagberg, J. The effects of aerobic training and age on plasma sICAM-1. Int. J. Sport. Med. 2013, 34, 253–257. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gu, J.W.; Gadonski, G.; Wang, J.; Makey, I.; Adair, T.H. Exercise increases endostatin in circulation of healthy volunteers. BMC Physiol. 2004, 4, 2. [Google Scholar]
- Bailey, A.P.; Shparago, M.; Gu, J.W. Exercise increases soluble vascular endothelial growth factor receptor-1 (sFlt-1) in circulation of healthy volunteers. Med. Sci. Monit. 2006, 12, CR45–CR50. [Google Scholar]
- Gliemann, L.; Gunnarsson, T.P.; Hellsten, Y.; Bangsbo, J. 10-20-30 training increases performance and lowers blood pressure and VEGF in runners. Scand. J. Med. Sci. Sport. 2015, 25, e479–e489. [Google Scholar] [CrossRef]
- Kraus, R.M.; Stallings, H.W.; Yeager, R.C.; Gavin, T.P. Circulating Plasma VEGF response to exercise in sedentary and endurance-trained men. J. Appl. Physiol. (1985) 2004, 96, 1445–1450. [Google Scholar] [CrossRef] [PubMed]
- Jürimäe, J.; Vaiksaar, S.; Purge, P. Circulating inflammatory cytokine responses to endurance exercise in female rowers. Int. J. Sport. Med. 2018, 39, 1041–1048. [Google Scholar] [CrossRef]
- Jürimäe, J.; Tillmann, V.; Purge, P.; Jürimäe, T. Body composition, maximal aerobic performance and inflammatory biomarkers in endurance-trained athletes. Clin. Physiol. Funct. Imaging 2017, 37, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Venta, R.; Cruz, E.; Valcarcel, G.; Terrados, N. Plasma vitamins, amino acids, and renal function in postexercise hyperhomocysteinemia. Med. Sci. Sport. Exerc. 2009, 41, 1645–1651. [Google Scholar] [CrossRef]



| Plasma Vitamin Concentration | Timepoint | 10K | M |
|---|---|---|---|
| Vitamin B12 (pmol/L) | Basal | 500.5 ± 86.1 1,a | 527.1 ± 75.5 1,a,b |
| Post0 | 547.1 ± 106.7 2,a | 584.2 ± 74.1 2,a,b | |
| Post24 | 472.0 ± 99.5 3 | 477.4 ± 72.1 3 | |
| Vitamin B6 (nmol/L) | Basal | 84.7 ± 52.9 | 111.4 ± 70.3 |
| Post0 | 70.2 ± 36.5 | 66.1 ± 25.9 | |
| Post24 | 63.6 ± 28.3 | 95.9 ± 56.0 | |
| Folate (nmol/L) | Basal | 7.6 ± 1.9 1,a | 10.9 ± 3.4 1,b |
| Post0 | 10.1 ± 3.0 2,3 | 11.2 ± 2.8 1,2 | |
| Post24 | 8.1 ± 2.7 1 | 8.9 ± 2.3.2 |
| Nutrient | Mean ± SD | Targets [48,49] |
|---|---|---|
| Energy intake (kcal/day) | 2671 ± 627 | - |
| Carbohydrates (% of Energy) | 45 ± 3 | 50–55 |
| Lipids (% of Energy) | 33 ± 5 | 30–35 |
| Proteins (g/day) | 90 ± 19 | 54 |
| Folates (µg) | 350 ± 128 | 400 |
| Vitamin B12 (µg) | 6.3 ± 2.9 | 2 |
| Vitamin B6 (mg) | 2.4 ± 1 | 1.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iglesias-Gutiérrez, E.; García-González, Á.; Montero-Bravo, A.; González-Medina, A.; Joglar, J.; Tomás-Zapico, C.; Fernández-García, B.; Fernández-Sanjurjo, M.; de Gonzalo-Calvo, D.; Díaz-Martínez, Á.E.; et al. Exercise-Induced Hyperhomocysteinemia Is Not Related to Oxidative Damage or Impaired Vascular Function in Amateur Middle-Aged Runners under Controlled Nutritional Intake. Nutrients 2021, 13, 3033. https://doi.org/10.3390/nu13093033
Iglesias-Gutiérrez E, García-González Á, Montero-Bravo A, González-Medina A, Joglar J, Tomás-Zapico C, Fernández-García B, Fernández-Sanjurjo M, de Gonzalo-Calvo D, Díaz-Martínez ÁE, et al. Exercise-Induced Hyperhomocysteinemia Is Not Related to Oxidative Damage or Impaired Vascular Function in Amateur Middle-Aged Runners under Controlled Nutritional Intake. Nutrients. 2021; 13(9):3033. https://doi.org/10.3390/nu13093033
Chicago/Turabian StyleIglesias-Gutiérrez, Eduardo, Ángela García-González, Ana Montero-Bravo, Antonio González-Medina, Judit Joglar, Cristina Tomás-Zapico, Benjamín Fernández-García, Manuel Fernández-Sanjurjo, David de Gonzalo-Calvo, Ángel Enrique Díaz-Martínez, and et al. 2021. "Exercise-Induced Hyperhomocysteinemia Is Not Related to Oxidative Damage or Impaired Vascular Function in Amateur Middle-Aged Runners under Controlled Nutritional Intake" Nutrients 13, no. 9: 3033. https://doi.org/10.3390/nu13093033
APA StyleIglesias-Gutiérrez, E., García-González, Á., Montero-Bravo, A., González-Medina, A., Joglar, J., Tomás-Zapico, C., Fernández-García, B., Fernández-Sanjurjo, M., de Gonzalo-Calvo, D., Díaz-Martínez, Á. E., & Úbeda, N. (2021). Exercise-Induced Hyperhomocysteinemia Is Not Related to Oxidative Damage or Impaired Vascular Function in Amateur Middle-Aged Runners under Controlled Nutritional Intake. Nutrients, 13(9), 3033. https://doi.org/10.3390/nu13093033







