Exocrine Pancreatic Function in Girls with Anorexia Nervosa
Abstract
:1. Introduction
2. Materials and Methods
- Cumulative dose of 13C-triglycerides recovered from exhaled air, defined as the percentage of 13CO2 excreted relative to the amount of 13C-triglycerides administered (% of cumulative dose (%CD);
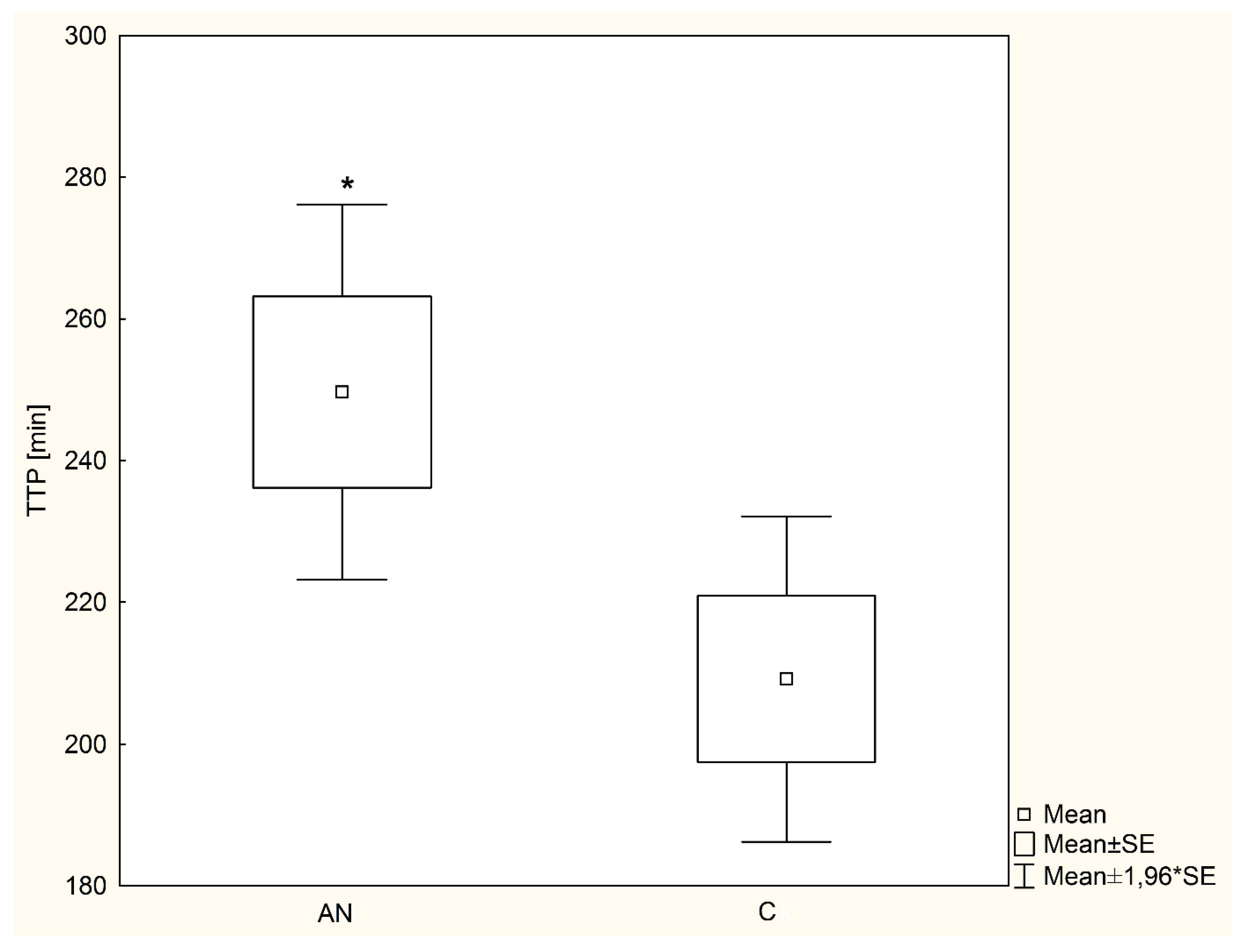
- Time from administration of 13C-triglycerides to obtaining the peak 13CO excretion (time to peak (TTP)).
Statistical Analysis
3. Results
4. Discussion
4.1. Leptin
4.2. Insulin
4.3. Ghrelin
4.4. Limitations of This Work and Directions for Further Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weterle-Smolińska, K.; Banasiuk, M.; Dziekiewicz, M.; Ciastoń, M.; Jagielska, G.; Banaszkiewicz, A. Zaburzenia motoryki przewodu pokarmowego u chorych na jadłowstręt psychiczny–przegląd piśmiennictwa [Gastrointestinal motility disorders in patients with anorexia nervosa–A review of the literature. Psychiatr. Pol. 2015, 49, 721–729. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Peterson, C.B. Anorexia Nervosa. N. Engl. J. Med. 2020, 382, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders(DSM-V); American Psychiatric Association: Washingon, DC, USA, 2013. [Google Scholar]
- Udo, T.; Grilo, C.M. Prevalence and correlates of DSM-5-defined eating disorders in a nationally representative sample of U.S. adults. Biol. Psychiatry 2018, 84, 345–354. [Google Scholar] [CrossRef]
- Treasure, J.; Zipfel, S.; Micali, N.; Wade, T.; Stice, E.; Claudino, A.; Schmidt, U.; Frank, G.K.; Bulik, C.M.; Wentz, E. Anorexia nervosa. Nat. Rev. Dis. Primers 2015, 1, 15074. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.; Golden, N.H.; Katzman, D.K.; Kreipe, R.E.; Rees, J.; Schebendach, J.; Sigman, G.; Ammerman, S.; Hoberman, H.M. Eating disorders in adolescents: A background paper. J. Adolesc. Health 1995, 16, 420–437. [Google Scholar] [CrossRef]
- Zipfel, S.; Giel, K.E.; Bulik, C.M.; Hay, P.; Schmidt, U. Anorexia nervosa: Aetiology, assessment, and treatment. Lancet Psychiatry 2015, 2, 1099–1111. [Google Scholar] [CrossRef]
- Berkowitz, S.A.; Witt, A.A.; Gillberg, C.; Rastam, M.; Wentz, E.; Lowe, M.R. Childhood body mass index in adolescent-onset anorexia nervosa. Int. J. Eat. Disord 2016, 49, 1002–1009. [Google Scholar] [CrossRef]
- Thornton, L.M.; Mazzeo, S.E.; Bulik, C.M. The heritability of eating disorders: Methods and current findings. Curr. Top. Behav. Neurosci. 2011, 6, 141–156. [Google Scholar]
- Grzelak, T.; Dutkiewicz, A.; Paszynska, E.; Dmitrzak-Weglarz, M.; Slopien, A.; Tyszkiewicz-Nwafor, M. Neurobiochemical and psychological factors influencing the eating behaviors and attitudes in anorexia nervosa. J. Physiol. Biochem. 2017, 73, 297–305. [Google Scholar] [CrossRef] [Green Version]
- Malczyk, Ż.; Oświęcimska, J. Powikłania gastroenterologiczne i zasady realimentacji pacjentów z jadłowstrętem psychicznym [Gastrointestinal complications and refeeding guidelines in patients with anorexia nervosa. Psychiatr. Pol. 2017, 51, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Winter, T.A.; Marks, T.; Callanan, M.; O’Keefe, S.J.; Bridger, S. Impaired pancreatic secretion in severely malnourished patients is a consequence of primary pancreatic dysfunction. Nutrition 2001, 17, 230–235. [Google Scholar] [CrossRef]
- Cleghorn, G.J.; Erlich, J.; Bowling, F.G.; Forrest, Y.; Greer, R.; Holt, T.L.; Shepherd, R.W. Exocrine pancreatic dysfunction in malnourished Australian aboriginal children. Med. J. 1991, 154, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Humphries, L.L.; Adams, L.J.; Eckfeldt, J.H.; Levitt, M.D.; McClain, C.J. Hyperamylasemia in patients with eating disorders. Ann Intern. Med. 1987, 106, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Nordgren, L.; von Schéele, C. Hepatic and pancreatic dysfunction in anorexia nervosa: A report of two cases. Biol. Psychiatry 1977, 12, 681–686. [Google Scholar] [PubMed]
- Backett, S.A. Acute pancreatitis and gastric dilatation in a patient with anorexia nervosa. Postgrad. Med. J. 1985, 61, 39–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-Olmos, M.A.; Peinó, R.; Prieto-Tenreiro, A.; Lage, M.; Nieto, L.; Lord, T.; Molina-Pérez, E.; Domínguez-Muñoz, J.E.; Casanueva, F.F. Intestinal absorption and pancreatic function are preserved in anorexia nervosa patients in both a severely malnourished state and after recovery. Eur. Eat. Disord. Rev. 2013, 21, 247–251. [Google Scholar] [CrossRef]
- Kobayashi, N.; Tamai, H.; Uehata, S.; Komaki, G.; Mori, K.; Matsubayashi, S.; Nakagawa, T. Pancreatic abnormalities in patients with eating disorders. Psychosom. Med. 1988, 50, 607–614. [Google Scholar] [CrossRef]
- Cox, K.L.; Cannon, R.A.; Ament, M.E.; Phillips, H.E.; Schaffer, C.B. Biochemical and ultrasonic abnormalities of the pancreas in anorexia nervosa. Dig. Dis. Sci. 1983, 28, 225–229. [Google Scholar] [CrossRef]
- Hempen, I.; Lehnert, P.; Fichter, M.; Teufel, J. Hyperamylasemia in anorexia nervosa and bulimia nervosa. Indication of a pancreatic disease? Dtsche Med. Wochenschr. 1989, 114, 1913–1916. [Google Scholar] [CrossRef]
- Misra, M.; Klibanski, A. Endocrine consequences of anorexia nervosa. Lancet Diabetes Endocrinol. 2014, 2, 581–592. [Google Scholar] [CrossRef] [Green Version]
- Chandra, R.; Liddle, R.A. Modulation of pancreatic exocrine and endocrine secretion. Curr. Opin. Gastroenterol. 2013, 29, 517–522. [Google Scholar] [CrossRef] [Green Version]
- Jaworek, J.; Nawrot-Porabka, K.; Leja-Szpak, A.; Konturek, S.J. Brain-gut axis in the modulation of pancreatic enzyme secretion. J. Physiol. Pharmacol. 2010, 61, 523–531. [Google Scholar]
- Murawska, S.; Kuczyńska, R.; Mierzwa, G.; Kulwas, A.; Rość, D.; Landowski, P.; Kamińska, B.; Czerwionka-Szaflarska, M. Ocena stężenia leptyny oraz rozpuszczalnej frakcji receptora dla niej u dzieci i młodzieży z chorobą Leśniowskiego-Crohna [Assessment of the concentration of leptin and its soluble fraction of its receptor in children and adolescents with Crohn’s disease]. Prz. Gastroenterol. 2009, 4, 262–272. [Google Scholar]
- Lammert, A.; Kiess, W.; Glasow, A.; Bottner, A.; Kratzsch, J. Different isoforms of the soluble leptin receptor determine the leptin binding activity of human circulating blood. Biochem. Biophys. Res. Commun. 2001, 283, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Owecki, M.; Nikisch, E.; Miczke, A.; Pupek-Musialik, D.; Sowinski, J. Leptin, soluble leptin receptors, free leptin index, and their relationship with insulin resistance and BMI: High normal BMI is the threshold for serum leptin increase in humans. Horm. Metab. Res. 2010, 42, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Ziora, K.; Geisler, G.; Dyduch, A.; Ostrowska, Z.; Schneiberg, B.; Oświęcimska, J. Stężenie leptyny w surowicy krwi u dziewcząt z jadłowstrętem psychicznym [Serum leptin concentration in girls with anorexia nervosa]. Endokrynol. Pediatric 2003, 1, 33–40. [Google Scholar]
- Misra, M.; Miller, K.K.; Almazan, C.; Ramaswamy, K.; Aggarwal, A.; Herzog, D.B.; Neubauer, G.; Breu, J.; Klibanski, A. Hormonal and body composition predictors of soluble leptin receptor, leptin, and free leptin index in adolescent girls with anorexia nervosa and controls and relation to insulin sensitivity. J. Clin. Endocrinol. Metab. 2004, 89, 3486–3495. [Google Scholar] [CrossRef] [Green Version]
- Baranowska-Bik, A.; Baranowska, B.; Martyńska, L.; Litwiniuk, A.; Kalisz, M.; Kochanowski, J.; Bik, W. Adipokine profile in patients with anorexia nervosa. Endokrynol. Pol. 2017, 68, 422–429. [Google Scholar] [CrossRef]
- Kapica, M.; Puzio, I.; Kato, I.; Kuwahara, A.; Zabielski, R. Role of feed-regulating peptides on pancreatic exocrine secretion. J. Physiol. Pharmacol. 2008, 59, 145–159. [Google Scholar] [PubMed]
- Elinson, N.; Amichay, D.; Birk, R.Z. Leptin directly regulates exocrine pancreas lipase and two related proteins in the rat. Br. J. Nutr 2006, 96, 691–696. [Google Scholar] [PubMed]
- Vacca, J.B.; Henke, W.J.; Knight, W.A., Jr. The exocrine pancreas in diabetes mellitus. Ann. Intern. Med. 1964, 61, 242–247. [Google Scholar] [CrossRef]
- Dostalova, I.; Smitka, K.; Papezova, H.; Kvasnickova, H.; Nedvidkova, J. Increased insulin sensitivity in patients with anorexia nervosa: The role of adipocytokines. Physiol. Res. 2007, 56, 587–594. [Google Scholar]
- Janas-Kozik, M. Rola wybranych czynników oreksygenicznych: Oreksyny A (OXA), oreksyny B (OXB), greliny (GRE) i anoreksygenicznych: Leptyny (LEP) w kontroli homeostazy organizmu w warunkach fizjologii [The role of selected orexigenic factors: Orexin A (OXA), orexin B (OXB), ghrelin (GRE) and anorexigenic factors: Leptin (LEP) in the control of homeostasis under physiological conditions]. Psychiatr. Psychol. Klin. 2005, 5, 87–94. [Google Scholar]
- Sedláčková, D.; Kopečková, J.; Papežová, H.; Vybíral, S.; Kvasničková, H.; Hill, M.; Nedvidkova, J. Changes of plasma obestatin, ghrelin and NPY in anorexia and bulimia nervosa patients before and after a high-carbohydrate breakfast. Physiol. Res. 2011, 60, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Germain, N.; Galusca, B.; Grouselle, D.; Frere, D.; Billard, S.; Epelbaum, J.; Estour, B. Ghrelin and obestatin circadian levels differentiate bingeing-purging from restrictive anorexia nervosa. J. Clin. Endocrinol. Metab. 2010, 95, 3057–3062. [Google Scholar] [CrossRef] [PubMed]
- Korek, E.; Krauss, H.; Gibas-Dorna, M.; Kupsz, J.; Piątek, M.; Piątek, J. Fasting and postprandial levels of ghrelin, leptin and insulin in lean, obese and anorexic subject. Prz. Gastroenterol. 2013, 8, 383–389. [Google Scholar] [CrossRef]
- Otto, B.; Cuntz, U.; Fruehauf, E.; Wawarta, R.; Folwaczny, C.; Riepl, R.L.; Heiman, M.L.; Lehnert, P.; Fichter, M.; Tschöp, M. Weight gain decreases elevated plasma ghrelin concentrations of patients with anorexia nervosa. Eur. J. Endocrinol. 2001, 145, 669–673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soriano-Guillén, L.; Vicente Barrios, V.; Campos-Barros, A.; Argente, J. Ghrelin levels in obesity and anorexia nervosa: Effect of weight reduction or recuperation. J. Pediatric 2004, 144, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Stock, S.; Leichner, P.; Wong, A.; Ghatei, M.; Kieffer, T.; Bloom, S.; Chanoine, G.-P. Ghrelin, peptide YY, glucose-dependent insulinotropic polypeptide, and hunger responses to a mixed meal in anorexic, obese, and control female adolescents. J. Clin. Endocrinol. Metab. 2005, 90, 2161–2168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korek, E.; Krauss, H.; Piątek, J.; Chęcińska, Z. Regulacja hormonalna łaknienia [Hormonal regulation of appetite]. Med. Ogólna Nauk. Zdrowiu 2013, 19, 211–217. [Google Scholar]
- Polińska, B.; Matowicka-Karna, J.; Kemona, H. Rola greliny w organizmie [The role of ghrelin in the body]. Postępy Hig. Med. Doswadczalnej 2011, 65, 1–7. [Google Scholar] [CrossRef]
- Schorr, M.; Miller, K.K. The endocrine manifestations of anorexia nervosa: Mechanisms and management. Nat. Rev. Endocrinol. 2017, 13, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Palczewska, I.; Niedźwiedzka, Z. Wskaźniki rozwoju somatycznego u dzieci i młodzieży warszawskiej [Somatic development indices in children and adolescents from Warsaw]. Med. Wieku Rozw. 2001, 5 (Suppl. 1), 1–112. [Google Scholar]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacker, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentration in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandhyamani, S.; Vijayakumari, A.; Balaraman Nair, M. Bonnet monkey model for pancreatic changes in induced malnutrition. Pancreas 1999, 18, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, W.R.; Vinijchaikul, K. The pancreas in kwashiorkor: An electron microscopic study. Lab. Investig. 1969, 20, 305–318. [Google Scholar]
- Braden, B. 13C breath tests for the assessment of exocrine pancreatic function. Pancreas 2010, 39, 955–959. [Google Scholar] [CrossRef] [PubMed]
- Maes, B.D.; Ghoos, Y.F.; Geypens, B.J.; Hiele, M.I.; Rutgeerts, P.J. Relation between gastric emptying rate and rate of intraluminal lipolysis. Gut 1996, 38, 23–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCallum, R.W.; Grill, B.B.; Lange, R.; Planky, M.; Glass, E.E.; Greenfeld, D.G. Definition of gastric emptying abnormality in patients with anorexia nervosa. Dig. Dis. Sci. 1985, 30, 713–722. [Google Scholar] [CrossRef]
- Abell, T.L.; Malagelada, J.R.; Lucas, A.R.; Brown, M.L.; Camilleri, M.; Go, V.L.; Azpiroz, F.; Callaway, C.W.; Kao, P.C.; Zinsmeister, A.R.; et al. Gastric electromechanical and neurohormonal function in anorexia nervosa. Gastroenterology 1987, 93, 958–965. [Google Scholar] [CrossRef]
- Jaworek, J.; Bonior, J.; Konturek, S.J.; Bilski, J.; Szlachcic, A.; Pawlik, W.W. Role of leptin in the control of postprandial pancreatic secretion. J. Physiol. Pharmacol. 2003, 54, 591–602. [Google Scholar]
- Matyjek, R.; Herzig, K.H.; Kato, S.; Zabielski, R. Exogenous lepton inhibits the secretion of pancreatic juice via a duodenal CCK1-vagal-dependent mechanism in anaesthetized rats. Regul. Pept. 2003, 114, 15–20. [Google Scholar] [CrossRef]
- Yadav, V.K.; Oury, F.; Suda, N.; Liu, Z.W.; Gao, X.B.; Confavreux, C.; Klemenhagen, K.C.; Tanaka, K.F.; Gingrich, J.A.; Guo, X.E.; et al. A serotonin-dependent mechanism explains the leptin regulation of bone mass, appetite, and energy expenditure. Cell 2009, 138, 976–989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tong, J.; D’Alessio, D. Eating disorders and gastrointestinal peptides. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 42–49. [Google Scholar] [CrossRef]
- Kentish, S.J.; O’Donnell, T.A.; Isaacs, N.J.; Young, R.L.; Li, H.; Harrington, A.M. Gastric vagal afferent modulation by leptin is influenced by food intake status. J. Physiol. 2013, 591, 1921–1934. [Google Scholar] [CrossRef] [PubMed]
- Barreto, S.G.; Carati, C.J.; Toouli, J.; Saccone, G.T. The islet-acinar axis of the pancreas: More than just insulin. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, 10–22. [Google Scholar] [CrossRef] [Green Version]
- Sjodin, L.; Holmberg, K.; Lyden, A. Insulin receptors on pancreatic acinar cells in guinea pigs. Endocrinology 1984, 115, 1102–1109. [Google Scholar] [CrossRef]
- Saisho, Y. Pancreas volume and fat deposition in diabetes and normal physiology: Consideration of the interplay between endocrine and exocrine pancreas. Rev. Diabet. Stud. 2016, 13, 132–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Chen, M.; Chen, Z.; Segura, B.J.; Mullholland, M.W. Inhibition of pancreatic protein secretion by ghrelin in therat. J. Physiol. 2001, 537, 231–236. [Google Scholar] [CrossRef]
- Schalla, M.A.; Stengel, A. The role of ghrelin in anorexia nervosa. Int. J. Mol. Sci. 2018, 19, 2117. [Google Scholar] [CrossRef] [Green Version]

| Parameter | AN (n = 31) | C (n = 38) | p (Cohen’s d Coefficient) |
|---|---|---|---|
| Mean ± SD (Min–Max) | |||
| Age (years) | 15.1 ± 1.6 (12.3–17.4) | 15.9 ± 0.9 (13.4–17.7) | 0.08 |
| Weight (kg) | 40.9 ± 6.4 (27.5–55.1) | 55.4 ± 7.2 (40.6–73.0) | <0.0001 (−2.26) |
| Weight-SDS | −1.61 ± 0.82 (–3.36 to 0.45) | −0.10 ± 0.87 (–2.04 to 2.05) | <0.0001 (−1.84) |
| Height (m) | 1.62 ± 0.78 (1.43–1.82) | 1.63 ± 0.62 (1.49–1.74) | 0.45 |
| BMI (kg/m2) | 15.38 ± 1.46 (10.7–17.58) | 20.59 ± 2.06 (15.66–25.40) | <0.0001 (–3.56) |
| BMI-SDS | −1.82 ± 0.61 (−3.88 to −0.81) | −0.08 ± 0.76 (−1.85 to 1.77) | <0.0001 (−2.85) |
| Max weight before illness (kg) | 58.0 ± 12.8 (32.00–90.00) | – | |
| Disease duration (months) | 9.5 ± 7.1 (2.0–34.0) | – | |
| Loss of weight (kg) | 17.1 ± 8.7 (4.5–37.2) | – | |
| Rate of loss of weight (kg/month) | 2.3 ± 1.4 (0.4–6.9) | – | |
| Duration of amenorrhea (months) | 5.4 ± 2.4 (1.0–10.0) | – | |
| Parameter | AN (n = 31) | C (n = 38) | p (Cohen’s d Coefficient) |
|---|---|---|---|
| Mean ± SD (Min–Max) | |||
| ALT (U/L) | 21.2 ± 18.8 (7.4–88.9) | 12.4 ± 4.7 (5.0–28.1) | 0.003 (0.46) |
| AST (U/L) | 22.5 ± 13.8 (12.8–84.6) | 18.0 ± 3.1 (13.8–28.2) | 0.21 |
| GGTP (U/L) | 14.4 ± 12.4 (6.0–70.0) | 12.3 ± 5.4 (6.0–35.0) | 0.57 |
| Total bilirubin (µmol/L) | 10.4 ± 6.4 (2.5–27.0) | 11.8 ± 11.2 (8.1–2.2) | 0.53 |
| Amylase (U/L) | 54.5 ± 25.4 (22.0–149.0) | 63.7 ± 21.8 (25.0–112.0) | 0.11 |
| Glucose (mg/dL) | 79.75 ± 7.34 (53.3–91.0) | 90.4 ± 8.7 (75.0–109.0) | <0.0001 (−1.45) |
| Insulin (µU/mL) | 4.62 ± 2.49 (1.32–12.90) | 10.82 ± 3.57 (4.61–18.62) | <0.0001 (−2.49) |
| HOMA-IR | 0.92 ± 0.53 (0.25–2.68) | 2.44 ± 088 (0.94–4.38) | <0.0001 (−2.87) |
| Leptin (ng/mL) | 1.26 ± 1.57 (0.12–7.42) | 11.51 ± 6.59 (1.16–25.28) | <0.0001 (−6.53) |
| sLR (ng/mL) | 12.13 ± 5.62 (6.79–33.42) | 6.27 ± 1.72 (3.57–13.32) | <0.0001 (1.04) |
| FLI | 0.12 ± 0.18 (0.01 –0.92) | 1.99 ± 1.35 (0.20–5.77) | <0.0001 (−10.39) |
| Acylated ghrelin (pg/mL) | 168.24 ± 259.73 (22.53–1420.95) | 52.01 ± 56.96 (18.93–289.92) | <0.0001 (0.45) |
| Desacylated ghrelin (pg/mL) | 741.15 ± 387.46 (143.79–1813.08) | 505.48 ± 178.74 (246.12–959.58) | <0.0001 (0.61) |
| Parameter | Group | |||
|---|---|---|---|---|
| AN (n = 31) | C (n = 38) | |||
| %CD (%) | TTP (min) | %CD (%) | TTP (min) | |
| Insulin (µU/mL) | r = −0.18 p = 0.31 | r = −0.11 p = 0.94 | r = −0.11 p = 0.50 | r = −0.36 p = 0.02 |
| HOMA-IR | r = −0.16 p = 0.36 | r = −0.01 p = 0.94 | r = −0.07 p = 0.68 | r = −0.36 p = 0.02 |
| Leptin (ng/mL) | r = −0.26 p = 0.14 | r = −0.13 p = 0.46 | r = −0.14 p = 0.40 | r = −0.21 p = 0.19 |
| sLR (ng/mL) | r = 0.01 p = 0.99 | r = −0.38 p = 0.03 | r = 0.02 p = 0.87 | r = −0.13 p = 0.42 |
| FLI | r = −0.77 p < 0.0001 | r = −0.36 p = 0.04 | r = −0.61 p < 0.0001 | r = −0.10 p = 0.55 |
| Acylated ghrelin (pg/mL) | r = 0.23 p = 0.20 | r = 0.15 p = 0.40 | r = −0.07 p = 0.67 | r = 0.25 p = 0.12 |
| Desacylated ghrelin (pg/mL) | r = −0.02 p = 0.88 | r = −0.23 p = 0.20 | r = −0.41 p = 0.01 | r = −0.01 p = 0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malczyk, Ż.; Roczniak, W.; Mazur, B.; Kwiecień, J.; Ziora, K.; Górska-Flak, K.; Oświęcimska, J. Exocrine Pancreatic Function in Girls with Anorexia Nervosa. Nutrients 2021, 13, 3280. https://doi.org/10.3390/nu13093280
Malczyk Ż, Roczniak W, Mazur B, Kwiecień J, Ziora K, Górska-Flak K, Oświęcimska J. Exocrine Pancreatic Function in Girls with Anorexia Nervosa. Nutrients. 2021; 13(9):3280. https://doi.org/10.3390/nu13093280
Chicago/Turabian StyleMalczyk, Żaneta, Wojciech Roczniak, Bogdan Mazur, Jarosław Kwiecień, Katarzyna Ziora, Karolina Górska-Flak, and Joanna Oświęcimska. 2021. "Exocrine Pancreatic Function in Girls with Anorexia Nervosa" Nutrients 13, no. 9: 3280. https://doi.org/10.3390/nu13093280






