1. Introduction
Prenatal alcohol exposure (PAE) can affect fetal development, resulting in altered neuroanatomy, neurophysiology, and increased risk of poor mental health outcomes. Fetal alcohol spectrum disorder (FASD) is the neurodevelopmental disability associated with PAE and the most common preventable cause of developmental disabilities in children [
1,
2,
3]. PAE is linked to aberrant structural and functional brain development, including globally reduced brain volume, reduced white and gray matter volume (see reviews [
4,
5]), altered cortical thickness [
6], altered white matter connectivity [
7,
8], and abnormal brain function [
9].
Approximately 90% of individuals with FASD have mental health problems [
10]. Evidence of mental health problems in infants with PAE can be observed as early as 2 years of age [
11] and continues to increase through childhood and adolescence, persisting into adulthood [
10]. Children with PAE experience more mental health problems than their ability-matched peers [
12]. Mental health problems in children can be thought of as internalizing (negative behaviors directed inwards, such as anxiety or depression) or externalizing (negative behaviors directed outwards, such as aggression and hyperactivity). Internalizing disorders frequently co-occur with FASD and are 4 and 11 times more common than in the general population, respectively [
13]. Similarly, externalizing disorders such as attention-deficit/hyperactivity disorder (ADHD) and oppositional defiant disorder are 10 and 13 times more common in individuals with PAE than in the general population, respectively [
13]. Unexposed youth and adults with internalizing and externalizing disorders tend to show volumetric abnormalities of limbic and basal ganglia structures [
14,
15,
16,
17,
18,
19,
20,
21,
22]. Youth with PAE have similar volumetric abnormalities [
23,
24,
25]; however, little research has investigated the neural underpinnings of internalizing or externalizing symptoms in individuals with PAE.
In the general population, brain iron, and in particular, iron deficiency anemia, has been linked to developmental disabilities and mental health problems in youth [
26] and adults [
27]. Early brain development requires iron for myelination [
28,
29], monoamine synthesis, and neurotransmitter metabolism [
29,
30]. In the first six months of life, infants depend on fetal iron stores accumulated during the third trimester of pregnancy [
31,
32]. Infants and children who have an iron deficiency show increased cognitive and affective problems (e.g., less social smiling and looking) [
33,
34]. Altered developmental and mental health outcomes associated with iron deficiency persist for years even after treatment [
35], highlighting the importance of sufficient iron availability throughout development.
Literature investigating brain iron in individuals with PAE is very limited. Prenatal maternal binge drinking increases the risk of iron deficiency anemia in infants at 12 months of age [
31]. In animal models, PAE alters brain iron bioavailability [
36], lowers fetal brain iron [
37], and behavioral deficits persist even after iron levels normalize [
38]. Blood iron deficiency in infancy is related to emotional withdrawal, which predicts affective problems in childhood [
39]. Of note, the relation between blood iron and brain iron is not well understood, and blood iron does not always correlate with brain iron, especially in children with neurodevelopmental disabilities [
40]. In other neurodevelopmental disabilities, preschoolers with autism have lower brain iron in the caudate nucleus [
41], and children with ADHD have lower brain iron in the caudate, putamen, and thalamus compared to controls [
40,
41,
42]. Although brain iron is associated with poor developmental and mental health outcomes often experienced by individuals with PAE, no studies to date have looked at brain iron in youth with PAE and how it is associated with internalizing and externalizing issues.
Transverse relaxation rates (
R2*) and susceptibility-weighted imaging (SWI) have both been validated as correlated to iron concentration [
43,
44,
45,
46], though they are indirectly related to brain iron and lack specificity [
47,
48]. Quantitative susceptibility mapping (QSM) measures tissue susceptibility, an intrinsic property that determines how tissue will interact with a magnetic field and provides a more reliable, reproducible, and valid measure of brain iron. Iron is paramagnetic (positive susceptibility) and has been validated post mortem as the primary susceptibility source in subcortical gray matter [
49,
50].
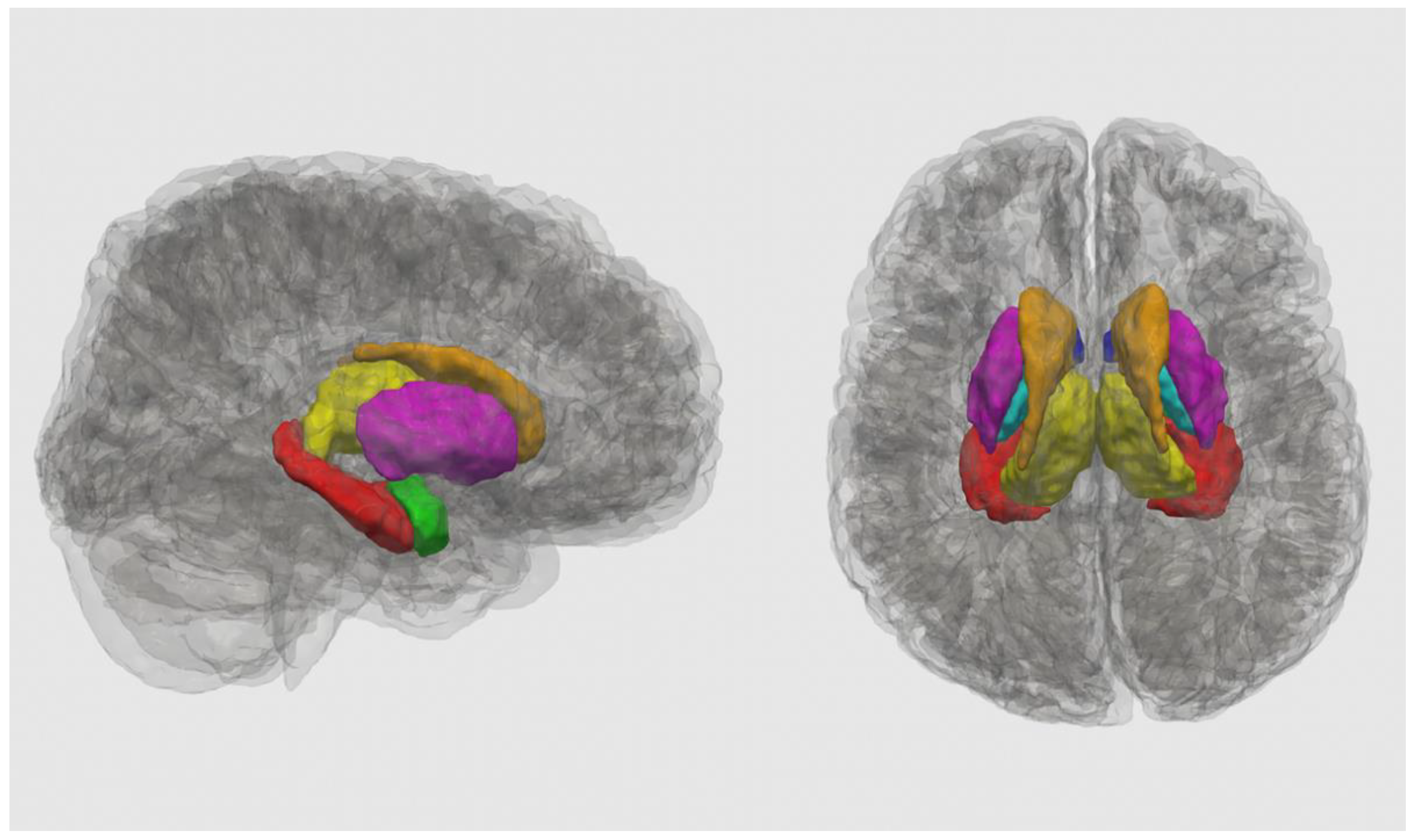
Here, we examined the association between brain iron and internalizing or externalizing symptoms in youth and determined how the group (exposed versus unexposed) moderates this association. Consistent with previous literature, we focused on key brain regions: the basal ganglia (nucleus accumbens, caudate, putamen, and pallidum), a primary site of iron accumulation [
50,
51], as well as limbic structures (thalamus, hippocampus, and amygdala) that have been implicated in both PAE and mental health problems [
5,
14,
15].
Based on previous research on brain iron in children with externalizing symptoms [
40,
41,
42] and adults with internalizing symptoms [
52,
53], it was expected that across diagnostic groups, a lower susceptibility of the caudate and putamen would be associated with more internalizing and externalizing symptoms. Further, a group–susceptibility interaction was anticipated, such that lower susceptibility in the thalamus would only be associated with more internalizing and externalizing symptoms in the PAE group. Consistent with Krueger and colleagues, who found a trend-level association between caudate volume and internalizing symptoms in children with PAE [
54], we hypothesized that the PAE group would have a lower caudate volume and that this would be associated with more internalizing symptoms.
4. Discussion
For the first time, we showed that brain iron is associated with internalizing and externalizing symptoms in youth and, in some cases, was moderated by PAE. Across groups, less brain iron (lower susceptibility) in the nucleus accumbens was associated with more internalizing symptoms, while in the amygdala, higher brain iron (greater susceptibility) was associated with more internalizing symptoms. This suggests that the effects of brain iron on mental health symptoms differ regionally. Our findings in the thalamus and putamen demonstrate that PAE not only alters brain iron but also disrupts the brain–behavior relationship. No associations between brain volumes and mental health symptoms were found, suggesting that more specific measures such as susceptibility may be helpful in determining what may be contributing to mental health problems in this population.
As expected, the PAE group had significantly more externalizing symptoms than unexposed controls. Externalizing symptoms are well-recognized in individuals with PAE; ADHD (an externalizing disorder) occurs 10 times more often in people with PAE than those unexposed, and it is the most common co-occurring disorder with FASD [
13]. Internalizing symptoms did not differ significantly between groups. Many studies report that externalizing symptoms are more affected by PAE than internalizing symptoms at this age [
12]. However, internalizing symptoms can be hard to measure with parent reports since internalizing symptoms are not as easily observed, which may contribute to an under-reporting of internalizing symptoms across one or both groups.
Iron is an essential nutrient for proper brain development and behavior. Alcohol consumption during pregnancy can affect fetal iron stores [
37], disrupting postnatal brain development via myelination, neurotransmitter synthesis and metabolism, or ATP synthesis. While PAE may affect iron in white matter, QSM is best suited to detecting iron in gray matter, and therefore we focused on gray matter structures. QSM can distinguish between paramagnetic and diamagnetic material [
68,
69]; however, it cannot identify the specific components contributing to susceptibility. In subcortical structures, myelin and calcium contribute diamagnetic (negative) susceptibility [
69,
70] and heme iron, FeS clusters, iron isotopes, transferrin, and ferritin contribute paramagnetic (positive) susceptibility. More than 97% of iron in cells (and up to 90% of the iron in the brain) is stored as ferritin [
70,
71], and post mortem studies show that ferritin is the primary susceptibility source in subcortical structures [
49,
50]. Increased ferritin iron stores may reflect sufficient brain iron available for use, stored iron that is unavailable, or an overload of brain iron. Raghunathan and colleagues [
72] propose that PAE alters brain vasculature, resulting in decreased brain vessel diameter, which may decrease heme iron. However, heme iron is not expected to contribute significantly to susceptibility [
73] in subcortical brain structures. Therefore, the present study findings likely reflect primarily contributions from ferritin in the subcortical brain regions.
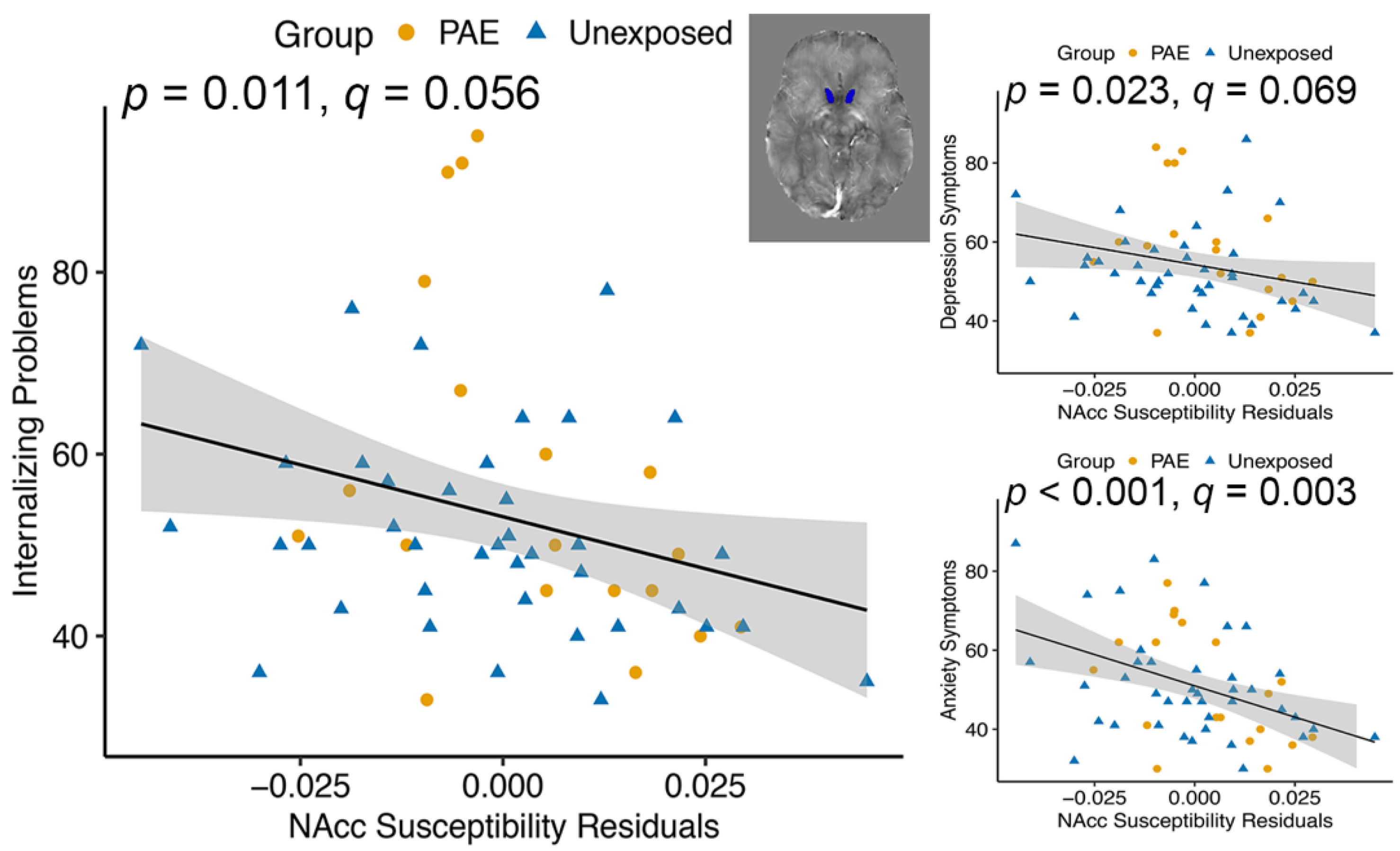
The nucleus accumbens plays an important role in motivation and emotional processing [
74] and is implicated in a number of psychological disorders, including depression and anxiety [
75]. Based on previous research, we had no hypothesis about brain iron in the nucleus accumbens and internalizing or externalizing symptoms, but found it was associated with internalizing symptoms, driven mostly by anxiety symptoms. The nucleus accumbens is part of the mesolimbic dopaminergic pathway, so low iron may impact behavior through dopamine metabolism. Iron-deficient rats show decreased dopamine transporter (DAT) in the striatum and nucleus accumbens, which can result in elevated dopamine, leading to desensitization of the DAT [
76]. Similarly, Youdim [
77] found decreased dopamine 2 (D2) receptors in the nucleus accumbens of iron-deficient rats; D2 receptors reduce acetylcholine release [
78], the main neurotransmitter of the parasympathetic nervous system. While this evidence suggests that iron in the nucleus accumbens affects dopamine synthesis and metabolism, other neurotransmitters such as GABA, glutamate, and serotonin may also be affected.
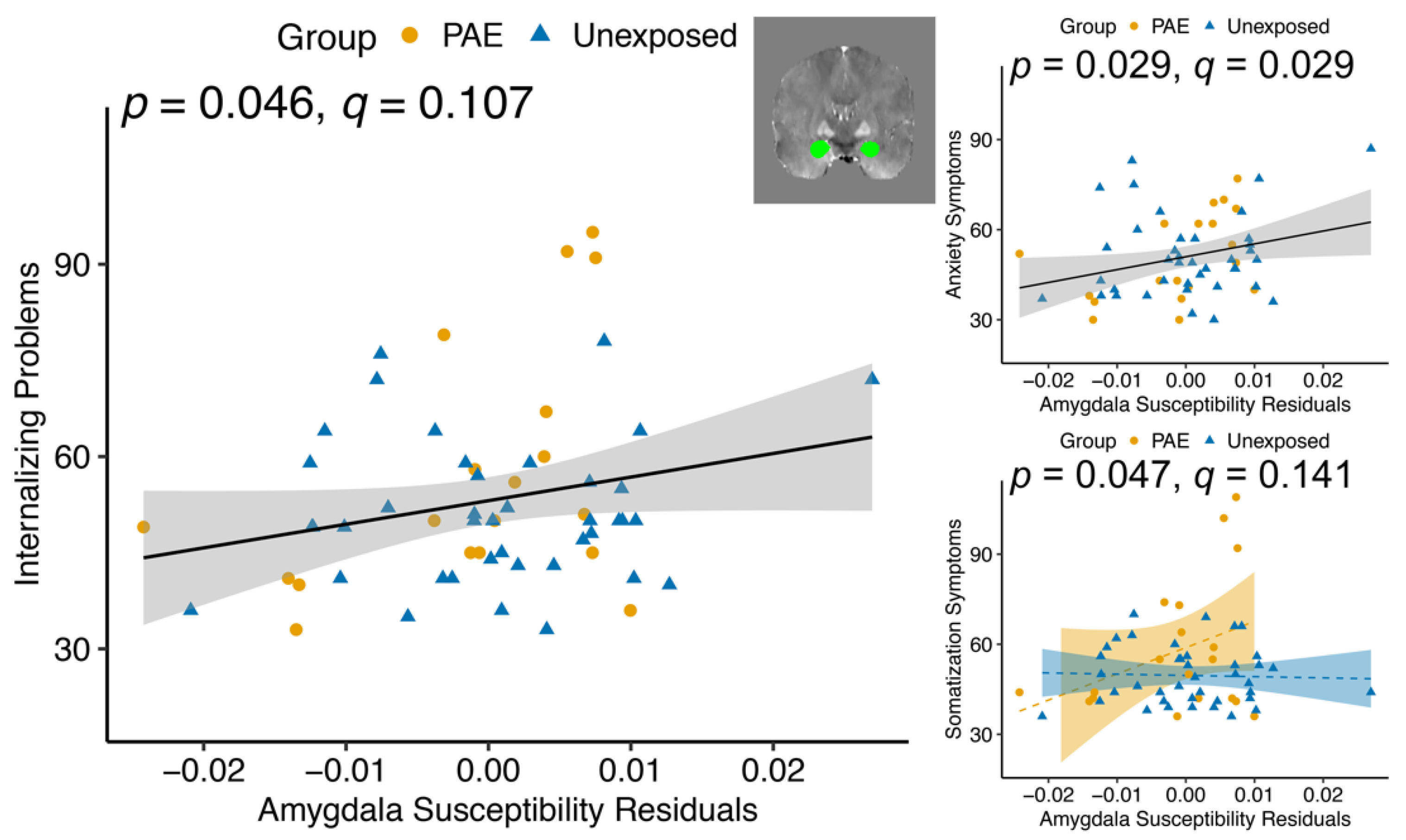
The amygdala plays an important role in limbic circuits and emotional responses, including fear and anxiety [
79]. Amygdala iron was positively related to Internalizing Problems and Anxiety symptoms in youth with and without PAE. We found a significant group–susceptibility interaction effect in the amygdala for Somatization symptoms. Post hoc testing showed that the PAE group had a more positive Somatization–susceptibility relationship, and the unexposed group had a slightly negative association, though neither of the relationships within groups was significant. Inhibitory circuits in the amygdala, which are highly dependent on GABA, are important for promoting and suppressing anxiety-like behavior [
80]. The amygdala tends to have stable, low levels of brain iron across the lifespan [
81]. It is possible that small perturbations in brain iron have a large effect on GABA metabolism and, therefore, on anxiety-like behavior in this brain region.
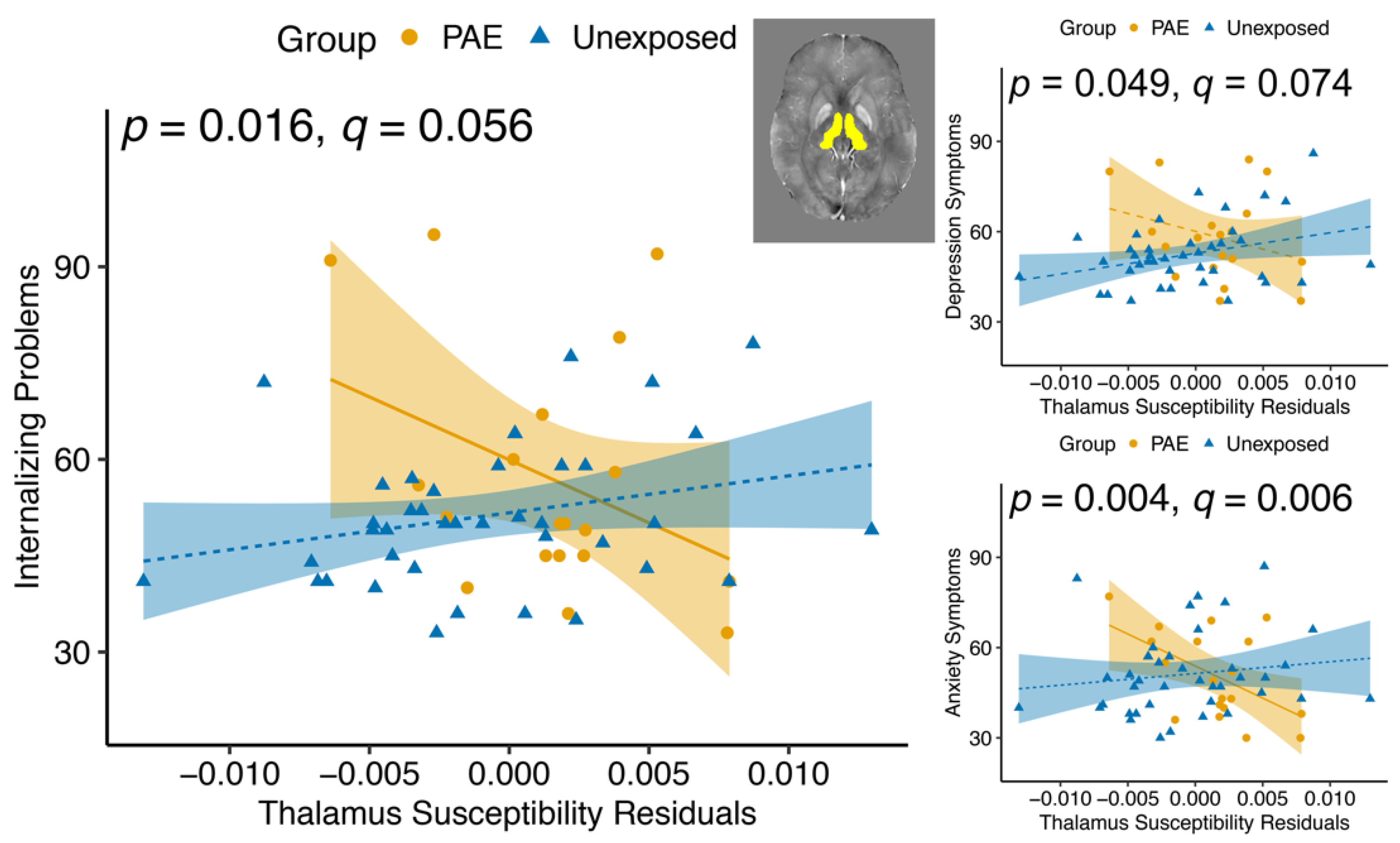
The thalamus is involved in limbic, cognitive, and sensorimotor circuits and acts primarily as a relay station [
82]. We found a group–susceptibility interaction effect in the thalamus, where thalamus susceptibility and internalizing symptoms (Internalizing Problems, Depression and Anxiety subscales) were more negatively associated in the PAE group than in the unexposed group. The post hoc within-group association was only significant in the PAE group for Internalizing Problems and Anxiety symptoms. The lack of relationships with externalizing symptoms is surprising given that prior studies report lower thalamus iron in youth with ADHD [
40,
42], an externalizing disorder that often co-occurs with PAE. That being said, participants in this study with ADHD were almost all taking psychostimulant medication, which is thought to normalize brain iron in the thalamus [
83]. Previous research in adults shows increased brain iron in the thalamus and putamen is related to depression severity [
52,
53]. We saw evidence of this trend in the unexposed group that had increasing internalizing symptoms with higher brain iron, while the opposite relationship was observed in youth with PAE. The thalamus is rich in dopamine [
84], and behavioral outcomes related to brain iron in the thalamus are likely related to dopamine metabolism.
The putamen is part of the striatum involved in reward, cognition, language, and addiction [
85]. We hypothesized that lower susceptibility in the putamen would be associated with more internalizing and externalizing symptoms. Instead, we found a group–susceptibility interaction effect such that the susceptibility in the putamen was more positively associated with Externalizing Problems in the PAE group than in the unexposed group. However, neither of the post hoc within-group relationships were significant. Previous research suggests that brain iron in the putamen is lower in children with ADHD [
40]; however, in our sample, more brain iron in the putamen was associated with more externalizing symptoms in the PAE group. Previous research has found that iron in the putamen is related to depression severity in adults [
52], but we did not find this in our sample. The putamen undergoes prolonged development [
86], and it is possible that the effects of susceptibility on the putamen and behavior would be better captured in older adolescence or young adulthood. Although the mechanism of how brain iron is related to internalizing and externalizing symptoms is not well understood, it is likely due to dopamine, serotonin, and norepinephrine metabolism [
87]. It is important to note that the concentration of brain iron required for healthy brain development differs by region [
62]. Therefore, while the caudate and pallidum have a high deposition of brain iron throughout development, other structures with lower brain iron, such as the hippocampus and amygdala, may be more sensitive to small perturbations in brain iron.
We found lower susceptibility in the hippocampus for the PAE group compared to the unexposed participants, but this did not survive correction for multiple comparisons. The hippocampus is highly vulnerable to iron status during gestation [
88], and while we did not find that hippocampus susceptibility was related to mental health symptoms, it may be associated with cognitive and learning difficulties that are common in individuals with PAE.
In contrast to the QSM results, there were no associations between brain volume and behavior. This differs from the research on mental health and brain structure in unexposed participants that has found associations between caudate volume, putamen development, amygdala, hippocampus, and thalamus abnormalities and internalizing and externalizing disorders [
14,
15,
16,
17,
18,
19,
20,
21,
22]. Only one study of children with PAE found a trend-level association between caudate volume and internalizing symptoms [
54]. Another study, in a sample overlapping with our study, found no associations between brain volumes and behavior [
89]. One explanation is that volume reductions in children with PAE may disrupt the etiology of mental health problems, obscuring the relationship that may be seen in unexposed individuals. However, further research to clarify these relationships is needed.
Nutritional interventions, such as choline supplementation, show promise for promoting neurodevelopment in infants and children with PAE [
90,
91,
92]. This opens the question of whether iron supplementation may be a viable intervention for individuals with PAE. Animal research has shown that iron supplementation can mitigate some of the effects of alcohol exposure during development [
38,
93,
94]. Future studies with prospective and large longitudinal samples are required to fully understand the impact of PAE on brain iron to assess iron supplementation as a potential nutritional intervention.
Limitations
This is the first study to examine brain iron and mental health in children and youth with PAE; however, some limitations in the study design and methodology exist. Gender was parent/caregiver reported, and biological sex was not collected. Future studies should collect both gender and sex when possible, as both variables provide distinct contributions to understanding the complex association between brain structure and mental health in children with PAE. QSM provides a novel way to study brain iron in children with PAE, though it cannot distinguish the components contributing to susceptibility. Given our small sample, this study had limited power. Information about PAE was collected as rigorously as possible; however, given the retrospective nature of the study, details around exposure amounts, trimester, and frequency were not always available. Youth with PAE are at a greater risk of experiencing other prenatal exposures (e.g., tobacco, cocaine, marijuana) [
95,
96] and postnatal adversities (e.g., neglect, deprivation, physical abuse, and caregiver changes) [
95,
97]; however, these factors were not considered in our analyses. Considering that prenatal exposures and postnatal adversities negatively impact brain development [
89,
95], these should be controlled in future studies. Lastly, mental health symptoms were measured using a caregiver report, which is less reliable when measuring internalizing symptoms [
98] and may be underestimating internalizing symptoms. Given the cognitive and emotional challenges youth with PAE face, a caregiver report was chosen to be the best method for assessment collection; however, future studies should use both parent and self-report outcome measures when possible.