Rainbow Trout (Oncorhynchus mykiss) as Source of Multifunctional Peptides with Antioxidant, ACE and DPP-IV Inhibitory Activities
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Sample Preparation
2.2. FPH Ultrafiltration
2.3. Delineation of Potential Biological Activities
2.4. Cell Culture
2.5. MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) Assay
2.6. TEAC (Trolox Equivalent Antioxidant Capacity) Assay
2.7. ORAC Assay
2.8. Determination of Ferric Reducing Antioxidant Power
2.9. Determination of DPPH Activity
2.10. Nitric Oxide Level Evaluation on Caco-2 Cells
2.11. Measurement of Intracellular ROS
2.12. Lipid Peroxidation (MDA) Assay
2.13. In Vitro Measurement of the ACE Inhibitory Activity
2.14. In Vitro DPP-IV Activityinhibition Assay
2.15. Evaluation of the Inhibitory Effect of FPH on Cellular DPP-IV Activity
2.16. Western Blot Analysis
2.17. Statistical Investigation
3. Results
3.1. Bioactive Peptides Prediction by BIOPEP
3.2. Characterization of the Biochemical Activities of FPH
3.2.1. Antioxidant Activity
3.2.2. ACE and DPP-IV Inhibitory Activities
3.3. Effect of FPH on the Caco-2 Cell Vitality
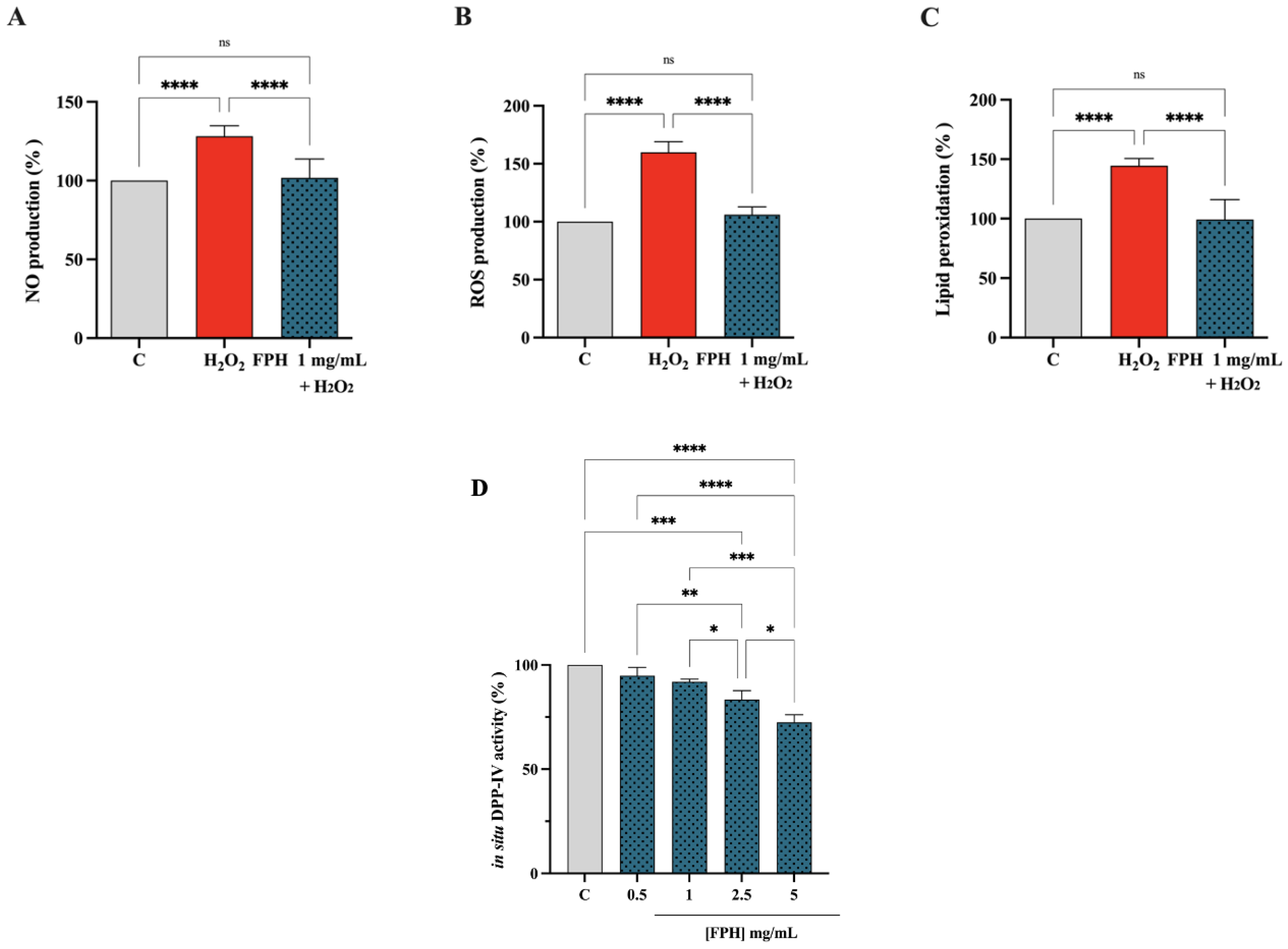
3.4. Characterization of Biological Activities of FPH at Cellular Levels: Antioxidant and DPP-IV Inhibitory Activities
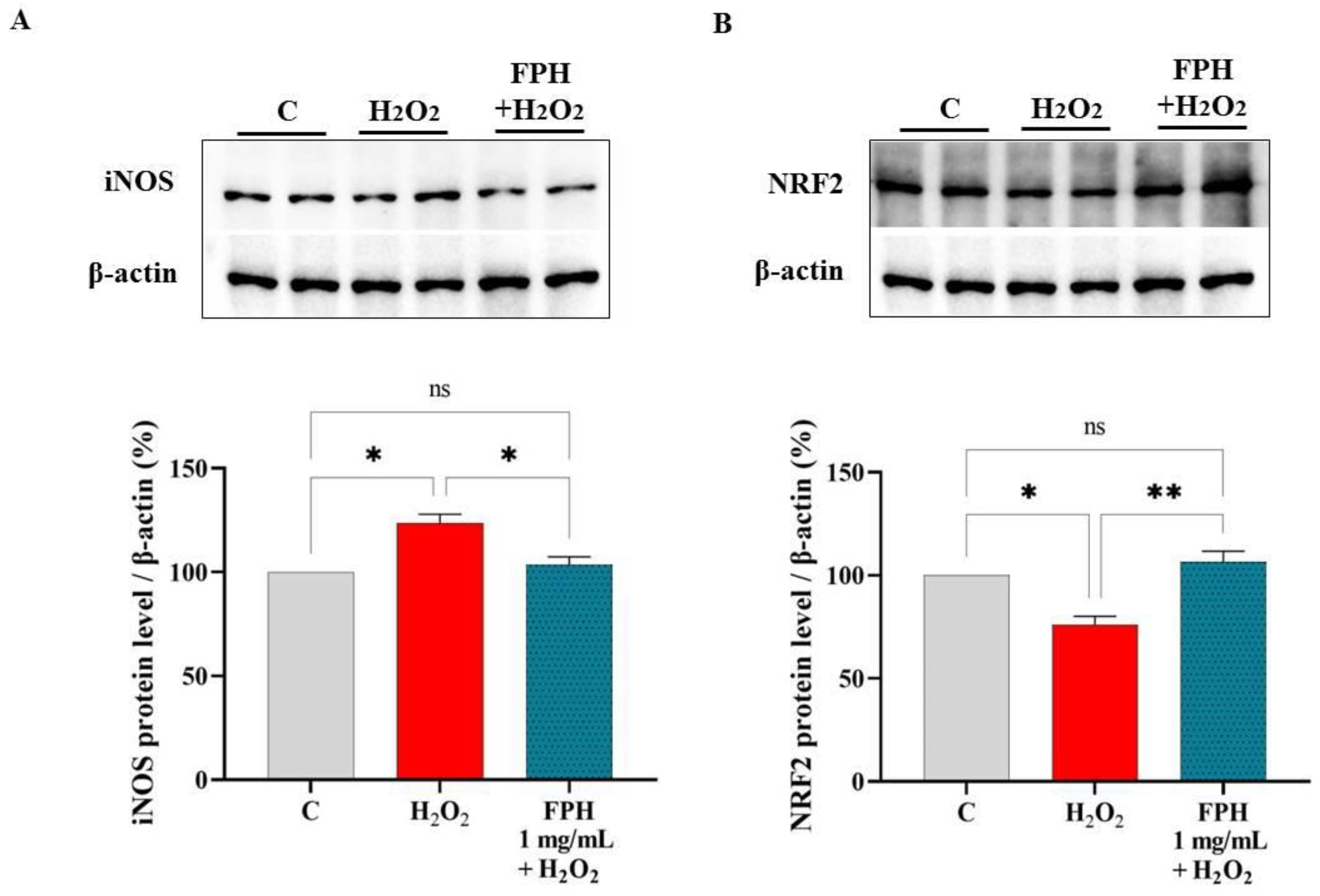
3.5. FPH Modulate the iNOS and NRF2 Protein Levels in Caco-2 Cells
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Akbarian, M.; Khani, A.; Eghbalpour, S.; Uversky, V.N. Bioactive Peptides: Synthesis, Sources, Applications, and Proposed Mechanisms of Action. Int. J. Mol. Sci. 2022, 23, 1445. [Google Scholar] [CrossRef]
- Harnedy, P.A.; FitzGerald, R.J. Bioactive peptides from marine processing waste and shellfish: A review. J. Funct. Foods 2012, 4, 6–24. [Google Scholar] [CrossRef]
- Rustad, T.; Storrø, I.; Slizyte, R. Possibilities for the utilisation of marine by-products. Int. J. Food Sci. Technol. 2011, 46, 2001–2014. [Google Scholar] [CrossRef]
- Hayes, M. Marine By-Products as a Source of Proteins for Potential Food, Pharma, and Agricultural Feed Use. Nov. Proteins Food Pharm. Agric. 2018, 163–182. [Google Scholar] [CrossRef]
- Ghaly, A.E.; Ramakrishnan, V.V.; Brooks, M.S.; Budge, S.M.; Dave, D. Fish Processing Wastes as a Potential Source of Proteins, Amino Acids and Oils: A Critical Review. J. Microb. Biochem. Technol. 2013, 5, 107–129. [Google Scholar] [CrossRef]
- Olsen, R.L.; Toppe, J.; Karunasagar, I. Challenges and realistic opportunities in the use of by-products from processing of fish and shellfish. Trends Food Sci. Technol. 2014, 36, 144–151. [Google Scholar] [CrossRef]
- Chalamaiah, M.; Dinesh Kumar, B.; Hemalatha, R.; Jyothirmayi, T. Fish protein hydrolysates: Proximate composition, amino acid composition, antioxidant activities and applications: A review. Food Chem. 2012, 135, 3020–3038. [Google Scholar] [CrossRef]
- Kim, S.-K.; Mendis, E. Bioactive compounds from marine processing byproducts—A review. Food Res. Int. 2006, 39, 383–393. [Google Scholar] [CrossRef]
- Abachi, S.; Pilon, G.; Marette, A.; Bazinet, L.; Beaulieu, L. Immunomodulatory effects of fish peptides on cardiometabolic syndrome associated risk factors: A review. Food Rev. Int. 2021, 1–44. [Google Scholar] [CrossRef]
- Shabir, U.; Ali, S.; Magray, A.R.; Ganai, B.A.; Firdous, P.; Hassan, T.; Nazir, R. Fish antimicrobial peptides (AMP’s) as essential and promising molecular therapeutic agents: A review. Microb. Pathog. 2018, 114, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Kumar, L.V.; Shakila, R.J.; Jeyasekaran, G. In Vitro Anti-Cancer, Anti-Diabetic, Anti-Inflammation and Wound Healing Properties of Collagen Peptides Derived from Unicorn Leatherjacket (Aluterus monoceros) at Different Hydrolysis. Turk. J. Fish. Aquat. Sci. 2019, 19, 551–560. [Google Scholar] [CrossRef]
- Wang, M.; Zhou, J.; Collado, M.; Barba, F. Accelerated Solvent Extraction and Pulsed Electric Fields for Valorization of Rainbow Trout (Oncorhynchus mykiss) and Sole (Dover sole) By-Products: Protein Content, Molecular Weight Distribution and Antioxidant Potential of the Extracts. Mar. Drugs 2021, 19, 207. [Google Scholar] [CrossRef] [PubMed]
- Nikoo, M.; Benjakul, S.; Yasemi, M.; Gavlighi, H.A.; Xu, X. Hydrolysates from rainbow trout (Oncorhynchus mykiss) processing by-product with different pretreatments: Antioxidant activity and their effect on lipid and protein oxidation of raw fish emulsion. LWT 2019, 108, 120–128. [Google Scholar] [CrossRef]
- Nikoo, M.; Regenstein, J.M.; Noori, F.; Gheshlaghi, S.P. Autolysis of rainbow trout (Oncorhynchus mykiss) by-products: Enzymatic activities, lipid and protein oxidation, and antioxidant activity of protein hydrolysates. LWT 2021, 140, 110702. [Google Scholar] [CrossRef]
- Nguyen, E.; Jones, O.; Kim, Y.H.B.; Martin-Gonzalez, F.S.; Liceaga, A.M. Impact of microwave-assisted enzymatic hydrolysis on functional and antioxidant properties of rainbow trout Oncorhynchus mykiss by-products. Fish. Sci. 2017, 83, 317–331. [Google Scholar] [CrossRef]
- Yaghoubzadeh, Z.; Ghadikolaii, F.P.; Kaboosi, H.; Safari, R.; Fattahi, E. Antioxidant Activity and Anticancer Effect of Bioactive Peptides from Rainbow Trout (Oncorhynchus mykiss) Skin Hydrolysate. Int. J. Pept. Res. Ther. 2019, 26, 625–632. [Google Scholar] [CrossRef]
- Wald, M.; Schwarz, K.; Rehbein, H.; Bußmann, B.; Beermann, C. Detection of antibacterial activity of an enzymatic hydrolysate generated by processing rainbow trout by-products with trout pepsin. Food Chem. 2016, 205, 221–228. [Google Scholar] [CrossRef]
- Priscilla, V.; Jose, E.Z. Optimization of Enzymatic Hydrolysis of Viscera Proteins of Rainbow Trout (Oncorhynchus mykiss). Adv. J. Food Sci. Technol. 2018, 16, 292–300. [Google Scholar] [CrossRef]
- Rajabzadeh, M.; Pourashouri, P.; Shabanpour, B.; Alishahi, A. Amino acid composition, antioxidant and functional properties of protein hydrolysates from the roe of rainbow trout (Oncorhynchus mykiss). Int. J. Food Sci. Technol. 2017, 53, 313–319. [Google Scholar] [CrossRef]
- Cheung, I.W.; Li-Chan, E.C. Enzymatic production of protein hydrolysates from steelhead (Oncorhynchus mykiss) skin gelatin as inhibitors of dipeptidyl-peptidase IV and angiotensin-I converting enzyme. J. Funct. Foods 2017, 28, 254–264. [Google Scholar] [CrossRef] [Green Version]
- Fan, W.; Tan, X.; Tu, M.; Jin, F.; Wang, Z.; Yu, C.; Qi, L.; Du, M. Preparation of the rainbow trout bone peptides directed by nutritional properties and flavor analyses. Food Sci. Nutr. 2018, 6, 925–933. [Google Scholar] [CrossRef]
- Kvangarsnes, K.; Dauksas, E.; Tolstorebrov, I.; Rustad, T.; Bartolomei, M.; Xu, R.; Lammi, C.; Cropotova, J. Physicochemical and Functional Properties of Rainbow Trout (Oncorhynchus mykiss) Hydrolysate. Article Submitted to Heliyon. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=4332924 (accessed on 6 January 2023).
- Howell, S.; Brewis, I.A.; Hooper, N.M.; Kenny, A.; Turner, A.J. Mosaic expression of membrane peptidases by confluent cultures of Caco-2 cells. FEBS Lett. 1993, 317, 109–112. [Google Scholar] [CrossRef]
- Bollati, C.; Xu, R.; Boschin, G.; Bartolomei, M.; Rivardo, F.; Li, J.; Arnoldi, A.; Lammi, C. Integrated Evaluation of the Multifunctional DPP-IV and ACE Inhibitory Effect of Soybean and Pea Protein Hydrolysates. Nutrients 2022, 14, 2379. [Google Scholar] [CrossRef]
- Lammi, C.; Zanoni, C.; Scigliuolo, G.M.; D’Amato, A.; Arnoldi, A. Lupin Peptides Lower Low-Density Lipoprotein (LDL) Cholesterol through an Up-regulation of the LDL Receptor/Sterol Regulatory Element Binding Protein 2 (SREBP2) Pathway at HepG2 Cell Line. J. Agric. Food Chem. 2014, 62, 7151–7159. [Google Scholar] [CrossRef] [PubMed]
- Bollati, C.; Cruz-Chamorro, I.; Aiello, G.; Li, J.; Bartolomei, M.; Santos-Sánchez, G.; Ranaldi, G.; Ferruzza, S.; Sambuy, Y.; Arnoldi, A.; et al. Investigation of the intestinal trans-epithelial transport and antioxidant activity of two hempseed peptides WVSPLAGRT (H2) and IGFLIIWV (H3). Food Res. Int. 2022, 152, 110720. [Google Scholar] [CrossRef] [PubMed]
- Santos-Sánchez, G.; Álvarez-López, A.I.; Ponce-España, E.; Carrillo-Vico, A.; Bollati, C.; Bartolomei, M.; Lammi, C.; Cruz-Chamorro, I. Hempseed (Cannabis sativa) protein hydrolysates: A valuable source of bioactive peptides with pleiotropic health-promoting effects. Trends Food Sci. Technol. 2022, 127, 303–318. [Google Scholar] [CrossRef]
- Bartolomei, M.; Capriotti, A.L.; Li, Y.; Bollati, C.; Li, J.; Cerrato, A.; Cecchi, L.; Pugliese, R.; Bellumori, M.; Mulinacci, N.; et al. Exploitation of Olive (Olea europaea L.) Seed Proteins as Upgraded Source of Bioactive Peptides with Multifunctional Properties: Focus on Antioxidant and Dipeptidyl-Dipeptidase—IV Inhibitory Activities, and Glucagon-like Peptide 1 Improved Modulation. Antioxidants 2022, 11, 1730. [Google Scholar] [CrossRef]
- Carmen, L.; Bollati, C.; Arnoldi, A. Antioxidant activity of soybean peptides on human hepatic HepG2 cells. J. Food Bioact. 2019, 7, 43–47. [Google Scholar] [CrossRef]
- Boschin, G.; Scigliuolo, G.M.; Resta, D.; Arnoldi, A. ACE-inhibitory activity of enzymatic protein hydrolysates from lupin and other legumes. Food Chem. 2014, 145, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Boschin, G.; Scigliuolo, G.M.; Resta, D.; Arnoldi, A. Optimization of the Enzymatic Hydrolysis of Lupin (Lupinus) Proteins for Producing ACE-Inhibitory Peptides. J. Agric. Food Chem. 2014, 62, 1846–1851. [Google Scholar] [CrossRef]
- Nasri, R.; Younes, I.; Jridi, M.; Trigui, M.; Bougatef, A.; Nedjar-Arroume, N.; Dhulster, P.; Nasri, M.; Karra-Châabouni, M. ACE inhibitory and antioxidative activities of Goby (Zosterissessor ophiocephalus) fish protein hydrolysates: Effect on meat lipid oxidation. Food Res. Int. 2013, 54, 552–561. [Google Scholar] [CrossRef]
- Ono, S.; Hosokawa, M.; Miyashita, K.; Takahashi, K. Inhibition properties of dipeptides from salmon muscle hydrolysate on angiotensin I-converting enzyme. Int. J. Food Sci. Technol. 2006, 41, 383–386. [Google Scholar] [CrossRef]
- Wu, S.; Feng, X.; Lan, X.; Xu, Y.; Liao, D. Purification and identification of Angiotensin-I Converting Enzyme (ACE) inhibitory peptide from lizard fish (Saurida elongata) hydrolysate. J. Funct. Foods 2015, 13, 295–299. [Google Scholar] [CrossRef]
- Bougatef, A.; Nedjar-Arroume, N.; Ravallec-Plé, R.; Leroy, Y.; Guillochon, D.; Barkia, A.; Nasri, M. Angiotensin I-converting enzyme (ACE) inhibitory activities of sardinelle (Sardinella aurita) by-products protein hydrolysates obtained by treatment with microbial and visceral fish serine proteases. Food Chem. 2008, 111, 350–356. [Google Scholar] [CrossRef]
- Elavarasan, K.; Shamasundar, B.; Badii, F.; Howell, N. Angiotensin I-converting enzyme (ACE) inhibitory activity and structural properties of oven- and freeze-dried protein hydrolysate from fresh water fish (Cirrhinus mrigala). Food Chem. 2016, 206, 210–216. [Google Scholar] [CrossRef]
- Himaya, S.; Ngo, D.-H.; Ryu, B.; Kim, S.-K. An active peptide purified from gastrointestinal enzyme hydrolysate of Pacific cod skin gelatin attenuates angiotensin-1 converting enzyme (ACE) activity and cellular oxidative stress. Food Chem. 2012, 132, 1872–1882. [Google Scholar] [CrossRef]
- Su, L.-J.; Zhang, J.-H.; Gomez, H.; Murugan, R.; Hong, X.; Xu, D.; Jiang, F.; Peng, Z.-Y. Reactive Oxygen Species-Induced Lipid Peroxidation in Apoptosis, Autophagy, and Ferroptosis. Oxidative Med. Cell. Longev. 2019, 2019, 5080843. [Google Scholar] [CrossRef]
- Khoubnasabjafari, M.; Ansarin, K.; Jouyban, A. Reliability of malondialdehyde as a biomarker of oxidative stress in psychological disorders. BioImpacts 2015, 5, 123–127. [Google Scholar] [PubMed]
- Lassoued, I.; Mora, L.; Nasri, R.; Aydi, M.; Toldrá, F.; Aristoy, M.-C.; Barkia, A.; Nasri, M. Characterization, antioxidative and ACE inhibitory properties of hydrolysates obtained from thornback ray (Raja clavata) muscle. J. Proteom. 2015, 128, 458–468. [Google Scholar] [CrossRef]
- Li, Z.; Wang, B.; Chi, C.; Gong, Y.; Luo, H.; Ding, G. Influence of average molecular weight on antioxidant and functional properties of cartilage collagen hydrolysates from Sphyrna lewini, Dasyatis akjei and Raja porosa. Food Res. Int. 2013, 51, 283–293. [Google Scholar] [CrossRef]
- Kumar, N.S.S.; Nazeer, R.A.; Jaiganesh, R. Purification and identification of antioxidant peptides from the skin protein hydrolysate of two marine fishes, horse mackerel (Magalaspis cordyla) and croaker (Otolithes ruber). Amino Acids 2012, 42, 1641–1649. [Google Scholar] [CrossRef] [PubMed]
- Sila, A.; Bougatef, A. Antioxidant peptides from marine by-products: Isolation, identification and application in food systems. A review. J. Funct. Foods 2016, 21, 10–26. [Google Scholar] [CrossRef]
- Ngo, D.-H. Marine Bioactive Peptides as Potential Antioxidants. Curr. Protein Pept. Sci. 2013, 14, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Halim, N.R.A.; Yusof, H.M.; Sarbon, N.M. Functional and bioactive properties of fish protein hydolysates and peptides: A comprehensive review. Trends Food Sci. Technol. 2016, 51, 24–33. [Google Scholar] [CrossRef]
- Mazzone, T.; Chait, A.; Plutzky, J. Cardiovascular disease risk in type 2 diabetes mellitus: Insights from mechanistic studies. Lancet 2008, 371, 1800–1809. [Google Scholar] [CrossRef]
- Hybertson, B.M.; Gao, B.; Bose, S.K.; McCord, J.M. Oxidative stress in health and disease: The therapeutic potential of Nrf2 activation. Mol. Asp. Med. 2011, 32, 234–246. [Google Scholar] [CrossRef]
- Kaneto, H.; Katakami, N.; Kawamori, D.; Miyatsuka, T.; Sakamoto, K.; Matsuoka, T.-A.; Matsuhisa, M.; Yamasaki, Y. Involvement of Oxidative Stress in the Pathogenesis of Diabetes. Antioxid. Redox Signal. 2007, 9, 355–366. [Google Scholar] [CrossRef]
- Sakai, K.; Matsumoto, K.; Nishikawa, T.; Suefuji, M.; Nakamaru, K.; Hirashima, Y.; Kawashima, J.; Shirotani, T.; Ichinose, K.; Brownlee, M.; et al. Mitochondrial reactive oxygen species reduce insulin secretion by pancreatic β-cells. Biochem. Biophys. Res. Commun. 2003, 300, 216–222. [Google Scholar] [CrossRef]
- Han, C.Y. Roles of Reactive Oxygen Species on Insulin Resistance in Adipose Tissue. Diabetes Metab. J. 2016, 40, 272–279. [Google Scholar] [CrossRef]
- Cryer, M.J.; Horani, T.; DiPette, D.J. Diabetes and Hypertension: A Comparative Review of Current Guidelines. J. Clin. Hypertens. 2016, 18, 95–100. [Google Scholar] [CrossRef] [Green Version]
- Harnedy, P.; Fitzgerald, R.J. In vitro assessment of the cardioprotective, anti-diabetic and antioxidant potential of Palmaria palmata protein hydrolysates. J. Appl. Phycol. 2013, 25, 1793–1803. [Google Scholar] [CrossRef]
- Neves, A.C.; Harnedy, P.A.; O’Keeffe, M.B.; FitzGerald, R.J. Bioactive peptides from Atlantic salmon (Salmo salar) with angiotensin converting enzyme and dipeptidyl peptidase IV inhibitory, and antioxidant activities. Food Chem. 2017, 218, 396–405. [Google Scholar] [CrossRef]
- Piepho, R.W. Overview of the Angiotensin-Converting-Enzyme Inhibitors. Am. J. Health-Syst. Pharm. 2000, 57, S3–S7. [Google Scholar] [CrossRef] [PubMed]
- Makrilakis, K. The Role of DPP-4 Inhibitors in the Treatment Algorithm of Type 2 Diabetes Mellitus: When to Select, What to Expect. Int. J. Environ. Res. Public Health 2019, 16, 2720. [Google Scholar] [CrossRef]
- Howell, S.; Kenny, A.J.; Turner, A.J. A survey of membrane peptidases in two human colonic cell lines, Caco-2 and HT-29. Biochem. J. 1992, 284, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Mentlein, R. Cell-Surface Peptidases. Int. Rev. Cytol. 2004, 235, 165–213. [Google Scholar] [CrossRef]
- Li, Y.; Aiello, G.; Fassi, E.; Boschin, G.; Bartolomei, M.; Bollati, C.; Roda, G.; Arnoldi, A.; Grazioso, G.; Lammi, C. Investigation of Chlorella pyrenoidosa Protein as a Source of Novel Angiotensin I-Converting Enzyme (ACE) and Dipeptidyl Peptidase-IV (DPP-IV) Inhibitory Peptides. Nutrients 2021, 13, 1624. [Google Scholar] [CrossRef]
- Lammi, C.; Bollati, C.; Gelain, F.; Arnoldi, A.; Pugliese, R. Enhancement of the Stability and Anti-DPPIV Activity of Hempseed Hydrolysates Through Self-Assembling Peptide-Based Hydrogels. Front. Chem. 2019, 6, 670. [Google Scholar] [CrossRef]
- Lammi, C.; Boschin, G.; Bollati, C.; Arnoldi, A.; Galaverna, G.; Dellafiora, L. A heuristic, computer-driven and top-down approach to identify novel bioactive peptides: A proof-of-principle on angiotensin I converting enzyme inhibitory peptides. Food Res. Int. 2021, 150, 110753. [Google Scholar] [CrossRef] [PubMed]
- Aiello, G.; Li, Y.; Boschin, G.; Bollati, C.; Arnoldi, A.; Lammi, C. Chemical and biological characterization of spirulina protein hydrolysates: Focus on ACE and DPP-IV activities modulation. J. Funct. Foods 2019, 63, 103592. [Google Scholar] [CrossRef]



| Sequence | Potential Bioactive Peptides | Biological Functions |
|---|---|---|
| (K)TEPGSLPEGKVK(I). | VK, GS, GK, EG, PG, TE, GKV, LP LP, EP, SL, EG, KV, PG, TE, VK | ACE inhibitor DPP-IV inhibitor |
| (K)IRLESDGSLLDVDEDDVEK(A) | IR IR, GS, DG, VE, EK, SDGS LL, EK, SL, ES, IR, RL, VD, VE | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)TELHFNHFAENSAFGIVPQPKSEDK(Q) | LH, EL, LHF AF, VP, GI, FG, TE, PQ, QP FA, VP, QP, AE, AF, FN, GI, HF, KS, LH, NH, PK, PQ, TE | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)DLKRTKVLLADAQIMLDHMK(N) | LK LA, KR, DA LA, LL, AD, IM, KR, KV, MK, ML, QI, TK, VL | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)QRPSSTTTDTGK(L) | RP, GK, TG, ST RP, PS, TD, TG, TT | ACE inhibitor DPP-IV inhibitor |
| (K)DCKKSRFSSDIVGPSDPQPDK(N) | RF, GP, VG, PQ, VGP, QP GP, QP, DP, KK, KS, PQ, PS, VG | ACE inhibitor DPP-IV inhibitor |
| (K)TPVESGASSAENRAADSTMTTSKPK(D) | KP RA, AA, GA, SG, KP, VE, TP, ST RA, TP, KP, GA, RA, AA, AD, AE, AS, ES, NR, PK, PV, SK, TM, TS, TT, VE | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)LCAEPVAESAKSEHAVTEESETK(D) | TE, AV VA, HA, EP, AE, AV, EH, ES, ET, KS, PV, TE, TK, VT | ACE inhibitor DPP-IV inhibitor |
| (K)RGGITCFLKVKCEEEMINDTMK(L) | LK VK, GI, GG, CF, RG FL, GG, GI, IN, KV, MI, MK, ND, RG, TM, VK | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)QHIIDGEKTIIQNPTDQQRKDHEK(A) | KD GE, DG, PT, EK NP, EK, DQ, GE, HE, HI, II, IQ, KT, PT, QH, QN, QQ, RK, TD, TI | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)SEHEVQDAELRTLLQSSASRKTQK(K) | EL DA, QK, EV, LQ, TQ, AEL, LR LL, AE, AS, EH, EV, HE, KT, QD, QS, RK, TL, TQ, VQ | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)IRCVEEKPVLSLPCVPHVAPPSNPK(A) | PHV, IR, KP IR, VAP, AP, VP, KP, VE, PP, EK, PH, LP PP, VA, AP, LP, VP, KP, NP, EK, SL, HV, IR, PH, PK, PS, PV, VE, VL | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)EVIRLEKDPEMLK(A) | KD, IR, LK RL, IR, EV, EK, LEK EK, DP, EV, IR, ML, RL, VI | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)ALYTQYLQFKENEIPLKETEK(S) | LY, LK LY, YL, PL, IP, EI, TE, LQ, TQ, EK, KE IP, EK, AL, PL, YT, EI, ET, KE, NE, QF, QY, TE, TQ, YL | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)MSHKSAVANGGGPGNHAYLTNK(E) | AY PG, GP, YL, AY, GP, GG, NG, PG, NK, HK, AV GP, VA, HA, AV, AY, GG, KS, LT, NG, NH, PG, SH, TN, YL | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)AISLNLNNYEK(E) | AI, NY, LN, EK, YE EK, SL, LN, NL, NN, NY, YE | ACE inhibitor DPP-IV inhibitor |
| (K)VSYECRVVSGKLVMGLDK(M) | GL, MG, GK, SG, SY, KL, YE, VM VV, GL, LV, MG, SY, VM, VS, YE, RV | ACE inhibitor DPP-IV inhibitor |
| (K)IVESYNTVSVLGVSK(S) | SVL GV, LG, SY, E, YN, LGV ES, GV, NT, SK, SV, SY, TV, VE, VL, VS, YN | Antioxidant ACE inhibitor DPP-IV inhibitor |
| (K)GSLGPFGVPGQVGPK(G) | LGP, GP, VP, VG, FG, GS, GV, GQ, LG, PG, VPG, QVGP GP, VP, SL, GV, PF, PG, PK, QV, VG, GPF | ACE inhibitor DPP-IV inhibitor |
| (K)GLQGSPGPMGKEGDVGPLGDAGGPGSKGEK(G) | GPL, PLG, GP, PL, VG, GL, AG, KG, DA, GS, MG, GK, GE, GG, QG, LG, GD, EG, PG, LQ, EK, KE, GPM, VGP, GP GP, SP, EK, GL, PL, AG, EG, GE, GG, KE, KG, MG, PG, PM, QG, SK, VG, GPM | ACE inhibitor DPP-IV inhibitor |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartolomei, M.; Cropotova, J.; Bollati, C.; Kvangarsnes, K.; d’Adduzio, L.; Li, J.; Boschin, G.; Lammi, C. Rainbow Trout (Oncorhynchus mykiss) as Source of Multifunctional Peptides with Antioxidant, ACE and DPP-IV Inhibitory Activities. Nutrients 2023, 15, 829. https://doi.org/10.3390/nu15040829
Bartolomei M, Cropotova J, Bollati C, Kvangarsnes K, d’Adduzio L, Li J, Boschin G, Lammi C. Rainbow Trout (Oncorhynchus mykiss) as Source of Multifunctional Peptides with Antioxidant, ACE and DPP-IV Inhibitory Activities. Nutrients. 2023; 15(4):829. https://doi.org/10.3390/nu15040829
Chicago/Turabian StyleBartolomei, Martina, Janna Cropotova, Carlotta Bollati, Kristine Kvangarsnes, Lorenza d’Adduzio, Jianqiang Li, Giovanna Boschin, and Carmen Lammi. 2023. "Rainbow Trout (Oncorhynchus mykiss) as Source of Multifunctional Peptides with Antioxidant, ACE and DPP-IV Inhibitory Activities" Nutrients 15, no. 4: 829. https://doi.org/10.3390/nu15040829
APA StyleBartolomei, M., Cropotova, J., Bollati, C., Kvangarsnes, K., d’Adduzio, L., Li, J., Boschin, G., & Lammi, C. (2023). Rainbow Trout (Oncorhynchus mykiss) as Source of Multifunctional Peptides with Antioxidant, ACE and DPP-IV Inhibitory Activities. Nutrients, 15(4), 829. https://doi.org/10.3390/nu15040829








