The Role of Dairy in Human Nutrition: Myths and Realities
Abstract
1. Introduction
2. Nutritional Composition of Milk and Dairy Products
2.1. Milk Proteins
2.2. Milk Fat
2.3. Milk Carbohydrates
2.4. Mineral Milk Fraction
2.5. Vitamin Content in Milk
3. Positive Effects of Dairy Consumption
3.1. Calcium Source
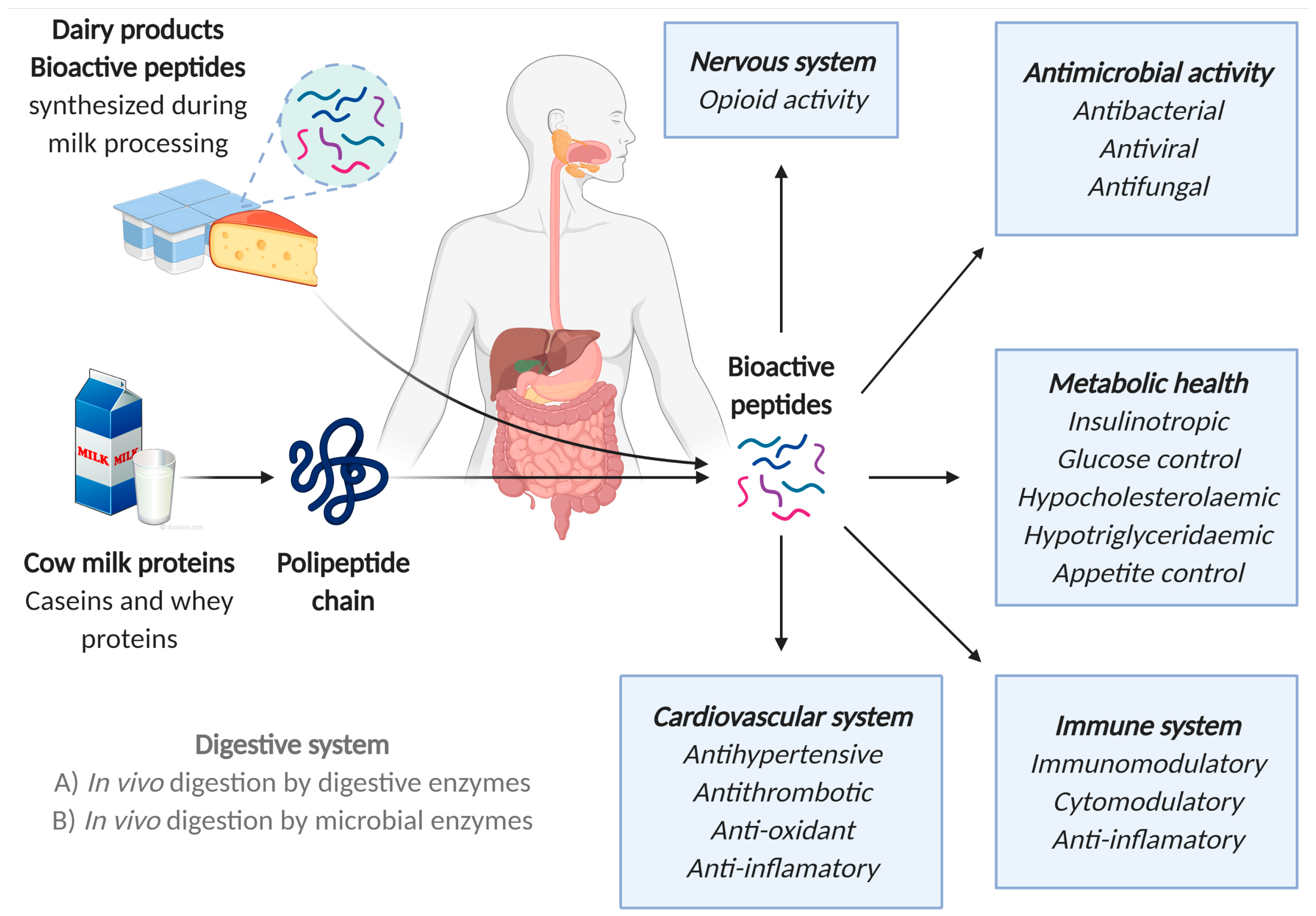
3.2. Source of Bioactive Peptides
3.3. Source of Prebiotics and Probiotics
3.4. CLA Source
3.5. Osteoporosis Prevention
3.6. Preventive Factors in CRC and Other Cancers
4. Negative Effects of Dairy Consumption
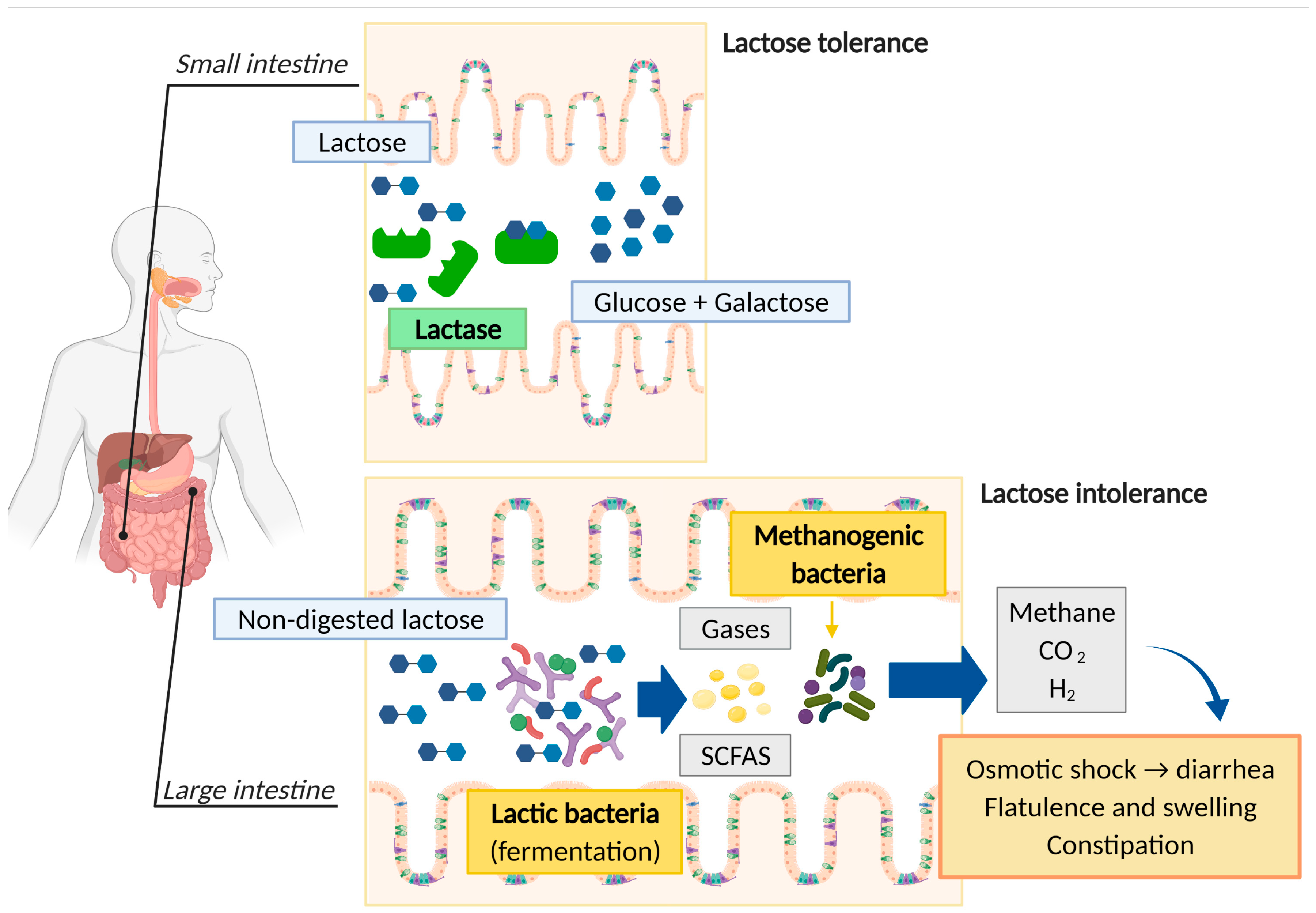
4.1. Lactose Intolerance
4.2. Cow Milk Allergy
5. Inconclusive Evidence on Dairy Consumption
5.1. Protection in Type II Diabetes Mellitus
5.2. Contributing to Weight Gain
5.3. Impaired Lipid Profile and Increased Risk of Cardiovascular Disease (CVD)
5.4. Risk Factors for Prostate Cancer Development
5.5. Autism Spectrum Disorder (ASD)
5.6. Source of miRNAs
6. Milk vs. Plant-Based Drinks: The Role of the Milk Fat Globule Membrane
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- CODEX STAN 206-1999; Food and Agriculture Administration Codex General Standard for the Use of Dairy Terms. FAO: Rome, Italy, 1999.
- González-Montaña, J.R.; Senís, E.; Gutiérrez, A.; Prieto, F. Cadmium and Lead in Bovine Milk in the Mining Area of the Caudal River (Spain). Environ. Monit. Assess. 2012, 184, 4029–4034. [Google Scholar] [CrossRef] [PubMed]
- Autio, M.; Sekki, S.; Autio, J.; Peltonen, K.; Niva, M. Toward De-Dairyfication of the Diet?—Consumers Downshifting Milk, yet Justifying Their Dairy Pleasures. Front. Sustain. 2023, 4, 975679. [Google Scholar] [CrossRef]
- Zingone, F.; Bucci, C.; Iovino, P.; Ciacci, C. Consumption of Milk and Dairy Products: Facts and Figures. Nutrition 2017, 33, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.C. Milk Nutritional Composition and Its Role in Human Health. Nutrition 2014, 30, 619–627. [Google Scholar] [CrossRef]
- Walsh, J.; Meyer, R.; Shah, N.; Quekett, J.; Fox, A.T. Differentiating Milk Allergy (IgE and Non-IgE Mediated) from Lactose Intolerance: Understanding the Underlying Mechanisms and Presentations. Br. J. Gen. Pract. 2016, 66, e609–e611. [Google Scholar] [CrossRef]
- Moreno Aznar, L.A.; Cervera Ral, P.; Ortega Anta, R.M.; Díaz Martín, J.J.; Baladia, E.; Basulto, J.; Bel Serrat, S.; Iglesia Altaba, I.; López-Sobaler, A.M.; Manera, M.; et al. [Scientific Evidence about the Role of Yogurt and Other Fermented Milks in the Healthy Diet for the Spanish Population]. Nutr. Hosp. 2013, 28, 2039–2089. [Google Scholar] [CrossRef]
- Mills, S.; Ross, R.P.; Hill, C.; Fitzgerald, G.F.; Stanton, C. Milk Intelligence: Mining Milk for Bioactive Substances Associated with Human Health. Int. Dairy J. 2011, 21, 377–401. [Google Scholar] [CrossRef]
- Lambrini, K.; Aikaterini, F.; Konstantinos, K.; Christos, I.; Ioanna, P.V.; Areti, T. Milk Nutritional Composition and Its Role in Human Health. J. Pharm. Pharmacol. 2020, 9, 8–13. [Google Scholar] [CrossRef]
- Maity, S.; Bhat, A.H.; Giri, K.; Ambatipudi, K. BoMiProt: A Database of Bovine Milk Proteins. J. Proteom. 2020, 215, 103648. [Google Scholar] [CrossRef]
- Borad, S.G.; Kumar, A.; Singh, A.K. Effect of Processing on Nutritive Values of Milk Protein. Crit. Rev. Food Sci. Nutr. 2017, 57, 3690–3702. [Google Scholar] [CrossRef]
- Visioli, F.; Strata, A. Milk, Dairy Products, and Their Functional Effects in Humans: A Narrative Review of Recent Evidence. Adv. Nutr. 2014, 5, 131–143. [Google Scholar] [CrossRef] [PubMed]
- German, J.B.; Dillard, C.J. Composition, Structure and Absorption of Milk Lipids: A Source of Energy, Fat-Soluble Nutrients and Bioactive Molecules. Crit. Rev. Food Sci. Nutr. 2006, 46, 57–92. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Pappenheim, S.; Yener, S.; Goselink, R.; Quintanilla-Carvajal, M.X.; van Valenberg, H.J.F.; Hettinga, K. Bovine Milk Fatty Acid and Triacylglycerol Composition and Structure Differ between Early and Late Lactation Influencing Milk Fat Solid Fat Content. Int. Dairy J. 2022, 131, 105370. [Google Scholar] [CrossRef]
- Jiang, X.; Zhao, Z.; Chen, X.; Miao, F.; Li, J.; Yu, H.; Jiang, P.; Lin, Z. Effects of Genetic Polymorphism in the IFI27 Gene on Milk Fat Traits and Relevance to Lipid Metabolism in Bovine Mammary Epithelial Cells. Animals 2024, 14, 3284. [Google Scholar] [CrossRef]
- El-Ganainy, S.M.M.; Shams, A.S.; Kandial, M.H.H.; Badr, A.M.M. Milk Production and Milk Fatty Acid Profile as a Response to Feeding Dairy Cows with Flax Products during the Persistence Period. J. Anim. Physiol. Anim. Nutr. 2023, 107, 1187–1197. [Google Scholar] [CrossRef]
- Liu, K.; Chen, M.; Huang, G.; Su, C.; Tang, W.; Li, N.; Yang, J.; Wu, X.; Si, B.; Zhao, S. Variations in the Milk Lipidomic Profile of Lactating Dairy Cows Fed the Diets Containing Alfalfa Hay versus Alfalfa Silage. Anim. Nutr. 2024, 19, 261–271. [Google Scholar] [CrossRef]
- Zou, Y.; Chen, Y.; Meng, Q.; Wang, Y.; Zhang, Y. Cow Milk Fatty Acid and Protein Composition in Different Breeds and Regions in China. Molecules 2024, 29, 5142. [Google Scholar] [CrossRef]
- Stergiadis, S.; Nørskov, N.P.; Purup, S.; Givens, I.; Lee, M.R.F. Comparative Nutrient Profiling of Retail Goat and Cow Milk. Nutrients 2019, 11, 2282. [Google Scholar] [CrossRef]
- Fan, H.; Xia, S.; Xiang, J.; Li, Y.; Ross, M.O.; Lim, S.A.; Yang, F.; Tu, J.; Xie, L.; Dougherty, U.; et al. Trans-Vaccenic Acid Reprograms CD8+ T Cells and Anti-Tumor Immunity. Nature 2023, 623, 1034–1043. [Google Scholar] [CrossRef]
- Song, J.; Wang, Y.; Fan, X.; Wu, H.; Han, J.; Yang, M.; Lu, L.; Nie, G. Trans-Vaccenic Acid Inhibits Proliferation and Induces Apoptosis of Human Nasopharyngeal Carcinoma Cells via a Mitochondrial-Mediated Apoptosis Pathway. Lipids Health Dis. 2019, 18, 46. [Google Scholar] [CrossRef]
- Morenga, L.T.; Montez, J.M. Health Effects of Saturated and Trans-Fatty Acid Intake in Children and Adolescents: Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0186672. [Google Scholar]
- Sanjulian, L.; Lamas, A.; Barreiro, R.; Martínez, I.; García-Alonso, L.; Cepeda, A.; Fente, C.; Regal, P. Investigating the Dietary Impact on Trans-Vaccenic Acid (Trans-C18: 1 n-7) and Other Beneficial Fatty Acids in Breast Milk and Infant Formulas. Foods 2024, 13, 2164. [Google Scholar] [CrossRef]
- Pimentel, L.; Gomes, A.; Pintado, M.; Rodríguez-Alcalá, L.M. Isolation and Analysis of Phospholipids in Dairy Foods. J. Anal. Methods Chem. 2016, 2016, 9827369. [Google Scholar]
- Graves, E.L.F.; Beaulieu, A.D.; Drackley, J.K. Factors Affecting the Concentration of Sphingomyelin in Bovine Milk. J. Dairy Sci. 2007, 90, 706–715. [Google Scholar] [CrossRef]
- Nilsson, Å.; Duan, R.D.; Ohlsson, L. Digestion and Absorption of Milk Phospholipids in Newborns and Adults. Front. Nutr. 2021, 8, 724006. [Google Scholar] [CrossRef]
- Contarini, G.; Povolo, M. Phospholipids in Milk Fat: Composition, Biological and Technological Significance, and Analytical Strategies. Int. J. Mol. Sci. 2013, 14, 2808–2831. [Google Scholar] [CrossRef]
- Martini, M.; Altomonte, I.; Sodi, I.; Vasylieva, Y.; Salari, F. Sterol, Tocopherol, and Bioactive Fatty Acid Differences between Conventional, High-Quality, and Organic Cow Milk. J. Dairy Sci. 2023, 106, 8239–8248. [Google Scholar] [CrossRef]
- Duong, S.; Strobel, N.; Buddhadasa, S.; Auldist, M.J.; Wales, W.J.; Moate, P.J.; Cox, G.; Orbell, J.D.; Cran, M.J. Modification of the Sterol Profile in Milk through Feeding. J. Dairy Sci. 2019, 102, 5933–5944. [Google Scholar] [CrossRef]
- Fox, P.F.; Uniacke-Lowe, T.; McSweeney, P.L.H.; O’Mahony, J.A. Dairy Chemistry and Biochemistry; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Costa, A.; Lopez-Villalobos, N.; Sneddon, N.W.; Shalloo, L.; Franzoi, M.; De Marchi, M.; Penasa, M. Invited Review: Milk Lactose—Current Status and Future Challenges in Dairy Cattle. J. Dairy Sci. 2019, 102, 5883–5898. [Google Scholar] [CrossRef]
- Jiang, Y.; Sun, T.; Lin, Y.; Liu, M.; Wang, X. Is It Possible to Obtain Substitutes for Human Milk Oligosaccharides from Bovine Milk, Goat Milk, or Other Mammal Milks? Compr. Rev. Food Sci. Food Saf. 2024, 23, e70018. [Google Scholar] [CrossRef]
- Robinson, R.C. Structures and Metabolic Properties of Bovine Milk Oligosaccharides and Their Potential in the Development of Novel Therapeutics. Front. Nutr. 2019, 6, 50. [Google Scholar] [CrossRef] [PubMed]
- Gopal, P.K.; Gill, H.S. Oligosaccharides and Glycoconjugates in Bovine Milk and Colostrum. Br. J. Nutr. 2000, 84, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Sundekilde, U.K.; Barile, D.; Meyrand, M.; Poulsen, N.A.; Larsen, L.B.; Lebrilla, C.B.; German, J.B.; Bertram, H.C. Natural Variability in Bovine Milk Oligosaccharides from Danish Jersey and Holstein-Friesian Breeds. J. Agric. Food Chem. 2012, 60, 6188–6196. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; McVeagh, P.; Petocz, P.; Brand-Miller, J. Brain Ganglioside and Glycoprotein Sialic Acid in Breastfed Compared with Formula-Fed Infants. Am. J. Clin. Nutr. 2003, 78, 1024–1029. [Google Scholar] [CrossRef]
- Rivero-Urgell, M.; Santamaria-Orleans, A. Oligosaccharides: Application in Infant Food. Early Hum. Dev. 2001, 65, S43–S52. [Google Scholar] [CrossRef]
- Boehm, G.; Stahl, B. Oligosaccharides from Milk. J. Nutr. 2007, 137, 847S–849S. [Google Scholar] [CrossRef]
- European Food Safety Authority. Dietary Reference Values for Nutrients Summary Report. EFSA Support. Publ. 2017, 14, e15121E. [Google Scholar] [CrossRef]
- Haug, A.; Høstmark, A.T.; Harstad, O.M. Bovine Milk in Human Nutrition—A Review. Lipids Health Dis. 2007, 6, 25. [Google Scholar] [CrossRef]
- Linehan, K.; Patangia, D.V.; Roos, R.P.; Stanton, C. Production, composition and nutritional properties of organic milk: A Critical review. Foods 2024, 13, 550. [Google Scholar] [CrossRef]
- Chouinard, P.Y.; Girard, C.L. Nutritional Interest of Milk and Dairy Products: Some Scientific Data to Fuel the Debate. Anim. Front. 2014, 4, 4–6. [Google Scholar] [CrossRef]
- Greupner, T.; Schneider, I.; Hahn, A. Calcium Bioavailability from Mineral Waters with Different Mineralization in Comparison to Milk and a Supplement. J. Am. Coll. Nutr. 2017, 36, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Rovira, R.F. Milk and Milk Products: Food Sources of Calcium. Nutr. Hosp. 2015, 31, 1–9. [Google Scholar] [CrossRef]
- Rangel-Galván, M.; Rangel-Galván, V.; Rangel-Huerta, A. T-Type Calcium Channel Modulation by Hydrogen Sulfide in Neuropathic Pain Conditions. Front. Pharmacol. 2023, 14, 1212800. [Google Scholar] [CrossRef] [PubMed]
- Martinez de Victoria, E. Calcium, Essential for Health. Nutr. Hosp. 2016, 33, 341. [Google Scholar]
- Mellander, O. The Physiological Importance of the Casein Phosphopeptide Calcium Salts. II. Peroral Calcium Dosage of Infants. Acta Soc. Med. Ups. 1950, 55, 247–257. [Google Scholar]
- Marcone, S.; Belton, O.; Fitzgerald, D.J. Milk-Derived Bioactive Peptides and Their Health Promoting Effects: A Potential Role in Atherosclerosis. Br. J. Clin. Pharmacol. 2017, 83, 152–162. [Google Scholar] [CrossRef]
- Rangel, A.H.d.N.; Bezerra, D.A.F.V.d.A.; Sales, D.C.; Araújo, E.d.O.M.; Lucena, L.M.d.; Porto, A.L.F.; Véras, Í.V.U.M.; Lacerda, A.F.; Ribeiro, C.V.D.M.; Anaya, K. An Overview of the Occurrence of Bioactive Peptides in Different Types of Cheeses. Foods 2023, 12, 4261. [Google Scholar] [CrossRef]
- Mohanty, D.P.; Mohapatra, S.; Misra, S.; Sahu, P.S. Milk Derived Bioactive Peptides and Their Impact on Human Health—A Review. Saudi J. Biol. Sci. 2016, 23, 577–583. [Google Scholar] [CrossRef]
- Sultan, S.; Huma, N.; Butt, M.S.; Aleem, M.; Abbas, M. Therapeutic Potential of Dairy Bioactive Peptides: A Contemporary Perspective. Crit. Rev. Food Sci. Nutr. 2018, 58, 105–115. [Google Scholar] [CrossRef]
- Crippa, G.; Bosi, M.; Cassi, A.; Fiorentini, L.; Rossi, F. Blood pressure lowering effect of dietary integration with grana padano cheese in hypertensive patients. J. Hypertens. 2011, 29, e27. [Google Scholar] [CrossRef]
- Auestad, N.; Layman, D.K. Dairy Bioactive Proteins and Peptides: A Narrative Review. Nutr. Rev. 2021, 79, 36–47. [Google Scholar]
- Mattar, R.; Mazo, D.F.d.C.; Carrilho, F.J. Lactose Intolerance: Diagnosis, Genetic, and Clinical Factors. Clin. Exp. Gastroenterol. 2012, 5, 113–121. [Google Scholar] [CrossRef]
- Aldredge, D.L.; Geronimo, M.R.; Hua, S.; Nwosu, C.C.; Lebrilla, C.B.; Barile, D. Annotation and Structural Elucidation of Bovine Milk Oligosaccharides and Determination of Novel Fucosylated Structures. Glycobiology 2013, 23, 664–676. [Google Scholar] [CrossRef]
- Yokoi, T.; Nishiyama, K.; Kushida, Y.; Uribayashi, K.; Kunihara, T.; Fujimoto, R.; Yamamoto, Y.; Ito, M.; Miki, T.; Haneda, T.; et al. O-Acetylesterase Activity of Bifidobacterium Bifidum Sialidase Facilities the Liberation of Sialic Acid and Encourages the Proliferation of Sialic Acid Scavenging Bifidobacterium Breve. Environ. Microbiol. Rep. 2022, 14, 637–645. [Google Scholar] [CrossRef]
- Sela, D.A.; Garrido, D.; Lerno, L.; Wu, S.; Tan, K.; Eom, H.J.; Joachimiak, A.; Lebrilla, C.B.; Mills, D.A. Bifidobacterium Longum Subsp. Infantis ATCC 15697 α-Fucosidases Are Active on Fucosylated Human Milk Oligosaccharides. Appl. Environ. Microbiol. 2012, 78, 795–803. [Google Scholar] [CrossRef]
- Garrido, D.; Ruiz-Moyano, S.; Kirmiz, N.; Davis, J.C.; Totten, S.M.; Lemay, D.G.; Ugalde, J.A.; German, J.B.; Lebrilla, C.B.; Mills, D.A. A Novel Gene Cluster Allows Preferential Utilization of Fucosylated Milk Oligosaccharides in Bifidobacterium Longum Subsp. Longum SC596. Sci. Rep. 2016, 6, 35045. [Google Scholar] [CrossRef]
- Le, T.K.C.; Hosaka, T.; Le, T.T.T.; Nguyen, T.G.; Tran, Q.B.; Le, T.H.H.; Da Pham, X. Oral Administration of Bifidobacterium Spp. Improves Insulin Resistance, Induces Adiponectin, and Prevents Inflammatory Adipokine Expressions. Biomed. Res. 2014, 35, 303–310. [Google Scholar] [CrossRef]
- Jena, P.K.; Sheng, L.; Nagar, N.; Wu, C.; Barile, D.; Mills, D.A.; Wan, Y.J.Y. Synbiotics Bifidobacterium Infantis and Milk Oligosaccharides Are Effective in Reversing Cancer-Prone Nonalcoholic Steatohepatitis Using Western Diet-Fed FXR Knockout Mouse Models. J. Nutr. Biochem. 2018, 57, 246–254. [Google Scholar] [CrossRef]
- Boudry, G.; Hamilton, M.K.; Chichlowski, M.; Wickramasinghe, S.; Barile, D.; Kalanetra, K.M.; Mills, D.A.; Raybould, H.E. Bovine Milk Oligosaccharides Decrease Gut Permeability and Improve Inflammation and Microbial Dysbiosis in Diet-Induced Obese Mice. J. Dairy Sci. 2017, 100, 2471–2481. [Google Scholar] [CrossRef]
- Wang, M.; Monaco, M.H.; Hauser, J.; Yan, J.; Dilger, R.N.; Donovan, S.M. Bovine Milk Oligosaccharides and Human Milk Oligosaccharides Modulate the Gut Microbiota Composition and Volatile Fatty Acid Concentrations in a Preclinical Neonatal Model. Microorganisms 2021, 9, 884. [Google Scholar] [CrossRef]
- Leeuwendaal, N.K.; Stanton, C.; O’toole, P.W.; Beresford, T.P. Fermented Foods, Health and the Gut Microbiome. Nutrients 2022, 14, 1527. [Google Scholar] [CrossRef]
- Abd El-Salam, M.H.; El-Shibiny, S.; Assem, F.M.; El-Sayyad, G.S.; Hasanien, Y.A.; Elfadil, D.; Soliman, T.N. Impact of Fermented Milk On Gut Microbiota And Human Health: A Comprehensive Review. Curr. Microbiol. 2025, 82, 107. [Google Scholar] [CrossRef]
- Hamet, M.F.; Medrano, M.; Pérez, P.F.; Abraham, A.G. Oral Administration of Kefiran Exerts a Bifidogenic Effect on BALB/c Mice Intestinal Microbiota. Benef. Microbes 2016, 7, 237–246. [Google Scholar] [CrossRef]
- Carasi, P.; Racedo, S.M.; Jacquot, C.; Romanin, D.E.; Serradell, M.A.; Urdaci, M.C. Impact of Kefir Derived Lactobacillus Kefiri on the Mucosal Immune Response and Gut Microbiota. J. Immunol. Res. 2015, 2015, 361604. [Google Scholar] [CrossRef]
- Ferlay, A.; Bernard, L.; Meynadier, A.; Malpuech-Brugère, C. Production of Trans and Conjugated Fatty Acids in Dairy Ruminants and Their Putative Effects on Human Health: A Review. Biochimie 2017, 141, 107–120. [Google Scholar] [CrossRef]
- Grażyna, C.; Hanna, C.; Adam, A.; Magdalena, B.M. Natural Antioxidants in Milk and Dairy Products. Int. J. Dairy Technol. 2017, 70, 165–178. [Google Scholar] [CrossRef]
- Ivanova, M. Conjugated linoleic acid-enriched dairy products: A review. J. Microbiol. Biotechnol. Food Sci. 2021, 10, e3609. [Google Scholar] [CrossRef]
- Pipoyan, D.; Stepanyan, S.; Stepanyan, S.; Beglaryan, M.; Costantini, L.; Molinari, R.; Merendino, N. The Effect of Trans Fatty Acids on Human Health: Regulation and Consumption Patterns. Foods 2021, 10, 2452. [Google Scholar] [CrossRef]
- Badawy, S.; Liu, Y.; Guo, M.; Liu, Z.; Xie, C.; Marawan, M.A.; Ares, I.; Lopez-Torres, B.; Martínez, M.; Maximiliano, J.E.; et al. Conjugated Linoleic Acid (CLA) as a Functional Food: Is It Beneficial or Not? Food Res. Int. 2023, 172, 113158. [Google Scholar]
- Asbaghi, O.; Shimi, G.; Naseri, K.; Saadati, S.; Kelishadi, M.R.; Doaei, S.; Haghighat, N. The Effects of Conjugated Linoleic Acid Supplementation on Blood Pressure and Endothelial Function in Adults: A Systematic Review and Dose-Response Meta-Analysis. Eur. J. Pharmacol. 2022, 931, 175162. [Google Scholar]
- Ghodoosi, N.; Rasaei, N.; Goudarzi, K.; Hashemzadeh, M.; Dolatshahi, S.; Omran, H.S.; Amirani, N.; Ashtary-larky, D.; Shimi, G.; Asbaghi, O. The Effects of Conjugated Linoleic Acid Supplementation on Glycemic Control, Adipokines, Cytokines, Malondialdehyde and Liver Function Enzymes in Patients at Risk of Cardiovascular Disease: A GRADE-Assessed Systematic Review and Dose–Response Meta-Analysis. Nutr. J. 2023, 22, 47. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.W.; Cheng, H.Y.; Lee, Y.H.; Liou, T.H.; Liao, C.D.; Huang, S.W. Effects of Conjugated Linoleic Acid and Exercise on Body Composition and Obesity: A Systematic Review and Meta-Analysis. Nutr. Rev. 2023, 81, 397–415. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, H.P.; Zhou, L.M.; Zhou, L.; Chen, T.; Qin, L.Q. Effect of Conjugated Linoleic Acid on Blood Pressure: A Meta-Analysis of Randomized, Double-Blind Placebo-Controlled Trials. Lipids Health Dis. 2015, 14, 11. [Google Scholar] [CrossRef] [PubMed]
- Rossini, M.; Adami, S.; Bertoldo, F.; Diacinti, D.; Gatti, D.; Giannini, S.; Giusti, A.; Malavolta, N.; Minisola, S.; Osella, G.; et al. Guidelines for the Diagnosis, Prevention and Management of Osteoporosis. Reumatismo 2016, 68, 1–39. [Google Scholar] [CrossRef]
- Polzonetti, V.; Pucciarelli, S.; Vincenzetti, S.; Polidori, P. Dietary Intake of Vitamin d from Dairy Products Reduces the Risk of Osteoporosis. Nutrients 2020, 12, 1743. [Google Scholar] [CrossRef]
- Rozenberg, S.; Body, J.J.; Bruyère, O.; Bergmann, P.; Brandi, M.L.; Cooper, C.; Devogelaer, J.P.; Gielen, E.; Goemaere, S.; Kaufman, J.M.; et al. Effects of Dairy Products Consumption on Health: Benefits and Beliefs—A Commentary from the Belgian Bone Club and the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases. Calcif. Tissue Int. 2016, 98, 1–17. [Google Scholar] [CrossRef]
- Matía-Martín, P.; Torrego-Ellacuría, M.; Larrad-Sainz, A.; Fernández-Pérez, C.; Cuesta-Triana, F.; Rubio-Herrera, M.Á. Effects of Milk and Dairy Products on the Prevention of Osteoporosis and Osteoporotic Fractures in Europeans and Non-Hispanic Whites from North America: A Systematic Review and Updated Meta-Analysis. Adv. Nutr. 2019, 10, S120–S143. [Google Scholar] [CrossRef]
- Cruz-Pierard, S.M.; Nestares, T.; Amaro-Gahete, F.J. Vitamin D and Calcium as Key Potential Factors Related to Colorectal Cancer Prevention and Treatment: A Systematic Review. Nutrients 2022, 14, 4934. [Google Scholar] [CrossRef]
- Song, M.; Garrett, W.S.; Chan, A.T. Nutrients, Foods, and Colorectal Cancer Prevention. Gastroenterology 2015, 148, 1244–1260.e16. [Google Scholar] [CrossRef]
- Lopez-Caleya, J.F.; Ortega-Valín, L.; Fernández-Villa, T.; Delgado-Rodríguez, M.; Martín-Sánchez, V.; Molina, A.J. The Role of Calcium and Vitamin D Dietary Intake on Risk of Colorectal Cancer: Systematic Review and Meta-Analysis of Case–Control Studies. Cancer Causes Control 2022, 33, 167–182. [Google Scholar] [CrossRef]
- Barrubés, L.; Babio, N.; Becerra-Tomás, N.; Rosique-Esteban, N.; Salas-Salvadó, J. Association between Dairy Product Consumption and Colorectal Cancer Risk in Adults: A Systematic Review and Meta-Analysis of Epidemiologic Studies. Adv. Nutr. 2019, 10, S190–S211. [Google Scholar] [CrossRef]
- Huncharek, M.; Muscat, J.; Kupelnick, B. Impact of Dairy Products and Dietary Calcium on Bone-Mineral Content in Children: Results of a Meta-Analysis. Bone 2008, 43, 312–321. [Google Scholar] [CrossRef]
- Ralston, R.A.; Truby, H.; Palermo, C.E.; Walker, K.Z. Colorectal Cancer and Nonfermented Milk, Solid Cheese, and Fermented Milk Consumption: A Systematic Review and Meta-Analysis of Prospective Studies. Crit. Rev. Food Sci. Nutr. 2014, 54, 1167–1179. [Google Scholar] [CrossRef] [PubMed]
- Carroll, C.; Cooper, K.; Papaioannou, D.; Hind, D.; Pilgrim, H.; Tappenden, P. Supplemental Calcium in the Chemoprevention of Colorectal Cancer: A Systematic Review and Meta-Analysis. Clin. Ther. 2010, 32, 789–803. [Google Scholar] [CrossRef] [PubMed]
- Lumsden, A.L.; Mulugeta, A.; Hyppönen, E. Milk Consumption and Risk of Twelve Cancers: A Large-Scale Observational and Mendelian Randomization Study. Clin. Nutr. 2023, 42, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R. Cow milk protein allergy and lactose intolerance. Curr. Sci. 2020, 118, 1378. [Google Scholar] [CrossRef]
- Malmir, H.; Larijani, B.; Esmaillzadeh, A. Consumption of Milk and Dairy Products and Risk of Osteoporosis and Hip Fracture: A Systematic Review and Meta-Analysis. Crit. Rev. Food Sci. Nutr. 2020, 60, e609–e611. [Google Scholar] [CrossRef]
- Flom, J.D.; Sicherer, S.H. Epidemiology of Cow’s Milk Allergy. Nutrients 2019, 11, 1051. [Google Scholar] [CrossRef]
- Taylor-Black, S.; Wang, J. The Prevalence and Characteristics of Food Allergy in Urban Minority Children. Ann. Allergy Asthma Immunol. 2012, 109, 431–437. [Google Scholar] [CrossRef]
- Zeng, J.; Wang, Q.; Yi, H.; Chen, C.; Du, C.; Xiong, G.; Wang, B.; Zhao, J.; Zhang, L.; Gong, P. Recent Insights in Cow’s Milk Protein Allergy: Clinical Relevance, Allergen Features, and Influences of Food Processing. Trends Food Sci. Technol. 2025, 156, 104830. [Google Scholar] [CrossRef]
- Jensen, S.A.; Fiocchi, A.; Baars, T.; Jordakieva, G.; Nowak-Wegrzyn, A.; Pali-Schöll, I.; Passanisi, S.; Pranger, C.L.; Roth-Walter, F.; Takkinen, K.; et al. World Allergy Organization (WAO) Diagnosis and Rationale for Action against Cow’s Milk Allergy (DRACMA) Guidelines Update—III—Cow’s Milk Allergens and Mechanisms Triggering Immune Activation. World Allergy Organ. J. 2022, 15, 100668. [Google Scholar] [CrossRef] [PubMed]
- Franco, C.; Fente, C.; Sánchez, C.; Lamas, A.; Cepeda, A.; Leis, R.; Regal, P. Cow’s Milk Antigens Content in Human Milk: A Scoping Review. Foods 2022, 11, 1783. [Google Scholar] [CrossRef] [PubMed]
- Shkembi, B.; Huppertz, T. Glycemic Responses of Milk and Plant-Based Drinks: Food Matrix Effects. Foods 2023, 12, 453. [Google Scholar] [CrossRef] [PubMed]
- Kawada, T. Dairy Product Subgroups and Risk of Type 2 Diabetes. Br. J. Nutr. 2018, 119, 965. [Google Scholar] [CrossRef]
- Díaz-López, A.; Bulló, M.; Martínez-González, M.A.; Corella, D.; Estruch, R.; Fitó, M.; Gómez-Gracia, E.; Fiol, M.; García de la Corte, F.J.; Ros, E.; et al. Dairy Product Consumption and Risk of Type 2 Diabetes in an Elderly Spanish Mediterranean Population at High Cardiovascular Risk. Eur. J. Nutr. 2016, 55, 349–360. [Google Scholar] [CrossRef]
- Gudi, S.K. Dairy Consumption and Risk of Type-2 Diabetes: The Untold Story. Ann. Pediatr. Endocrinol. Metab. 2021, 26, 14–18. [Google Scholar] [CrossRef]
- Abargouei, A.S.; Janghorbani, M.; Salehi-Marzijarani, M.; Esmaillzadeh, A. Effect of Dairy Consumption on Weight and Body Composition in Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Clinical Trials. Int. J. Obes. 2012, 36, 1485–1493. [Google Scholar] [CrossRef]
- Chen, M.; Pan, A.; Malik, V.S.; Hu, F.B. Effects of Dairy Intake on Body Weight and Fat: A Meta-Analysis of Randomized Controlled Trials. Am. J. Clin. Nutr. 2012, 96, 735–747. [Google Scholar] [CrossRef]
- Lanou, A.J.; Barnard, N.D. Dairy and Weight Loss Hypothesis: An Evaluation of the Clinical Trials. Nutr. Rev. 2008, 66, 272–279. [Google Scholar] [CrossRef]
- Wrotniak, B.H.; Georger, L.; Hill, D.L.; Zemel, B.S.; Stettler, N. Association of Dairy Intake with Weight Change in Adolescents Undergoing Obesity Treatment. J. Public Health 2019, 41, 338–345. [Google Scholar] [CrossRef]
- Lovegrove, J.A.; Hobbs, D.A. New Perspectives on Dairy and Cardiovascular Health. Proc. Nutr. Soc. 2016, 75, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Huth, P.J.; Park, K.M. Influence of Dairy Product and Milk Fat Consumption on Cardiovascular Disease Risk: A Review of the Evidence. Adv. Nutr. 2012, 3, 266–285. [Google Scholar]
- Zhuang, P.; Liu, X.; Li, Y.; Ao, Y.; Wu, Y.; Ye, H.; Wan, X.; Zhang, L.; Meng, D.; Tian, Y.; et al. A Global Analysis of Dairy Consumption and Incident Cardiovascular Disease. Nat. Commun. 2025, 16, 437. [Google Scholar] [CrossRef] [PubMed]
- Yoshinari, M.; Ohkuma, T.; Iwase, M.; Kitazono, T. Milk and Yogurt Consumption and Its Association with Cardiometabolic Risk Factors in Patients with Type 2 Diabetes: The Fukuoka Diabetes Registry. Nutr. Metab. Cardiovasc. Dis. 2025, 35, 103772. [Google Scholar] [CrossRef] [PubMed]
- Abid, Z.; Cross, A.J.; Sinha, R. Meat, Dairy, and Cancer. Am. J. Clin. Nutr. 2014, 100, 386S–393S. [Google Scholar] [CrossRef]
- López-Plaza, B.; Bermejo, L.M.; Santurino, C.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Gómez-Candela, C. Milk and Dairy Product Consumption and Prostate Cancer Risk and Mortality: An Overview of Systematic Reviews and Meta-Analyses. Adv. Nutr. 2019, 10, S212–S223. [Google Scholar] [CrossRef]
- Steck, S.E.; Omofuma, O.O.; Su, L.J.; Maise, A.A.; Woloszynska-Read, A.; Johnson, C.S.; Zhang, H.; Bensen, J.T.; Fontham, E.T.H.; Mohler, J.L.; et al. Calcium, Magnesium, and Whole-Milk Intakes and High-Aggressive Prostate Cancer in the North Carolina-Louisiana Prostate Cancer Project (PCaP). Am. J. Clin. Nutr. 2018, 107, 799–807. [Google Scholar] [CrossRef]
- Pal, M.; Hodge, A.M.; Papa, N.; MacInnis, R.J.; Bassett, J.K.; Bolton, D.; Davis, I.D.; Millar, J.; English, D.R.; Hopper, J.L.; et al. Body Size and Dietary Risk Factors for Aggressive Prostate Cancer: A Case–Control Study. Cancer Causes Control 2019, 30, 1301–1312. [Google Scholar] [CrossRef]
- Zhao, Z.; Wu, D.; Gao, S.; Zhou, D.; Zeng, X.; Yao, Y.; Xu, Y.; Zeng, G. The Association between Dairy Products Consumption and Prostate Cancer Risk: A Systematic Review and Meta-Analysis. Br. J. Nutr. 2023, 129, 1714–1731. [Google Scholar] [CrossRef]
- Travis, R.C.; Appleby, P.N.; Martin, R.M.; Holly, J.M.P.; Albanes, D.; Black, A.; Bueno-De-Mesquita, H.B.; Chan, J.M.; Chen, C.; Chirlaque, M.D.; et al. A Meta-Analysis of Individual Participant Data Reveals an Association between Circulating Levels of IGF-I and Prostate Cancer Risk. Cancer Res. 2016, 76, 2288–2300. [Google Scholar] [CrossRef]
- Elsabbagh, M.; Divan, G.; Koh, Y.; Kim, Y.S.; Kauchali, S.; Marcín, C.; Montiel-Nava, C.; Patel, V.; Paula, C.S.; Wang, C. Global Prevalence of Autism and Other Pervasive Developmental Disorders. Autism Res. 2012, 5, 160–179. [Google Scholar] [CrossRef] [PubMed]
- Karhu, E.; Zukerman, R.; Eshraghi, R.S.; Mittal, J.; Deth, R.C.; Castejon, A.M.; Trivedi, M.; Mittal, R.; Eshraghi, A.A. Nutritional Interventions for Autism Spectrum Disorder. Nutr. Rev. 2020, 78, 515–531. [Google Scholar] [CrossRef] [PubMed]
- Hartman, R.E.; Patel, D. Dietary Approaches to the Management of Autism Spectrum Disorders. In Personalized Food Intervention and Therapy for Autism Spectrum Disorder Management; Springer: Berlin/Heidelberg, Germany, 2020; pp. 547–571. [Google Scholar]
- Gogou, M.; Kolios, G. Are Therapeutic Diets an Emerging Additional Choice in Autism Spectrum Disorder Management? World J. Pediatr. 2018, 14, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Sanctuary, M.R.; Kain, J.N.; Angkustsiri, K.; German, J.B. Dietary Considerations in Autism Spectrum Disorders: The Potential Role of Protein Digestion and Microbial Putrefaction in the Gut-Brain Axis. Front. Nutr. 2018, 5, 40. [Google Scholar] [CrossRef]
- Fernández-Rico, S.; Mondragón, A.D.C.; López-Santamarina, A.; Cardelle-Cobas, A.; Regal, P.; Lamas, A.; Ibarra, I.S.; Cepeda, A.; Miranda, J.M. A2 Milk: New Perspectives for Food Technology and Human Health. Foods 2022, 11, 2387. [Google Scholar] [CrossRef]
- Jianqin, S.; Leiming, X.; Lu, X.; Yelland, G.W.; Ni, J.; Clarke, A.J. Effects of Milk Containing Only A2 Beta Casein versus Milk Containing Both A1 and A2 Beta Casein Proteins on Gastrointestinal Physiology, Symptoms of Discomfort, and Cognitive Behavior of People with Self-Reported Intolerance to Traditional Cows’ Milk. Nutr. J. 2015, 15, 35. [Google Scholar] [CrossRef]
- Bell, S.J.; Grochoski, G.T.; Clarke, A.J. Health Implications of Milk Containing β-Casein with the A2 Genetic Variant. Crit. Rev. Food Sci. Nutr. 2006, 46, 93–100. [Google Scholar] [CrossRef]
- Kamiński, S.; Cieślińska, A.; Kostyra, E. Polymorphism of Bovine Beta-Casein and Its Potential Effect on Human Health. J. Appl. Genet. 2007, 48, 189–198. [Google Scholar] [CrossRef]
- Rani, P.; Yenuganti, V.R.; Shandilya, S.; Onteru, S.K.; Singh, D. MiRNAs: The Hidden Bioactive Component of Milk. Trends Food Sci. Technol. 2017, 65, 94–102. [Google Scholar] [CrossRef]
- Benmoussa, A.; Provost, P. Milk MicroRNAs in Health and Disease. Compr. Rev. Food Sci. Food Saf. 2019, 18, 703–722. [Google Scholar] [CrossRef]
- Izumi, H.; Tsuda, M.; Sato, Y.; Kosaka, N.; Ochiya, T.; Iwamoto, H.; Namba, K.; Takeda, Y. Bovine Milk Exosomes Contain MicroRNA and MRNA and Are Taken up by Human Macrophages. J. Dairy Sci. 2015, 98, 2920–2933. [Google Scholar] [CrossRef] [PubMed]
- Mohr, A.M.; Mott, J.L. Overview of MicroRNA Biology. Semin. Liver Dis. 2015, 35, 3–11. [Google Scholar] [CrossRef]
- Zempleni, J.; Baier, S.R.; Howard, K.M.; Cui, J. Gene Regulation by Dietary MicroRNAs. Can. J. Physiol. Pharmacol. 2015, 93, 1097–1102. [Google Scholar] [CrossRef]
- Baier, S.R.; Nguyen, C.; Xie, F.; Wood, J.R.; Zempleni, J. MicroRNAs Are Absorbed in Biologically Meaningful Amounts from Nutritionally Relevant Doses of Cow Milk and Affect Gene Expression in Peripheral Blood Mononuclear Cells, HEK-293 Kidney Cell Cultures, and Mouse Livers. J. Nutr. 2014, 144, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Herwijnen, M.J.C.v.; Driedonks, T.A.P.; Snoek, B.L.; Kroon, A.M.T.; Kleinjan, M.; Jorritsma, R.; Pieterse, C.M.J.; Hoen, E.N.M.N.; Wauben, M.H.M. Abundantly Present MiRNAs in Milk-Derived Extracellular Vesicles Are Conserved Between Mammals. Front. Nutr. 2018, 5, 81. [Google Scholar] [CrossRef] [PubMed]
- Zempleni, J. Milk Exosomes: Beyond Dietary MicroRNAs. Genes Nutr. 2017, 12, 12. [Google Scholar] [CrossRef]
- Alsaweed, M.; Lai, C.T.; Hartmann, P.E.; Geddes, D.T.; Kakulas, F. Human Milk MiRNAs Primarily Originate from the Mammary Gland Resulting in Unique MiRNA Profiles of Fractionated Milk. Sci. Rep. 2016, 6, 20680. [Google Scholar] [CrossRef]
- Melnik, B.C. Milk Disrupts P53 and DNMT1, the Guardians of the Genome: Implications for Acne Vulgaris and Prostate Cancer. Nutr. Metab. 2017, 14, 55. [Google Scholar] [CrossRef]
- Melnik, B.C. Milk Exosomal MiRNAs: Potential Drivers of AMPK-To-MTORC1 Switching in β-Cell de-Differentiation of Type 2 Diabetes Mellitus. Nutr. Metab. 2019, 16, 85. [Google Scholar] [CrossRef]
- Cai, M.; He, H.; Jia, X.; Chen, S.; Wang, J.; Shi, Y.; Liu, B.; Xiao, W.; Lai, S. Genome-Wide MicroRNA Profiling of Bovine Milk-Derived Exosomes Infected with Staphylococcus Aureus. Cell Stress Chaperones 2018, 23, 663–672. [Google Scholar] [CrossRef]
- Palmer, J.D.; Soule, B.P.; Simone, B.A.; Zaorsky, N.G.; Jin, L.; Simone, N.L. MicroRNA Expression Altered by Diet: Can Food Be Medicinal? Aging Res. Rev. 2014, 17, 16–24. [Google Scholar] [CrossRef]
- Korpal, M.; Lee, E.S.; Hu, G.; Kang, Y. The MiR-200 Family Inhibits Epithelial–Mesenchymal Transition and Cancer Cell Migration by Direct Targeting of E-Cadherin Transcriptional Repressors ZEB1 and ZEB2. J. Biol. Chem. 2008, 283, 14910–14914. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B.C.; John, S.M.; Schmitz, G. Milk: An Epigenetic Inducer of FoxP3 Expression. J. Allergy Clin. Immunol. 2016, 138, 937–938. [Google Scholar] [CrossRef] [PubMed]
- Van Hese, I.; Goossens, K.; Vandaele, L.; Opsomer, G. Invited Review: MicroRNAs in Bovine Colostrum—Focus on Their Origin and Potential Health Benefits for the Calf. J. Dairy Sci. 2020, 103, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhang, G.; Wu, J.H.; Jiang, C.P. Diverse Roles of MiR-29 in Cancer (Review). Oncol. Rep. 2014, 31, 1509–1516. [Google Scholar] [CrossRef]
- Kriegel, A.J.; Liu, Y.; Fang, Y.; Ding, X.; Liang, M. The MiR-29 Family: Genomics, Cell Biology, and Relevance to Renal and Cardiovascular Injury. Physiol. Genom. 2012, 44, 237–244. [Google Scholar] [CrossRef]
- Xiong, Y.; Fang, J.H.; Yun, J.P.; Yang, J.; Zhang, Y.; Jia, W.H.; Zhuang, S.M. Effects of Microrna-29 on Apoptosis, Tumorigenicity, and Prognosis of Hepatocellular Carcinoma. Hepatology 2010, 51, 836–845. [Google Scholar] [CrossRef]
- Lozano, C.; Duroux-Richard, I.; Firat, H.; Schordan, E.; Apparailly, F. MicroRNAs: Key Regulators to Understand Osteoclast Differentiation? Front. Immunol. 2019, 10, 375. [Google Scholar] [CrossRef]
- Rossi, M.; Pitari, M.R.; Amodio, N.; Di Martino, M.T.; Conforti, F.; Leone, E.; Botta, C.; Paolino, F.M.; Del Giudice, T.; Iuliano, E.; et al. MiR-29b Negatively Regulates Human Osteoclastic Cell Differentiation and Function: Implications for the Treatment of Multiple Myeloma-Related Bone Disease. J. Cell. Physiol. 2013, 228, 1506–1515. [Google Scholar] [CrossRef]
- Melnik, B.C. Milk: An Epigenetic Amplifier of FTO-Mediated Transcription? Implications for Western Diseases. J. Transl. Med. 2015, 13, 385. [Google Scholar] [CrossRef]
- Walther, B.; Guggisberg, D.; Badertscher, R.; Egger, L.; Portmann, R.; Dubois, S.; Haldimann, M.; Kopf-Bolanz, K.; Rhyn, P.; Zoller, O.; et al. Comparison of Nutritional Composition between Plant-Based Drinks and Cow’s Milk. Front. Nutr. 2022, 9, 988707. [Google Scholar] [CrossRef] [PubMed]
- Luque-Uría, A.; Calvo, M.V.; Visioli, F.; Fontecha, J. Milk Fat Globule Membrane and Their Polar Lipids: Reviewing Preclinical and Clinical Trials on Cognition. Food Funct. 2024, 15, 6783–6797. [Google Scholar] [CrossRef] [PubMed]
- Keenan, T.W.; Mather, I.H. Intracellular Origin of Milk Fat Globules and the Nature of the Milk Fat Globule Membrane. In Advanced Dairy Chemistry; Springer: Boston, MA, USA, 2009; Volume 2. [Google Scholar]
- Ma, Z.; Gong, H.; Liu, B.; Mao, X. The Compositions of Milk Fat Globule Membrane Determine the Interfacial Behavior, Digestive Properties, and Bioactivities: Natural Versus Processed Forms. Trends Food Sci. Technol. 2024, 154, 104786. [Google Scholar] [CrossRef]
- Fong, B.Y.; Norris, C.S.; MacGibbon, A.K.H. Protein and Lipid Composition of Bovine Milk-Fat-Globule Membrane. Int. Dairy J. 2007, 17, 275–288. [Google Scholar] [CrossRef]
- Sánchez, C.; Franco, L.; Regal, P.; Lamas, A.; Cepeda, A.; Fente, C. Breast Milk: A Source of Functional Compounds with Potential Application in Nutrition and Therapy. Nutrients 2021, 13, 1026. [Google Scholar] [CrossRef]
- Colombo, J.; Harris, C.L.; Wampler, J.L.; Zhuang, W.; Shaddy, D.J.; Liu, B.Y.; Wu, S.S. Improved Neurodevelopmental Outcomes at 5.5 Years of Age in Children Who Received Bovine Milk Fat Globule Membrane and Lactoferrin in Infant Formula Through 12 Months: A Randomized Controlled Trial. J. Pediatr. 2023, 261, 113483. [Google Scholar] [CrossRef]
- Li, F.; Wu, S.S.; Berseth, C.L.; Harris, C.L.; Richards, J.D.; Wampler, J.L.; Zhuang, W.; Cleghorn, G.; Rudolph, C.D.; Liu, B.; et al. Improved Neurodevelopmental Outcomes Associated with Bovine Milk Fat Globule Membrane and Lactoferrin in Infant Formula: A Randomized, Controlled Trial. J. Pediatr. 2019, 215, 24–31.e8. [Google Scholar] [CrossRef]
- Davies, N.; Frampton, C.; Fuad, M.; Slykerman, R. The Effect of Supplementation with Milk Fat Globule Membranes on Psychological Health: A Randomized Clinical Trial in Healthy Adults with Moderate Stress. J. Funct. Foods 2023, 105, 105585. [Google Scholar] [CrossRef]
- Calvo, M.V.; Loria Kohen, V.; Díaz-Mardomingo, C.; García-Herranz, S.; Baliyan, S.; Tomé-Carneiro, J.; Colmenarejo, G.; Visioli, F.; Venero, C.; Fontecha, J. Milk Fat Globule Membrane-Enriched Milk Improves Episodic Memory: A Randomized, Parallel, Double-Blind, Placebo-Controlled Trial in Older Adults. J. Funct. Foods 2023, 111, 105849. [Google Scholar] [CrossRef]
- Mondragon Portocarrero, A.D.C.; Lopez-Santamarina, A.; Lopez, P.R.; Ortega, I.S.I.; Duman, H.; Karav, S.; Miranda, J.M. Substitutive Effects of Milk vs. Vegetable Milk on the Human Gut Microbiota and Implications for Human Health. Nutrients 2024, 16, 3108. [Google Scholar] [CrossRef]
- Li, T.; Gao, J.; Du, M.; Mao, X. Milk Fat Globule Membrane Supplementation Modulates the Gut Microbiota and Attenuates Metabolic Endotoxemia in High-Fat Diet-Fed Mice. J. Funct. Foods 2018, 47, 56–65. [Google Scholar] [CrossRef]
- Kosmerl, E.; Rocha-Mendoza, D.; Ortega-Anaya, J.; Jiménez-Flores, R.; García-Cano, I. Improving Human Health with Milk Fat Globule Membrane, Lactic Acid Bacteria, and Bifidobacteria. Microorganisms 2021, 9, 341. [Google Scholar] [CrossRef] [PubMed]

| Nutrient | Whole Milk | Semi-Skimmed Milk | Skim Milk | Whole Natural Yogurt | Whole Greek Yogurt | Skimmed Natural Yogurt | Burgos Cheese | Cured Manchego Cheese | Cheddar Cheese | Swiss Cheese | Ricotta Cheese | Parmesan Cheese |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Energy (Kcal) | 65.4/274 | 47.6/199 | 37/155 | 61.4/257 | 95 | 44.9/188 | 200/837 | 476/1992 | 409 | 393 | 158 | 420 |
| Proteins (g) | 3.10 | 3.50 | 3.90 | 4 | 8.78 | 4.30 | 14 | 38 | 23.30 | 27 | 7.81 | 29.60 |
| Total lipids (g) | 3.80 | 1.60 | 0.20 | 2.60 | 4.39 | 0.32 | 14.90 | 35.80 | 34 | 31 | 11 | 28 |
| Saturated fatty acids (g) | 2.30 | 1.10 | 0.09 | 1.50 | 2.39 | 0.11 | 8.80 | 18.70 | 19.2 | 18.2 | 6.97 | 15.5 |
| Monosaturated fatty acids (g) | 1.10 | 0.45 | 0.06 | 0.72 | 0.96 | 0.15 | 4.30 | 8.40 | 7.44 | 7.26 | 2.56 | 6.40 |
| Polyunsaturated fatty acids (g) | 0.13 | 0.04 | 0.01 | 0.13 | 0.11 | 0 | 0.89 | 6.20 | 1.18 | 1.14 | 0.39 | 1.20 |
| Cholesterol (g) | 14 | 6.30 | 2.60 | 10.20 | 17 | 1 | 14.50 | 74.40 | 100 | 93 | 48 | 87 |
| Carbohydrates (g) | 4.70 | 4.80 | 4.90 | 5.50 | 4.75 | 6.30 | 2.50 | 0.51 | 2.44 | 1.44 | 6.86 | 12.40 |
| Water (g) | 88.40 | 90.10 | 91 | 87.90 | 81.30 | 89.10 | 68.60 | 25.70 | 36.60 | 37.60 | 72.90 | 22.80 |
| Calcium (mg) | 124 | 125 | 121 | 142 | 111 | 140 | 191 | 848 | 707 | 890 | 224 | 884 |
| Iron (mg) | 0.09 | 0.09 | 0.09 | 0.09 | <0.10 | 0.09 | 0.62 | 0.75 | 0.16 | 0.13 | 0.10 | 0.45 |
| Iodine (µg) | 9 | 8.60 | 11.10 | 3.70 | 42.30 | 5.30 | 4.80 | 34 | N.D | N.D | N.D | N.D |
| Magnesium (mg) | 11.60 | 11.90 | 28.60 | 14.30 | 10.70 | 13.70 | 24.40 | 33.50 | 26.80 | 33.40 | 19.70 | 34.90 |
| Zinc (mg) | 0.38 | 0.52 | 0.54 | 0.59 | 0.47 | 0.44 | 2 | 3.20 | 3.67 | 4.37 | 0.56 | 4.33 |
| Sodium (mg) | 48 | 47 | 53 | 80 | 34 | 57 | 294 | 742 | 654 | 185 | 105 | 1750 |
| Potassium (mg) | 157 | 155 | 150 | 280 | 147 | 187 | 200 | 100 | 77 | 71 | 230 | 184 |
| Phosphorus (mg) | 92 | 91 | 97 | 170 | 126 | 109 | 600 | 560 | 458 | 574 | 162 | 634 |
| Selenium (µg) | 1.40 | 1.50 | 1.60 | 2 | N.D | 1 | 14.50 | 1.60 | 28.30 | 30.1 | 5.50 | 35 |
| Thiamine (mg) | 0.04 | 0.04 | 0.04 | 0.04 | 0.06 | 0.04 | 0.02 | 0.04 | 0.03 | 0.01 | 0.04 | 0.03 |
| Riboflavin (mg) | 0.19 | 0.19 | 0.17 | 0.18 | 0.24 | 0.19 | 0.17 | 0.33 | 0.44 | 0.30 | 0.33 | 0.35 |
| Niacin equivalents (mg) | 0.73 | 0.71 | 0.90 | 0.44 | 0.23 | 1.20 | 4.10 | 7.20 | 0.05 | 0.06 | 0.17 | 0.08 |
| Vitamin B6 (mg) | 0.04 | 0.06 | 0.04 | 0.05 | 0.04 | 0.08 | 0.08 | 0.20 | 0.07 | 0.07 | 0.10 | 0.08 |
| Folate (µg) | 5.50 | 2.70 | 5.30 | 3.70 | N.D | 4.70 | 14.30 | 21.80 | 21 | 9 | 4 | 6 |
| Vitamin B12 (µg) | 0.30 | 0.30 | 0.30 | 0.20 | N.D | 0.40 | 0.66 | 1.50 | 1.06 | 3.02 | 0.78 | 1.35 |
| Vitamin C (mg) | 1.40 | 0.52 | 1.70 | 0.70 | N.D | 1.60 | 0 | 0 | N.D | N.D | N.D | N.D |
| Vitamin A (retinol equivalents, µg) | 46 | 18.90 | Traces | 9.80 | 38 | 0.80 | 261 | 234 | 316 | 292 | 127 | 228 |
| Vitamin D (µg) | 0.03 | 0.02 | Traces | 0.06 | <0.01 | 0 | 0 | 0.19 | 0 | N.D | N.D | |
| Vitamin E (mg) | 0.1 | 0.04 | Traces | 0.05 | N.D | Traces | 0.56 | 0.61 | 0.75 | 0.6 | 0.27 | 0.51 |
| Amino Acids | Protein Consumption Pattern (mg/g) | Milk Protein (mg/g) |
|---|---|---|
| Histidine | 18 | 25 |
| Isoleucine | 25 | 56 |
| Leucine | 55 | 92 |
| Lysine | 51 | 72 |
| Methionine + Cysteine | 25 | 30 |
| Phenylalanine + Tyrosine | 47 | 56 |
| Threonine | 27 | 41 |
| Tryptophan | 7 | 13 |
| Valine | 32 | 62 |
| Fatty Acid | % of Total Fatty Acids |
|---|---|
| Butyric 4:0 | 3.30 |
| Capric 10:0 | 2.60 |
| Caprylic 8:0 | 1.20 |
| Caproic 6:0 | 1.90 |
| Stearic 18:0 | 11.00 |
| Lauric 12:0 | 3.50 |
| Linoleic 18:2 | 2.40 |
| α–linolenic 18:3 | 0.50 |
| Myristic 14:0 | 11.50 |
| Oleic 18:1 | 28.00 |
| Palmitic 16:0 | 25.80 |
| Palmitoleic 16:1 | 2.90 |
| Food | Ration Size (g) | Ca Content (mg) | Fractional Absorption | Estimated Absorbable Ca (mg) | Rations Equivalent to One Serving of Milk |
|---|---|---|---|---|---|
| Milk | 240 | 300 | 32.10 | 96.30 | 1 |
| Yogurt | 240 | 300 | 32.10 | 96.30 | 1 |
| Cheddar cheese | 42 | 303 | 32.10 | 97.20 | 1 |
| Pinto bean | 86 | 44.70 | 26.70 | 11.90 | 8.10 |
| Red bean | 172 | 40.50 | 24.40 | 9.90 | 9.70 |
| White bean | 110 | 113 | 21.80 | 24.70 | 3.90 |
| Broccoli | 71 | 35 | 61.30 | 21.50 | 4.50 |
| Kale | 85 | 61 | 49.30 | 30.10 | 3.20 |
| Spinach | 85 | 115 | 5.10 | 5.90 | 16.30 |
| Sweet potato | 164 | 44 | 22.20 | 9.80 | 9.80 |
| Tofu with calcium | 126 | 258 | 31 | 80 | 1.20 |
| Nutrient | Whole Milk | Soy-Based Drink | Oat-Based Drink | Almond-Based Drink |
|---|---|---|---|---|
| Energy (Kcal) | 274 | 38 | 48 | 15 |
| Proteins (g) | 3.10 | 3.55 | 0.80 | 0.55 |
| Total lipids (g) | 3.80 | 2.12 | 2.75 | 1.22 |
| Saturated fatty acids (g) | 2.30 | 0.31 | N.D | 0.10 |
| Monosaturated fatty acids (g) | 1.10 | 0.42 | N.D | 0.73 |
| Polyunsaturated fatty acids (g) | 0.13 | 1.15 | N.D | 0.28 |
| Cholesterol (g) | 14 | - | - | - |
| Carbohydrates (g) | 4.70 | 1.29 | 5.10 | 0.34 |
| Water (g) | 88.40 | 92.40 | 90.60 | 97.40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanjulián, L.; Fernández-Rico, S.; González-Rodríguez, N.; Cepeda, A.; Miranda, J.M.; Fente, C.; Lamas, A.; Regal, P. The Role of Dairy in Human Nutrition: Myths and Realities. Nutrients 2025, 17, 646. https://doi.org/10.3390/nu17040646
Sanjulián L, Fernández-Rico S, González-Rodríguez N, Cepeda A, Miranda JM, Fente C, Lamas A, Regal P. The Role of Dairy in Human Nutrition: Myths and Realities. Nutrients. 2025; 17(4):646. https://doi.org/10.3390/nu17040646
Chicago/Turabian StyleSanjulián, Laura, Salvador Fernández-Rico, Nerea González-Rodríguez, Alberto Cepeda, José Manuel Miranda, Cristina Fente, Alexandre Lamas, and Patricia Regal. 2025. "The Role of Dairy in Human Nutrition: Myths and Realities" Nutrients 17, no. 4: 646. https://doi.org/10.3390/nu17040646
APA StyleSanjulián, L., Fernández-Rico, S., González-Rodríguez, N., Cepeda, A., Miranda, J. M., Fente, C., Lamas, A., & Regal, P. (2025). The Role of Dairy in Human Nutrition: Myths and Realities. Nutrients, 17(4), 646. https://doi.org/10.3390/nu17040646











