Vitamin D Deficiency Exacerbates Poor Sleep Outcomes with Endocrine-Disrupting Chemicals Exposure: A Large American Population Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participant
2.2. Exposure Information
2.3. Sleep Time Survey
2.4. Serum 1,25(OH)2D3 Analyses
2.5. Assessment of Covariates
2.6. Statistical Analysis
2.6.1. Elastic Net Model and Generalized Linear Model
2.6.2. WQS Regression Model and Quantile g-Computation Model
2.6.3. Bayesian Kernel Machine Regression
3. Results
3.1. Population Characteristics
3.2. Distribution, Correlation, and Selection of Environmental Endocrine Disruptors in Urine
3.3. Correlation between Urinary EDC Metabolites and Sleep Duration
3.4. Correlation between Urinary EDC Metabolites and Vitamin D Deficiency
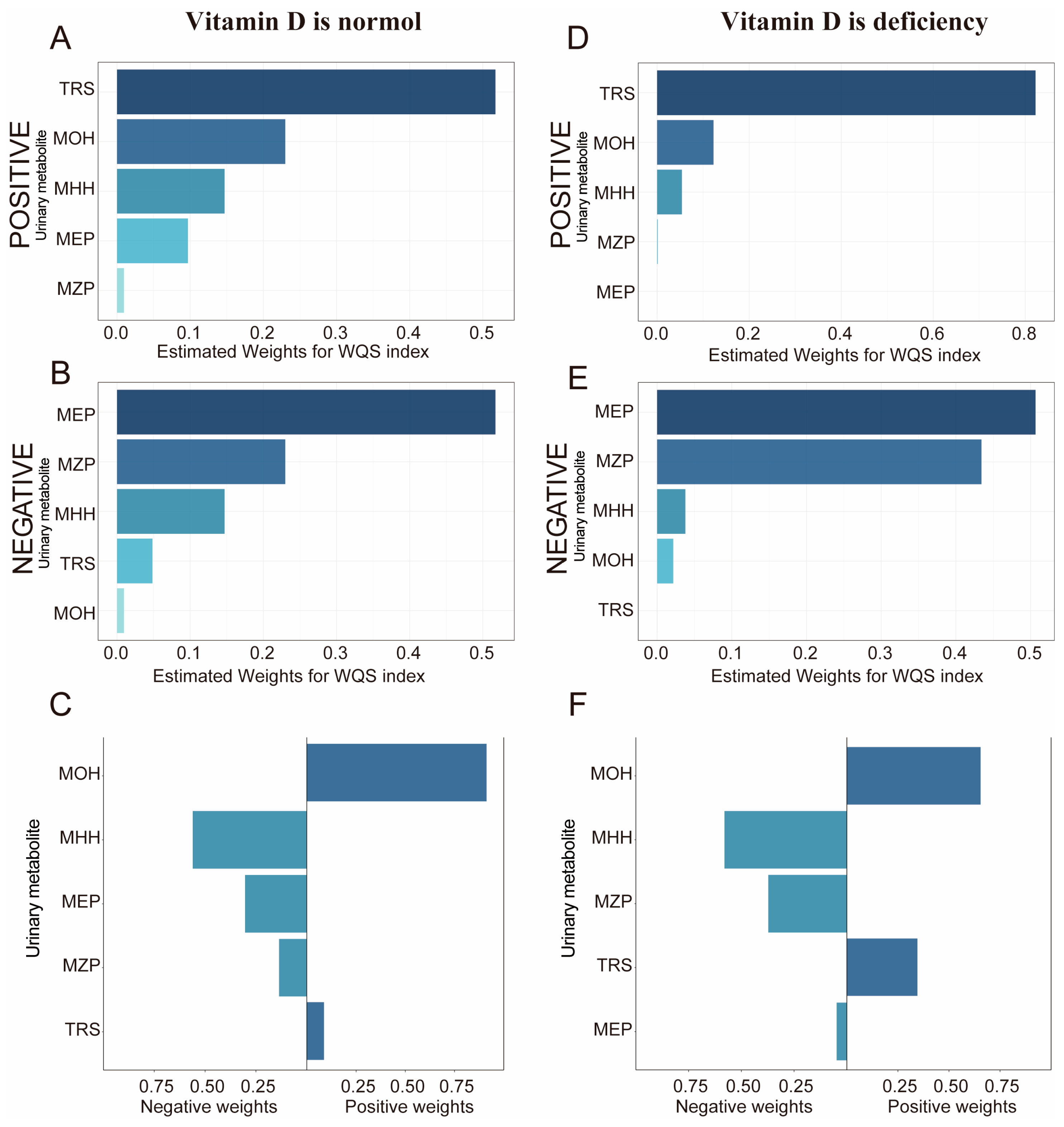
3.5. Stratified Analysis by Vitamin D Level
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yilmaz, B.; Terekeci, H.; Sandal, S.; Kelestimur, F. Endocrine disrupting chemicals: Exposure, effects on human health, mechanism of action, models for testing and strategies for prevention. Rev. Endocr. Metab. Disord. 2020, 21, 127–147. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M. Early-life exposure to EDCs: Role in childhood obesity and neurodevelopment. Nat. Reviews. Endocrinol. 2017, 13, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Woods, N.F.; Mitchell, E.S. Sleep symptoms during the menopausal transition and early postmenopause: Observations from the Seattle Midlife Women’s Health Study. Sleep 2010, 33, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, Y.; Tao, Y.; Guo, X.; Cui, Y.; Li, Z. Phthalates (PAEs) and reproductive toxicity: Hypothalamic-pituitary-gonadal (HPG) axis aspects. J. Hazard. Mater. 2023, 459, 132182. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Chen, S.; Zeng, C.; Fan, Y.; Ge, W.; Chen, W. Estrogenic and non-estrogenic effects of bisphenol A and its action mechanism in the zebrafish model: An overview of the past two decades of work. Environ. Int. 2023, 176, 107976. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, H.; Nielsen, O.; Koch, H.M.; Skakkebaek, N.E.; Juul, A.; Jørgensen, N.; Andersson, A.M. Changes in urinary excretion of phthalates, phthalate substitutes, bisphenols and other polychlorinated and phenolic substances in young Danish men; 2009–2017. Int. J. Hyg. Environ. Health 2020, 223, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Bi, M.; Liu, W.; Luan, X.; Li, M.; Liu, M.; Liu, W.; Cui, Z. Production, Use, and Fate of Phthalic Acid Esters for Polyvinyl Chloride Products in China. Environ. Sci. Technol. 2021, 55, 13980–13989. [Google Scholar] [CrossRef] [PubMed]
- Kahn, L.G.; Philippat, C.; Nakayama, S.F.; Slama, R.; Trasande, L. Endocrine-disrupting chemicals: Implications for human health. Lancet. Diabetes Endocrinol. 2020, 8, 703–718. [Google Scholar] [CrossRef]
- Yang, T.C.; Jovanovic, N.; Chong, F.; Worcester, M.; Sakhi, A.K.; Thomsen, C.; Garlantézec, R.; Chevrier, C.; Jensen, G.; Cingotti, N.; et al. Interventions to Reduce Exposure to Synthetic Phenols and Phthalates from Dietary Intake and Personal Care Products: A Scoping Review. Curr. Environ. Health Rep. 2023, 10, 184–214. [Google Scholar] [CrossRef] [PubMed]
- Kabel, A.M.; Al Thumali, A.M.; Aldowiala, K.A.; Habib, R.D.; Aljuaid, S.S.; Alharthi, H.A. Sleep disorders in adolescents and young adults: Insights into types, relationship to obesity and high altitude and possible lines of management. Diabetes Metab. Syndr. 2018, 12, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Bin Heyat, M.B.; Akhtar, F.; Ansari, M.A.; Khan, A.; Alkahtani, F.; Khan, H.; Lai, D. Progress in Detection of Insomnia Sleep Disorder: A Comprehensive Review. Curr. Drug Targets 2021, 22, 672–684. [Google Scholar] [PubMed]
- Vgontzas, A.N.; Kales, A. Sleep and its disorders. Annu. Rev. Med. 1999, 50, 387–400. [Google Scholar] [CrossRef] [PubMed]
- Nesan, D.; Feighan, K.M.; Antle, M.C.; Kurrasch, D.M. Gestational low-dose BPA exposure impacts suprachiasmatic nucleus neurogenesis and circadian activity with transgenerational effects. Sci. Adv. 2021, 7, eabd1159. [Google Scholar] [CrossRef] [PubMed]
- Kinch, C.D.; Ibhazehiebo, K.; Jeong, J.H.; Habibi, H.R.; Kurrasch, D.M. Low-dose exposure to bisphenol A and replacement bisphenol S induces precocious hypothalamic neurogenesis in embryonic zebrafish. Proc. Natl. Acad. Sci. USA 2015, 112, 1475–1480. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.; Ferrini, M.G.; Han, G.; Jellyman, J.K.; Ross, M.G. In vivo maternal and in vitro BPA exposure effects on hypothalamic neurogenesis and appetite regulators. Environ. Res. 2018, 164, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.; Goodwin, B.; Shockley, K.; Xia, M.; Huang, R.; Norris, J.; Merrick, B.A.; Jetten, A.M.; Austin, C.P.; Tice, R.R. Bisphenol A affects androgen receptor function via multiple mechanisms. Chem. Biol. Interact. 2013, 203, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Zamora, A.N.; Peterson, K.E.; Téllez-Rojo, M.M.; Song, P.X.K.; Meeker, J.D.; Cantoral, A.; Goodrich, J.M.; Dolinoy, D.C.; Jansen, E.C. Urinary phthalates, phenols, and parabens in relation to sleep health markers among a cohort of Mexican adolescents. Sci. Total Environ. 2023, 861, 160651. [Google Scholar] [CrossRef] [PubMed]
- Zamora, A.N.; Peterson, K.E.; Goodrich, J.M.; Téllez-Rojo, M.M.; Song, P.X.K.; Meeker, J.D.; Dolinoy, D.C.; Torres-Olascoaga, L.; Cantoral, A.; Jansen, E.C. Associations between exposure to phthalates, phenols, and parabens with objective and subjective measures of sleep health among Mexican women in midlife: A cross-sectional and retrospective analysis. Environ. Sci. Pollut. Res. Int. 2023, 30, 65544–65557. [Google Scholar] [CrossRef] [PubMed]
- Norman, A.W. The history of the discovery of vitamin D and its daughter steroid hormone. Ann. Nutr. Metab. 2012, 61, 199–206. [Google Scholar] [CrossRef]
- Holick, M.F. McCollum Award Lecture, 1994: Vitamin D—New horizons for the 21st century. Am. J. Clin. Nutr. 1994, 60, 619–630. [Google Scholar] [CrossRef]
- McCarty, D.E.; Chesson, A.L., Jr.; Jain, S.K.; Marino, A.A. The link between vitamin D metabolism and sleep medicine. Sleep Med. Rev. 2014, 18, 311–319. [Google Scholar] [CrossRef]
- Huiberts, L.M.; Smolders, K. Effects of vitamin D on mood and sleep in the healthy population: Interpretations from the serotonergic pathway. Sleep Med. Rev. 2021, 55, 101379. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhou, T.; Li, X.; Ma, H.; Liang, Z.; Fonseca, V.A.; Heianza, Y.; Qi, L. Baseline Vitamin D Status, Sleep Patterns, and the Risk of Incident Type 2 Diabetes in Data From the UK Biobank Study. Diabetes Care 2020, 43, 2776–2784. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Wang, H.; Zhu, Z.; Ye, Q.; Lin, F.; Cai, G. Association between exposure to phenols and parabens and cognitive function in older adults in the United States: A cross-sectional study. Sci. Total Environ. 2023, 858 Pt 3, 160129. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Pan, C.; Su, W.; Vinturache, A.; Hu, Y.; Dong, X.; Ding, G. Associations between exposure to a mixture of phenols, parabens, and phthalates and sex steroid hormones in children 6–19 years from NHANES, 2013–2016. Sci. Total Environ. 2022, 822, 153548. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Kuehnle, T.; Juda, M.; Kantermann, T.; Allebrandt, K.; Gordijn, M.; Merrow, M. Epidemiology of the human circadian clock. Sleep Med. Rev. 2007, 11, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- AlQuaiz, A.M.; Kazi, A.; Youssef, R.M.; Alshehri, N.; Alduraywish, S.A. Association between standardized vitamin 25(OH)D and dyslipidemia: A community-based study in Riyadh, Saudi Arabia. Environ. Health Prev. Med. 2020, 25, 4. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Wu, B.; Hu, W.; Wang, X.; Su, W.; Meng, J.; Lowe, S.; Zhao, D.; Huang, C.; Liang, M.; et al. Associations of perchlorate, nitrate, and thiocyanate with metabolic syndrome and its components among US adults: A cross-sectional study from NHANES. Sci. Total Environ. 2023, 879, 163083. [Google Scholar] [CrossRef] [PubMed]
- Che, Z.; Jia, H.; Chen, R.; Pan, K.; Fan, Z.; Su, C.; Wu, Z.; Zhang, T. Associations between exposure to brominated flame retardants and metabolic syndrome and its components in U.S. adults. Sci. Total Environ. 2023, 858, 159935. [Google Scholar] [CrossRef]
- Palmer, M.K.; Toth, P.P. Trends in Lipids, Obesity, Metabolic Syndrome, and Diabetes Mellitus in the United States: An NHANES Analysis (2003–2004 to 2013–2014). Obesity 2019, 27, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Xue, Q.; Wen, Y.; Huang, Y.; Wang, Y.; Mahai, G.; Yan, T.; Liu, Y.; Rong, T.; Wang, Y.; et al. Environmental polycyclic aromatic hydrocarbon exposure in relation to metabolic syndrome in US adults. Sci. Total Environ. 2022, 840, 156673. [Google Scholar] [CrossRef]
- Zou, H.; Zhang, H.H. On the Adaptive Elastic-Net with a Diverging Number of Parameters. Ann. Stat. 2009, 37, 1733–1751. [Google Scholar] [CrossRef] [PubMed]
- King, L.; Wang, Q.; Xia, L.; Wang, P.; Jiang, G.; Li, W.; Huang, Y.; Liang, X.; Peng, X.; Li, Y.; et al. Environmental exposure to perchlorate, nitrate and thiocyanate, and thyroid function in Chinese adults: A community-based cross-sectional study. Environ. Int. 2023, 171, 107713. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Wang, Z.; Pan, Y.; Wang, H.; Sun, L.; Liu, L.; Zhong, W.J.; Zhang, W.B. Associations between mixed urinary phenols and parabens metabolites and bone mineral density: Four statistical models. Chemosphere 2022, 311, 137065. [Google Scholar] [CrossRef] [PubMed]
- Keil, A.P.; Buckley, J.P.; O’Brien, K.M.; Ferguson, K.K.; Zhao, S.; White, A.J. A Quantile-Based g-Computation Approach to Addressing the Effects of Exposure Mixtures. Environ. Health Perspect. 2020, 128, 47004. [Google Scholar] [CrossRef] [PubMed]
- Bobb, J.F.; Claus Henn, B.; Valeri, L.; Coull, B.A. Statistical software for analyzing the health effects of multiple concurrent exposures via Bayesian kernel machine regression. Environ. Health 2018, 17, 67. [Google Scholar] [CrossRef] [PubMed]
- Messerlian, C.; Martinez, R.M.; Hauser, R.; Baccarelli, A.A. ‘Omics’ and endocrine-disrupting chemicals—New paths forward. Nat. Reviews. Endocrinol. 2017, 13, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Darbre, P.D. Chemical components of plastics as endocrine disruptors: Overview and commentary. Birth Defects Res. 2020, 112, 1300–1307. [Google Scholar] [CrossRef] [PubMed]
- Nieves, J.W.; Gennings, C.; Factor-Litvak, P.; Hupf, J.; Singleton, J.; Sharf, V.; Oskarsson, B.; Fernandes Filho, J.A.; Sorenson, E.J.; D’Amico, E.; et al. Association Between Dietary Intake and Function in Amyotrophic Lateral Sclerosis. JAMA Neurol. 2016, 73, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Morsi, R.; Bilal, M.; Iqbal, H.M.N.; Ashraf, S.S. Laccases and peroxidases: The smart, greener and futuristic biocatalytic tools to mitigate recalcitrant emerging pollutants. Sci. Total Environ. 2020, 714, 136572. [Google Scholar] [CrossRef]
- Ghassabian, A.; Vandenberg, L.; Kannan, K.; Trasande, L. Endocrine-Disrupting Chemicals and Child Health. Annu. Rev. Pharmacol. Toxicol. 2022, 62, 573–594. [Google Scholar] [CrossRef]
- Sears, C.G.; Braun, J.M. Urinary phthalate metabolite concentrations and adolescent sleep duration. Environ. Epidemiol. 2021, 5, e134. [Google Scholar] [CrossRef]
- Zheng, J.; Reynolds, J.E.; Long, M.; Ostertag, C.; Pollock, T.; Hamilton, M.; Dunn, J.F.; Liu, J.; Martin, J.; Grohs, M.; et al. The effects of prenatal bisphenol A exposure on brain volume of children and young mice. Environ. Res. 2022, 214 Pt 3, 114040. [Google Scholar] [CrossRef]
- Zhang, S.; Feng, X. Effect of 17β-trenbolone exposure during adolescence on the circadian rhythm in male mice. Chemosphere 2022, 288 Pt 1, 132496. [Google Scholar] [CrossRef]
- Motta, G.; Thangaraj, S.V.; Padmanabhan, V. Developmental Programming: Impact of Prenatal Exposure to Bisphenol A on Senescence and Circadian Mediators in the Liver of Sheep. Toxics 2023, 12, 15. [Google Scholar] [CrossRef]
- Kim, J.K.; Khan, A.; Cho, S.; Na, J.; Lee, Y.; Bang, G.; Yu, W.J.; Jeong, J.S.; Jee, S.H.; Park, Y.H. Effect of developmental exposure to bisphenol A on steroid hormone and vitamin D3 metabolism. Chemosphere 2019, 237, 124469. [Google Scholar] [CrossRef]
- Johns, L.E.; Ferguson, K.K.; Cantonwine, D.E.; McElrath, T.F.; Mukherjee, B.; Meeker, J.D. Urinary BPA and Phthalate Metabolite Concentrations and Plasma Vitamin D Levels in Pregnant Women: A Repeated Measures Analysis. Environ. Health Perspect. 2017, 125, 087026. [Google Scholar] [CrossRef]
- Elkafas, H.; Ali, M.; Elmorsy, E.; Kamel, R.; Thompson, W.E.; Badary, O.; Al-Hendy, A.; Yang, Q. Vitamin D3 Ameliorates DNA Damage Caused by Developmental Exposure to Endocrine Disruptors in the Uterine Myometrial Stem Cells of Eker Rats. Cells 2020, 9, 1459. [Google Scholar] [CrossRef]
- Otsuka, H.; Sugimoto, M.; Ikeda, S.; Kume, S. Effects of bisphenol A administration to pregnant mice on serum Ca and intestinal Ca absorption. Anim. Sci. J. 2012, 83, 232–237. [Google Scholar] [CrossRef]
- Al-Griw, M.A.; Marwan, Z.M.; Hdud, I.M.; Shaibi, T. Vitamin D mitigates adult onset diseases in male and female mice induced by early-life exposure to endocrine disruptor BPA. Open Vet. J. 2021, 11, 407–417. [Google Scholar] [CrossRef]
- Gao, Q.; Kou, T.; Zhuang, B.; Ren, Y.; Dong, X.; Wang, Q. The Association between Vitamin D Deficiency and Sleep Disorders: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 1395. [Google Scholar] [CrossRef]
- McCarty, D.E. Resolution of hypersomnia following identification and treatment of vitamin d deficiency. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2010, 6, 605–608. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Barrea, L.; Scannapieco, M.; Di Somma, C.; Scacchi, M.; Aimaretti, G.; Savastano, S.; Colao, A.; Marzullo, P. The lullaby of the sun: The role of vitamin D in sleep disturbance. Sleep Med. 2019, 54, 262–265. [Google Scholar] [CrossRef]
- Wong, D.; Bellyou, M.; Li, A.; Prado, M.A.M.; Beauchet, O.; Annweiler, C.; Montero-Odasso, M.; Bartha, R. Magnetic resonance spectroscopy in the hippocampus of adult APP/PS1 mice following chronic vitamin D deficiency. Behav. Brain Res. 2024, 457, 114713. [Google Scholar] [CrossRef]
- Jung, J.; Kang, J.; Kim, T. Attenuation of homeostatic sleep response and rest-activity circadian rhythm in vitamin D deficient mice. Chronobiol. Int. 2023, 40, 1097–1110. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; Miller, M.A. Sleep and Cardio-Metabolic Disease. Curr. Cardiol. Rep. 2017, 19, 110. [Google Scholar] [CrossRef]
- Plante, D.T. The Evolving Nexus of Sleep and Depression. Am. J. Psychiatry 2021, 178, 896–902. [Google Scholar] [CrossRef]
- Bubu, O.M.; Brannick, M.; Mortimer, J.; Umasabor-Bubu, O.; Sebastião, Y.V.; Wen, Y.; Schwartz, S.; Borenstein, A.R.; Wu, Y.; Morgan, D.; et al. Sleep, Cognitive impairment, and Alzheimer’s disease: A Systematic Review and Meta-Analysis. Sleep 2017, 40, zsw032. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Monti, J.M.; Burman, D.; Karthikeyan, R.; BaHammam, A.S.; Spence, D.W.; Brown, G.M.; Narashimhan, M. Clarifying the role of sleep in depression: A narrative review. Psychiatry Res. 2020, 291, 113239. [Google Scholar] [CrossRef]
- Nie, L.; Chu, H.; Liu, C.; Cole, S.R.; Vexler, A.; Schisterman, E.F. Linear regression with an independent variable subject to a detection limit. Epidemiology 2010, 21 (Suppl. S4), S17–S24. [Google Scholar] [CrossRef] [PubMed]






| Characteristics | Total |
|---|---|
| Gender n(%) | |
| Male | 2709 (48.8) |
| Female | 2767 (51.2) |
| Age n (%) | |
| 20–39 years | 1856 (36.1) |
| 40–59 years | 1919 (40.2) |
| 60–80 years | 1701 (23.7) |
| Race n (%) | |
| Mexican American | 767 (7.8) |
| Other Hispanic | 536 (5.5) |
| Non-Hispanic White | 2560 (70.4) |
| Non-Hispanic Black | 1095 (10.2) |
| Other Race—Including Multi-Racial | 518 (6.1) |
| Educational level n (%) | |
| Less Than 9th Grade | 510 (5.2) |
| 9–11th Grade (Includes 12th grade with no diploma) | 808 (11.2) |
| High School Grad/GED or Equivalent | 1276 (22.6) |
| Some College or AA degree | 1574 (30.8) |
| College Graduate or above | 1308 (30.3) |
| Marital Status n (%) | |
| Married | 2882 (57.1) |
| Widowed | 432 (5.6) |
| Divorced | 597 (10.2) |
| Separated | 164 (2.0) |
| Never married | 995 (18.0) |
| Living with partner | 406 (7.1) |
| Ratio of family income to poverty n (%) | |
| <1.3 | 1751 (21.3) |
| 1.3–3.5 | 1966 (34.6) |
| >3.5 | 1759 (44.1) |
| Smoke n (%) | |
| Yes | 2473 (44.3) |
| No | 3003 (55.7) |
| Alcohol drinking n (%) | |
| Yes | 4042 (78.7) |
| No | 1434 (21.3) |
| Recreational activities n(%) | |
| Yes | 2636 (54.6) |
| No | 2840 (45.4) |
| Body mass index (BMI) n (%) | |
| <18.5 kg/m2 | 82 (1.4) |
| 18.5 to <25 kg/m2 | 1509 (28.8) |
| 25 to <30 kg/m2 | 1829 (33.4) |
| ≥30 kg/m2 | 2056 (36.4) |
| Sleep duration (mean ± SD) (h) | 6.87 ± 1.34 |
| Is vitamin D deficient? n (%) | |
| Yes | 5006 (96.0) |
| No | 470 (4.0) |
| Chemicals. | Model 1 | Model 2 | ||
|---|---|---|---|---|
| β (95%CI) | p | β (95%CI) | p | |
| Single substance | ||||
| CNP | −0.004 (−0.046, 0.033) | 0.760 | −0.014 (−0.061, 0.018) | 0.288 |
| ECP | 0.003 (−0.033, 0.043) | 0.808 | −0.012 (−0.057, 0.021) | 0.366 |
| MBP | −0.012 (−0.056, 0.022) | 0.389 | −0.024 (−0.075, 0.005) | 0.087 |
| MC1 | −0.009 (−0.046, 0.024) | 0.528 | −0.016 (−0.056, 0.013) | 0.230 |
| MEP | −0.039 (−0.063, −0.012) | 0.004 | −0.050 (−0.073, −0.022) | <0.001 |
| MHH | −0.016 (−0.057, 0.015) | 0.248 | −0.026 (−0.071, 0.001) | 0.055 |
| MHP | −0.009 (−0.048, 0.024) | 0.509 | −0.020 (−0.063, 0.008) | 0.131 |
| MOH | −0.002 (−0.041, 0.035) | 0.891 | −0.013 (−0.056, 0.020) | 0.350 |
| MZP | −0.038 (−0.089, −0.016) | 0.005 | −0.034 (−0.085, −0.010) | 0.013 |
| BP3 | −0.003 (−0.021, 0.017) | 0.821 | 0.028 (0.001, 0.035) | 0.040 |
| BPH | −0.031 (−0.091, −0.008) | 0.020 | −0.040 (−0.104, −0.020) | 0.004 |
| TRS | 0.046 (0.015, 0.054) | 0.001 | 0.027 (0.000, 0.040) | 0.047 |
| MPB | −0.009 (−0.029, 0.015) | 0.523 | −0.041 (−0.058, −0.010) | 0.005 |
| PPB | 0.001 (−0.016, 0.017) | 0.963 | −0.030 (−0.037, 0.000) | 0.045 |
| Multi-material | ||||
| CNP | −0.013 (−0.066, 0.028) | 0.424 | −0.014 (−0.068, 0.025) | 0.371 |
| ECP | −0.023 (−0.071, 0.006) | 0.819 | 0.031 (−0.052, 0.140) | 0.369 |
| MBP | −0.009 (−0.058, 0.031) | 0.548 | −0.016 (−0.069, 0.021) | 0.291 |
| MC1 | −0.008 (−0.052, 0.032) | 0.637 | −0.007 (−0.052, 0.032) | 0.645 |
| MEP | −0.035 (−0.060, −0.007) | 0.015 | −0.037 (−0.063, −0.009) | 0.009 |
| MHH | −0.237 (−0.465, −0.175) | <0.001 | −0.222 (−0.446, −0.156) | <0.001 |
| MHP | 0.005 (−0.031, 0.044) | 0.728 | −0.003 (−0.042, 0.033) | 0.819 |
| MOH | 0.177 (0.095, 0.412) | 0.002 | 0.186 (0.108, 0.424) | 0.001 |
| MZP | −0.059 (−0.181, −0.001) | 0.039 | −0.030 (−0.079, −0.003) | 0.033 |
| BP3 | −0.003 (−0.021, 0.017) | 0.844 | 0.022 (−0.003, 0.032) | 0.107 |
| BPH | −0.040 (−0.071, 0.006) | 0.049 | −0.040 (−0.071, −0.006) | 0.049 |
| TRS | 0.043 (0.012, 0.052) | 0.002 | 0.029 (0.001, 0.041) | 0.038 |
| MPB | −0.021 (−0.056, 0.022) | 0.382 | −0.023 (−0.079, 0.008) | 0.113 |
| PPB | 0.030 (−0.011, 0.048) | 0.216 | 0.013 (−0.022, 0.038) | 0.586 |
| Chemicals | Model 1 | Model 2 | ||
|---|---|---|---|---|
| OR (95%CI) | p | OR (95%CI) | p | |
| Single substance | ||||
| BP3 | 0.833 (0.793, 0.874) | <0.001 | 0.859 (0.813, 0.906) | <0.001 |
| TRS | 0.909 (0.863, 0.957) | <0.001 | 0.954 (0.904, 0.997) | 0.049 |
| MPB | 1.007 (0.954, 1.064) | 0.799 | 1.023 (0.962, 1.087) | 0.464 |
| PPB | 0.990 (0.950, 1.032) | 0.647 | 0.991 (0.945, 1.040) | 0.716 |
| CNP | 0.805 (0.726, 0.892) | <0.001 | 0.833 (0.751, 0.925) | 0.001 |
| ECP | 1.094 (0.995, 1.203) | 0.062 | 0.959 (0.867, 1.060) | 0.412 |
| MBP | 1.044 (0.946, 1.151) | 0.391 | 1.043 (0.940, 1.157) | 0.428 |
| MC1 | 0.844 (0.770, 0.925) | <0.001 | 0.864 (0.787, 0.949) | 0.002 |
| MEP | 1.051 (0.988, 1.119) | 0.116 | 1.069 (1.002, 1.141) | 0.044 |
| MHH | 0.964 (0.881, 1.055) | 0.425 | 0.996 (0.908, 1.093) | 0.936 |
| MHP | 0.991 (0.906, 1.083) | 0.840 | 1.016 (0.928, 1.113) | 0.724 |
| MOH | 0.938 (0.852, 1.033) | 0.196 | 0.964 (0.873, 1.066) | 0.477 |
| MZP | 1.132 (1.032, 1.240) | 0.008 | 1.051 (0.954, 1.158) | 0.314 |
| Multi-material | ||||
| BP3 | 0.834 (0.792, 0.878) | <0.001 | 0.857 (0.810, 0.906) | <0.001 |
| TRS | 1.101 (1.000, 1.213) | 0.042 | 0.931 (0.882, 0.982) | 0.009 |
| MPB | 0.912 (0.811, 1.025) | 0.121 | 0.957 (0.906, 1.012) | 0.122 |
| PPB | 0.975 (0.908, 1.047) | 0.488 | 0.940 (0.872, 1.014) | 0.112 |
| CNP | 0.902 (0.798, 1.020) | 0.101 | 0.885 (0.780, 0.994) | 0.047 |
| ECP | 0.890 (0.704, 1.125) | 0.330 | 1.006 (0.772, 1.311) | 0.965 |
| MBP | 1.019 (0.897, 1.158) | 0.769 | 1.039 (0.912, 1.184) | 0.568 |
| MC1 | 0.897 (0.800, 0.995) | 0.032 | 0.910 (0.825, 0.992) | 0.046 |
| MEP | 1.139 (1.008, 1.301) | 0.044 | 1.080 (1.007, 1.158) | 0.031 |
| MHH | 1.298 (0.891, 1.892) | 0.175 | 1.453 (1.275, 2.166) | 0.046 |
| MHP | 1.078 (0.964, 1.207) | 0.188 | 1.121 (0.976, 1.288) | 0.107 |
| MOH | 0.735 (0.493, 1.096) | 0.130 | 0.610 (0.399, 0.933) | 0.023 |
| MZP | 1.159 (1.042, 1.290) | 0.007 | 1.059 (1.009, 1.133) | 0.043 |
| Chemicals | Model 1 | Model 2 | ||
|---|---|---|---|---|
| β (95%CI) | p | β (95%CI) | p | |
| Vitamin D is normal | ||||
| TRS | 0.038 (0.007,0.048) | 0.008 | 0.023 (−0.004, 0.038) | 0.112 |
| MEP | −0.042 (−0.067, −0.013) | 0.004 | −0.050 (−0.075, −0.021) | 0.001 |
| MHH | −0.200 (−0.415, −0.119) | <0.001 | −0.193 (−0.406, −0.109) | 0.001 |
| MOH | 0.205 (0.133, 0.447) | <0.001 | 0.187 (0.106, 0.422) | 0.001 |
| MZP | −0.021 (−0.069, 0.012) | 0.173 | −0.015 (−0.062, 0.021) | 0.332 |
| Vitamin D deficiency | ||||
| TRS | 0.135 (0.037, 0.187) | 0.004 | 0.128 (0.029, 0.183) | 0.007 |
| MEP | −0.012 (−0.106, 0.081) | 0.797 | −0.018 (−0.113, 0.077) | 0.709 |
| MHH | −0.246 (−0.809, 0.067) | 0.097 | −0.266 (−0.842, 0.040) | 0.074 |
| MOH | 0.242 (−0.080, 0.856) | 0.104 | 0.265 (−0.050, 0.898) | 0.079 |
| MZP | −0.145 (−0.345, −0.074) | 0.003 | −0.143 (−0.349, −0.066) | 0.004 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, R.; Chen, Z.; Yang, T.; Gu, H.; Yang, X.; Cheng, S. Vitamin D Deficiency Exacerbates Poor Sleep Outcomes with Endocrine-Disrupting Chemicals Exposure: A Large American Population Study. Nutrients 2024, 16, 1291. https://doi.org/10.3390/nu16091291
Zhou R, Chen Z, Yang T, Gu H, Yang X, Cheng S. Vitamin D Deficiency Exacerbates Poor Sleep Outcomes with Endocrine-Disrupting Chemicals Exposure: A Large American Population Study. Nutrients. 2024; 16(9):1291. https://doi.org/10.3390/nu16091291
Chicago/Turabian StyleZhou, Ruiqi, Zhongwen Chen, Tingting Yang, Huiwen Gu, Xiaohong Yang, and Shuqun Cheng. 2024. "Vitamin D Deficiency Exacerbates Poor Sleep Outcomes with Endocrine-Disrupting Chemicals Exposure: A Large American Population Study" Nutrients 16, no. 9: 1291. https://doi.org/10.3390/nu16091291
APA StyleZhou, R., Chen, Z., Yang, T., Gu, H., Yang, X., & Cheng, S. (2024). Vitamin D Deficiency Exacerbates Poor Sleep Outcomes with Endocrine-Disrupting Chemicals Exposure: A Large American Population Study. Nutrients, 16(9), 1291. https://doi.org/10.3390/nu16091291






