The Effect of Prebiotics and Oral Anti-Diabetic Agents on Gut Microbiome in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomised Controlled Trials
Abstract
:1. Introduction
1.1. Description of the Intervention
1.2. How This Intervention Might Work
1.3. Why It Is Important to Do This Review
1.4. Research Questions
2. Methods
2.1. Studies Included
2.2. Participants of Interest
2.3. Types of Interventions
2.4. Outcome Measures
2.5. Search Strategy
3. Collection of Data and Analysis
3.1. Study Selection
3.2. Data Extraction and Management
4. Data Analysis
4.1. Network Meta-Analysis (NMA)
4.2. Meta-Analysis
4.3. Effect Size
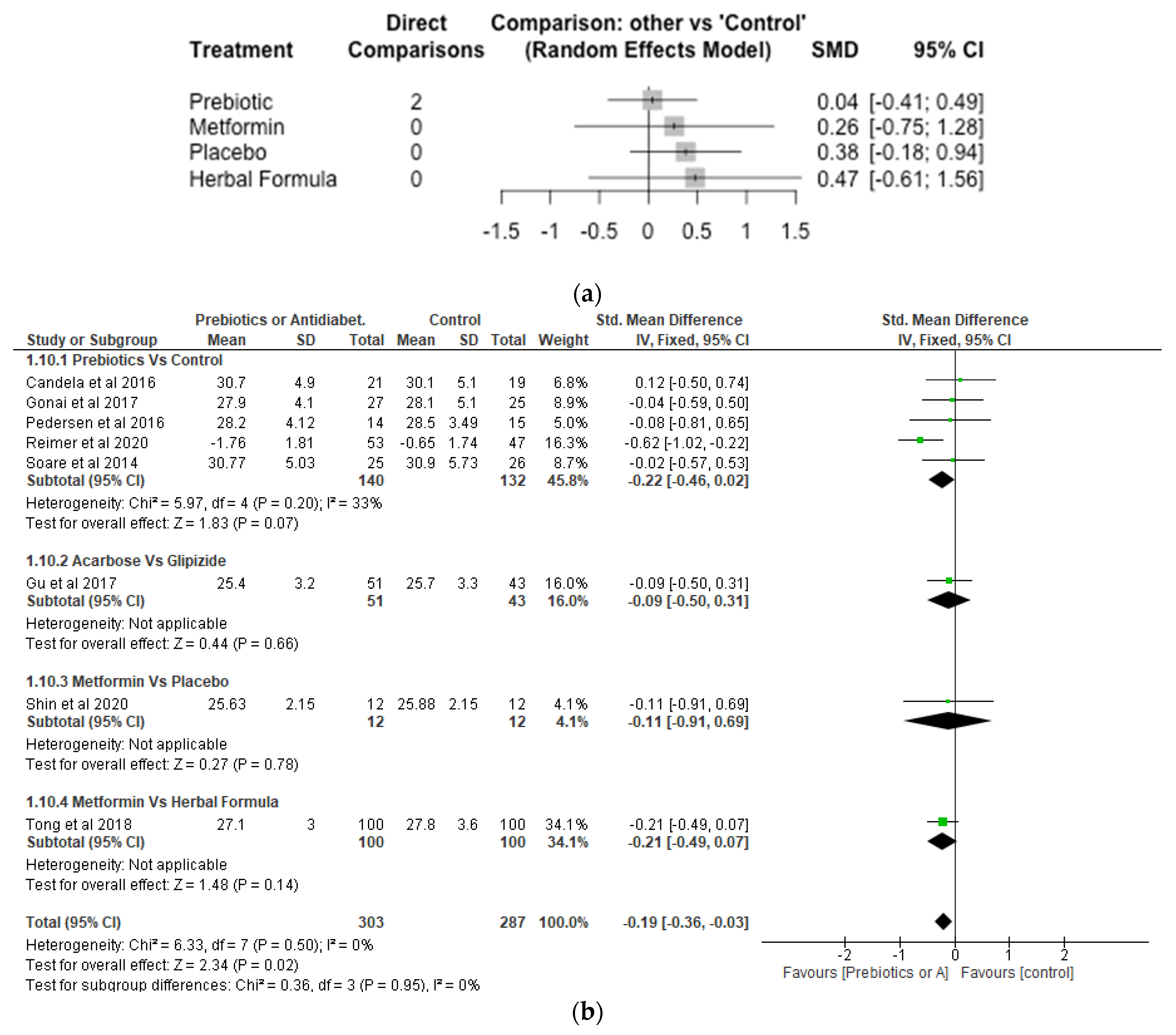
5. Results
6. Risk of Bias of Studies Included
7. Effects of Interventions
8. Bifidobacterium
9. Lactobaccilus
10. Akkermansia
11. Glycaemic Control
12. Body Mass Index (BMI)
13. Discussion
14. Limitations
15. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Roshanravan, N.; Alamdari, N.M.; Jafarabadi, M.A.; Mohammadi, A.; Shabestari, B.R.; Nasirzadeh, N.; Asghari, S.; Mansoori, B.; Akbarzadeh, M.; Ghavami, A.; et al. Effects of oral butyrate and inulin supplementation on inflammation-induced pyroptosis pathway in type 2 diabetes: A randomized, double-blind, placebo-controlled trial. Cytokine 2020, 131, 155101. [Google Scholar] [CrossRef] [PubMed]
- Fallucca, F.; Porrata, C.; Fallucca, S.; Pianesi, M. Influence of diet on gut microbiota, inflammation and type 2 diabetes mellitus. First experience with macrobiotic Ma-Pi 2 diet. Diabetes/Metab. Res. Rev. 2014, 30, 48–54. [Google Scholar] [CrossRef]
- Aliasgharzadeh, A.; Aliloo, A.; Ghotaslou, R.; Arbabi, S. Comparison of bifidobacterium spp. and lactobacillus spp. count in faeces of patients with type 2 diabetes mellitus and healthy people. Middle East J. Fam. Med. 2018, 16, 102–106. [Google Scholar]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ojo, O.; Ojo, O.O.; Zand, N.; Wang, X. The Effect of Dietary Fibre on Gut Microbiota, Lipid Profile, and Inflammatory Markers in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2021, 13, 1805. [Google Scholar] [CrossRef]
- Fu, J.; Xu, K.; Ni, X.; Li, X.; Zhu, X.; Xu, W. Habitual Dietary Fiber Intake, Fecal Microbiota, and Hemoglobin A1c Level in Chinese Patients with Type 2 Diabetes. Nutrients 2022, 14, 1003. [Google Scholar] [CrossRef]
- Fallucca, F.; Fontana, L.; Fallucca, S.; Pianesi, M. Gut microbiota and Ma-Pi 2 macrobiotic diet in the treatment of type 2 diabetes. World J. Diabetes 2015, 6, 403–411. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Han, H.; Fu, X.; Liu, A.; Zhan, Y.; Qiu, H.; Ma, L.; Zhang, X.; Wang, X. Metformin Reduces Blood Glucose in Treatment-Naive Type 2 Diabetes by Altering the Gut Microbiome. Can. J. Diabetes 2022, 46, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-E.; Choi, Y.; Jun, J.E.; Lee, Y.-B.; Jin, S.-M.; Hur, K.Y.; Ko, G.P.; Lee, M.-K. Additional effect of dietary fiber in patients with type 2 diabetes mellitus using metformin and sulfonylurea: An open-label, pilot trial. Diabetes Metab. J. 2019, 43, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Rezende, E.S.V.; Lima, G.C.; Naves, M.M.V. Dietary fibers as beneficial microbiota modulators: A proposed classification by prebiotic categories. Nutrition 2021, 89, 111217. [Google Scholar] [CrossRef] [PubMed]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elbere, I.; Kalnina, I.; Silamikelis, I.; Konrade, I.; Zaharenko, L.; Sekace, K.; Radovica-Spalvina, I.; Fridmanis, D.; Gudra, D.; Pirags, V.; et al. Association of metformin administration with gut microbiome dysbiosis in healthy volunteers. PLoS ONE 2018, 13, e0204317. [Google Scholar] [CrossRef] [Green Version]
- Lv, Y.; Zhao, X.; Guo, W.; Gao, Y.; Yang, S.; Li, Z.; Wang, G. The Relationship between Frequently Used Glucose-Lowering Agents and Gut Microbiota in Type 2 Diabetes Mellitus. J. Diabetes Res. 2018, 2018, 1890978. [Google Scholar] [CrossRef] [PubMed]
- Almugadam, B.S.; Liu, Y.; Chen, S.M.; Wang, C.H.; Shao, C.Y.; Ren, B.W.; Tang, L. Alterations of Gut Microbiota in Type 2 Diabetes Individuals and the Confounding Effect of Antidiabetic Agents. J. Diabetes Res. 2020, 2020, 7253978. [Google Scholar] [CrossRef] [PubMed]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for enriching next-generation health-promoting gut bacteria through prebiotics and other dietary components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Luo, Z.; Zhou, J.; Sun, B. Gut Microbiota and Antidiabetic Drugs: Perspectives of Personalized Treatment in Type 2 Diabetes Mellitus. Front. Cell. Infect. Microbiol. 2022, 12, 853771. [Google Scholar] [CrossRef]
- Wu, H.; Esteve, E.; Tremaroli, V.; Khan, M.T.; Caesar, R.; Mannerås-Holm, L.; Ståhlman, M.; Olsson, L.M.; Serino, M.; Planas-Fèlix, M.; et al. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat. Med. 2017, 23, 850–858. [Google Scholar] [CrossRef]
- Ghorbani, Y.; Schwenger, K.J.P.; Allard, J.P. Manipulation of intestinal microbiome as potential treatment for insulin resistance and type 2 diabetes. Eur. J. Nutr. 2021, 60, 2361–2379. [Google Scholar] [CrossRef]
- Zhang, W.; Tang, Y.; Huang, J.; Yang, Y.; Yang, Q.; Hu, H. Efficacy of inulin supplementation in improving insulin control, HbA1c and HOMA-IR in patients with type 2 diabetes: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Biochem. Nutr. 2020, 66, 176–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahboobi, S.; Rahimi, F.; Jafarnejad, S. Effects of Prebiotic and Synbiotic Supplementation on Glycaemia and Lipid Profile in Type 2 Diabetes: A Meta-Analysis of Randomized Controlled Trials. Adv. Pharm. Bull. 2018, 8, 565–574. [Google Scholar] [CrossRef]
- Wang, L.; Yang, H.; Huang, H.; Zhang, C.; Zuo, H.-X.; Xu, P.; Niu, Y.-M.; Wu, S.-S. Inulin-type fructans supplementation improves glycemic control for the prediabetes and type 2 diabetes populations: Results from a GRADE-assessed systematic review and dose–response meta-analysis of 33 randomized controlled trials. J. Transl. Med. 2019, 17, 410. [Google Scholar] [CrossRef]
- Akbari, V.; Hendijani, F. Effects of probiotic supplementation in patients with type 2 diabetes: Systematic review and meta-analysis. Nutr. Rev. 2016, 74, 774–784. [Google Scholar] [CrossRef]
- Merkevicius, K.; Kundelis, R.; Maleckas, A.; Velickiene, D. Microbiome Changes after Type 2 Diabetes Treatment: A Systematic Review. Medicina 2021, 57, 1084. [Google Scholar] [CrossRef] [PubMed]
- Bock, P.M.; Telo, G.H.; Ramalho, R.; Sbaraini, M.; Leivas, G.; Martins, A.F.; Schaan, B.D. The effect of probiotics, prebiotics or synbiotics on metabolic outcomes in individuals with diabetes: A systematic review and meta-analysis. Diabetologia 2021, 64, 26–41. [Google Scholar] [CrossRef]
- Ojo, O.; Feng, Q.-Q.; Ojo, O.O.; Wang, X.-H. The Role of Dietary Fibre in Modulating Gut Microbiota Dysbiosis in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2020, 12, 3239. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.A.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-analyses of Health Care Interventions: Checklist and Explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [Green Version]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, 71. [Google Scholar] [CrossRef] [PubMed]
- Methley, A.M.; Campbell, S.; Chew-Graham, C.; McNally, R.; Cheraghi-Sohi, S. PICO, PICOS and SPIDER: A comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv. Res. 2014, 14, 579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Wiley-Blackwell: Hoboken, NJ, USA, 2009. [Google Scholar]
- Medina-Vera, I.; Sanchez-Tapia, M.; Noriega-López, L.; Granados-Portillo, O.; Guevara-Cruz, M.; Flores-López, A.; Avila-Nava, A.; Fernández, M.L.; Tovar, A.R.; Torres, N. A dietary intervention with functional foods reduces metabolic endotoxaemia and attenuates biochemical abnormalities by modifying faecal microbiota in people with type 2 diabetes. Diabetes Metab. 2019, 45, 122–131. [Google Scholar] [CrossRef]
- Pedersen, C.; Wu, H.; Jaiyeola, E.; Diribe, O.; La Ragione, R.; Robertson, M.D.; Wright, J.; Gallagher, E.; Horton, F.; Hinton, P.; et al. Host–microbiome interactions in human type 2 diabetes following prebiotic fibre (galacto-oligosaccharide) intake. Br. J. Nutr. 2016, 116, 1869–1877. [Google Scholar] [CrossRef]
- Mitchell, M.; Muftakhidinov, B.; Winchen, T. Engauge Digitizer Software. 2020. Available online: http://markummitchell.github.io/engauge-digitizer (accessed on 21 August 2022).
- The Nordic Cochrane Centre. Review Manager (RevMan) [Computer Program]; Version 5.3; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 2014. [Google Scholar]
- Rücker, G.; Krahn, U.; König, J.; Efthimiou, O.; Davies, A.; Papakonstantinou, T.; Schwarzer, G. Netmeta: Network Meta-Analysis Using Frequentist Methods. R Package Version 2.1-0. 2022. Available online: https://cran.r-project.org/web/packages/netmeta/netmeta.pdf (accessed on 2 November 2022).
- Arias-Córdova, Y.; Ble-Castillo, J.L.; García-Vázquez, C.; Olvera-Hernández, V.; Ramos-García, M.; Navarrete-Cortes, A.; Jiménez-Domínguez, G.; Juárez-Rojop, I.E.; Tovilla-Zárate, C.A.; Martínez-López, M.C.; et al. Resistant Starch Consumption Effects on Glycemic Control and Glycemic Variability in Patients with Type 2 Diabetes: A Randomized Crossover Study. Nutrients 2021, 13, 4052. [Google Scholar] [CrossRef]
- Birkeland, E.; Gharagozlian, S.; Birkeland, K.I.; Valeur, J.; Mage, I.; Rud, I.; Aas, A.M. Prebiotic effect of inulin-type fructans on faecal microbiota and short-chain fatty acids in type 2 diabetes: A randomised controlled trial. Eur. J. Nutr. 2020, 59, 3325–3338. [Google Scholar] [CrossRef] [PubMed]
- Candela, M.; Biagi, E.; Soverini, M.; Consolandi, C.; Quercia, S.; Severgnini, M.; Peano, C.; Turroni, S.; Rampelli, S.; Pozzilli, P.; et al. Modulation of gut microbiota dysbioses in type 2 diabetic patients by macrobiotic Ma-Pi 2 diet. Br. J. Nutr. 2016, 116, 80–93. [Google Scholar] [CrossRef] [Green Version]
- Gonai, M.; Shigehisa, A.; Kigawa, I.; Kurasaki, K.; Chonan, O.; Matsuki, T.; Yoshida, Y.; Aida, M.; Hamano, K.; Terauchi, Y. Galacto-oligosaccharides ameliorate dysbiotic Bifidobacteriaceae decline in Japanese patients with type 2 diabetes. Benef. Microbes 2017, 8, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Wang, X.; Li, J.; Zhang, Y.; Zhong, H.; Liu, R.; Zhang, D.; Feng, Q.; Xie, X.; Hong, J.; et al. Analyses of gut microbiota and plasma bile acids enable stratification of patients for antidiabetic treatment. Nat. Commun. 2017, 8, 1785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reimer, R.A.; Wharton, S.; Green, T.J.; Manjoo, P.; Ramay, H.R.; Lyon, M.R.; Gahler, R.J.; Wood, S. Effect of a functional fibre supplement on glycemic control when added to a year-long medically supervised weight management program in adults with type 2 diabetes. Eur. J. Nutr. 2021, 60, 1237–1251. [Google Scholar] [CrossRef]
- Shin, N.R.; Gu, N.; Choi, H.S.; Kim, H. Combined effects of Scutellaria baicalensis with metformin on glucose tolerance of patients with type 2 diabetes via gut microbiota modulation. Am. J. Physiol. Metab. Endocrinol. Metab. 2020, 318, E52–E61. [Google Scholar] [CrossRef]
- Soare, A.; Khazrai, Y.M.; Del Toro, R.; Roncella, E.; Fontana, L.; Fallucca, S.; Angeletti, S.; Formisano, V.; Capata, F.; Ruiz, V.; et al. The effect of the macrobiotic Ma-Pi 2 diet vs. the recommended diet in the management of type 2 diabetes: The randomized controlled MADIAB trial. Nutr. Metab. 2014, 11, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soare, A.; Del Toro, R.; Khazrai, Y.M.; Di Mauro, A.; Fallucca, S.; Angeletti, S.; Skrami, E.; Gesuita, R.; Tuccinardi, D.; Manfrini, S.; et al. A 6-month follow-up study of the randomized controlled Ma-Pi macrobiotic dietary intervention (MADIAB trial) in type 2 diabetes. Nutr. Diabetes 2016, 6, e222. [Google Scholar] [CrossRef] [Green Version]
- Su, B.; Liu, H.; Li, J.; Sunli, Y.; Liu, B.; Liu, D.; Zhang, P.; Meng, X. Acarbose treatment affects the serum levels of inflammatory cytokines and the gut content of bifidobacteria in Chinese patients with type 2 diabetes mellitus. J. Diabetes 2015, 7, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Tong, X.; Xu, J.; Lian, F.; Yu, X.; Zhao, Y.; Xu, L.; Zhang, M.; Zhao, X.; Shen, J.; Wu, S.; et al. Structural alteration of gut microbiota during the amelioration of human type 2 diabetes with hyperlipidemia by metformin and a traditional chinese herbal formula: A multicenter, randomized, open label clinical trial. mBio 2018, 9, e02392-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Bommel, E.J.M.; Herrema, H.; Davids, M.; Kramer, M.H.H.; Nieuwdorp, M.; van Raalte, D.H. Effects of 12-week treatment with dapagliflozin and gliclazide on faecal microbiome: Results of a double-blind randomized trial in patients with type 2 diabetes. Diabetes Metab. 2020, 46, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, B.R.; Jaiswal, S.; Ravindra, P.V. Modulation of gut microbiota by bioactive compounds for prevention and management of type 2 diabetes. Biomed. Pharmacother. 2022, 152, 113148. [Google Scholar] [CrossRef] [PubMed]
- Lê, K.-A.; Li, Y.; Xu, X.; Yang, W.; Liu, T.; Zhao, X.; Tang, Y.G.; Cai, D.H.; Go, V.L.W.; Pandol, S.; et al. Alterations in fecal Lactobacillus and Bifidobacterium species in type 2 diabetic patients in Southern China population. Front. Physiol. 2013, 3, 496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robertson, M.D. Prebiotics and type 2 diabetes: Targeting the gut microbiota for improved glycaemic control? Pract. Diabetes 2020, 37, 133–137. [Google Scholar] [CrossRef]
- Prattichizzo, F.; Giuliani, A.; Mensà, E.; Sabbatinelli, J.; De Nigris, V.; Rippo, M.R.; La Sala, L.; Procopio, A.D.; Olivieri, F.; Ceriello, A. Pleiotropic effects of metformin: Shaping the microbiome to manage type 2 diabetes and postpone ageing. Ageing Res. Rev. 2018, 48, 87–98. [Google Scholar] [CrossRef]








| Patient/Population | Intervention | Outcome (Primary) | Study Designs | Combining Search Terms |
|---|---|---|---|---|
| Patients with diabetes | Prebiotics OR Oral anti-diabetic agents | Gut microbiome | Randomised controlled trial | |
| Diabetes mellitus, type 2 OR Diabetes complications OR Patients with diabetes OR diabetes mellitus OR type 2 diabetes OR Diabetes | Prebiotics OR Dietary fibre OR Fibre OR Polysaccharide OR Dietary carbohydrate OR Resistant Starch OR carbohydrate OR Oral anti-diabetic agents OR metformin or gliclazide OR acarbose | Microbiome OR Gastrointestinal microbiota OR Gut microbiota OR Microbiota | #1 Randomized OR Randomised controlled trial OR placebo OR controlled clinical trial OR therapy OR randomly OR drug OR trial OR groups #2 “Animals” NOT “Humans” #3 #1 NOT #2 | 1st Column + 2nd Column + 3rd Column + 4th Column |
| Citation/Country of Study | Type of Study | Aim | Participants | Sample Size | Mean Age (Years) | Mean Diabetes Duration (Years) | Interventions | Results/Findings |
|---|---|---|---|---|---|---|---|---|
| Arias-Córdova et al. [36] Mexico | RCT | To assess the effects of NBS and HMS on GC and GV in patients with T2D when treatments were matched for digestible starch content. | All participants with T2D treated with metformin or a combination of glibenclamide and metformin | n = 10 | 48.5 ± 9.12 | Not Applicable | NBS, HMS and DMS. including three treatment phases, each with a duration of 4 days, and washout period between treatments of 9-day. | The intake of NBS showed a reduction in fasting glycemia compared to DMS. |
| Birkeland et al. [37] Norway | RCT | To examine the effect of inulin-type fructans on faecal microbiota and SCFAs in patients with T2D. | T2D, with 2/3 of participants receiving glucose-lowering drugs | n = 25 | 4.7 (0.2–20.0) | 16 g Inulin-type fructans versus 16 g maltodextrin. There was 4-week washout which separated the 6 weeks treatment period. | There was moderate, but significant increase in faecal levels of bifidobacteria in the group supplemented daily with inulin-type fructans. | |
| Candela et al. [38] Italy | RCT | To explore the effect of microbiotic Ma-Pi 2 diet in modulating gut microbiota dysbiosis in patients with T2D. | Patients with T2D | Ma-Pi 2 diet: n = 21 Control diet: n = 19 | 66 | Not Applicable | Fibre rich microbiotic Ma-Pi 2 diet is enriched with complex carbohydrates, legumes, fermented products, sea salt and green tea. | FBG and PBG were reduced significantly in both Ma-Pi 2 and control diets, although this was significantly higher in the Ma-Pi 2 diet compared to control. Both diets were also effective in supporting the recovery of health promoting SCFA producing bacteria including Faecalibacterium, Roseburia, Bacteroides and Akkermansia. Increases in Collinsella and Streptococcus were only counteracted by Ma-Pi 2 diet. |
| Gonai et al. [39] Japan | RCT | To explore the effects of GOS on on glycaemia, gut microbiota and metabolitic parameters in patients with T2D. | Patients with T2D | GOS group: 28 Placebo group: 27 | GOS group: 55 ± 11 Placebo group: 54 ± 12 | GOS group: 10 ± 8 Placebo group: 6 ± 5 | 10 g/d GOS syrup versus 10 g/d maltodextrin syrup. 4 weeks of treatment. | After consumption of GOS, Bifidobacteriaceae was significantly restored in patients with T2D, whereas lipopolysaccharide binding protein and glucose tolerance did not show improvement. |
| Gu et al. [40] China | RCT | To compare the effect of Acarbose versus sulfonylurea Glipizide on metabolic parameters, (e.g., glycaemic, control plasma BAs), and the intestinal microbiota, and discriminate such changes from disease-dependent alterations. | Patients with T2D | Acarbose group: 51 Glipizide group: 43 | Acarbose group: 53 ± 7 Glipizide group: 54 ± 7 | Not Applicable | Acarbose treatment versus Glipizide treatment. A 3-month treatment period. | Both the acarbose and glipizide groups improved glycemic control, with no significant differences. Acarbose increased the relative abundances of Lactobacillus and Bifidobacterium and depleted Bacteroides. However, Glipizide treatment did not affect the relative abundances at species-level. After 3 months of treatment, reductions in BW and BMI were more significant in the Acarbose group compared to the Glipizide group. |
| Medina-Vera et al. [31] Mexico | RCT | To assess functional food-based dietary intervention on biochemical parameters and faecal microbiota in patients with T2D. | Patients with T2D | DP group: n = 28 Placebo group: n = 25 | DP group: 50.4 ± 8.7 Placebo group: 49.8 ± 10.6 | DP group: 4.1 ± 3.5 Placebo group: 4.4 ± 3.9 | A dietary portfolio, DP (14 g of dehydrated nopal, 4 g of chia seeds, 30 g of soy protein and 4 g of inulin) versus placebo (28 g of calcium caseinate and 15 g of maltodextrin). The treatment period was for 3 months. | Consumption of DP promoted the abundance of Bifidobacterium longum which has been reported to improve insulin sensitivity. There was significant reduction in the levels of HbA1c in patients with T2D in the DP group. |
| Pedersen et al. [32] | RCT | To investigate the effects of prebiotic supplementation on intestinal bacteria in patients with type 2 diabetes | Patients with type 2 diabetes | Prebiotic group: n = 14 Placebo group: n= 15 | Prebiotic group (56.7 ± 6.0) Placebo group (58.1 ± 6.6) | Prebiotic group (4.6 ± 2.2) Placebo group (4.0 ± 3.1) | Prebiotic (galacto-oligosaccharide mixture) or placebo (maltodextrin supplements each given 5.5g/day for 12 weeks. | Prebiotic fibre supplementation did not improve glucose control or abundance of bacteria compared with control. |
| Reimer et al. [41] Canada | RCT | To assess the effect of the soluble viscous fibre PGX on glycemic control in adults withT2D. | T2D patients | PGX group: n = 147; Placebo group: n = 143 | PGX group: 56.2 ± 8.6 Placebo group: 53.4 ± 9.9 | Not Applicable | PGX (15–20 g/day) versus placebo (rice flour, 6.4–8.6 g/day) 52 weeks of treatment | PGX group increased Roseburia and led to a sustained reduction in HbA1c and FBG compared to placebo. |
| Shin et al. [42] Korea | RCT | To investigate whether the combination of SB and metformin influenced T2D symptoms. | T2D on 500 mg/day metformin | n = 12 | SB + Metformin: 63.1 Placebo: 63.1 | Not Applicable | SB (3.52 g SB extract) + metformin versus placebo+ metformin. A 4-week washout separated the 8 weeks of treatment | Lactobacillus and Akkermansia, showed significant increases after SB + metformin treatment. The glucose, HbA1c and BMI were not changed after 8 weeks of SB and placebo treatment. |
| Soare et al. [43] Italy | RCT | To compare the effects of the Ma-Pi 2 diet and the dietary guidelines for T2D recommended by professional societies in Italy on T2D patients. | Overweight or obese (BMI:27–45 kg/m2), aged 40–75 years affected by T2D | Ma-Pi 2 diet:25 Control diet:26 | Ma-Pi 2 diet: 67 ± 8.16 Control diet: 65 ± 7.28 | Ma-Pi 2 diet:7 ± 7.793 Control diet:4.5 ± 8.845 | Fibre-rich Ma-Pi 2 macrobiotic diet versus recommended diet of type 2 diabetes by professional societies. 3 weeks of treatment. | The patients that received Ma-Pi 2 diet showed significant reduction in FBG, PBG, HbA1c, and BMI compared to those receiving the recommended diet for T2D. |
| Soare et al. [44] Italy | RCT | To investigate whether the benefits of Ma-Pi 2 extended beyond the 21-day intensive dietary intervention. | Overweight or obese (BMI 27–45 kg/m2), aged 40–75 years affected by T2D. | Ma-Pi 4 diet: 65 ± 8.89 Control diet:64 ± 8.15 | Ma-Pi 4 diet: 7 ± 7.41 Control diet: 4 ± 6.67 | Fibre-rich Ma-Pi 4 macrobiotic diet versus recommended diet of T2D diabetes by professional societies. 6 months of treatment. | The Ma-Pi 4 diet had great improvement in glycemic control, compared with the control group. Body weight loss was also observed in Ma-Pi 4 group, but was not significantly different compared to the control group. | |
| Su et al. [45] China | RCT | To evaluate the effects of acarbose add-on therapy on gut microbiota and inflammatory cytokines among Chinese patients with T2D. | Patients with T2D that did not receive acarbose for at least 1 month. | Acarbose group: 59 Control group: 36 | Acarbose group: 55.7 ± 11.0 Control group: 56.5 ± 10.2 | Not Applicable | 50 mg acarbose (t.i.d) a day with meals together with oral antidiabetic drugs and/or insulin or insulin analogs versus similar antidiabetic treatment to interventional group but without acarbose. Four weeks of treatment. | Treatment with acarbose can increase the abundance of Bifidobacterium longum in patients with T2D and improve glycemic control. |
| Tong et al. [46] China | RCT | To evaluate the role of gut microbiota during improvements in hyperglycemia and hyperlipidemia by two drugs: metformin and AMC for diabetic patients with hyperlipidemia. | Patients with T2D and Hyperlipidemia. | Metformin group: 100 AMC group: 100 | Metformin group: 58.55 ± 9.17 AMC group: 59.00 ± 9.46 | Not Applicable | AMC twice daily versus metformin tablets 0.25 g/time and 3 times/day. 12 weeks of treatment. | The effect of AMC in regulating the microbes in the gut and in improving HOMA-IR and triglyceride levels was more profound compared with metformin. |
| van Bommel et al. [47] Netherlands | RCT | To examine the effects of 12-week treatment with the SGLT2 inhibitor dapagliflozin and sulphonylurea gliclazide on gut microbiome composition in patients with T2D treated with metformin. | All participants with T2D treated with metformin as monotherapy | Dapagliflozin group: n = 24; Gliclazide group: n = 17. | Dapagliflozin group: 63 ± 7 Gliclazide group: 63 ± 7 | Dapagliflozingroup: 9.8 ± 4.1 Gliclazide group: 10.7 ± 7.3 | 10 mg dapagliflozin and 30 mg gliclazide. Treatment for 12 weeks. | Both dapagliflozin and gliclazide reduced HbA1c and FBG, while BMI was reduced by dapagliflozin, but increased by gliclazide. |
| Wu et al. [18] Spain | RCT | To investigate the effect of metformin on the composition and function of the microbiota. | Individuals with type 2 diabetes | Metformin group: n = 22 Placebo group: n = 18 | Metformin group: 52.6 ± 9.4 Placebo group: 54.9 ± 8.1 | Not applicable | A start dose of 425 mg/day of metformin and increased progressively to reach 1700 mg/day or placebo (calorie restricted diet). Treatment was for four months. | Metformin and not calorie restricted diet had significant effect on composition and function of the gut microbiota and reduction in HbA1c and fasting blood glucose levels. |
| Zhao et al. [48] China | RCT | To examine the effect of dietary fibre on SCFA-producing strains in patients with type 2 diabetes. | Individuals with type 2 diabetes | High fibre diet group: n = 27 Control group: n = 16 | High fibre diet group: 58.4 ± 32.2 Control group: 59.7 ± 24.0 | High fibre diet group: 8.0 ± 30.1 Control group: 7.9 ± 20 | High fibre diet composed of whole grains, traditional Chinese medicinal foods, and prebiotics. | The presence of SCFA producers in greater diversity and abundance by fibre. Participants had better improvement in HbA1c levels. |
| Studies | Bifidobacterium | Lactobacillus | Roseburia | Bacteroides | Ruminococcus | Clostridium | Akkermansia |
|---|---|---|---|---|---|---|---|
| Birkeland et al. [37] Norway | There was moderate, but significant increase in faecal levels of bifidobacteria in the group supplemented daily with inulin-type fructans. | N/A | N/A | Bacteroides ovatus was enriched by the prebiotic fibre | N/A | N/A | N/A |
| Candela et al. [38] Italy | N/A | N/A | Ma-Pi 2 diet and control were effective in supporting the recovery of Roseburia | Ma-Pi 2 diet and control were effective in supporting the recovery of Bacteroides | Ma-Pi 2 diet and control supported the reduction of Ruminococcus | N/A | Ma-Pi 2 diet and control resulted in the increase of Akkermansia |
| Gonai et al. [39] Japan | Bifidobacterium was significantly restored after consumption of GOS | N/A | N/A | N/A | Ruminococcus was significantly lower after consumption of GOS | N/A | N/A |
| Gu et al. [40] China | The relative abundances of Bifidobacterium species increased in Acarbose group. | Acarbose group increased the relative abundances of Lactobacillus species. | N/A | The intervention of Acarbose depleted the relative abundances of Bacteroides species. | N/A | Acarbose group depleted the relative abundances of Clostridium species. | N/A |
| Medina-Vera et al. [31] Mexico | Consumption of dietary portfolio stimulated the abundance of Bifidobacterium longum. | N/A | N/A | N/A | N/A | N/A | Dietary portfolio increased Akkermansia muciniphila |
| Pedersen et al. [32] | The effect of prebiotic treatment on Bifidobacterium was not significant. | The effect of prebiotic treatment on Lactobacillus was not significant. | The effect of prebiotic treatment on Roseburia was not significant. | N/A | N/A | The effect of prebiotic treatment on Clostridium was not significant. | N/A |
| Reimer et al. [41] Canada | Bifidobacterium Spp. changed significantly over time after PGX. | Lactobacillus was greater in the placebo compared with the PolyGlycopleX | The relative abundance of Roseburia was significantly increased by the soluble viscous fibre PolyGlycopleX | N/A | N/A | Clostridium coccoides changed significantly over time after PGX | Akkermansia muciniphila changed significantly over time after PGX |
| Shin et al. [42] Korea | The relative abundance of Bifidobacterium was significantly lower in the scutellaria baicalensis and metformin group compared to placebo. | Scutellaria baicalensis and metformin increased Lactobacillus significantly compared to placebo. | N/A | N/A | N/A | N/A | Scutellaria baicalensis and metformin increased Akkermansia significantly compared to placebo. |
| Su et al. [45] China | Acarbose treatment can increase the content of gut Bifidobacterium longum in type 2 diabetes mellitus patients. | N/A | N/A | N/A | N/A | N/A | N/A |
| Tong et al. [46] China | N/A | N/A | Roseburia was enhanced by herbal formula | N/A | N/A | N/A | There was decrease in Akkermansia in the metformin treated group |
| van Bommel et al. [47] Netherlands | N/A | N/A | N/A | N/A | N/A | N/A | Akkermansia muciniphila was not significantly affected by Dapagliflozin or Gliclazide treatment. |
| Wu et al. [18] Spain | There was increase in Bifidobacterium adolescentis after metformin treatment | N/A | N/A | N/A | N/A | N/A | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ojo, O.; Wang, X.; Ojo, O.O.; Brooke, J.; Jiang, Y.; Dong, Q.; Thompson, T. The Effect of Prebiotics and Oral Anti-Diabetic Agents on Gut Microbiome in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomised Controlled Trials. Nutrients 2022, 14, 5139. https://doi.org/10.3390/nu14235139
Ojo O, Wang X, Ojo OO, Brooke J, Jiang Y, Dong Q, Thompson T. The Effect of Prebiotics and Oral Anti-Diabetic Agents on Gut Microbiome in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomised Controlled Trials. Nutrients. 2022; 14(23):5139. https://doi.org/10.3390/nu14235139
Chicago/Turabian StyleOjo, Omorogieva, Xiaohua Wang, Osarhumwese Osaretin Ojo, Joanne Brooke, Yiqing Jiang, Qingqing Dong, and Trevor Thompson. 2022. "The Effect of Prebiotics and Oral Anti-Diabetic Agents on Gut Microbiome in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomised Controlled Trials" Nutrients 14, no. 23: 5139. https://doi.org/10.3390/nu14235139
APA StyleOjo, O., Wang, X., Ojo, O. O., Brooke, J., Jiang, Y., Dong, Q., & Thompson, T. (2022). The Effect of Prebiotics and Oral Anti-Diabetic Agents on Gut Microbiome in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomised Controlled Trials. Nutrients, 14(23), 5139. https://doi.org/10.3390/nu14235139







