Effects of Long-Term Administration of Bovine Bone Gelatin Peptides on Myocardial Hypertrophy in Spontaneously Hypertensive Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Bovine Bone Gelatin Peptides
2.3. Animal Experiment and Sample Collection
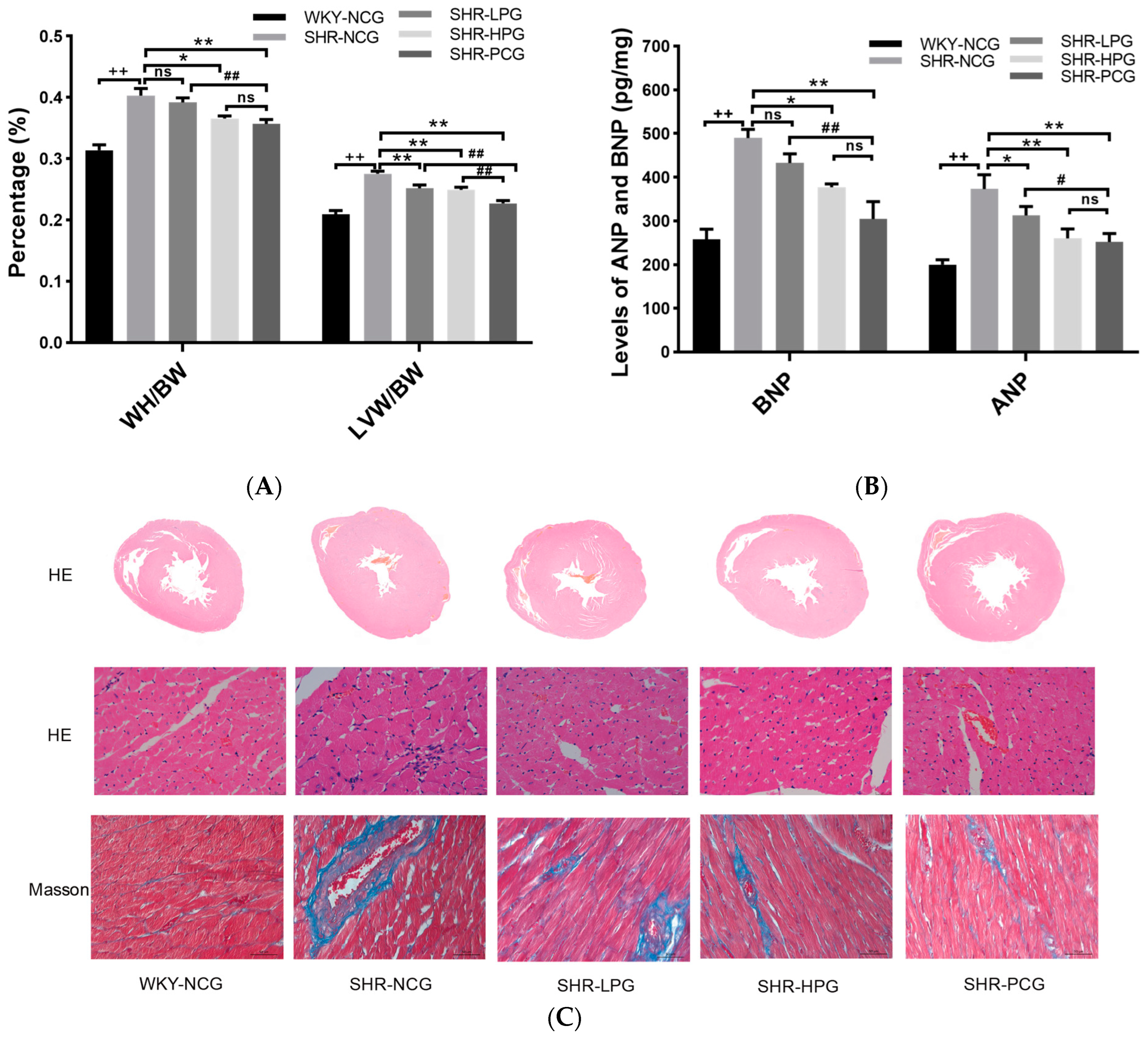
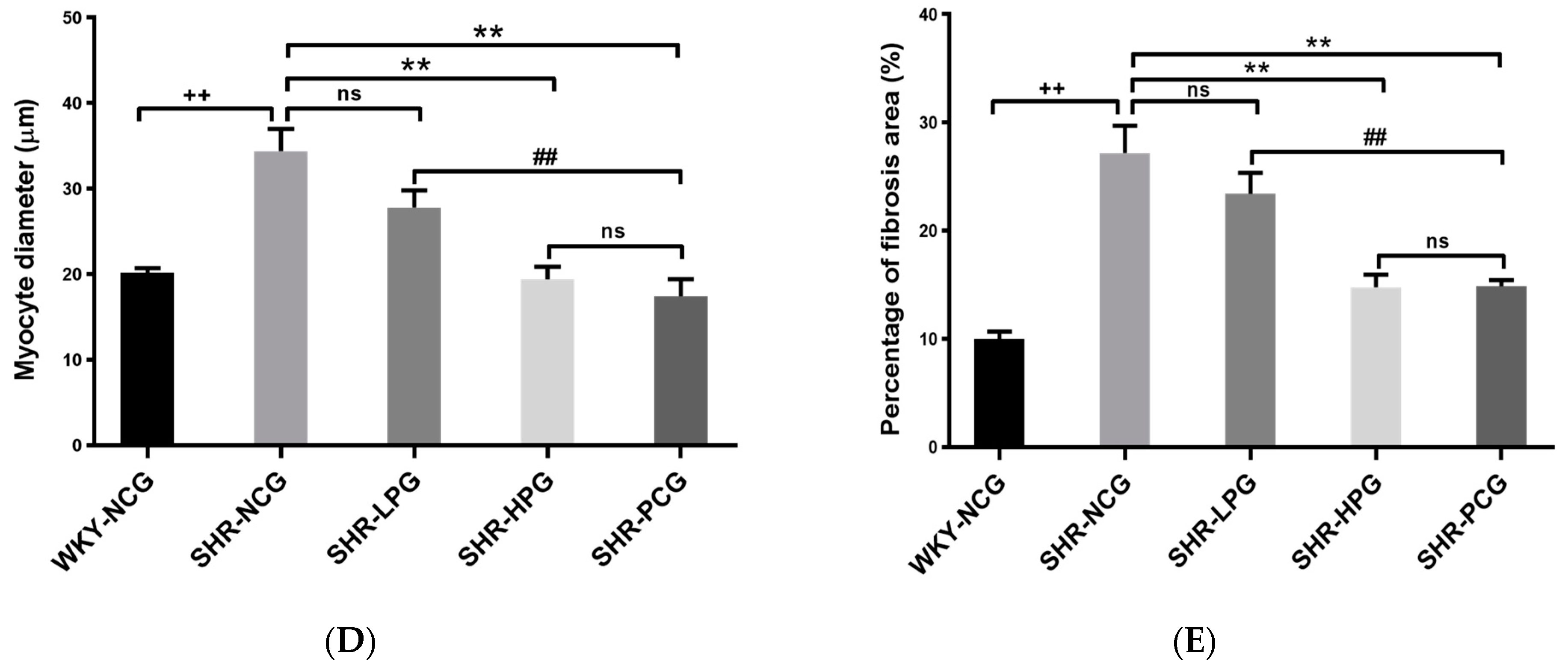
2.4. Histological Measurement
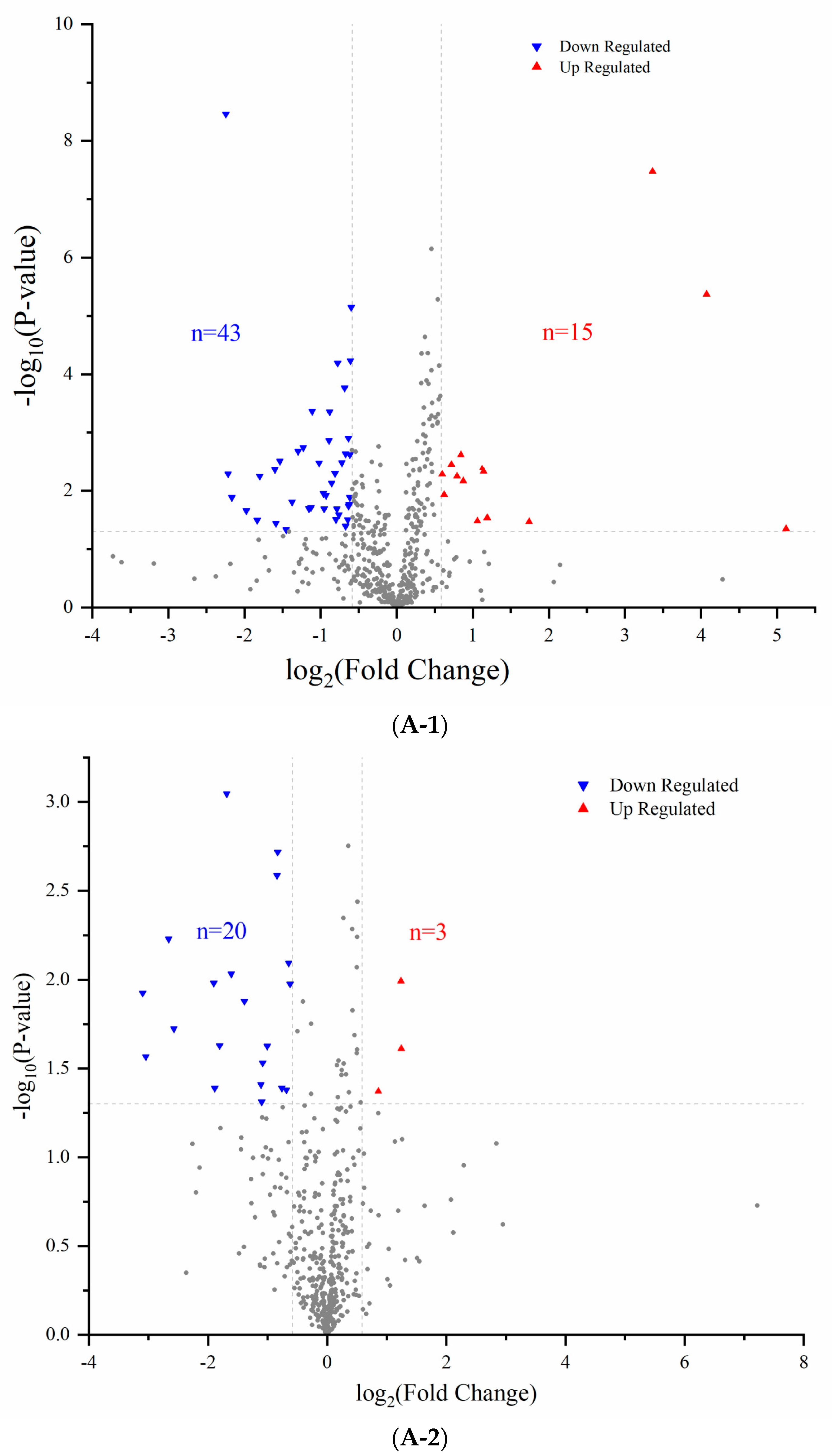
2.5. Label-Free Proteomics Analysis
2.6. Western Blotting
2.7. Enzyme-Linked Immuno Sorbent Assay (ELISA)
2.8. Statistical Analysis
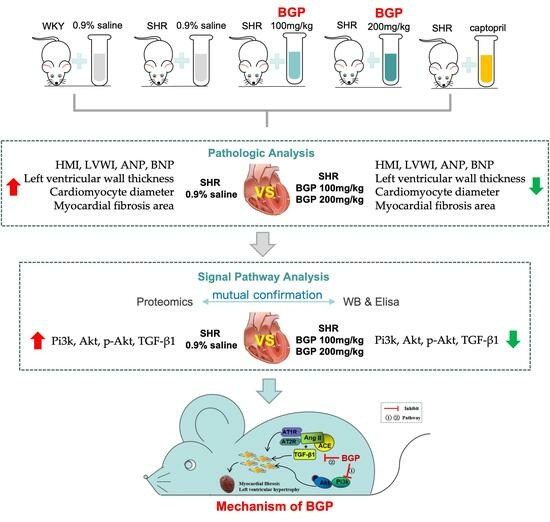
3. Results
3.1. Effects of BGP on Myocardial Hypertrophy and Myocardial Fibrosis in SHR
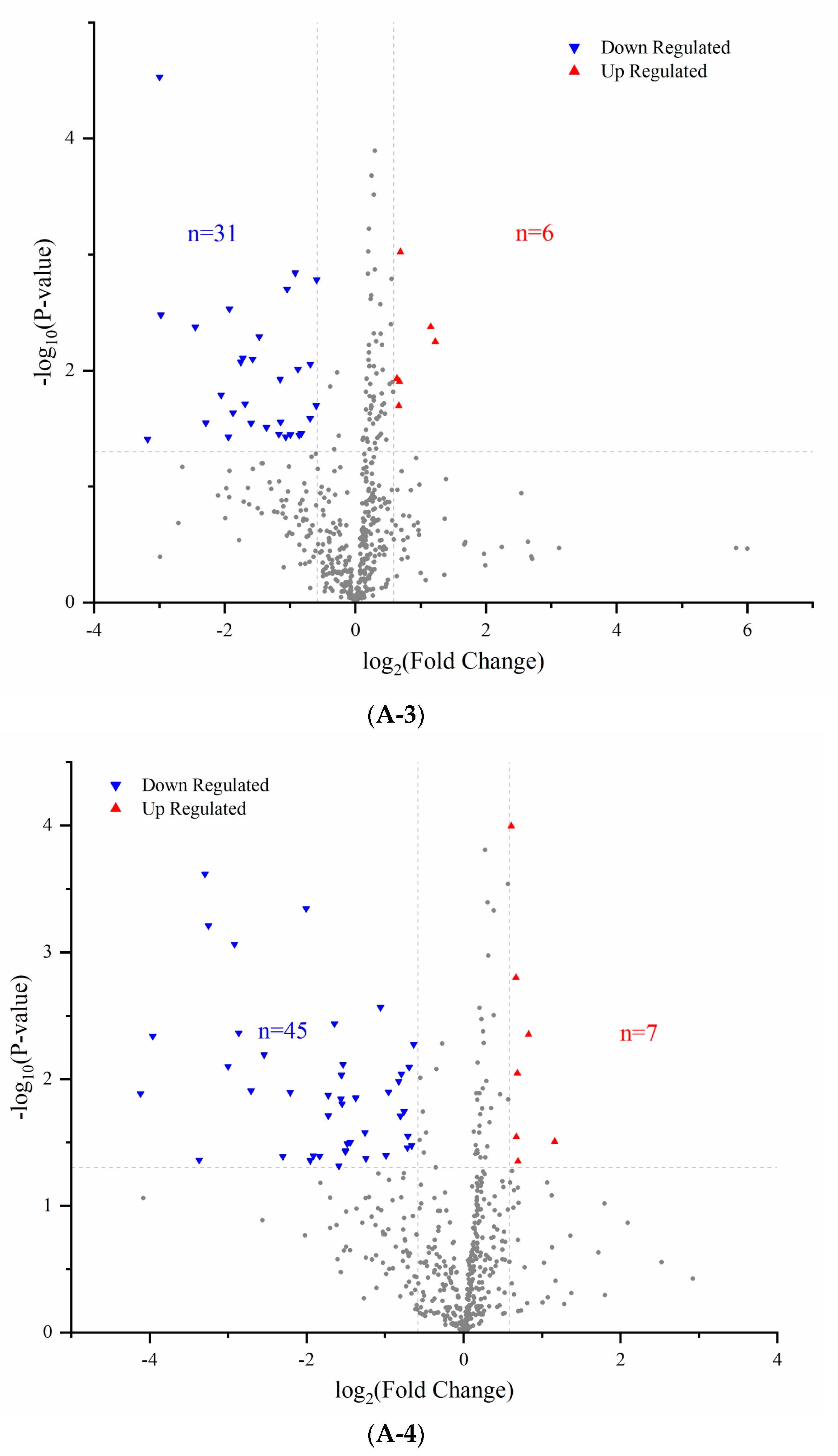
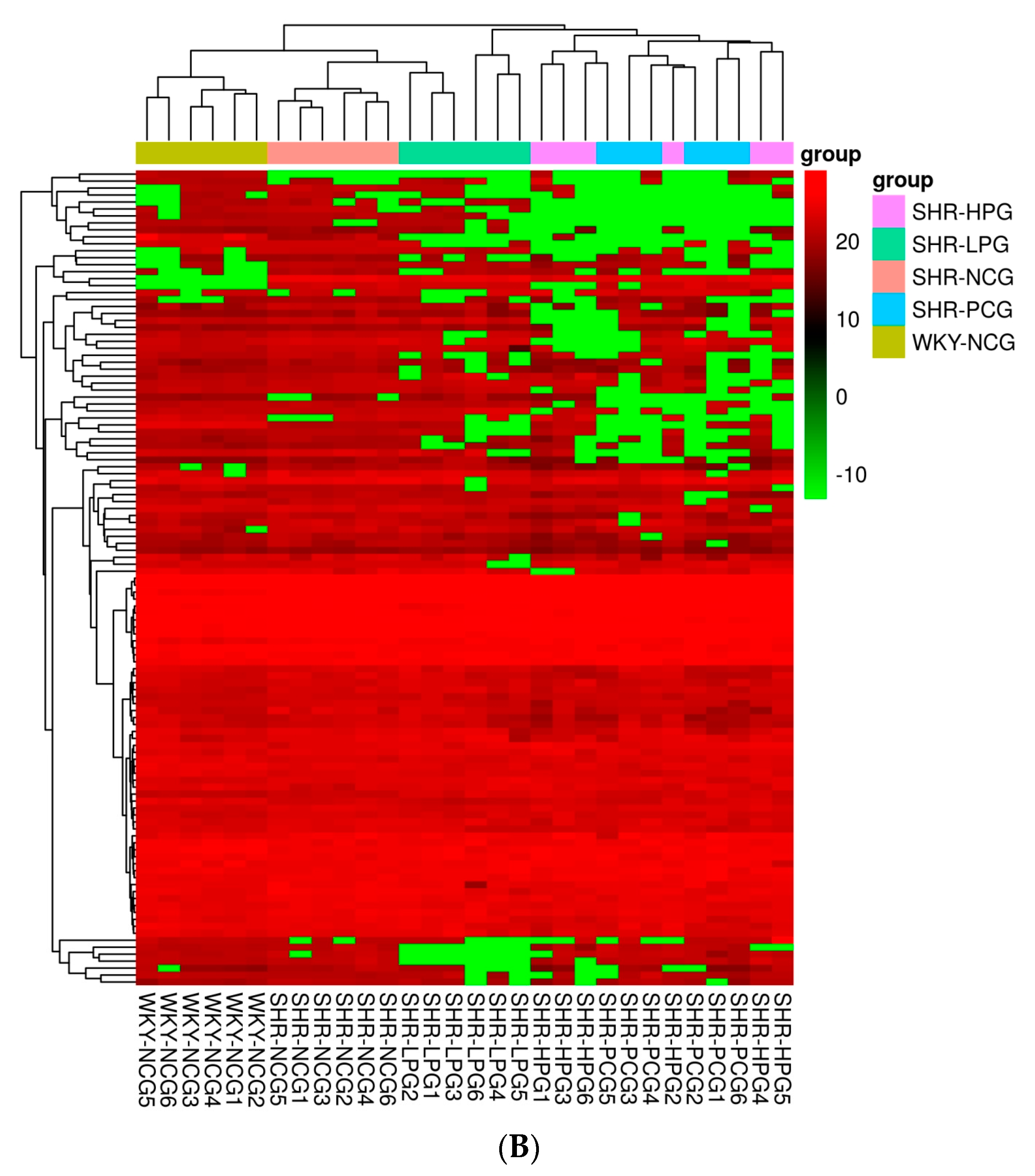
3.2. Comparison of Proteomics Profiles by Label-Free MS/MS
3.3. GO Annotations Analysis
3.4. Pathway Analysis
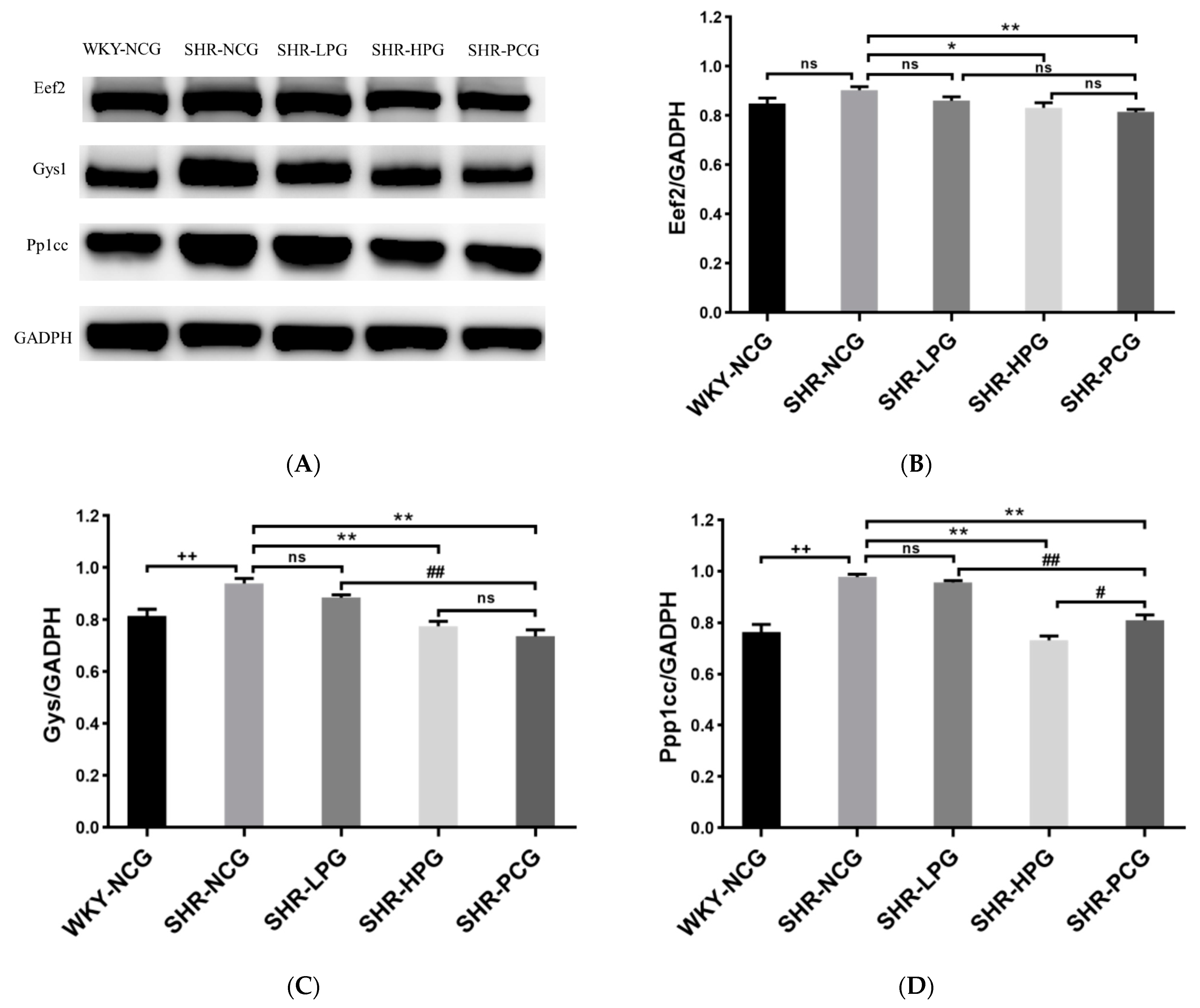
3.5. Western Blotting Validation
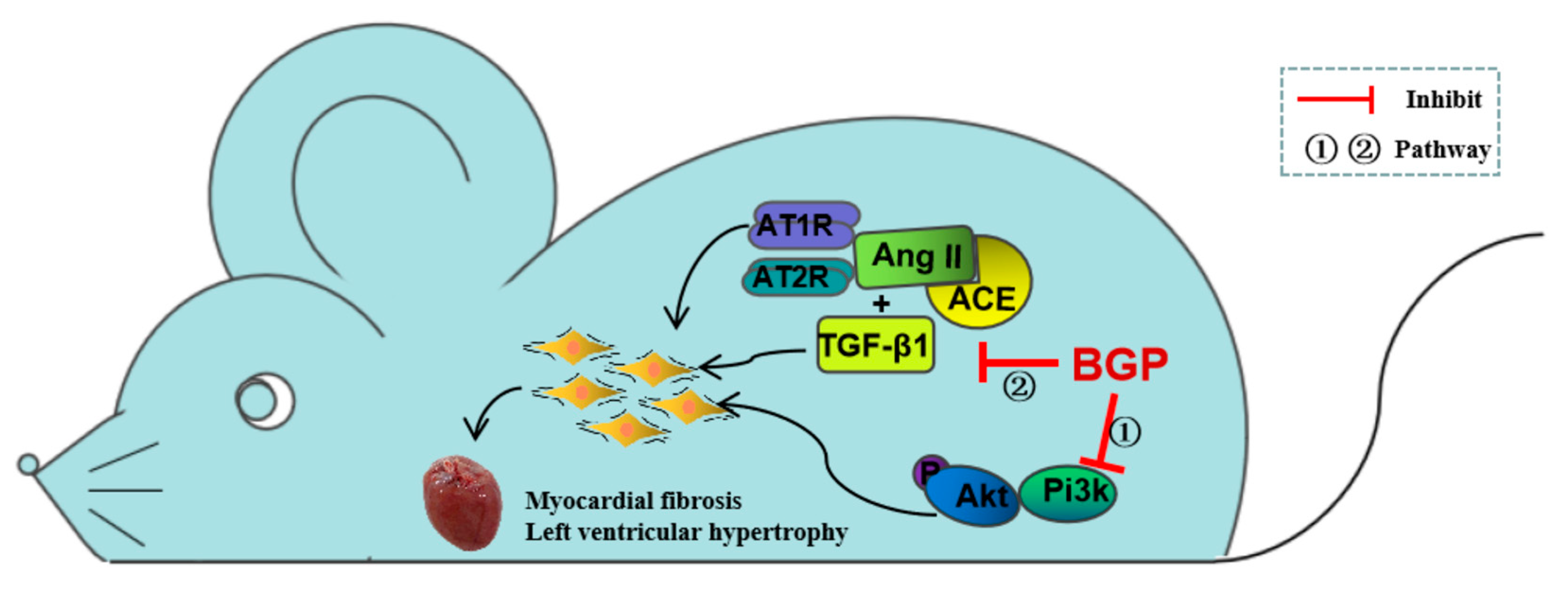
3.6. Levels of Pi3k/Akt Signaling Pathway and TGF-β1 in SHR Rats
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Shenasa, M.; Shenasa, H. Hypertension, left ventricular hypertrophy, and sudden cardiac death. Int. J. Cardiol. 2017, 237, 60–63. [Google Scholar] [CrossRef]
- You, J.; Wu, J.; Zhang, Q.; Ye, Y.; Wang, S.; Huang, J.; Liu, H.; Wang, X.; Zhang, W.; Bu, L.; et al. Differential cardiac hypertrophy and signaling pathways in pressure versus volume overload. Am. J. Physiol. Circ. Physiol. 2017, 314, H552–H562. [Google Scholar] [CrossRef]
- Zhao, L.; Wu, D.; Sang, M.; Xu, Y.; Liu, Z.; Wu, Q. Stachydrine ameliorates isoproterenol-induced cardiac hypertrophy and fibrosis by suppressing inflammation and oxidative stress through inhibiting NF-κB and JAK/STAT signaling pathways in rats. Int. Immunopharmacol. 2017, 48, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Berk, B.C.; Fujiwara, K.; Lehoux, S. ECM remodeling in hypertensive heart disease. J. Clin. Investig. 2007, 117, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.-X.; Zhang, P.; Zhang, X.-J.; Zhao, Y.-C.; Deng, K.-Q.; Jiang, X.; Wang, P.-X.; Huang, Z.; Li, H. The ubiquitin E3 ligase TRAF6 exacerbates pathological cardiac hypertrophy via TAK1-dependent signalling. Nat. Commun. 2016, 7, 11267. [Google Scholar] [CrossRef]
- Zou, Y.; Akazawa, H.; Qin, Y.; Sano, M.; Takano, H.; Minamino, T.; Makita, N.; Iwanaga, K.; Zhu, W.; Kudoh, S.; et al. Mechanical stress activates angiotensin II type 1 receptor without the involvement of angiotensin II. Nat. Cell Biol. 2004, 6, 499–506. [Google Scholar] [CrossRef]
- Nakamura, M.; Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat. Rev. Cardiol. 2018, 15, 387–407. [Google Scholar] [CrossRef]
- Ferrario, C.M. Cardiac remodelling and RAS inhibition. Ther. Adv. Cardio. Dis. 2016, 10, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, I.; Minamino, T. Physiological and pathological cardiac hypertrophy. J. Mol. Cell. Cardiol. 2016, 97, 245–262. [Google Scholar] [CrossRef]
- Devereux, R.B. Therapeutic options in minimizing left ventricular hypertrophy. Am. Heart J. 2000, 139, s9–s14. [Google Scholar] [CrossRef]
- Julian, D.G.; Pocock, S.J. Effects of long-term use of cardiovascular drugs. Lancet 2015, 385, 325. [Google Scholar] [CrossRef]
- Xing, L.J.; Li, G.H.; Toldra, F.; Zhang, W.G. The physiological activity of bioactive peptides obtained from meat and meat by-products. Adv. Food Nutr. Res. 2021, 97, 97–125. [Google Scholar]
- Cao, S.; Wang, Y.; Hao, Y.; Zhang, W.; Zhou, G. Antihypertensive effects in vitro and in vivo of novel angiotensin-converting enzyme inhibitory peptides from bovine bone gelatin hydrolysate. J. Agr. Food Chem. 2020, 68, 759–768. [Google Scholar] [CrossRef]
- Fu, Y.; Therkildsen, M.; Aluko, R.E.; Lametsch, R. Exploration of collagen recovered from animal by-products as a precursor of bioactive peptides: Successes and challenges. Crit. Rev. Food Sci. 2019, 59, 2011–2027. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Wang, Y.; Xing, L.J.; Zhang, W.G.; Zhou, G.H. Structure and physical properties of gelatin from bovine bone collagen influenced by acid pretreatment and pepsin. Food Bioprod. Process. 2020, 121, 213–223. [Google Scholar] [CrossRef]
- Yildiz, M.; Oktay, A.A.; Stewart, M.H.; Milani, R.V.; Ventura, H.O.; Lavie, C.J. Left ventricular hypertrophy and hypertension. Prog. Cardiovasc. Dis. 2020, 63, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Wu, W.; Tian, X.; Hou, M.; Dai, R.; Li, X. Unraveling proteo123me changes of Holstein beef M. semitendinosus and its relationship to meat discoloration during post-mortem storage analyzed by label-free mass spectrometry. J. Proteomics 2017, 154, 85–93. [Google Scholar] [CrossRef]
- Cao, S.M.; Wang, Z.X.; Xing, L.J.; Zhou, L.; Zhang, W.G. Bovine bone gelatin-derived peptides: Food processing characteristics and evaluation of anti-hypertensive and antihyperlipidemic activities. J. Agric. Food Chem. 2022, 70, 9877–9887. [Google Scholar] [CrossRef]
- Edhager, A.V.; Povlsen, J.A.; Løfgren, B.; Bøtker, H.E.; Palmfeldt, J. Proteomics of the rat myocardium during development of Type 2 diabetes mellitus reveals progressive alterations in major metabolic pathways. J. Proteome Res. 2018, 17, 2521–2532. [Google Scholar] [CrossRef]
- Azibani, F.; Fazal, L.; Chatziantoniou, C.; Samuel, J.; Delcayre, C. Aldosterone mediates cardiac fibrosis in the setting of hypertension. Curr. Hypertens. Rep. 2013, 15, 395–400. [Google Scholar] [CrossRef]
- Kong, P.; Christia, P.; Frangogiannis, N.G. The pathogenesis of cardiac fibrosis. Cell. Mol. Life Sci. 2014, 71, 549–574. [Google Scholar] [CrossRef]
- Leask, A. Getting to the heart of the matter. Circ. Res. 2015, 116, 1269–1276. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, Z.; Wang, H. Fusaric acid (FA) protects heart failure induced by isoproterenol (ISP) in mice through fibrosis prevention via TGF-β1/SMADs and PI3K/AKT signaling pathways. Biomed. Pharmacother. 2017, 93, 130–145. [Google Scholar] [CrossRef] [PubMed]
- Loffredo, F.S.; Steinhauser, M.L.; Jay, S.M.; Gannon, J.; Pancoast, J.R.; Yalamanchi, P.; Sinha, M.; Dall Osso, C.; Khong, D.; Shadrach, J.L.; et al. Growth differentiation factor 11 is a circulating factor that reverses age-related cardiac hypertrophy. Cell 2013, 153, 828–839. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Masuyama, T.; Sakata, Y.; Nishikawa, N.; Mano, T.; Yoshida, J.; Miwa, T.; Sugawara, M.; Yamaguchi, Y.; Ookawara, T.; et al. Myocardial stiffness is determined by ventricular fibrosis, but not by compensatory or excessive hypertrophy in hypertensive heart. Cardiovasc. Res. 2002, 55, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Geske, J.B.; McKie, P.M.; Ommen, S.R.; Sorajja, P. B-type natriuretic peptide and survival in hypertrophic cardiomyopathy. J. Am. Coll. Cardiol. 2013, 61, 2456–2460. [Google Scholar] [CrossRef]
- Yang, J.; Wang, H.; Zhang, Y.; Yang, Y.; Lu, M.; Zhang, J.; Li, S.; Zhang, S.; Li, G. Astragaloside IV attenuates inflammatory cytokines by inhibiting TLR4/NF-κB signaling pathway in isoproterenol-induced myocardial hypertrophy. J. Ethnopharmacol. 2013, 150, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.A.; Olson, E.N. Cardiac plasticity. New Engl. J. Med. 2008, 358, 1370–1380. [Google Scholar] [CrossRef]
- Rapacciuolo, A.; Esposito, G.; Caron, K.; Mao, L.; Thomas, S.A.; Rockman, H.A. Important role of endogenous norepinephrine and epinephrine in the development of in vivo pressure-overload cardiac hypertrophy. J. Am. Coll. Cardiol. 2001, 38, 876–882. [Google Scholar] [CrossRef]
- Gelinas, R.; Mailleux, F.; Dontaine, J.; Bultot, L.; Demeulder, B.; Ginion, A.; Daskalopoulos, E.P.; Esfahani, H.; Dubois-Deruy, E.; Lauzier, B.; et al. AMPK activation counteracts cardiac hypertrophy by reducing O-GlcNAcylation. Nat. Commun. 2018, 9, 374. [Google Scholar] [CrossRef]
- Naga, P.S.; Esposito, G.; Mao, L.; Koch, W.J.; Rockman, H.A. Gbetagamma-dependent phosphoinositide 3-kinase activation in hearts with in vivo pressure overload hypertrophy. J. Biol. Chem. 2000, 275, 4693–4698. [Google Scholar] [CrossRef]
- Molkentin, J.D.; Dorn, G.W. Cytoplasmic signaling pathways that regulate cardiac hypertrophy. Annu. Rev. Physiol. 2001, 63, 391–426. [Google Scholar] [CrossRef] [PubMed]
- Rockman, H.A.; Koch, W.J.; Lefkowitz, R.J. Seven-transmembrane-spanning receptors and heart function. Nature 2002, 415, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Chien, K.R. Meeting Koch’s postulates for calcium signaling in cardiac hypertrophy. J. Clin. Investig. 2000, 105, 1339–1342. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chien, K.R. Stress pathways and heart failure. Cell 1999, 98, 555–558. [Google Scholar] [CrossRef] [PubMed]
- Frey, N.; Katus, H.A.; Olson, E.N.; Hill, J.A. Hypertrophy of the heart. Circulation 2004, 109, 1580–1589. [Google Scholar] [CrossRef]
- Kana, K.; Song, H.; Laschinger, C.; Zandstra, P.W.; Radisic, M. PI3K phosphorylation is linked to improved electrical excitability in an in vitro engineered heart tissue disease model system. Tissue Eng. Part A 2015, 21, 2379–2389. [Google Scholar] [CrossRef] [PubMed]
- Shiojima, I. Disruption of coordinated cardiac hypertrophy and angiogenesis contributes to the transition to heart failure. J. Clin. Investig. 2005, 115, 2108–2118. [Google Scholar] [CrossRef]
- Nagoshi, T.; Matsui, T.; Aoyama, T.; Leri, A.; Anversa, P.; Li, L.; Ogawa, W.; del Monte, F.; Gwathmey, J.K.; Grazette, L.; et al. PI3K rescues the detrimental effects of chronic Akt activation in the heart during ischemia/reperfusion injury. J. Clin. Investig. 2005, 115, 2128–2138. [Google Scholar] [CrossRef]
- Wohlschlaeger, J.; Schmitz, K.J.; Palatty, J.; Takeda, A.; Takeda, N.; Vahlhaus, C.; Levkau, B.; Stypmann, J.; Schmid, C.; Schmid, K.W.; et al. Roles of cyclooxygenase-2 and phosphorylated Akt (Thr308) in cardiac hypertrophy regression mediated by left-ventricular unloading. J. Thorac. Cardiov. Sur. 2007, 133, 37–43. [Google Scholar] [CrossRef]
- Ma, Y.; Li, W.; Xie, Q. Rosuvastatin inhibits TGF-beta1 expression and alleviates myocardial fibrosis in diabetic rats. Die Pharm. 2013, 68, 355–358. [Google Scholar]
- Leask, A. Potential therapeutic targets for cardiac fibrosis TGF beta, angiotensin, endothelin, CCN2, and PDGF, partners in fibroblast activation. Circ. Res. 2010, 106, 1675–1680. [Google Scholar] [CrossRef] [PubMed]
- Schultz, J.E.J.; Witt, S.A.; Glascock, B.J.; Nieman, M.J.; Reiser, P.J.; Nix, S.L.; Kimball, T.R.; Doetschman, T. TGF-β1 mediates the hypertrophic cardiomyocyte growth induced by angiotensin II. J. Clin. Investig. 2002, 109, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Chung, A.C.; Huang, X.R.; Lan, H.Y. Angiotensin II induces connective tissue growth factor and collagen I expression via transforming growth factor-β-dependent and -independent smad pathways: The role of Smad3. Hypertension 2009, 54, 877–884. [Google Scholar] [CrossRef]
- Gao, X.; He, X.; Luo, B.; Peng, L.; Lin, J.; Zuo, Z. Angiotensin II increases collagen I expression via transforming growth factor-beta1 and extracellular signal-regulated kinase in cardiac fibroblasts. Eur. J. Pharmacol. 2009, 606, 115–120. [Google Scholar] [CrossRef]
- Dahlöf, B. Left ventricular hypertrophy and angiotensin II antagonists. Am. J. Hypertens. 2001, 14, 174–182. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, S.; Wang, X.; Xing, L.; Zhang, W. Effects of Long-Term Administration of Bovine Bone Gelatin Peptides on Myocardial Hypertrophy in Spontaneously Hypertensive Rats. Nutrients 2023, 15, 5021. https://doi.org/10.3390/nu15245021
Cao S, Wang X, Xing L, Zhang W. Effects of Long-Term Administration of Bovine Bone Gelatin Peptides on Myocardial Hypertrophy in Spontaneously Hypertensive Rats. Nutrients. 2023; 15(24):5021. https://doi.org/10.3390/nu15245021
Chicago/Turabian StyleCao, Songmin, Xinyu Wang, Lujuan Xing, and Wangang Zhang. 2023. "Effects of Long-Term Administration of Bovine Bone Gelatin Peptides on Myocardial Hypertrophy in Spontaneously Hypertensive Rats" Nutrients 15, no. 24: 5021. https://doi.org/10.3390/nu15245021
APA StyleCao, S., Wang, X., Xing, L., & Zhang, W. (2023). Effects of Long-Term Administration of Bovine Bone Gelatin Peptides on Myocardial Hypertrophy in Spontaneously Hypertensive Rats. Nutrients, 15(24), 5021. https://doi.org/10.3390/nu15245021