Abstract
Osteoporosis results in reduced bone strength and an elevated risk of fractures. Both overweight and underweight have been associated with osteoporosis; however, few studies have examined associations between osteoporosis and indices related to obesity. Therefore, the aim of this study was to investigate the associations of obesity-related indices, including body mass index (BMI), waist–hip ratio (WHR), waist-to-height ratio (WHtR), body roundness index (BRI), body adiposity index (BAI), abdominal volume index (AVI), lipid accumulation product (LAP), and visceral adiposity index (VAI), with baseline and change in calcaneus ultrasound T-score between baseline and follow-up (ΔT-score). T-score was measured using ultrasound. A total of 26,983 subjects were enrolled (mean age 51.2 ± 10.4 years). Multivariable analysis showed significant associations between low BMI (per 1 kg/m2; β, 0.065), WHR (per 1%; β, 0.012), WHtR (per 1%; β, 0.024), BRI (per 1; β, 0.079), BAI (per 1; β, 0.032), AVI (per 1; β, 0.049), and LAP (per 1; β, 0.005) with low baseline T-scores (all p < 0.001). Furthermore, there were significant associations between low BMI (per 1 kg/m2; β, 0.005; p = 0.036), BAI (per 1; β, 0.010; p < 0.001), and VAI (per 1; β, 0.017; p = 0.002) with low ΔT-scores. A low baseline T-score was significantly associated with low values of LAP, AVI, BAI, BMI, BRI, WHR, and WHtR but not VAI. In addition, low BMI, BAI, and VAI were significantly associated with low ΔT-scores, representing a rapidly decreasing T-score. Consequently, avoiding being underweight may help prevent osteoporosis in the Taiwanese population.
1. Introduction
Osteoporosis is a serious health issue worldwide, characterized by reduced bone mass, disturbance of the microarchitecture, and skeletal fragility, leading to a reduction in bone strength and elevated fracture risk [1]. The presence of fragility fractures, particularly of the spine, wrist, hip, humerus, rib, and pelvis or T-scores ≤ −2.5 standard deviations at any site based on bone mineral density (BMD) measurements using dual-energy X-ray absorptiometry (DXA), can be used to make a clinical diagnosis of osteoporosis [2]. Known risk factors for osteoporosis included being overweight, smoking tobacco, excess alcohol intake, insufficient physical activity, malnutrition, and certain medications such as long-term steroid therapy [3]. Known non-modifiable risk factors include sex, age, race, and genetic characteristics [4]. The social and economic burden of osteoporosis is steadily increasing because of the aging global population. According to the United States health care system, osteoporotic fractures cost approximately USD 17 billion annually, a figure which is expected to approach USD 50 billion by 2040 [5]. Fractures and their clinical sequelae, including pain, disability, and death, are the main complications of osteoporosis [6]. Hip fractures have been associated with a 15–20% increase in mortality within 1 year, and a 2.5-fold increased risk of future fractures [6]. The disability resulting from fractures causes a heavy burden on society [7], and consequently, determining the risk factors for osteoporosis in order to decrease the prevalence of osteoporosis-related fractures is an important topic. Hence, early detection, diagnosis, and treatment of osteoporosis would improve patient treatment prognosis and quality of life. Therefore, we need indices to help us to diagnose osteoporosis early.
Many simple and conveniently obtained anthropometric indicators have been introduced to assess the central obesity-related risk of metabolic syndrome, including body mass index (BMI), waist–hip ratio (WHR), waist-to-height ratio (WHtR), body roundness index (BRI), body adiposity index (BAI), abdominal volume index (AVI), lipid accumulation product (LAP), and visceral adiposity index (VAI) [8,9]. These indicators can be calculated using routinely obtained parameters such as body weight (BW) and height (BH), waist (WC) and hip circumference (HC), triglycerides (TG), and high-density lipoprotein cholesterol (HDL-C). These obesity-related indices can evaluate obesity, which is defined by an excess accumulation of adipose tissue. Previous research has suggested the relationship between obesity with insulin resistance and type 2 diabetes, which is that nonesterified fatty acids secreted from adipose tissue in obese people may lead to insulin resistance and β-cell dysfunction [10]. Prior studies revealed that these obesity-related indices are predictors of hypertension [11,12,13]. Our recent research revealed that these obesity indices were associated with fatty liver [14], albuminuria, advanced kidney disease [15], lung function [16], osteoporosis [17], hypertension [18], peripheral artery disease [19], and dementia [20]. Moreover, although both overweight and underweight have been associated with osteoporosis [21], few studies have explored the relationship between osteoporosis and indices related to obesity. The relationship between BMI and the risk of fracture has been demonstrated to be inverse and nonlinear, with the greatest risk in subjects with BMI < 20 kg/m2 and only a small risk reduction in those with a BMI > 25 kg/m2 [22,23]. Another systematic review and meta-analysis indicated that adult obese patients, defined as BMI > 30 kg/m2 or >28 kg/m2 in China, had higher lumbar spine and femoral neck BMD than patients with a healthy weight [24]. A systematic review found that low BMI (20–25 kg/m2) was an important and modifiable risk factor for the development of osteoporosis and osteoporotic fractures [25]. Therefore, this longitudinal study aimed to investigate the relationships of indices related to obesity with baseline and change in calcaneus ultrasound T-score (ΔT-score).
2. Materials and Methods
2.1. Taiwan Biobank
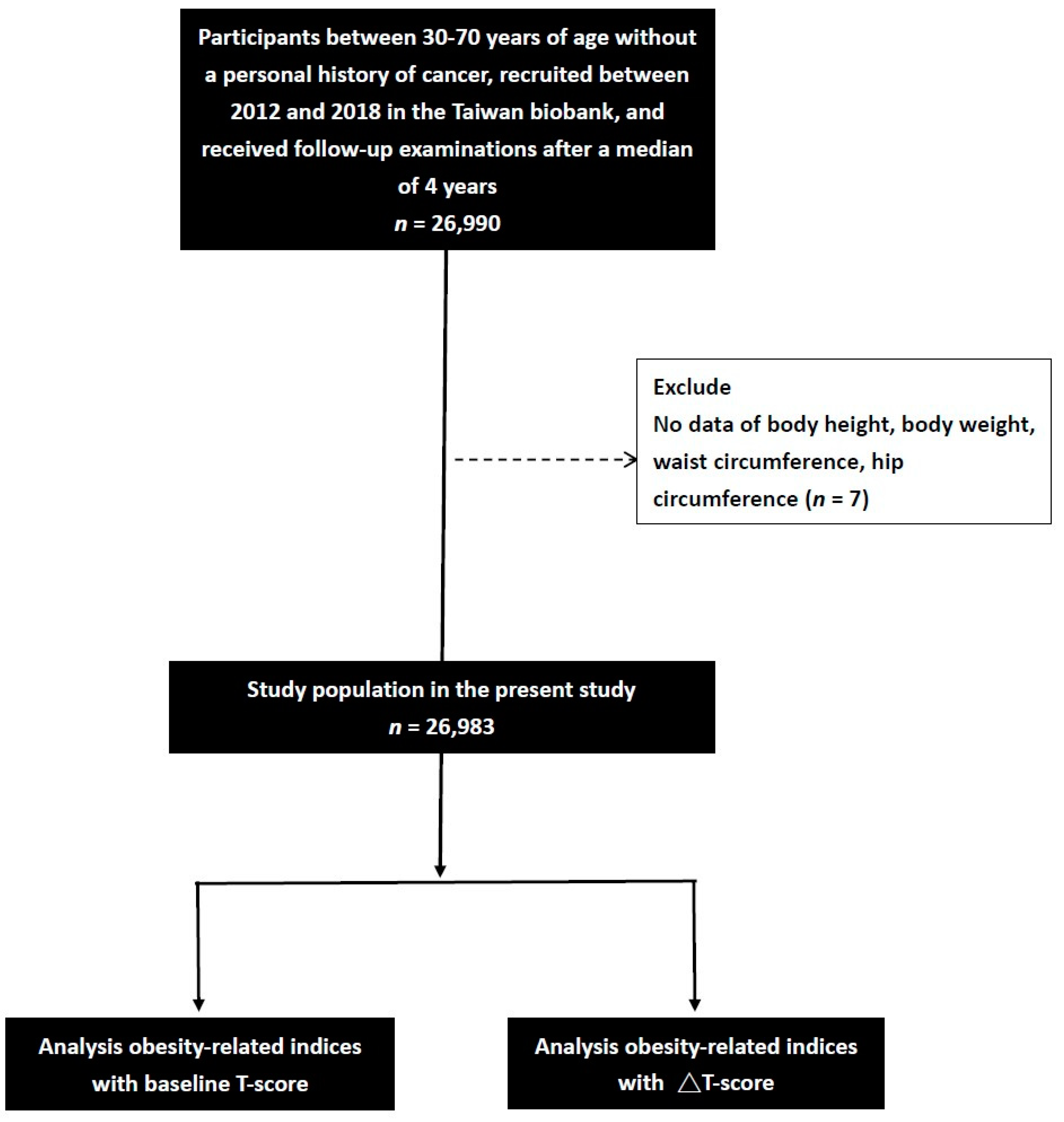
A total of 26,990 enrollees in the Taiwan Biobank had a median of 4 years of follow-up data. Those with no data on WC (n = 1), HC (n = 1), BH (n = 1), and BW (n = 4) were excluded, and a total of 26,983 participants (mean age 51.2 ± 10.4 years; men: 9543; women: 17,440) were included for analysis (Figure 1).

Figure 1.
Flowchart of study population. The change in T-score (ΔT-score) was calculated as the T-score at follow-up minus that at baseline.
The Taiwan Biobank was launched by the Taiwan government in 2012 as a prospective study of citizens aged 30–70 recruited from centers around Taiwan for epidemiological and biomedical research purposes. For each enrollee in the Taiwan Biobank, comprehensive genomic and phenotypic data are collected and recorded at enrolment and during follow-up through urine and blood tests (for uric acid, fasting glucose, hemoglobin, TG, total/HDL/low-density lipoprotein (LDL) cholesterol), physical examination (for BH, BW, HC, and WC), and structured questionnaires (for age, sex, and medical history of hypertension and diabetes mellitus (DM)) [26,27]. Ethical approval for the Taiwan Biobank was given by the Ethics and Governance Council of the Taiwan Biobank and Institutional Review Board on Biomedical Science Research, Academia Sinica, Taiwan. All participants in the Taiwan Biobank provided written informed consent before enrollment.
Body mass index was calculated as BW/BH2, and the estimated glomerular filtration rate (eGFR) was calculated using the 4-variable MDRD equation [28]. Trained personnel performed blood pressure (BP) measurements digitally with the participants abstaining from exercise, caffeine, and smoking for a minimum of 30 min before measurements. All measurements were taken three times with a 1–2 min break between measurements, with average values being used for analysis. The “Physical Fitness 333 Plan” criteria were used to define regular exercise, as promoted by the Ministry of Education in Taiwan, as at least 30 min three times a week [29].
This study was conducted according to the Declaration of Helsinki and approved by the Institutional Review Board of Kaohsiung Medical University Hospital (KMUHIRB-E(I)-20210058) on 8 April 2021.
2.2. Assessment of Calcaneus Ultrasound T-Score
The calcaneus of the non-dominant foot was used for ultrasound measurements (Achilles InSight, GE, Madison Heights, MI, USA) to calculate the T-score as (the participant’s T-score minus the mean T-score in a normal young adult population) divided by the SD of the normal young-adult population. The change in T-score (ΔT-score) was calculated as the T-score at follow-up minus that at baseline.
2.3. Calculation of Obesity-Related Indices
The calculation of obesity-related indices is shown in Table 1.

Table 1.
Calculation formula of each obesity-related index.
2.4. Statistical Analysis
Data are shown as number (%) and mean (±SD), as appropriate. The chi-square and independent t tests were used to examine differences in categorical and continuous variables, respectively. Significant variables in univariable analysis were used for multivariable analysis, which was used to evaluate the association of each obesity-related index with baseline T-score and ΔT-score. Results were considered significant at p < 0.05. SPSS version 19.0 for Windows (IBM Corp., Armonk, NY, USA) was used for the statistical analysis.
3. Results
The 26,983 enrolled participants were divided into two groups by baseline calcaneus T-score: ≥ −2.5 (n = 24,932; 92.4%) and <−2.5 (n = 2051; 7.6%).
3.1. Clinical Characteristics of the Study Groups
The T-score < −2.5 group had a higher prevalence of hypertension, regular exercise, and smoking, were older, predominantly male, and had higher systolic and diastolic BPs, higher menstruation status, higher BW and HC, lower T-scores at follow-up and baseline, higher fasting glucose and hemoglobin, lower eGFR, BMI, and BAI, and higher WHR compared to the T-score ≥ −2.5 group (Table 2).

Table 2.
Comparison of clinical characteristics among participants according to T score ≥ −2.5 or <−2.5.
3.2. Factors Associated with Baseline Calcaneus T-Score Using Univariable Analysis
Univariable linear regression analysis showed that a low baseline T-score was significantly associated with male sex, hypertension, regular exercise, older age, DM, smoking, drinking alcohol, high systolic and diastolic BPs, menopause, high hemoglobin, LDL-C, TG, uric acid, fasting glucose and total cholesterol, and low HDL-C and eGFR (Table 3).

Table 3.
Determinants of baseline T-score using univariable linear regression analysis.
3.3. Multivariable Analysis to Analyze the Relationships between the Indices Related to Obesity and Baseline Calcaneus Ultrasound T-Score
The following multivariable linear regression analysis models were conducted to determine relationships among the indices related to obesity with baseline T-score (Table 4).

Table 4.
Determinants of baseline T-score using multivariable linear regression analysis.
The analysis showed that a low baseline T-score was significantly associated with low values of BMI (per 1 kg/m2; unstandardized coefficient (β), 0.065; 95% confidence interval (CI), 0.058 to 0.072; p < 0.001), WHR (per 1%; β, 0.012; 95% CI, 0.008 to 0.016; p < 0.001), WHtR (per 1%; β, 0.024; 95% CI, 0.020 to 0.029; p < 0.001), BRI (per 1; β, 0.079; 95% CI, 0.066 to 0.093; p < 0.001), BAI (per 1; β, 0.032; 95% CI, 0.025 to 0.038; p < 0.001), AVI (per 1; β, 0.049; 95% CI, 0.041 to 0.057; p < 0.001), and LAP (per 1; β, 0.005; 95% CI, 0.004 to 0.0064; p = 0.022). However, no association between VAI and baseline T-score was found.
3.4. Factors Associated with ΔT-Score Using Univariable Analysis
Univariable linear regression analysis showed that a low ΔT-score was significantly associated with uric acid, female sex, older age, low diastolic BP, regular exercise, no alcohol or smoking history, menopause, hemoglobin, low fasting glucose, TG, and total/HDL-C (Table 5).

Table 5.
Determinants of ΔT-score using univariable linear regression analysis.
3.5. Relationships among the Indices Related to Obesity with ΔT-Score in Multivariable Analysis
The following multivariable linear regression analysis models were conducted to determine relationships among the indices related to obesity with T-score (Table 6).

Table 6.
Determinants of ΔT-score using multivariable linear regression analysis.
The analysis showed that a low ΔT-score was significantly associated with low values of BMI (per 1 kg/m2; β, 0.005; 95% CI, 0 to 0.011; p = 0.036), BAI (per 1; β, 0.010; 95% CI, 0.005 to 0.014; p < 0.001), and VAI (per 1; β, 0.017; 95% CI, 0.006 to 0.027; p = 0.002). However, no associations between LAP, WHR, WHtR, BRI, or AVI with ΔT-score were found.
4. Discussion
The results of this study showed that low LAP, AVI, BAI, BMI, BRI, WHR, and WHtR were associated with low baseline T-score. In addition, we found significant associations between a low ΔT-score with low BMI, BAI, and VAI.
An interesting finding was that a low baseline T-score was associated with low values of all the indices related to obesity studied except VAI. Low weight is a well-known risk factor for the future, whereas a high BMI has been suggested to have a protective effect [35,36,37,38,39,40]. From a public health point of view, however, the story is more complicated. Obesity is associated with increased morbidity from diabetes, hypertension, and cardiovascular diseases, and is also associated with increased mortality [23,41]. It is important, therefore, to quantify the association between BMI and fracture risk and to explore its relationship to age, sex, and BMD with the aim of being able to give balanced advice on lifestyle to patients. A previous meta-analysis found that without information on BMD, a decreased BMI in both women and men was correlated with a significantly higher age-specific fracture risk, and that those with increased BMI values had a decreased fracture risk [23]. However, when including information on BMD, the risk ratio per unit increase in BMI was found to change remarkably and to remain significantly different only for hip fractures in women [23]. Obesity has been shown to have a protective effect against fractures at some locations [22,42]. For example, obese postmenopausal women and men have been reported to have significantly reduced risks of fractures of the hip, pelvis, and wrist [43]. Another report from Catalonia also reported an association between obesity with lower risks of spine and hip fractures [44]. For osteoporotic fracture, a high BMI had a greater and significant protective effect in the absence of BMD [42]. With regard to health and mortality, WC and WHR are indicators of central adiposity and negatively impact both regardless of BMI [45,46]. The WC and WHR have been suggested by the World Health Organization as useful indicators of illness risk [47]. In addition, a cross-sectional study from Egypt reported a positive correlation between bone health and BMI, fat mass, and its distribution and basal metabolic rate, especially at the femoral neck, in women pre- and post-menopause, suggesting that overweight/obesity plays a protective role in bone health [48]. In a Chinese study, 5.75% of 9135 male participants had osteoporosis, among whom large body mass, including BMI, BRI, WHtR, WHR, a body shape index, and WC, was negatively associated with osteoporosis in older and middle-aged groups, and BMI was the strongest predictor of osteoporosis [49]. Moreover, a cross-sectional study of the United Kingdom Biobank including 3674 participants with magnetic resonance imaging and DXA findings found positive associations among abdominal subcutaneous adipose tissue, visceral adipose tissue, and total body adiposity with BMD [50]. A possible explanation for the association of obesity with high BMD is that obesity may cause an increase in BMD due to an elevated 17β-estradiol level with increased mechanical load, which may have a protective effect on bone [51]. Other studies have suggested that increased daily protein intake could positively affect BMD and functional performance [52,53,54,55].
We also found that low BMI, BAI, and VAI were significantly correlated with low ΔT-score, indicating a rapid decline in T-score. Several studies have reported greater bone loss in elderly underweight subjects compared to control subjects, with T-score values below −2.5 [56,57]. The mechanisms underlying this condition could be muscle weakness brought on by protein or vitamin D nutritional deficits, a loss of cushioning over the greater trochanter, or an increased risk of falling [58,59,60]. Underweight individuals have been shown to be at an increased risk of malnutrition [56]. Malnutrition, in particular with regard to protein, has been correlated with a higher risk of fractures related to osteoporosis through a decrease in bone mass affecting muscle strength [61]. Malnutrition, physical inactivity, and various other factors can cause tissue loss, a condition known as sarcopenia. Etiological factors include certain cytokines which can affect bone restructuring and proteolysis, and subsequently osteoclast metabolism. Mechanisms resulting in the release of cytokines such as inflammation and stress have been associated with unfavorable restructuring of bone and protein mass loss, resulting in worse muscular function prognosis [62,63,64]. Further studies are needed to elucidate the etiology; however, it may be partially due to the acceleration of bone mineral loss with repeated fracture episodes, which is more likely to occur in those who are underweight due to a decrease in muscle strength, malnutrition, and low BMD [21]. Weight bearing has been shown to enhance bone density through an effect on cells [65], and animal studies have shown that osteocytes are particularly sensitive to biomechanical stress [66]. Several reports have shown that osteoclasts underwent apoptosis in the absence of loading, but that this process was inhibited if the osteoclasts detected shear stress signals [67,68]. At the same time, osteoclast activity was inhibited and osteoblast differentiation was promoted. These findings are compatible with the present study, in that low values of the obesity-related indices, which would cause lower loading on osteocytes by weight bearing, were associated with low BMD and a rapid decline in T-score. These findings have important implications for the identification of possible cases based on clinical risk factors, and they suggest that avoiding being underweight may be important in helping to prevent osteoporosis in Taiwan.
Despite the inclusion of a large participant cohort with complete follow-up data, some limitations should be mentioned. First, we did not have prescription data for the participants, and the prevention and development of osteoporosis could have been affected by some medications. Second, we did not use DXA to confirm the presence of osteoporosis but rather ultrasound. The current diagnostic criteria are based on BMD, which is most commonly measured using DXA. Nevertheless, use of the T-score is recommended in research of osteoporosis pharmaceutical interventions. Moreover, ultrasound is relatively cost-efficient and can avoid radiation exposure. Furthermore, a previous study on women in China reported that the ultrasound system used in this study could identify osteoporosis as determined by axial BMD on DXA [69]. Another limitation is that as only approximately half of the Taiwan Biobank enrollees returned for follow-up examinations, sample bias cannot be ruled out, and this may have affected our findings. Finally, further studies may be necessary to verify our findings in other ethnicities, as all enrollees in the Taiwan Biobank are ethnically Chinese.
5. Conclusions
Low values of all indices related to obesity analyzed in this study except VAI were associated with a low baseline T-score. Furthermore, a low ΔT-score, representing a rapid decrease in T-score, was associated with low BMI, BAI, and VAI. Hence, avoiding being underweight may be important in helping to prevent osteoporosis in Taiwan.
Author Contributions
Conceptualization, L.-H.C., Y.-H.L., S.-C.C. and H.-M.S.; methodology, S.-C.C. and H.-M.S.; formal analysis, S.-C.C.; investigation, L.-H.C., Y.-H.L., S.-C.C. and H.-M.S.; data curation, S.-C.C. and H.-M.S.; writing—original draft, L.-H.C.; writing—review and editing, S.-C.C.; supervision, S.-C.C. and H.-M.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported partially by the Research Center for Precision Environmental Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan, from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan and by Kaohsiung Medical University Research Center Grant (KMU-TC111A01 and KMUTC111IFSP01).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Kaohsiung Medical University Hospital (protocol code KMUHIRB-E(I)-20210058).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data underlying this study are from the Taiwan Biobank. Due to restrictions placed on the data by the Personal Information Protection Act of Taiwan, the minimal data set cannot be made publicly available. Data may be available upon request to interested researchers. Please send data requests to: Szu-Chia Chen, PhD, MD. Division of Nephrology, Department of Internal Medicine, Kaohsiung Medical University Hospital, Kaohsiung Medical University.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pouresmaeili, F.; Kamalidehghan, B.; Kamarehei, M.; Goh, Y.M. A comprehensive overview on osteoporosis and its risk factors. Ther. Clin. Risk Manag. 2018, 14, 2029–2049. [Google Scholar] [CrossRef]
- Kanis, J.A. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: Synopsis of a WHO report. WHO Study Group. Osteoporos. Int. 1994, 4, 368–381. [Google Scholar] [CrossRef] [PubMed]
- Chavda, S.; Chavda, B.; Dube, R. Osteoporosis Screening and Fracture Risk Assessment Tool: Its Scope and Role in General Clinical Practice. Cureus 2022, 14, e26518. [Google Scholar] [CrossRef] [PubMed]
- Salari, N.; Ghasemi, H.; Mohammadi, L.; Behzadi, M.H.; Rabieenia, E.; Shohaimi, S.; Mohammadi, M. The global prevalence of osteoporosis in the world: A comprehensive systematic review and meta-analysis. J. Orthop. Surg. Res. 2021, 16, 609. [Google Scholar] [CrossRef]
- Nancy, E.; Lane, E. Etiology, and Diagnosis of Osteoporosis. Am. J. Obstet. Gynecol. 2006, 194, S3–S11. [Google Scholar]
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef]
- Chandran, M.; Lau, T.C.; Gagnon-Arpin, I.; Dobrescu, A.; Li, W.; Leung, M.Y.M.; Patil, N.; Zhao, Z. The health and economic burden of osteoporotic fractures in Singapore and the potential impact of increasing treatment rates through more pharmacological options. Arch. Osteoporos. 2019, 14, 114. [Google Scholar] [CrossRef]
- Wu, L.; Zhu, W.; Qiao, Q.; Huang, L.; Li, Y.; Chen, L. Novel and traditional anthropometric indices for identifying metabolic syndrome in non-overweight/obese adults. Nutr. Metab. 2021, 18, 3. [Google Scholar] [CrossRef]
- Jao, H.F.; Wung, C.H.; Yu, H.C.; Lee, M.Y.; Chen, P.C.; Chen, S.C.; Chang, J.M. Sex Difference in the Associations among Obesity-Related Indices with Metabolic Syndrome in Patients with Type 2 Diabetes Mellitus. Int. J. Med. Sci. 2021, 18, 3470–3477. [Google Scholar] [CrossRef]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–846. [Google Scholar] [CrossRef]
- Chiu, T.H.; Huang, Y.C.; Chiu, H.; Wu, P.Y.; Chiou, H.C.; Huang, J.C.; Chen, S.C. Comparison of Various Obesity-Related Indices for Identification of Metabolic Syndrome: A Population-Based Study from Taiwan Biobank. Diagnostics 2020, 10, 1081. [Google Scholar] [CrossRef] [PubMed]
- Janghorbani, M.; Aminorroaya, A.; Amini, M. Comparison of Different Obesity Indices for Predicting Incident Hypertension. High Blood Press. Cardiovasc. Prev. 2017, 24, 157–166. [Google Scholar] [CrossRef]
- Lin, Y.A.; Chen, Y.J.; Tsao, Y.C.; Yeh, W.C.; Li, W.C.; Tzeng, I.S.; Chen, J.Y. Relationship between obesity indices and hypertension among middle-aged and elderly populations in Taiwan: A community-based, cross-sectional study. BMJ Open 2019, 9, e031660. [Google Scholar] [CrossRef]
- Lin, I.T.; Lee, M.Y.; Wang, C.W.; Wu, D.W.; Chen, S.C. Gender Differences in the Relationships among Metabolic Syndrome and Various Obesity-Related Indices with Nonalcoholic Fatty Liver Disease in a Taiwanese Population. Int. J. Environ. Res. Public Health 2021, 18, 857. [Google Scholar] [CrossRef]
- Ou, Y.L.; Lee, M.Y.; Lin, I.T.; Wen, W.L.; Hsu, W.H.; Chen, S.C. Obesity-related indices are associated with albuminuria and advanced kidney disease in type 2 diabetes mellitus. Ren. Fail. 2021, 43, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.E.; Chen, S.C.; Geng, J.H.; Wu, D.W.; Wu, P.Y.; Huang, J.C. Obesity-Related Indices Are Associated with Longitudinal Changes in Lung Function: A Large Taiwanese Population Follow-Up Study. Nutrients 2021, 13, 4055. [Google Scholar] [CrossRef] [PubMed]
- Wung, C.H.; Chung, C.Y.; Wu, P.Y.; Huang, J.C.; Tsai, Y.C.; Chen, S.C.; Chiu, Y.W.; Chang, J.M. Associations between Metabolic Syndrome and Obesity-Related Indices and Bone Mineral Density T-Score in Hemodialysis Patients. J. Pers. Med. 2021, 11, 775. [Google Scholar] [CrossRef]
- Lee, W.C.; Wu, P.Y.; Huang, J.C.; Tsai, Y.C.; Chiu, Y.W.; Chen, S.C.; Chang, J.M.; Chen, H.C. Sex Difference in the Associations among Obesity-Related Indices with Incident Hypertension in a Large Taiwanese Population Follow-Up Study. J. Pers. Med. 2022, 12, 972. [Google Scholar] [CrossRef] [PubMed]
- Wung, C.H.; Lee, M.Y.; Wu, P.Y.; Huang, J.C.; Chen, S.C. Obesity-Related Indices Are Associated with Peripheral Artery Occlusive Disease in Patients with Type 2 Diabetes Mellitus. J. Pers. Med. 2021, 11, 533. [Google Scholar] [CrossRef]
- Huang, S.H.; Chen, S.C.; Geng, J.H.; Wu, D.W.; Li, C.H. Metabolic Syndrome and High-Obesity-Related Indices Are Associated with Poor Cognitive Function in a Large Taiwanese Population Study Older than 60 Years. Nutrients 2022, 14, 1535. [Google Scholar] [CrossRef]
- Nielson, C.M.; Srikanth, P.; Orwoll, E.S. Obesity and fracture in men and women: An epidemiologic perspective. J. Bone Miner. Res. 2012, 27, 1–10. [Google Scholar] [CrossRef]
- Compston, J. Obesity and bone. Curr. Osteoporos. Rep. 2013, 11, 30–35. [Google Scholar] [CrossRef]
- De Laet, C.; Kanis, J.A.; Oden, A.; Johanson, H.; Johnell, O.; Delmas, P.; Eisman, J.A.; Kroger, H.; Fujiwara, S.; Garnero, P.; et al. Body mass index as a predictor of fracture risk: A meta-analysis. Osteoporos. Int. 2005, 16, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Qiao, D.; Li, Y.; Liu, X.; Zhang, X.; Qian, X.; Zhang, H.; Zhang, G.; Wang, C. Association of obesity with bone mineral density and osteoporosis in adults: A systematic review and meta-analysis. Public Health 2020, 180, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Espallargues, M.; Sampietro-Colom, L.; Estrada, M.D.; Sola, M.; del Rio, L.; Setoain, J.; Granados, A. Identifying bone-mass-related risk factors for fracture to guide bone densitometry measurements: A systematic review of the literature. Osteoporos. Int. 2001, 12, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Yang, J.H.; Chiang, C.W.K.; Hsiung, C.N.; Wu, P.E.; Chang, L.C.; Chu, H.W.; Chang, J.; Song, I.W.; Yang, S.L.; et al. Population structure of Han Chinese in the modern Taiwanese population based on 10,000 participants in the Taiwan Biobank project. Hum. Mol. Genet. 2016, 25, 5321–5331. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.T.; Hung, T.H.; Yeh, C.K. Taiwan Regulation of Biobanks. J. Law Med. Ethics 2015, 43, 816–826. [Google Scholar] [CrossRef]
- Levey, A.S.; Bosch, J.P.; Lewis, J.B.; Greene, T.; Rogers, N.; Roth, D. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of Diet in Renal Disease Study Group. Ann. Intern. Med. 1999, 130, 461–470. [Google Scholar] [CrossRef]
- Ministry of Education. Physical Fitness 333 Plan; Ministry of Education: Taiwan, China, 1999.
- Thomas, D.M.; Bredlau, C.; Bosy-Westphal, A.; Mueller, M.; Shen, W.; Gallagher, D.; Maeda, Y.; McDougall, A.; Peterson, C.M.; Ravussin, E.; et al. Relationships between body roundness with body fat and visceral adipose tissue emerging from a new geometrical model. Obesity (Silver Spring) 2013, 21, 2264–2271. [Google Scholar] [CrossRef] [PubMed]
- Bergman, R.N.; Stefanovski, D.; Buchanan, T.A.; Sumner, A.E.; Reynolds, J.C.; Sebring, N.G.; Xiang, A.H.; Watanabe, R.M. A better index of body adiposity. Obesity (Silver Spring) 2011, 19, 1083–1089. [Google Scholar] [CrossRef]
- Guerrero-Romero, F.; Rodriguez-Moran, M. Abdominal volume index. An anthropometry-based index for estimation of obesity is strongly related to impaired glucose tolerance and type 2 diabetes mellitus. Arch. Med. Res. 2003, 34, 428–432. [Google Scholar] [CrossRef]
- Kahn, H.S. The “lipid accumulation product” performs better than the body mass index for recognizing cardiovascular risk: A population-based comparison. BMC Cardiovasc. Disord. 2005, 5, 26. [Google Scholar] [CrossRef]
- Amato, M.C.; Giordano, C.; Galia, M.; Criscimanna, A.; Vitabile, S.; Midiri, M.; Galluzzo, A. Visceral Adiposity Index: A reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care 2010, 33, 920–922. [Google Scholar] [CrossRef]
- Cummings, S.R.; Nevitt, M.C.; Browner, W.S.; Stone, K.; Fox, K.M.; Ensrud, K.E.; Cauley, J.; Black, D.; Vogt, T.M. Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N. Engl. J. Med. 1995, 332, 767–773. [Google Scholar] [CrossRef]
- Osteoporosis: Review of the evidence for prevention, diagnosis and treatment and cost-effectiveness analysis. Osteoporos. Int. 1998, 8 (Suppl. 4), S7–S80. [CrossRef]
- Honkanen, R.J.; Honkanen, K.; Kröger, H.; Alhava, E.; Tuppurainen, M.; Saarikoski, S. Risk factors for perimenopausal distal forearm fracture. Osteoporos. Int. 2000, 11, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Galvard, H.; Elmståhl, S.; Elmståhl, B.; Samuelsson, S.M.; Robertsson, E. Differences in body composition between female geriatric hip fracture patients and healthy controls: Body fat is more important as explanatory factor for the fracture than body weight and lean body mass. Aging 1996, 8, 282–286. [Google Scholar] [CrossRef]
- Joakimsen, R.M.; Fønnebø, V.; Magnus, J.H.; Tollan, A.; Søgaard, A.J. The Tromsø Study: Body height, body mass index and fractures. Osteoporos. Int. 1998, 8, 436–442. [Google Scholar] [CrossRef]
- Van der Voort, D.J.; Geusens, P.P.; Dinant, G.J. Risk factors for osteoporosis related to their outcome: Fractures. Osteoporos. Int. 2001, 12, 630–638. [Google Scholar] [CrossRef]
- Kopelman, P.G. Obesity as a medical problem. Nature 2000, 404, 635–643. [Google Scholar] [CrossRef]
- Ha, J.; Baek, K.H. Body mass index at the crossroads of osteoporosis and type 2 diabetes. Korean J. Intern. Med. 2020, 35, 1333–1335. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Alhambra, D.; Premaor, M.O.; Fina Aviles, F.; Hermosilla, E.; Martinez-Laguna, D.; Carbonell-Abella, C.; Nogues, X.; Compston, J.E.; Diez-Perez, A. The association between fracture and obesity is site-dependent: A population-based study in postmenopausal women. J. Bone Miner. Res. 2012, 27, 294–300. [Google Scholar] [CrossRef]
- Premaor, M.O.; Compston, J.E.; Fina Avilés, F.; Pagès-Castellà, A.; Nogués, X.; Díez-Pérez, A.; Prieto-Alhambra, D. The association between fracture site and obesity in men: A population-Based cohort study. J. Bone Miner. Res. 2013, 28, 1771–1777. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Klimentidis, Y.C.; Bea, J.W.; Ernst, K.C.; Hu, C.; Jackson, R.; Thomson, C.A. Body Mass Index, Waist Circumference, and Mortality in a Large Multiethnic Postmenopausal Cohort-Results from the Women’s Health Initiative. J. Am. Geriatr. Soc. 2017, 65, 1907–1915. [Google Scholar] [CrossRef] [PubMed]
- Padwal, R.; Leslie, W.D.; Lix, L.M.; Majumdar, S.R. Relationship Among Body Fat Percentage, Body Mass Index, and All-Cause Mortality: A Cohort Study. Ann. Intern. Med. 2016, 164, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Nishida, C.; Ko, G.T.; Kumanyika, S. Body fat distribution and noncommunicable diseases in populations: Overview of the 2008 WHO Expert Consultation on Waist Circumference and Waist-Hip Ratio. Eur. J. Clin. Nutr. 2010, 64, 2–5. [Google Scholar] [CrossRef]
- Hassan, N.E.; El-Masr, S.A.; El Bann, R.A.; Al-Tohamy, M.; El-Lebedy, D.; Abdel, D.A.; Amin, D.; Megahed, S.; Khalil, A. Bone Health and its Relation to Energy Intake, Fat Mass and its Distribution. Pak. J. Biol. Sci. 2020, 23, 1075–1085. [Google Scholar] [CrossRef]
- Qin, K.; He, M.; Cao, X.T.; Yang, H.W.; Yang, Y.; Wang, Y.J.; Yu, C.; An, Z.M.; Li, S.Y. Obesity and Osteoporosis in Men Aged Above 50. Sichuan Da Xue Xue Bao Yi Xue Ban 2017, 48, 17–22. [Google Scholar]
- Bland, V.L.; Klimentidis, Y.C.; Bea, J.W.; Roe, D.J.; Funk, J.L.; Going, S.B. Cross-sectional associations between adipose tissue depots and areal bone mineral density in the UK Biobank imaging study. Osteoporos. Int. 2022, 33, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; He, C.; He, W.; Yang, M.; Luo, X.; Li, C. Obesity and Bone Health: A Complex Link. Front. Cell Dev. Biol. 2020, 8, 600181. [Google Scholar] [CrossRef]
- Karunananthan, S.; Bergman, H.; Vedel, I.; Retornaz, F. Frailty: Searching for a relevant clinical and research paradigm. Rev. Med. Interne 2009, 30, 105–109. [Google Scholar] [CrossRef]
- Gaffney-Stomberg, E.; Insogna, K.L.; Rodriguez, N.R.; Kerstetter, J.E. Increasing dietary protein requirements in elderly people for optimal muscle and bone health. J. Am. Geriatr. Soc. 2009, 57, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Corti, M.C.; Guralnik, J.M.; Salive, M.E.; Sorkin, J.D. Serum albumin level and physical disability as predictors of mortality in older persons. Jama 1994, 272, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Tengstrand, B.; Cederholm, T.; Söderqvist, A.; Tidermark, J. Effects of protein-rich supplementation and nandrolone on bone tissue after a hip fracture. Clin. Nutr. 2007, 26, 460–465. [Google Scholar] [CrossRef]
- Coin, A.; Sergi, G.; Beninca, P.; Lupoli, L.; Cinti, G.; Ferrara, L.; Benedetti, G.; Tomasi, G.; Pisent, C.; Enzi, G. Bone mineral density and body composition in underweight and normal elderly subjects. Osteoporos. Int. 2000, 11, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Montalcini, T.; Romeo, S.; Ferro, Y.; Migliaccio, V.; Gazzaruso, C.; Pujia, A. Osteoporosis in chronic inflammatory disease: The role of malnutrition. Endocrine 2013, 43, 59–64. [Google Scholar] [CrossRef]
- Bischoff, H.A.; Stahelin, H.B.; Dick, W.; Akos, R.; Knecht, M.; Salis, C.; Nebiker, M.; Theiler, R.; Pfeifer, M.; Begerow, B.; et al. Effects of vitamin D and calcium supplementation on falls: A randomized controlled trial. J. Bone Miner. Res. 2003, 18, 343–351. [Google Scholar] [CrossRef]
- Wootton, R.; Brereton, P.J.; Clark, M.B.; Hesp, R.; Hodkinson, H.M.; Klenerman, L.; Reeve, J.; Slavin, G.; Tellez-Yudilevich, M. Fractured neck of femur in the elderly: An attempt to identify patients at risk. Clin. Sci. 1979, 57, 93–101. [Google Scholar] [CrossRef]
- Nilsson, B.E. Spinal osteoporosis and femoral neck fracture. Clin. Orthop. Relat. Res. 1970, 68, 93–95. [Google Scholar] [CrossRef]
- Rizzoli, R.; Bonjour, J.P. Malnutrition and osteoporosis. Z. Gerontol. Geriatr. 1999, 32 (Suppl. 1), I31–I37. [Google Scholar] [CrossRef]
- Rolland, Y.; Vellas, B. Sarcopenia. Rev. Med. Interne 2009, 30, 150–160. [Google Scholar] [CrossRef]
- Lang, T.; Streeper, T.; Cawthon, P.; Baldwin, K.; Taaffe, D.R.; Harris, T.B. Sarcopenia: Etiology, clinical consequences, intervention, and assessment. Osteoporos. Int. 2010, 21, 543–559. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Baumgartner, R.N.; Roubenoff, R.; Mayer, J.; Nair, K.S. Sarcopenia. J. Lab. Clin. Med. 2001, 137, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, P.; Body, J.J.; Boonen, S.; Boutsen, Y.; Devogelaer, J.P.; Goemaere, S.; Kaufman, J.; Reginster, J.Y.; Rozenberg, S. Loading and skeletal development and maintenance. J. Osteoporos. 2010, 2011, 786752. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; van der Plas, A.; Semeins, C.M.; Ajubi, N.E.; Frangos, J.A.; Nijweide, P.J.; Burger, E.H. Sensitivity of osteocytes to biomechanical stress in vitro. FASEB J. 1995, 9, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, J.I.; Plotkin, L.I.; Stewart, S.A.; Weinstein, R.S.; Parfitt, A.M.; Manolagas, S.C.; Bellido, T. Osteocyte apoptosis is induced by weightlessness in mice and precedes osteoclast recruitment and bone loss. J. Bone Miner. Res. 2006, 21, 605–615. [Google Scholar] [CrossRef]
- Gu, G.; Mulari, M.; Peng, Z.; Hentunen, T.A.; Vaananen, H.K. Death of osteocytes turns off the inhibition of osteoclasts and triggers local bone resorption. Biochem. Biophys. Res. Commun. 2005, 335, 1095–1101. [Google Scholar] [CrossRef]
- Jin, N.; Lin, S.; Zhang, Y.; Chen, F. Assess the discrimination of Achilles InSight calcaneus quantitative ultrasound device for osteoporosis in Chinese women: Compared with dual energy X-ray absorptiometry measurements. Eur. J. Radiol. 2010, 76, 265–268. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).