The Potential of the Adzuki Bean (Vigna angularis) and Its Bioactive Compounds in Managing Type 2 Diabetes and Glucose Metabolism: A Narrative Review
Abstract
:1. Introduction
2. Methods
3. Background for Adzuki Beans
4. The Role of Adzuki Bean Supplements in Managing T2D Outcomes
4.1. Effectiveness of Adzuki Bean Flour on T2D Outcomes
4.2. Effectiveness of Adzuki Bean Polyphenols on T2D Outcomes
4.3. Effectiveness of Adzuki Bean Polysaccharides on T2D Outcomes
4.4. Effectiveness of Extruded Adzuki Bean Proteins on T2D Outcomes
4.5. The Potential for Modifying the Antidiabetic Capacity of Adzuki Bean Supplements via Different Food Processing Methods
4.5.1. Effectiveness of Cooking on the Antidiabetic Capacity of Adzuki Bean Supplements
4.5.2. Effectiveness of Extrusion Cooking on the Antidiabetic Capacity of Adzuki Bean Supplements
4.5.3. Effectiveness of Germination on the Antidiabetic Capacity of Adzuki Bean Supplements
4.6. The Antidiabetic Capacity of Adzuki Bean Supplements, Comparing Their Capacity to Other Means Used to Manage T2D
4.6.1. Effectiveness of Adzuki Bean Hot Water Extract on T2D Outcomes when Compared to Sulfonylurea
4.6.2. Effectiveness of Adzuki Bean Polysaccharides on T2D Outcomes when Compared to Metformin
4.6.3. Effectiveness of Adzuki Bean Convenience Food on T2D Outcomes when Compared to Traditional Diabetic Low-Glycemic-Index Diets
4.7. The Antidiabetic Capacity of Adzuki Bean Supplementation via Improving Liver and Pancreatic Function
4.7.1. Effectiveness of Adzuki Bean Supplementation on Improving Pancreatic Function
4.7.2. Effectiveness of Adzuki Bean Supplementation on Improving Liver Function
4.8. The Role of Adzuki Bean Supplements on Insulin Signaling and Glucose Metabolism Pathways
5. Role of Adzuki Bean Supplementation in Managing Obesity Outcomes
5.1. Effectiveness of Adzuki Bean Flour on Obesity Outcomes
5.2. Effectiveness of Adzuki Bean Extract on Obesity Outcomes
5.3. Effectiveness of Adzuki Bean Polysaccharides and Protein on Obesity Outcomes
5.4. The Role of Adzuki Bean Supplementation in Adipogenesis and Lipolysis
5.4.1. Effectiveness of Adzuki Bean Supplementation on Adipogenesis
5.4.2. Effectiveness of Adzuki Bean Supplementation on Lipolysis
6. The Role of Adzuki Bean Supplementation on Dyslipidemia Outcomes
6.1. Effectiveness of Adzuki Bean Flour on Dyslipidemia Outcomes
6.2. Effectiveness of Adzuki Bean Extract on Dyslipidemia Outcomes
6.3. Effectiveness of Adzuki Bean Polysaccharides on Dyslipidemia Outcomes
6.4. Effectiveness of Adzuki Bean Protein on Dyslipidemia Outcomes
7. Effect of Adzuki Bean Supplementation on Gut Microbiota and Oxidative Stress
7.1. Effectiveness of Adzuki Bean Supplementation on Gut Microbiota Outcomes
7.2. Effectiveness of Adzuki Bean on Antioxidative Outcomes
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| T2D | Type 2 diabetes |
| PI3K | Phosphoinositide 3-kinases |
| AKT | Protein kinase B |
| HbA1c | Hemoglobin A1C |
| BW | Body weight |
| GABA | γ-aminobutyric acid |
| SOD1 | Cu/Zn superoxide dismutase |
| HOMA-IR | Homeostatic Model Assessment for Insulin Resistance |
| Insr | Insulin receptor |
| Irs-1 | Insulin receptor substrate 1 |
| Pi3k | Phosphoinositide 3-kinases |
| Akt | Proteinkinase B |
| Glut-2 | Glucose transporter 2 |
| Ppar-γ | Peroxisome proliferator-activated receptor-γ |
| C/ebp-α | CCAAT-enhancer-binding proteins-α |
| Lpl | Lipoprotein lipase |
| Cd36 | Cluster of differentiation 36 |
| Atgl | Adipose triglyceride lipase |
| Hsl | Hormone-sensitive lipase |
| Ppar-α | Peroxisome proliferator-activated receptor-α |
| Cpt-1α | Carnitine palmitoyltransferase 1α |
| Mcad | Medium-chain acyl-CoA dehydrogenase |
| LDL | Low-density lipoprotein cholesterol |
| VLDL | Very-low-density lipoprotein cholesterol |
| TG | Triglyceride/triacylglycerol |
| HDL | High-density lipoprotein cholesterol |
References
- World Health Organization. Noncommunicable Diseases. Available online: https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases (accessed on 22 August 2023).
- Centers for Disease Control and Prevention. Type 2 Diabetes. Available online: https://www.cdc.gov/diabetes/basics/type2.html (accessed on 21 August 2023).
- Centers for Disease Control and Prevention. How Type 2 Diabetes Affects Your Workforce. Available online: https://www.cdc.gov/diabetes/prevention/how-type2-affects-workforce.htm (accessed on 21 August 2023).
- International Diabetes Federation. IDF Diabetes Atlas, 10th ed.; International Diabetes Federation: Brussels, Belgium, 2021. [Google Scholar]
- Smushkin, G.; Vella, A. What Is Type 2 Diabetes? Medicine 2010, 38, 597–601. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association Professional Practice Committee 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2022. Diabetes Care 2022, 45, S17–S38. [Google Scholar] [CrossRef] [PubMed]
- Tomic, D.; Shaw, J.E.; Magliano, D.J. The Burden and Risks of Emerging Complications of Diabetes Mellitus. Nat. Rev. Endocrinol. 2022, 18, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.-X.; Ma, X.-N.; Guan, C.-H.; Li, Y.-D.; Mauricio, D.; Fu, S.-B. Cardiovascular Disease in Type 2 Diabetes Mellitus: Progress toward Personalized Management. Cardiovasc. Diabetol. 2022, 21, 74. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.J.; Adams, L.A.; Canbay, A.; Syn, W.-K. Extrahepatic Complications of Nonalcoholic Fatty Liver Disease. Hepatology 2014, 59, 1174–1197. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Hou, D.; Laraib, Y.; Xue, Y.; Shen, Q. Comparison of the Effects of Raw and Cooked Adzuki Bean on Glucose/Lipid Metabolism and Liver Function in Diabetic Mice. Cereal Chem. 2021, 98, 1081–1090. [Google Scholar] [CrossRef]
- Jiang, S.; Young, J.L.; Wang, K.; Qian, Y.; Cai, L. Diabetic-Induced Alterations in Hepatic Glucose and Lipid Metabolism: The Role of Type 1 and Type 2 Diabetes Mellitus. Mol. Med. Rep. 2020, 22, 603–611. [Google Scholar] [CrossRef]
- Dludla, P.V.; Mabhida, S.E.; Ziqubu, K.; Nkambule, B.B.; Mazibuko-Mbeje, S.E.; Hanser, S.; Basson, A.K.; Pheiffer, C.; Kengne, A.P. Pancreatic β-Cell Dysfunction in Type 2 Diabetes: Implications of Inflammation and Oxidative Stress. World J. Diabetes 2023, 14, 130–146. [Google Scholar] [CrossRef]
- Khin, P.P.; Lee, J.H.; Jun, H.-S. Pancreatic Beta-Cell Dysfunction in Type 2 Diabetes. Eur. J. Inflamm. 2023, 21, 1721727X231154152. [Google Scholar] [CrossRef]
- Kim, W.-H.; Lee, J.W.; Suh, Y.H.; Hong, S.H.; Choi, J.S.; Lim, J.H.; Song, J.H.; Gao, B.; Jung, M.H. Exposure to Chronic High Glucose Induces β-Cell Apoptosis Through Decreased Interaction of Glucokinase With Mitochondria: Downregulation of Glucokinase in Pancreatic β-Cells. Diabetes 2005, 54, 2602–2611. [Google Scholar] [CrossRef]
- Wang, K.; Wang, H.; Liu, Y.; Shui, W.; Wang, J.; Cao, P.; Wang, H.; You, R.; Zhang, Y. Dendrobium Officinale Polysaccharide Attenuates Type 2 Diabetes Mellitus via the Regulation of PI3K/Akt-Mediated Glycogen Synthesis and Glucose Metabolism. J. Funct. Foods 2018, 40, 261–271. [Google Scholar] [CrossRef]
- Ren, C.; Zhang, Y.; Cui, W.; Lu, G.; Wang, Y.; Gao, H.; Huang, L.; Mu, Z. A Polysaccharide Extract of Mulberry Leaf Ameliorates Hepatic Glucose Metabolism and Insulin Signaling in Rats with Type 2 Diabetes Induced by High Fat-Diet and Streptozotocin. Int. J. Biol. Macromol. 2015, 72, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-Y.; Shi, C.-X.; Gao, R.; Sun, H.-J.; Xiong, X.-Q.; Ding, L.; Chen, Q.; Li, Y.-H.; Wang, J.-J.; Kang, Y.-M.; et al. Irisin Inhibits Hepatic Gluconeogenesis and Increases Glycogen Synthesis via the PI3K/Akt Pathway in Type 2 Diabetic Mice and Hepatocytes. Clin. Sci. 2015, 129, 839–850. [Google Scholar] [CrossRef]
- Zorena, K.; Jachimowicz-Duda, O.; Ślęzak, D.; Robakowska, M.; Mrugacz, M. Adipokines and Obesity. Potential Link to Metabolic Disorders and Chronic Complications. Int. J. Mol. Sci. 2020, 21, 3570. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.; Keyes, D. Lifestyle Modification for Diabetes and Heart Disease Prevention. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Viguiliouk, E.; Stewart, S.E.; Jayalath, V.H.; Ng, A.P.; Mirrahimi, A.; de Souza, R.J.; Hanley, A.J.; Bazinet, R.P.; Blanco Mejia, S.; Leiter, L.A.; et al. Effect of Replacing Animal Protein with Plant Protein on Glycemic Control in Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2015, 7, 9804–9824. [Google Scholar] [CrossRef] [PubMed]
- Diabetes Canada Clinical Practice Guidelines Expert Committee; Sievenpiper, J.L.; Chan, C.B.; Dworatzek, P.D.; Freeze, C.; Williams, S.L. Nutrition Therapy. Can. J. Diabetes 2018, 42 (Suppl. S1), S64–S79. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, A.H.; Appel, L.J.; Vadiveloo, M.; Hu, F.B.; Kris-Etherton, P.M.; Rebholz, C.M.; Sacks, F.M.; Thorndike, A.N.; Van Horn, L.; Wylie-Rosett, J. 2021 Dietary Guidance to Improve Cardiovascular Health: A Scientific Statement From the American Heart Association. Circulation 2021, 144, e472–e487. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Kendall, C.W.C.; Augustin, L.S.A.; Mitchell, S.; Sahye-Pudaruth, S.; Blanco Mejia, S.; Chiavaroli, L.; Mirrahimi, A.; Ireland, C.; Bashyam, B.; et al. Effect of Legumes as Part of a Low Glycemic Index Diet on Glycemic Control and Cardiovascular Risk Factors in Type 2 Diabetes Mellitus: A Randomized Controlled Trial. Arch. Intern. Med. 2012, 172, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Nartea, A.; Kuhalskaya, A.; Fanesi, B.; Orhotohwo, O.L.; Susek, K.; Rocchetti, L.; Di Vittori, V.; Bitocchi, E.; Pacetti, D.; Papa, R. Legume Byproducts as Ingredients for Food Applications: Preparation, Nutrition, Bioactivity, and Techno-Functional Properties. Compr. Rev. Food Sci. Food Saf. 2023, 22, 1953–1985. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, W.; Deng, Z.; Li, H.; Zhang, B. The Composition and Antioxidant Activity of Bound Phenolics in Three Legumes, and Their Metabolism and Bioaccessibility of Gastrointestinal Tract. Foods 2020, 9, 1816. [Google Scholar] [CrossRef]
- Singh, N.; Jain, P.; Ujinwal, M.; Langyan, S. Escalate Protein Plates from Legumes for Sustainable Human Nutrition. Front. Nutr. 2022, 9, 977986. [Google Scholar] [CrossRef]
- Atkinson, F.S.; Brand-Miller, J.C.; Foster-Powell, K.; Buyken, A.E.; Goletzke, J. International Tables of Glycemic Index and Glycemic Load Values 2021: A Systematic Review. Am. J. Clin. Nutr. 2021, 114, 1625–1632. [Google Scholar] [CrossRef]
- Becerra-Tomás, N.; Díaz-López, A.; Rosique-Esteban, N.; Ros, E.; Buil-Cosiales, P.; Corella, D.; Estruch, R.; Fitó, M.; Serra-Majem, L.; Arós, F.; et al. Legume Consumption Is Inversely Associated with Type 2 Diabetes Incidence in Adults: A Prospective Assessment from the PREDIMED Study. Clin. Nutr. 2018, 37, 906–913. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Q.; Li, S.; Yue, Y.; Ma, Y.; Ren, G. Convenient Food Made of Extruded Adzuki Bean Attenuates Inflammation and Improves Glycemic Control in Patients with Type 2 Diabetes: A Randomized Controlled Trial. Ther. Clin. Risk Manag. 2018, 14, 871–884. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yang, T.; Liu, R.; Redden, B.; Maalouf, F.; Zong, X. Food Legume Production in China. Crop J. 2017, 5, 115–126. [Google Scholar] [CrossRef]
- Kitano-Okada, T.; Ito, A.; Koide, A.; Nakamura, Y.; Han, K.-H.; Shimada, K.; Sasaki, K.; Ohba, K.; Sibayama, S.; Fukushima, M. Anti-Obesity Role of Adzuki Bean Extract Containing Polyphenols: In Vivo and in Vitro Effects. J. Sci. Food Agric. 2012, 92, 2644–2651. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Liu, Z.; Zhu, Y.; Wang, H.; Dai, Z.; Yang, X.; Ren, X.; Xue, Y.; Shen, Q. Cooked Adzuki Bean Reduces High-Fat Diet-Induced Body Weight Gain, Ameliorates Inflammation, and Modulates Intestinal Homeostasis in Mice. Front. Nutr. 2022, 9, 918696. [Google Scholar] [CrossRef]
- WebMD. Editorial Contributors Health Benefits of Adzuki Beans. Available online: https://www.webmd.com/diet/health-benefits-of-adzuki-beans (accessed on 28 August 2023).
- Liu, R. Anti-Obesity Effects of Flavonoids and Saponins from Adzuki Bean; Hong Kong Baptist University: Hong Kong, China, 2014. [Google Scholar]
- Mukai, Y.; Sato, S. Polyphenol-Containing Azuki Bean (Vigna angularis) Seed Coats Attenuate Vascular Oxidative Stress and Inflammation in Spontaneously Hypertensive Rats. J. Nutr. Biochem. 2011, 22, 16–21. [Google Scholar] [CrossRef]
- Zhao, Q.; Hou, D.; Fu, Y.; Xue, Y.; Guan, X.; Shen, Q. Adzuki Bean Alleviates Obesity and Insulin Resistance Induced by a High-Fat Diet and Modulates Gut Microbiota in Mice. Nutrients 2021, 13, 3240. [Google Scholar] [CrossRef]
- Wu, G.; Bai, Z.; Wan, Y.; Shi, H.; Huang, X.; Nie, S. Antidiabetic Effects of Polysaccharide from Azuki Bean (Vigna angularis) in Type 2 Diabetic Rats via Insulin/PI3K/AKT Signaling Pathway. Food Hydrocoll. 2020, 101, 105456. [Google Scholar] [CrossRef]
- Lee, J.; Lee, B.W.; Kim, K.E.; An, H.S.; Jeong, E.A.; Shin, H.J.; Song, S.B.; Roh, G.S. Adzuki Bean MY59 Extract Reduces Insulin Resistance and Hepatic Steatosis in High-Fat-Fed Mice via the Downregulation of Lipocalin-2. Nutrients 2022, 14, 5049. [Google Scholar] [CrossRef]
- Kim, M.; Kim, D.K.; Cha, Y.-S. Black Adzuki Bean (Vigna angularis) Extract Protects Pancreatic β Cells and Improves Glucose Tolerance in C57BL/6J Mice Fed a High-Fat Diet. J. Med. Food 2016, 19, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.-J.; Liu, D.; Wan, Y.-J.; Huang, X.-J.; Nie, S.-P. Comparison of Hypoglycemic Effects of Polysaccharides from Four Legume Species. Food Hydrocoll. 2019, 90, 299–304. [Google Scholar] [CrossRef]
- Itoh, T.; Kobayashi, M.; Horio, F.; Furuichi, Y. Hypoglycemic Effect of Hot-Water Extract of Adzuki (Vigna angularis) in Spontaneously Diabetic KK-A(y) Mice. Nutrition 2009, 25, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Itoh, T.; Kita, N.; Kurokawa, Y.; Kobayashi, M.; Horio, F.; Furuichi, Y. Suppressive Effect of a Hot Water Extract of Adzuki Beans (Vigna angularis) on Hyperglycemia after Sucrose Loading in Mice and Diabetic Rats. Biosci. Biotechnol. Biochem. 2004, 68, 2421–2426. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Ren, G. Suppressive Effect of Extruded Adzuki Beans (Vigna angularis) on Hyperglycemia after Sucrose Loading in Rats. Ind. Crops Prod. 2014, 52, 228–232. [Google Scholar] [CrossRef]
- Yao, Y.; Cheng, X.; Ren, G. α-Glucosidase Inhibitory Activity of Protein-Rich Extracts from Extruded Adzuki Bean in Diabetic KK-Ay Mice. Food Funct. 2014, 5, 966–971. [Google Scholar] [CrossRef]
- Zhao, Q.; Fu, Y.; Zhang, F.; Wang, C.; Yang, X.; Bai, S.; Xue, Y.; Shen, Q. Heat-Treated Adzuki Bean Protein Hydrolysates Reduce Obesity in Mice Fed a High-Fat Diet via Remodeling Gut Microbiota and Improving Metabolic Function. Mol. Nutr. Food Res. 2022, 66, e2100907. [Google Scholar] [CrossRef]
- Jiang, X.; Xu, Q.; Zhang, A.; Liu, Y.; Li, Z.; Tang, H.; Cao, D.; Zhang, D. Revealing the Hypoglycemic Effects and Mechanism of GABA-Rich Germinated Adzuki Beans on T2DM Mice by Untargeted Serum Metabolomics. Front. Nutr. 2021, 8, 791191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Jiang, X.; Ge, Y.; Xu, Q.; Li, Z.; Tang, H.; Cao, D.; Zhang, D. The Effects of GABA-Rich Adzuki Beans on Glycolipid Metabolism, as Well as Intestinal Flora, in Type 2 Diabetic Mice. Front. Nutr. 2022, 9, 849529. [Google Scholar] [CrossRef]
- Liu, R.; Zheng, Y.; Cai, Z.; Xu, B. Saponins and Flavonoids from Adzuki Bean (Vigna angularis L.) Ameliorate High-Fat Diet-Induced Obesity in ICR Mice. Front. Pharmacol. 2017, 8, 687. [Google Scholar] [CrossRef] [PubMed]
- Sabarathinam, S. A Glycemic Diet Improves the Understanding of Glycemic Control in Diabetes Patients during Their Follow-Up. Future Sci. OA 2023, 9, FSO843. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, Y.N.; Takahashi, Y.; Yamaki, K. Phenolic Antioxidants in Some Vigna Species of Legumes and Their Distinct Inhibitory Effects on α-Glucosidase and Pancreatic Lipase Activities. J. Food Sci. 2012, 77, C927–C933. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.-S.; Teng, C.-Y.; Hsieh, K.-F.; Chiou, Y.-S.; Wu, J.-C.; Lu, T.-J.; Pan, M.-H. Adzuki Bean Water Extract Attenuates Obesity by Modulating M2/M1 Macrophage Polarization and Gut Microbiota Composition. Mol. Nutr. Food Res. 2019, 63, e1900626. [Google Scholar] [CrossRef] [PubMed]
- Yook, J.-S.; Kim, K.-A.; Kim, M.; Cha, Y.-S. Black Adzuki Bean (Vigna angularis) Attenuates High-Fat Diet-Induced Colon Inflammation in Mice. J. Med. Food 2017, 20, 367–375. [Google Scholar] [CrossRef]
- Choi, J.M.; Lee, S.I.; Cho, E.J. Effect of Vigna angularis on High-Fat Diet-Induced Memory and Cognitive Impairments. J. Med. Food 2020, 23, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Song, S.-B.; Cha, Y.-S. Effects of Black Adzuki Bean (Vigna angularis, Geomguseul) Extract on Body Composition and Hypothalamic Neuropeptide Expression in Rats Fed a High-Fat Diet. Food Nutr. Res. 2015, 59, 27719. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Pichiah, P.B.T.; Kim, D.K.; Cha, Y.-S. Black Adzuki Bean (Vigna angularis) Extract Exerts Phenotypic Effects on White Adipose Tissue and Reverses Liver Steatosis in Diet-Induced Obese Mice. J. Food Biochem. 2017, 41, e12333. [Google Scholar] [CrossRef]
- Kim, M.; Park, J.-E.; Song, S.-B.; Cha, Y.-S. Effects of Black Adzuki Bean (Vigna angularis) Extract on Proliferation and Differentiation of 3T3-L1 Preadipocytes into Mature Adipocytes. Nutrients 2015, 7, 277–292. [Google Scholar] [CrossRef]
- Van Gaal, L.; Scheen, A. Weight Management in Type 2 Diabetes: Current and Emerging Approaches to Treatment. Diabetes Care 2015, 38, 1161–1172. [Google Scholar] [CrossRef]
- Saad, B.; Kmail, A.; Haq, S.Z.H. Anti-Diabesity Middle Eastern Medicinal Plants and Their Action Mechanisms. Evid. Based Complement. Alternat. Med. 2022, 2022, 2276094. [Google Scholar] [CrossRef]
- Medhi, G.K.; Dutta, G.; Borah, P.; Lyngdoh, M.; Sarma, A. Prevalence of Diabetes and Its Relationship with Body Mass Index Among Elderly People in a Rural Area of Northeastern State of India. Cureus 2021, 13, e12747. [Google Scholar] [CrossRef]
- Gupta, S.; Bansal, S. Does a Rise in BMI Cause an Increased Risk of Diabetes?: Evidence from India. PLoS ONE 2020, 15, e0229716. [Google Scholar] [CrossRef]
- Ruze, R.; Liu, T.; Zou, X.; Song, J.; Chen, Y.; Xu, R.; Yin, X.; Xu, Q. Obesity and Type 2 Diabetes Mellitus: Connections in Epidemiology, Pathogenesis, and Treatments. Front. Endocrinol. 2023, 14, 1161521. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, X.-P.; Yuan, J.; Cai, B.; Wang, X.-L.; Wu, X.-L.; Zhang, Y.-H.; Zhang, X.-Y.; Yin, T.; Zhu, X.-H.; et al. Association of Body Mass Index and Age with Incident Diabetes in Chinese Adults: A Population-Based Cohort Study. BMJ Open 2018, 8, e021768. [Google Scholar] [CrossRef] [PubMed]
- Teufel, F.; Seiglie, J.A.; Geldsetzer, P.; Theilmann, M.; Marcus, M.E.; Ebert, C.; Arboleda, W.A.L.; Agoudavi, K.; Andall-Brereton, G.; Aryal, K.K.; et al. Body-Mass Index and Diabetes Risk in 57 Low-Income and Middle-Income Countries: A Cross-Sectional Study of Nationally Representative, Individual-Level Data in 685,616 Adults. Lancet 2021, 398, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Karin, A.; Jon, E.; Martin, A.; Lena, B.; Martin, L.; Naveed, S.; Marcus, L.; Maria, Å.; Annika, R. Body Mass Index in Adolescence, Risk of Type 2 Diabetes and Associated Complications: A Nationwide Cohort Study of Men. EClinicalMedicine 2022, 46, 101356. [Google Scholar] [CrossRef] [PubMed]
- Schnurr, T.M.; Jakupović, H.; Carrasquilla, G.D.; Ängquist, L.; Grarup, N.; Sørensen, T.I.A.; Tjønneland, A.; Overvad, K.; Pedersen, O.; Hansen, T.; et al. Obesity, Unfavourable Lifestyle and Genetic Risk of Type 2 Diabetes: A Case-Cohort Study. Diabetologia 2020, 63, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Haider, N.; Larose, L. Harnessing Adipogenesis to Prevent Obesity. Adipocyte 2019, 8, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Jakab, J.; Miškić, B.; Mikšić, Š.; Juranić, B.; Ćosić, V.; Schwarz, D.; Včev, A. Adipogenesis as a Potential Anti-Obesity Target: A Review of Pharmacological Treatment and Natural Products. Diabetes Metab. Syndr. Obes. 2021, 14, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Mao, S.; Chen, S.; Zhang, W.; Liu, C. PPARs-Orchestrated Metabolic Homeostasis in the Adipose Tissue. Int. J. Mol. Sci. 2021, 22, 8974. [Google Scholar] [CrossRef]
- Jung, S.J.; Choi, Y.J.; Park, T.K.; Woo, S.E.; Kim, B.-Y.; Yoon, J.S.; Jang, S.Y. Wnt Signalling Inhibits Adipogenesis in Orbital Fibroblasts from Patients with Graves’ Orbitopathy. Br. J. Ophthalmol. 2022, 106, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Lass, A.; Zimmermann, R.; Oberer, M.; Zechner, R. Lipolysis—A Highly Regulated Multi-Enzyme Complex Mediates the Catabolism of Cellular Fat Stores. Prog. Lipid Res. 2011, 50, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Wu, Y.; Rong, X.; Zheng, C.; Guo, J. Anti-Lipolysis Induced by Insulin in Diverse Pathophysiologic Conditions of Adipose Tissue. Diabetes Metab. Syndr. Obes. 2020, 13, 1575–1585. [Google Scholar] [CrossRef] [PubMed]
- Franz, M.J. Weight Management: Obesity to Diabetes. Diabetes Spectr. 2017, 30, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Berberich, A.J.; Hegele, R.A. A Modern Approach to Dyslipidemia. Endocr. Rev. 2022, 43, 611–653. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Chen, X.; Wang, B. Pathology of Metabolically-Related Dyslipidemia. Clin. Chim. Acta 2021, 521, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Mooradian, A.D. Dyslipidemia in Type 2 Diabetes Mellitus. Nat. Clin. Pract. Endocrinol. Metab. 2009, 5, 150–159. [Google Scholar] [CrossRef]
- Kaze, A.D.; Santhanam, P.; Musani, S.K.; Ahima, R.; Echouffo-Tcheugui, J.B. Metabolic Dyslipidemia and Cardiovascular Outcomes in Type 2 Diabetes Mellitus: Findings From the Look AHEAD Study. J. Am. Heart Assoc. 2021, 10, e016947. [Google Scholar] [CrossRef]
- Kamrul-Hasan, A.B.M.; Alam, M.S.; Zarin, N.; Kabir, M.A.; Gaffar, A.J.; Hossain, M.F.; Talukder, S.K.; Raunak, A.I.B.; Nabi, M.M.U.; Asaduzzaman, M.; et al. Prevalence and Patterns of Dyslipidemia among Lipid-Lowering Drug-Naïve Patients with Type 2 Diabetes Mellitus—A Countrywide Study in Bangladesh. Endocr. Metab. Sci. 2023, 13, 100152. [Google Scholar] [CrossRef]
- Ahmmed, M.S.; Shuvo, S.D.; Paul, D.K.; Karim, M.R.; Kamruzzaman, M.; Mahmud, N.; Ferdaus, M.J.; Elahi, M.T. Prevalence of Dyslipidemia and Associated Risk Factors among Newly Diagnosed Type-2 Diabetes Mellitus (T2DM) Patients in Kushtia, Bangladesh. PLoS Glob. Public Health 2021, 1, e0000003. [Google Scholar] [CrossRef] [PubMed]
- Bhowmik, B.; Siddiquee, T.; Mujumder, A.; Afsana, F.; Ahmed, T.; Mdala, I.A.; Moreira, N.C.d.V.; Khan, A.K.A.; Hussain, A.; Holmboe-Ottesen, G.; et al. Serum Lipid Profile and Its Association with Diabetes and Prediabetes in a Rural Bangladeshi Population. Int. J. Environ. Res. Public Health 2018, 15, 1944. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.-W.; Deng, F.-Y.; Lei, S.-F. Meta-Analysis of Atherogenic Index of Plasma and Other Lipid Parameters in Relation to Risk of Type 2 Diabetes Mellitus. Prim. Care Diabetes 2015, 9, 60–67. [Google Scholar] [CrossRef]
- Iatcu, C.O.; Steen, A.; Covasa, M. Gut Microbiota and Complications of Type-2 Diabetes. Nutrients 2021, 14, 166. [Google Scholar] [CrossRef] [PubMed]
- Tangvarasittichai, S. Oxidative Stress, Insulin Resistance, Dyslipidemia and Type 2 Diabetes Mellitus. World J. Diabetes 2015, 6, 456–480. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Meckling, K.A.; Marcone, M.F.; Kakuda, Y.; Proulx, A.; Tsao, R. In Vitro Antioxidant Synergism and Antagonism between Food Extracts Can Lead to Similar Activities in H2O2-Induced Cell Death, Caspase-3 and MMP-2 Activities in H9c2 Cells. J. Sci. Food Agric. 2012, 92, 2983–2993. [Google Scholar] [CrossRef]
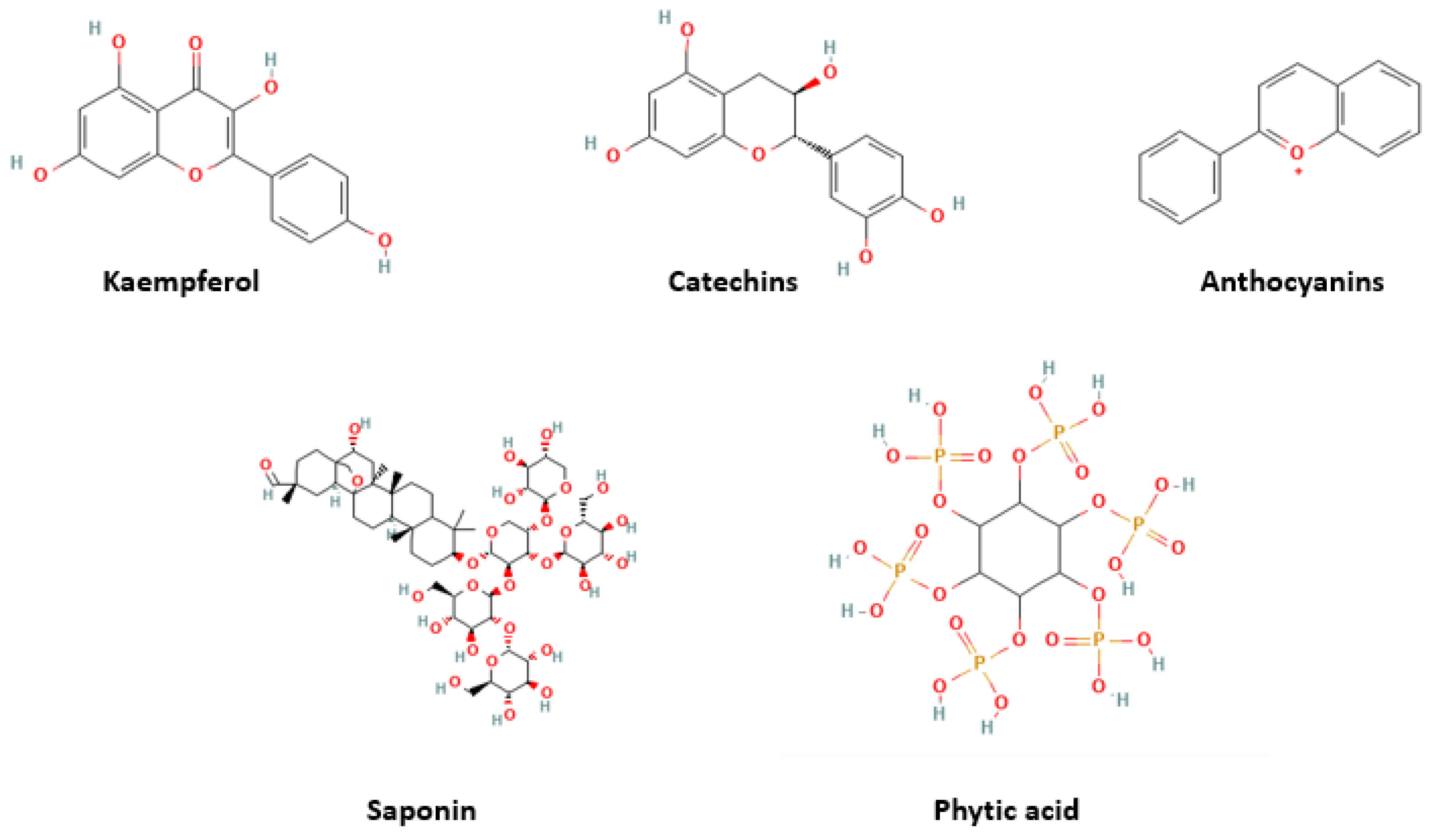
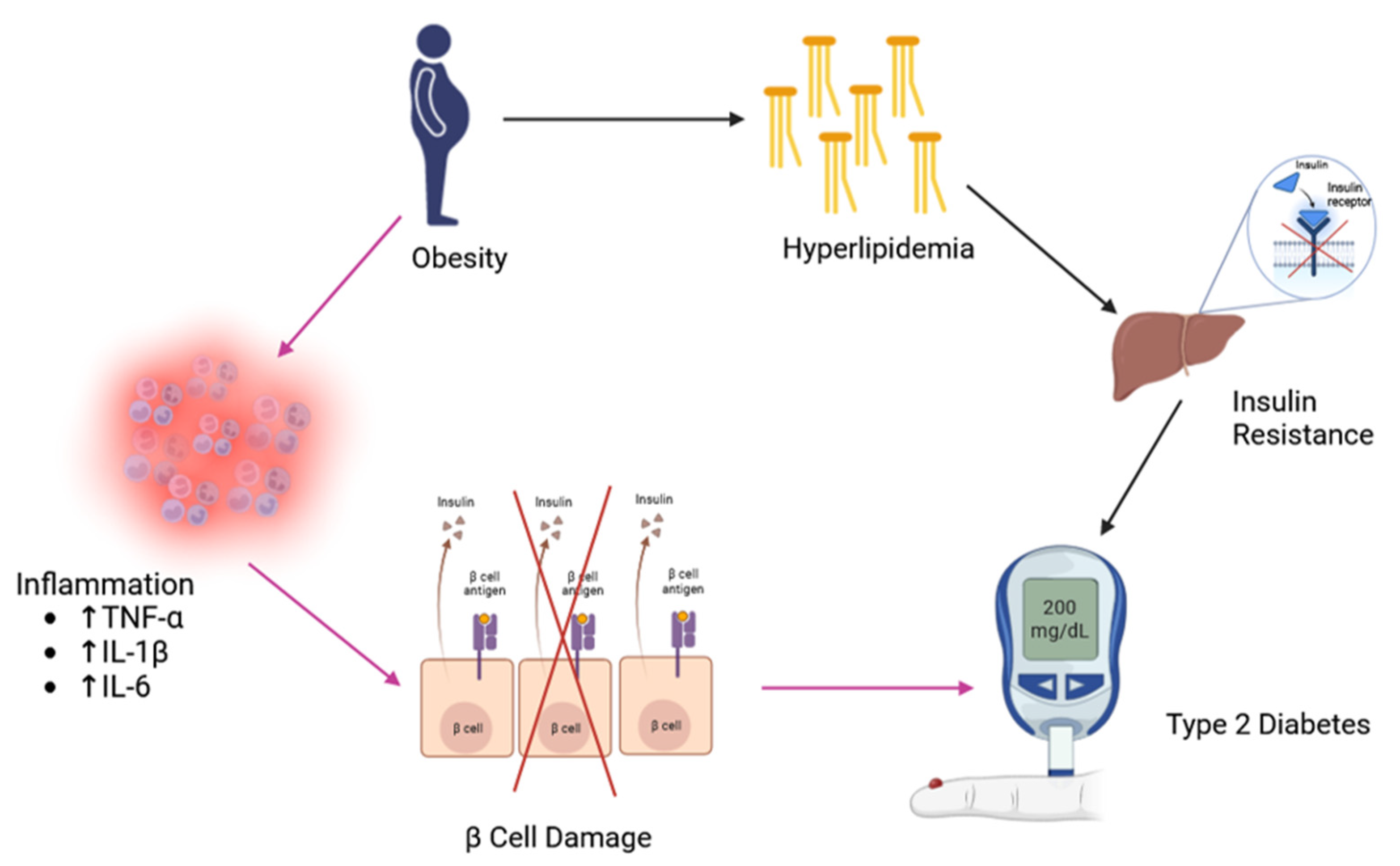
| Country | Name | Description | Image | Ingredients |
|---|---|---|---|---|
| Japan | Steamed Adzuki Snack-Sweet Red Bean | Steamed adzuki bean |  | Organic adzuki beans, organic sugar, and water |
| Taiwan | Taitan Adzuki Bean with Black Glutinous Rice and Quinoa Instant Cereals | Adzuki bean and black glutinous rice mix |  | Adzuki bean, milk powder, black glutinous rice, quinoa, and other ingredients |
| Taiwan | Formosa Yay Imperial Mochi (Red Bean Filling) | Mochi with adzuki bean paste |  | Rice and adzuki bean paste |
| Japan | Pure Azuki Bean Tea | Adzuki bean tea bag |  | Adzuki bean |
| Japan | Tenzan Honpo Yokan Tsubuan | Japanese adzuki bean pastry |  | Adzuki bean |
| Japan | Ogura Komachi | Bread with adzuki bean jam added |  | No details provided |
| Japan | Tohato All Azuki (Red Bean) and Matcha (Green Tea) Snack | Pastry with adzuki bean and matcha added |  | No details provided |
| Japan | Morokoshi | Molded cake made from adzuki bean flour |  | Caster sugar, adzuki bean powder, and white bean paste |
| Japan | Oshiruko | Freeze-dried sweets adzuki (red bean) soup |  | No details provided |
| Japan | Dorayaki | Japanese pancake with adzuki bean paste filling |  | Sugar, egg, wheat, trehalose, adzuki beans, water, sorbitol, reduced sugar syrup, rice flour, glutinous rice flour, modified food starch, vegetable oil, baking powder, and honey |
| Japan | UHA High Concentrated Milk 8.2 Azuki Milk | Cow milk with adzuki bean flavor added |  | No details provided |
| Japan | Yamasan Kyoto Uji Japanese Mochi Candy | Candy with adzuki bean flavor added |  | No details provided |
| Treatment | Outcomes Related to Diabetes or Glucose Metabolism/Pathways | Reference | |||
|---|---|---|---|---|---|
| Dose | Duration | Glucose-related | Insulin-related | Liver/pancreatic function-related and potential mechanism of action | |
| Animal studies | |||||
| 150 g adzuki bean flour/kg diet/day | 12 weeks |
|
|
| (Zhao, Hou, Fu, et al., 2021) [36] |
| 200 mg adzuki bean extract/kg BW/day | 12 weeks |
|
|
| (Lee et al., 2022) [38] |
| 100 mg ABP/kg BW/day 200 mg ABP/kg BW/day 400 mg ABP/kg body BW/day | 4 weeks | Medium and high doses of ABP:
|
|
| (Wu et al., 2020) [37] |
| 1 g BAB extract and 0 g kaempferol/100 g diet/day 0 g BAB extract and 0.08 g kaempferol/100 g diet/day | 12 weeks |
|
|
| (Kim et al., 2016) [39] |
| 400 mg ABP/kg BW/day | 4 weeks | 3- and 4-week supplementation:
|
|
| (Wu et al., 2019) [40] |
| 300 g raw adzuki bean flour/kg BW/day 300 g cooked adzuki bean flour/kg BW/day | 8 weeks | 300 g/kg raw adzuki bean flour:
|
| (Zhao, Hou, Laraib, et al., 2021) [10] | |
| 500 mg of cellulose/kg BW/day 5000 mg of cellulose/kg BW/day 500 mg of EtEx. 40/kg BW/day 5000 mg of EtEx.40/kg BW/day | 7 weeks 4 weeks 1 week | 500 mg EtEx. 40 only:
| 5000 mg EtEx. 40 vs. 5000 mg cellulose:
|
| (Itoh et al., 2009) [41] |
| 100 mg EtEx.40/kg BW/day 500 mg EtEx.40/kg BW/day | 120 min |
|
|
| (Itoh et al., 2004) [42] |
| 200 mg ABE/kg BW/day 200 mg EABE/kg BW/day | 120 min |
|
| (Yao, Ren, et al., 2014) [43] | |
| 1% of EA 2% of EA | 6 weeks |
|
| (Yao et al., 2014) [44] | |
| 6 g AP/100 g diet/day | 12 weeks |
|
|
| (Zhao et al., 2022) [45] |
| 15 g GABA/100 g diet/day 25 g GABA/100 g diet/day 35 g GABA/100 g diet/day 35 g Adzuki bean/100 g diet/day 0.1 g GABA/kg diet/day 0.1 g Metformin/kg diet/day | 6 weeks |
|
|
| (Jiang et al., 2021) [46] |
| (Zhang et al., 2022) [47] | ||||
| Human study | |||||
| 44.8 g ABE/day | 4 weeks |
|
|
| (Liu et al., 2018) [29] |
| In vitro studies | |||||
| 0.025 mg/mL BAB ethanolic extract 0.05 mg/mL BAB ethanolic extract 0.1 mg/mL BAB ethanolic extract 0.2 mg/mL BAB ethanolic extract | 96 h |
|
| (Kim et al., 2016) [39] | |
| 0.25 mg/mL ABTE, ABF, or ABS 0.50 mg/mL ABTE, ABF, or ABS 0.75 mg/mL ABTE, ABF, or ABS 1 mg/mL ABTE, ABF, or ABS | N/A |
|
| (Liu et al., 2017) [48] | |
| Treatment | Outcomes Related to Obesity and Dyslipidemia | Reference | ||
|---|---|---|---|---|
| Dose | Duration | Outcomes related to obesity | Outcomes related to dyslipidemia | |
| Animal studies | ||||
| 150 g adzuki bean flour/kg diet/day | 12 weeks |
|
| (Zhao, Hou, Fu, et al., 2021) [36] |
| 200 mg adzuki bean extract/kg BW/day | 12 weeks |
| (Lee et al., 2022) [38] | |
| 100 mg ABP/kg BW/day 200 mg ABP/kg BW/day 400 mg ABP/kg BW/day | 4 weeks |
| Low and middle doses of ABP:
| (Wu et al., 2020) [37] |
| 1 g BAB extract and 0 g kaempferol/100 g diet/day 0 g BAB extract and 0.08 g kaempferol/100 g diet/day | 12 weeks |
| (Kim et al., 2016) [39] | |
| 400 mg ABP/kg BW/day | 28 days |
| (Wu et al., 2019) [40] | |
| 300 g raw adzuki bean flour/kg BW/day 300 g cooked adzuki bean flour/kg BW/day | 8 weeks | Raw adzuki bean flour:
| Raw adzuki bean flour:
| (Zhao, Hou, Laraib, et al., 2021) [10] |
| 500 mg of cellulose/kg of BW/day 5000 mg of cellulose/kg of BW/day 500 mg of EtEx. 40/kg of BW/day 5000 mg of EtEx.40/kg of BW/day | 7 weeks 4 weeks 1 week | Weeks 1–7 (500 mg EtEx. 40 or 5000 mg EtEx. 40/day):
| Weeks 1, 4, and 7 (500 mg EtEx. 40 or 5000 mg EtEx. 40/day):
| (Itoh et al., 2009) [41] |
| 1% of EA 2% of EA | 6 weeks |
|
| (Yao et al., 2014) [44] |
| 1% HWE 5% HWE | 13 weeks |
| (Lee et al., 2019) [51] | |
| 1% HWE | 4 weeks |
|
| (Kitano-Okada et al., 2012) [31] |
| 1% BAB extract 2% BAB extract | 12 weeks |
| (Yook et al., 2017) [52] | |
| 150 g adzuki bean/kg diet/day | 12 weeks |
|
| (Zhao, Liu, et al., 2022) [32] |
| 100 mg ABE/kg BW/day 200 mg ABE/kg BW/day | 4 weeks | 200 mg ABE/kg/day:
| (Choi et al., 2020) [53] | |
| 1 g BAB extract/100 g diet/day 2 g BAB extract/100 g diet/day | 8 weeks | 1 g BAB extract/100 g/day:
| 1 g BAB extract/100 g/day:
| (Kim, Song, et al., 2015) [54] |
| 6 g AP/100 g diet/day | 12 weeks |
|
| (Zhao, Fu, et al., 2022) [45] |
| 60 mg/kg BW/day of ABTE, ABF, or ABS 300 mg/kg BW/day of ABTE, ABF, or ABS | 4 weeks |
|
| (Liu et al., 2017) [48] |
| 15 g GABA/100 g diet/day 25 g GABA/100 g diet/day 35 g GABA/100 g diet/day 35 g adzuki bean/100 g diet/day 0.1 g GABA/kg diet/day 0.1 g Metformin/kg diet/day | 6 weeks |
|
| (Jiang et al., 2021) [46] |
| (Zhang et al., 2022) [47] | ||||
| 1% freeze-dried BAB powder 0.08% kaempferol | 12 weeks |
|
| (Kim et al., 2017) [55] |
| Human study | ||||
| 44.8 g ABE/day | 4 weeks |
| (Liu et al., 2018) [29] | |
| In vitro study | ||||
| 250 µg/mL adzuki bean polyphenols 500 µg/mL adzuki bean polyphenols 750 µg/mL adzuki bean polyphenols | 48 h |
|
| (Kitano-Okada et al., 2012) [31] |
| 0.5 mg/mL BAB ethanolic extract 1 mg/mL BAB ethanolic extract 1.6 mg/mL BAB ethanolic extract 2 mg/mL BAB ethanolic extract 2.5 mg/mL BAB ethanolic extract 4 mg/mL BAB ethanolic extract | 72 h |
|
| (Kim, Park, et al., 2015) [56] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwan, S.H.; Gonzalez de Mejia, E. The Potential of the Adzuki Bean (Vigna angularis) and Its Bioactive Compounds in Managing Type 2 Diabetes and Glucose Metabolism: A Narrative Review. Nutrients 2024, 16, 329. https://doi.org/10.3390/nu16020329
Kwan SH, Gonzalez de Mejia E. The Potential of the Adzuki Bean (Vigna angularis) and Its Bioactive Compounds in Managing Type 2 Diabetes and Glucose Metabolism: A Narrative Review. Nutrients. 2024; 16(2):329. https://doi.org/10.3390/nu16020329
Chicago/Turabian StyleKwan, Shu Hang, and Elvira Gonzalez de Mejia. 2024. "The Potential of the Adzuki Bean (Vigna angularis) and Its Bioactive Compounds in Managing Type 2 Diabetes and Glucose Metabolism: A Narrative Review" Nutrients 16, no. 2: 329. https://doi.org/10.3390/nu16020329
APA StyleKwan, S. H., & Gonzalez de Mejia, E. (2024). The Potential of the Adzuki Bean (Vigna angularis) and Its Bioactive Compounds in Managing Type 2 Diabetes and Glucose Metabolism: A Narrative Review. Nutrients, 16(2), 329. https://doi.org/10.3390/nu16020329







