Omega-3 Fatty Acids and Inflammatory Processes
Abstract
:1. Introduction
- An increased blood supply to the site of inflammation;
- Increased capillary permeability caused by retraction of endothelial cells. This permits larger molecules, not normally capable of traversing the endothelium, to do so and thus delivers soluble mediators to the site of inflammation;
- Leukocyte migration from the capillaries into the surrounding tissue. This is promoted by release of chemoattractants from the site of inflammation and by the upregulation of adhesion molecules on the endothelium. Once in the tissue the leukocytes move to the site of inflammation;
- Release of mediators from leukocytes at the site of inflammation. These may include lipid mediators (e.g., prostaglandins (PGs), leukotrienes (LTs)), peptide mediators (e.g., cytokines), reactive oxygen species (e.g., superoxide), amino acid derivatives (e.g., histamine), and enzymes (e.g., matrix proteases) depending upon the cell type involved, the nature of the inflammatory stimulus, the anatomical site involved, and the stage during the inflammatory response. These mediators normally would play a role in host defense, but when produced inappropriately or in an unregulated fashion they can cause damage to host tissues, leading to disease. Several of these mediators may act to amplify the inflammatory process acting, for example, as chemoattractants. Some of the inflammatory mediators may escape the inflammatory site into the circulation and from there they can exert systemic effects. For example, the cytokine interleukin (IL)-6 induces hepatic synthesis of the acute phase protein C-reactive protein, while the cytokine tumour necrosis factor (TNF)-α elicits metabolic effects within skeletal muscle, adipose tissue and bone.
2. Fatty Acid Composition of Cells Involved in Inflammation and its Modification by Marine n-3 Fatty Acids

3. Mechanisms by which Polyunsaturated Fatty Acids can Influence Inflammatory Cell Function
- PUFA intake can influence complex lipid, lipoprotein, metabolite and hormone concentrations that in turn influence inflammation;
- Non-esterified PUFAs can act directly on inflammatory cells via surface or intracellular “fatty acid receptors” – the latter may include transcription factors like peroxisome proliferator activated receptors (PPARs);
- PUFAs can be oxidized (enzymatically or non-enzymatically) and the oxidized derivatives can act directly on inflammatory cells via surface or intracellular receptors – oxidation can occur to the non-esterified form of the PUFA or to PUFAs esterified into more complex lipids including circulating or cell membrane phospholipids and intact lipoproteins such as low density lipoprotein (LDL);
- PUFAs can be incorporated into the phospholipids of inflammatory cell membranes(as described above). Here they play important roles assuring the correct environment for membrane protein function, maintaining membrane order (“fluidity”) and influencing lipid raft formation [26]. Membrane phospholipids are substrates for the generation of second messengers like diacylglycerol and it has been demonstrated that the fatty acid composition of such second messengers, which is determined by that of the precursor phospholipid, can influence their activity [27]. In addition, membrane phospholipids are substrates for the release of (non-esterfied) PUFAs intracellularly – the released PUFAs can act as signaling molecules, ligands (or precursors of ligands) for transcription factors, or precursors for biosynthesis of lipid mediators which are involved in regulation of many cell and tissue responses, including aspects of inflammation and immunity (see below). Thus, changes in membrane phospholipid fatty acid composition, as described above, can influence the function of cells involved in inflammation via:
- ○ alterations in the physical properties of the membrane such as membrane order and raft structure;
- ○ effects on cell signaling pathways, either through modifying the expression, activity or avidity of membrane receptors or modifying intracellular signal transduction mechanisms that lead to altered transcription factor activity and changes in gene expression;
- ○ alterations in the pattern of lipid mediators produced, with the different mediators having different biological activities and potencies (see below).
4. Lipid Mediators: Biosynthesis, Roles in Inflammation, and the Impact of Marine n-3 fatty acids
4.1. Eicosanoids Generated from Arachidonic Acid

4.2. Fatty Acid Modification of Eicosanoid Profiles
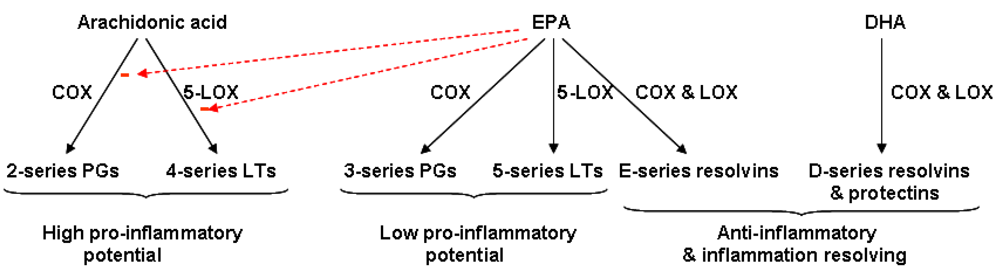
4.3. Resolvins: Novel Anti-Inflammatory and Inflammation Resolving Mediators Produced from EPA and DHA
5. Influence of Marine n-3 Fatty Acids on Leukocyte Chemotaxis

6. Influence of Marine n-3 Fatty Acids on Adhesion Molecules and Adhesive Interactions
7. Influence of Marine n-3 Fatty Acids on Inflammatory Cytokines
7.1. Transcription Factors Involved in Regulating Inflammatory Gene Expression
7.2. Fatty Acid Modulation of Inflammatory Cytokine Production and of Transcription Factor Activation
8. Anti-Inflammatory Effects of Marine n-3 Fatty Acids Suggest a Therapeutic Value
- decrease production of eicosanoid mediators from arachidonic acid, many of which have pro-inflammatory roles;
- increase production of weakly inflammatory or anti-inflammatory eicosanoids from EPA;
- increase production of anti-inflammatory and inflammation resolving resolvins from EPA and DHA;
- decrease chemotactic responses of leukocytes;
- decrease adhesion molecule expression on leukocytes and on endothelial cells and decrease intercellular adhesive interactions;
- decrease production of pro-inflammatory cytokines and other pro-inflammatory proteins induced via the NFκB system.
| Disease/condition |
| Rheumatoid arthritis |
| Crohn’s disease |
| Ulcerative colitis |
| Lupus |
| Type-1 diabetes |
| Cystic fibrosis |
| Childhood asthma |
| Adult asthma |
| Allergic disease |
| Chronic obstructive pulmonary disease |
| Psoriasis |
| Multiple sclerosis |
| Atherosclerosis |
| Acute cardiovascular events |
| Obesity |
| Neurodegenerative diseases of ageing |
| Systemic inflammatory response to surgery, trauma and critical illness |
9. Conclusions
References
- Calder, P.C.; Albers, R.; Antoine, J.M.; Blum, S.; Bourdet-Sicard, R.; Ferns, G.A.; Folkerts, G.; Friedmann, P.S.; Frost, G.S.; Guarner, F.; Løvik, M.; Macfarlane, S.; Meyer, P.D.; M'Rabet, L.; Serafini, M.; van Eden, W.; van Loo, J.; Vas Dias, W.; Vidry, S.; Winklhofer-Roob, B.M.; Zhao, J. Inflammatory disease processes and interactions with nutrition. Brit. J. Nutr. 2009, 101, S1–S45. [Google Scholar]
- Calder, P.C.; Yaqoob, P.; Harvey, D.J.; Watts, A.; Newsholme, E.A. The incorporation of fatty acids by lymphocytes and the effect on fatty acid composition and membrane fluidity. Biochem. J. 1994, 300, 509–518. [Google Scholar]
- Yaqoob, P.; Newsholme, E.A.; Calder, P.C. Influence of cell culture conditions on diet-induced changes in lymphocyte fatty acid composition. Biochimica et Biophysica Acta 1995, 1225, 333–340. [Google Scholar]
- Lokesh, B.R.; Hsieh, H.L.; Kinsella, J.E. Peritoneal macrophages from mice fed dietary (n-3) polyunsaturated fatty acids secrete low levels of prostaglandins. J. Nutr. 1986, 116, 2547–2552. [Google Scholar]
- Chapkin, R.S.; Akoh, C.C.; Lewis, R.E. Dietary fish oil modulation of in vivo peritoneal macrophage leukotriene production and phagocytosis. J. Nutr. Biochem. 1992, 3, 599–604. [Google Scholar] [CrossRef]
- Brouard, C.; Pascaud, M. Effects of moderate dietary supplementations with n-3 fatty acids on macrophage and lymphocyte phospholipids and macrophage eicosanoid synthesis in the rat. Biochimica et Biophysica Acta 1990, 1047, 19–28. [Google Scholar]
- Surette, M.E.; Whelan, J.; Lu, G.; Hardard'ottir, I.; Kinsella, J.E. Dietary n - 3 polyunsaturated fatty acids modify Syrian hamster platelet and macrophage phospholipid fatty acyl composition and eicosanoid synthesis: a controlled study. Biochimica et Biophysica Acta 1995, 1255, 185–191. [Google Scholar] [PubMed]
- Calder, P.C.; Bond, J.A.; Harvey, D.J.; Gordon, S.; Newsholme, E.A. Uptake and incorporation of saturated and unsaturated fatty acids into macrophage lipids and their effect upon macrophage adhesion and phagocytosis. Biochem. J. 1990, 269, 807–814. [Google Scholar]
- Fritsche, K.L.; Alexander, D.W.; Cassity, N.A.; Huang, S.C. Maternally-supplied fish oil alters piglet immune cell fatty acid profile and eicosanoid production. Lipids 1993, 28, 677–682. [Google Scholar]
- Palombo, J.D.; DeMichele, S.J.; Lydon, E.; Bistrian, B.R. Cyclic vs continuous enteral feeding with omega-3 and gamma-linolenic fatty acids: effects on modulation of phospholipid fatty acids in rat lung and liver immune cells. JPEN J. Parent. Enter. Nutr. 1997, 21, 123–132. [Google Scholar]
- Careaga-Houck, M.; Sprecher, H. Effect of a fish oil diet on the composition of rat neutrophil lipids and the molecular species of choline and ethanolamine glycerophospholipids. J. Lipid Res. 1989, 30, 77–87. [Google Scholar]
- James, M.J.; Cleland, L.G.; Gibson, R.A.; Hawkes, J.S. Interaction between fish and vegetable oils in relation to rat leucocyte leukotriene production. J. Nutr. 1991, 121, 631–637. [Google Scholar]
- de La Puerta Vázquez, R.; Martínez-Domínguez, E.; Sánchez Perona, J.; Ruiz-Gutiérrez, V. Effects of different dietary oils on inflammatory mediator generation and fatty acid composition in rat neutrophils. Metabolism 2004, 53, 59–65. [Google Scholar]
- Lee, T.H.; Hoover, R.L.; Williams, J.D.; Sperling, R.I.; Ravalese, J.; Spur, B.W.; Robinson, D.R.; Corey, E.J.; Lewis, R.A.; Austen, K.F. Effects of dietary enrichment with eicosapentaenoic acid and docosahexaenoic acid on in vitro neutrophil and monocyte leukotriene generation and neutrophil function. N. Engl. J. Med. 1985, 312, 1217–1224. [Google Scholar] [PubMed]
- Endres, S.; Ghorbani, R.; Kelley, V.E.; Georgilis, K.; Lonnemann, G.; van der Meer, J.M.W.; Cannon, J.G.; Rogers, T.S.; Klempner, M.S.; Weber, P.C.; Schaeffer, E.J.; Wolff, S.M.; Dinarello, C.A. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 1989, 320, 265–271. [Google Scholar]
- Sperling, R.I.; Benincaso, A.I.; Knoell, C.T.; Larkin, J.K.; Austen, K.F.; Robinson, D.R. Dietary ω-3 polyunsaturated fatty acids inhibit phosphoinositide formation and chemotaxis in neutrophils. J. Clin. Investig. 1993, 91, 651–960. [Google Scholar]
- Gibney, M.J.; Hunter, B. The effects of short- and long-term supplementation with fish oil on the incorporation of n-3 polyunsaturated fatty acids into cells of the immune system in healthy volunteers. Eur. J. Clin. Nutr. 1993, 47, 255–259. [Google Scholar]
- Caughey, G.E.; Mantzioris, E.; Gibson, R.A.; Cleland, L.G.; James, M.J. The effect on human tumor necrosis factor α and interleukin 1β production of diets enriched in n-3 fatty acids from vegetable oil or fish oil. Am. J. Clin. Nutr. 1996, 63, 116–122. [Google Scholar]
- Yaqoob, P.; Pala, H.S.; Cortina-Borja, M.; Newsholme, E.A.; Calder, P.C. Encapsulated fish oil enriched in α-tocopherol alters plasma phospholipid and mononuclear cell fatty acid compositions but not mononuclear cell functions. J. Clin. Investig. 2000, 30, 260–274. [Google Scholar]
- Healy, D.A.; Wallace, F.A.; Miles, E.A.; Calder, P.C.; Newsholme, P. The effect of low to moderate amounts of dietary fish oil on neutrophil lipid composition and function. Lipids 2000, 35, 763–768. [Google Scholar]
- Thies, F.; Nebe-von-Caron, G.; Powell, J.R.; Yaqoob, P.; Newsholme, E.A.; Calder, P.C. Dietary supplementation with γ-linolenic acid or fish oil decreases T lymphocyte proliferation in healthy older humans. J. Nutr. 2001, 131, 1918–1927. [Google Scholar]
- Kew, S.; Banerjee, T.; Minihane, A.M.; Finnegan, Y.E.; Williams, C.M.; Calder, P.C. Relation between the fatty acid composition of peripheral blood mononuclear cells and measures of immune cell function in healthy, free-living subjects aged 25–72 y. Am. J. Clin. Nutr. 2003, 77, 1278–1286. [Google Scholar]
- Miles, E.A.; Banerjee, T.; Calder, P.C. The influence of different combinations of gamma-linolenic, stearidonic and eicosapentaenoic acids on the fatty acid composition of blood lipids and mononuclear cells in human volunteers. Prostagland. Leuk. Essent. Fatty Acids 2004, 70, 529–538. [Google Scholar]
- Kew, S.; Mesa, M.D.; Tricon, S.; Buckley, R.; Minihane, A.M.; Yaqoob, P. Effects of oils rich in eicosapentaenoic and docosahexaenoic acids on immune cell composition and function in healthy humans. Am. J. Clin. Nutr. 2004, 79, 674–681. [Google Scholar]
- Rees, D.; Miles, E.A.; Banerjee, T.; Wells, S.J.; Roynette, C.E.; Wahle, K.W.J.W.; Calder, P.C. Dose-related effects of eicosapentaenoic acid on innate immune function in healthy humans: a comparison of young and older men. Am. J. Clin. Nutr. 2006, 83, 331–342. [Google Scholar]
- Yaqoob, P. The nutritional significance of lipid rafts. Annu. Rev. Nutr. 2009, 29, 257–282. [Google Scholar]
- Miles, E.A.; Calder, P.C. Modulation of immune function by dietary fatty acids. Proc. Nutr. Soc. 1998, 57, 277–292. [Google Scholar]
- Hughes, D.A.; Southon, S.; Pinder, A.C. (n-3) Polyunsaturated fatty acids modulate the expression of functionally associated molecules on human monocytes in vitro. J. Nutr. 1996, 126, 603–610. [Google Scholar] [PubMed]
- Babcock, T.A.; Novak, T.; Ong, E.; Jho, D.H.; Helton, W.S.; Espat, N.J. Modulation of lipopolysaccharide-stimulated macrophage tumor necrosis factor-α production by ω-3 fatty acid is associated with differential cyclooxygenase-2 protein expression and is independent of interleukin-10. J. Surg. Res. 2002, 107, 135–139. [Google Scholar]
- Lo, C.J.; Chiu, K.C.; Fu, M.; Lo, R.; Helton, S. Fish oil decreases macrophage tumor necrosis factor gene transcription by altering the NF kappa B activity. J. Surg. Res. 1999, 82, 216–221. [Google Scholar]
- Novak, T.E.; Babcock, T.A.; Jho, D.H.; Helton, W.S.; Espat, N.J. NF-kappa B inhibition by omega -3 fatty acids modulates LPS-stimulated macrophage TNF-alpha transcription. Am. J. Physiol. 2003, 284, L84–L89. [Google Scholar]
- Lo, C.J.; Chiu, K.C.; Fu, M.; Chu, A.; Helton, S. Fish oil modulates macrophage P44/P42 mitogen-activated protein kinase activity induced by lipopolysaccharide. JPEN 2000, 24, 159–163. [Google Scholar]
- Bates, E.J.; Ferrante, A.; Harvey, D.P.; Nandoskar, M.; Poulos, A. Docosahexanoic acid (22:6, n-3) but not eicosapentaenoic acid (20:5, n-3) can induce neutrophil-mediated injury of cultured endothelial cells: involvement of neutrophil elastase. J. Leukocyte Biol. 1993, 54, 590–598. [Google Scholar]
- Ferrante, A.; Goh, D.; Harvey, D.P.; Robinson, B.S.; Hii, C.S.; Bates, E.J.; Hardy, S.J.; Johnson, D.W.; Poulos, A. Neutrophil migration inhibitory properties of polyunsaturated fatty acids. The role of fatty acid structure, metabolism, and possible second messenger systems. J. Clin. Investig. 1994, 93, 1063–1070. [Google Scholar] [CrossRef]
- Ferrante, J.V.; Huang, Z.H.; Nandoskar, M.; Hii, C.S.; Robinson, B.S.; Rathjen, D.A.; Poulos, A.; Morris, C.P.; Ferrante, A. Altered responses of human macrophages to lipopolysaccharide by hydroperoxy eicosatetraenoic acid, hydroxy eicosatetraenoic acid, and arachidonic acid. Inhibition of tumor necrosis factor production. J. Clin. Investig. 1997, 99, 1445–1452. [Google Scholar] [CrossRef]
- Moghaddami, N.; Irvine, J.; Gao, X.; Grover, P.K.; Costabile, M.; Hii, C.S.; Ferrante, A. Novel action of n-3 polyunsaturated fatty acids: inhibition of arachidonic acid-induced increase in tumor necrosis factor receptor expression on neutrophils and a role for proteases. Arthritis Rheum. 2007, 56, 799–808. [Google Scholar]
- De Caterina, R.; Cybulsky, M.I.; Clinton, S.K.; Gimbrone, M.A.; Libby, P. The omega-3 fatty acid docosahexaenoate reduces cytokine-induced expression of proatherogenic and proinflammatory proteins in human endothelial cells. Arteriosclerosis Thromb. 1994, 14, 1829–1836. [Google Scholar]
- De Caterina, R.; Libby, P. Control of endothelial leukocyte adhesion molecules by fatty acids. Lipids 1996, 31, S57–S63. [Google Scholar]
- Collie-Duguid, E.S.; Wahle, K.W. Inhibitory effect of fish oil n-3 polyunsaturated fatty acids on the expression of endothelial cell adhesion molecules. Biochem. Biophys. Res. Commun. 1996, 220, 969–974. [Google Scholar]
- Ferrante, J.V.; Ferrante, A. Novel role of lipoxygenases in the inflammatory response: promotion of TNF mRNA decay by 15-hydroperoxyeicosatetraenoic acid in a monocytic cell line. J. Immunol. 2005, 174, 3169–3172. [Google Scholar]
- Sethi, S.; Ziouzenkova, O.; Ni, H.; Wagner, D.D.; Plutzky, J.; Mayadas, T.N. Oxidized omega-3 fatty acids in fish oil inhibit leukocyte-endothelial interactions through activation of PPAR alpha. Blood 2002, 100, 1340–1346. [Google Scholar]
- Mishra, A.; Chaudhary, A.; Sethi, S. Oxidized omega-3 fatty acids inhibit NF-kappaB activation via a PPARalpha-dependent pathway. Arteriosclerosis Thromb. Vasc. Biol. 2004, 24, 1621–1627. [Google Scholar]
- De Sanctis, J.B.; Blanca, I.; Bianco, N.E. Expression of different lipoprotein receptors in natural killer cells and their effect on natural killer proliferative and cytotoxic activity. Immunology 1995, 86, 399–407. [Google Scholar]
- Jeffery, N.M.; Yaqoob, P.; Wiggins, D.; Gibbons, G.F.; Newsholme, E.A.; Calder, P.C. Characterisation of lipoprotein composition in rats fed different dietary lipids and the effect of lipoproteins upon lymphocyte proliferation. J. Nutr. Biochem. 1996, 7, 282–292. [Google Scholar]
- Mahoney, E.M.; Khoo, J.C.; Steinberg, D. Lipoprotein lipase secretion by human monocytes and rabbit alveolar macrophages in culture. PNAS 1982, 79, 1639–1642. [Google Scholar]
- Calder, P.C.; Yaqoob, P.; Newsholme, E.A. Triacylglycerol metabolism by lymphocytes and the effect of triacylglycerols on lymphocyte proliferation. Biochem. J. 1994, 298, 605–611. [Google Scholar]
- Lewis, R.A.; Austen, K.F.; Soberman, R.J. Leukotrienes and other products of the 5-lipoxygenase pathway: biochemistry and relation to pathobiology in human diseases. N. Engl. J. Med. 1990, 323, 645–655. [Google Scholar]
- Tilley, S.L.; Coffman, T.M.; Koller, B.H. Mixed messages: modulation of inflammation and immune responses by prostaglandins and thromboxanes. J. Clin. Investig. 2001, 108, 15–23. [Google Scholar]
- Levy, B.D.; Clish, C.B.; Schmidt, B.; Gronert, K.; Serhan, C.N. Lipid mediator class switching during acute inflammation: signals in resolution. Nature Immunol. 2001, 2, 612–619. [Google Scholar]
- Vachier, I.; Chanez, P.; Bonnans, C.; Godard, P.; Bousquet, J.; Chavis, C. Endogenous anti-inflammatory mediators from arachidonate in human neutrophils. Biochem. Biophys. Res. Commun. 2002, 290, 219–224. [Google Scholar]
- Gewirtz, A.T.; Collier-Hyams, L.S.; Young, A.N.; Kucharzik, T.; Guilford, W.J.; Parkinson, J.F.; Williams, I.R.; Neish, A.S.; Madara, J.L. Lipoxin A4 analogs attenuate induction of intestinal epithelial proinflammatory gene expression and reduce the severity of dextran sodium sulfate-induced colitis. J. Immunol. 2002, 168, 5260–5267. [Google Scholar]
- Serhan, C.N.; Jain, A.; Marleau, S.; Clish, C.; Kantarci, A.; Behbehani, B.; Colgan, S.P.; Stahl, G.L.; Merched, A.; Petasis, N.A.; Chan, L.; Van Dyke, T.E. Reduced inflammation and tissue damage in transgenic rabbits overexpressing 15-lipoxygenase and endogenous anti-inflammatory lipid mediators. J. Immunol. 2003, 171, 6856–6865. [Google Scholar]
- Peterson, L.D.; Jeffery, N.M.; Thies, F.; Sanderson, P.; Newsholme, E.A.; Calder, P.C. Eicosapentaenoic and docosahexaenoic acids alter rat spleen leukocyte fatty acid composition and prostaglandin E2 production but have different effects on lymphocyte functions and cell-mediated immunity. Lipids 1998, 33, 171–180. [Google Scholar]
- Yaqoob, P.; Calder, P.C. Effects of dietary lipid manipulation upon inflammatory mediator production by murine macrophages. Cell. Immunol. 1995, 163, 120–128. [Google Scholar]
- Chapkin, R.S.; Akoh, C.C.; Miller, C.C. Influence of dietary n-3 fatty acids on macrophage glycerophospholipid molecular species and peptidoleukotriene synthesis. J. Lipid Res. 1991, 32, 1205–1213. [Google Scholar]
- Meydani, S.N.; Endres, S.; Woods, M.M,; Goldin, B.R.; Soo, C.; Morrill-Labrode, A.; Dinarello, C.; Gorbach, S.L. Oral (n-3) fatty acid supplementation suppresses cytokine production and lymphocyte proliferation: comparison between young and older women. J. Nutr. 1991, 121, 547–555. [Google Scholar] [PubMed]
- Von Schacky, C.; Kiefl, R.; Jendraschak, E.; Kaminski, W.E. N-3 fatty acids and cysteinyl-leukotriene formation in humans in vitro, ex vivo and in vivo. J. Lab. Clin. Med. 1993, 121, 302–309. [Google Scholar] [PubMed]
- Goldman, D.W.; Pickett, W.C.; Goetzl, E.J. Human neutrophil chemotactic and degranulating activities of leukotriene B5 (LTB5) derived from eicosapentaenoic acid. Biochem. Biophys. Res. Commun. 1983, 117, 282–288. [Google Scholar]
- Lee, T.H.; Mencia-Huerta, J.M.; Shih, C.; Corey, E.J.; Lewis, R.A.; Austen, K.F. Characterization and biologic properties of 5,12-dihydroxy derivatives of eicosapentaenoic acid, including leukotriene-B5 and the double lipoxygenase product. J. Biol. Chem. 1984, 259, 2383–2389. [Google Scholar]
- Tull, S.P.; Yates, C.M.; Maskrey, B.H.; O'Donnell, V.B.; Madden, J.; Grimble, R.F.; Calder, P.C.; Nash, G.B.; Rainger, G.E. Omega-3 Fatty acids and inflammation: novel interactions reveal a new step in neutrophil recruitment. PLoS Biol. 2009, 7, e1000177. [Google Scholar]
- Dooper, M.M.B.W.; Wassink, L.; M’Rabet, L.; Graus, Y.M.F. The modulatory effects of prostaglandin-E on cytokine production by human peripheral blood mononuclear cells are independent of the prostaglandin subtype. Immunology 2002, 107, 152–159. [Google Scholar]
- Miles, E.A.; Allen, E.; Calder, P.C. In vitro effects of eicosanoids derived from different 20-carbon fatty acids on production of monocyte-derived cytokines in human whole blood cultures. Cytokine 2002, 20, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Clish, C.B.; Brannon, J.; Colgan, S.P.; Chiang, N.; Gronert, K. Novel functional sets of lipid-derived mediators with antinflammatory actions generated from omega-3 fatty acids via cyclooxygenase 2-nonsteroidal antiinflammatory drugs and transcellular processing. J. Exp. Med. 2000, 192, 1197–1204. [Google Scholar]
- Serhan, C.N.; Hong, S.; Gronert, K.; Colgan, S.P.; Devchand, P.R.; Mirick, G.; Moussignac, R-L. Resolvins: a family of bioactive products of omega-3 fatty acid transformation circuits initiated by aspirin treatment that counter pro-inflammation signals. J. Exp. Med. 2002, 196, 1025–1037. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N; Chiang, N.; van Dyke, T.E. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nature Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef]
- Schmidt, E.B.; Pedersen, J.O.; Ekelund, S.; Grunnet, N.; Jersild, C.; Dyerberg, J. Cod liver oil inhibits neutrophil and monocyte chemotaxis in healthy males. Atherosclerosis 1989, 77, 53–57. [Google Scholar]
- Schmidt, E.B.; Varming, K.; Perdersen, J.O.; Lervang, H.H.; Grunnet, N.; Jersild, C.; Dyerberg, J. Long term supplementation with n-3 fatty acids. ii. Effect on neutrophil and monocyte chemotaxis. Scandanavian J. Clin. Lab. Investig. 1992, 52, 229–236. [Google Scholar] [CrossRef]
- Luostarinen, R.; Siegbahn, A.; Saldeen, T. Effect of dietary fish oil supplemented with different doses of vitamin E on neutrophil chemotaxis in healthy volunteers. Nutr. Res. 1992, 12, 1419–1430. [Google Scholar]
- Schmidt, E.B.; Pedersen, J.O.; Varming, K.; Ernst, E.; Jersild, C.; Grunnet, N.; Dyerberg, J. N-3 fatty acids and leukocyte chemotaxis: effects in hyperlipidemia, and dose-response studies in healthy males. Arteriosclerosis Thromb. 1991, 11, 429–435. [Google Scholar] [CrossRef]
- Schmidt, E.B.; Varming, K.; Moller, J.M.; Bulow Pederson, I.; Madsen, P.; Dyerberg, J. No effect of a very low dose of n-3 fatty acids on monocyte function in healthy humans. Scandinavian J. Clin. Investig. 1996, 56, 87–92. [Google Scholar]
- Hill, A.M.; Worthley, C.; Murphy, K.J.; Buckley, J.D.; Ferrante, A.; Howe, P.R. n-3 Fatty acid supplementation and regular moderate exercise: differential effects of a combined intervention on neutrophil function. Brit. J. Nutr. 2007, 98, 300–309. [Google Scholar]
- Miles, E.A.; Wallace, F.A.; Calder, P.C. Dietary fish oil reduces intercellular adhesion molecule 1 and scavenger receptor expression on murine macrophages. Atherosclerosis 2000, 152, 43–50. [Google Scholar]
- Sanderson, P.; Calder, P.C. Dietary fish oil diminishes lymphocyte adhesion to macrophage and endothelial cell monolayers. Immunology 1998, 94, 79–87. [Google Scholar]
- Hughes, D.A.; Pinder, A.C.; Piper, Z.; Johnson, I.T.; Lund, E.K. Fish oil supplementation inhibits the expression of major histocompatibility complex class II molecules and adhesion molecules on human monocytes. Am. J. Clin. Nutr. 1996, 63, 267–272. [Google Scholar]
- Miles, E.A.; Thies, F.; Wallace, F.A.; Powell, J.R.; Hirst, T.L.; Newsholme, E.A.; Calder, P.C. Influence of age and dietary fish oil on plasma soluble adhesion molecule concentrations. Clin. Sci. 2001, 100, 91–100. [Google Scholar]
- Kumar, A.; Takada, Y.; Boriek, A.M.; Aggarwal, B.B. Nuclear factor-kappaB: its role in health and disease. J. Mol. Med. 2004, 82, 434–448. [Google Scholar]
- Sigal, L.H. Basic science for the clinician 39: NF-kappaB-function, activation, control, and consequences. J. Clin. Rheumatol. 2006, 12, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Perkins, N.D. Integrating cell-signalling pathways with NF-kappaB and IκK function. Nature Rev. Mol. Cell Biol. 2007, 8, 49–62. [Google Scholar]
- Van den Berghe, W.; Vermeulen, L.; Delerive, P.; De Bosscher, K.; Staels, B.; Haegeman, G. A paradigm for gene regulation: inflammation, NF-kappaB and PPAR. Adv. Exp. Med. Biol. 2003, 544, 181–196. [Google Scholar]
- Khalfoun, B.; Thibault, F.; Watier, H.; Bardos, P.; Lebranchu, Y. Docosahexaenoic and eicosapentaenoic acids inhibit in vitro human endothelial cell production of interleukin-6. Adv. Exp. Med. Biol. 1997, 400, 589–597. [Google Scholar]
- Xi, S.; Cohen, D.; Barve, S.; Chen, L.H. Fish oil suppressed cytokines and nuclear factor κB induced by murine AIDS virus infection. Nutr. Res. 2001, 21, 865–878. [Google Scholar]
- Billiar, T.; Bankey, P.; Svingen, B.; Curran, R.D.; West, M.A.; Holman, R.T.; Simmons, R.L.; Cerra, F.B. Fatty acid uptake and Kupffer Cell function: fish oil alters eicosanoid and monokine production to endotoxin stimulation. Surgery 1988, 104, 343–349. [Google Scholar]
- Renier, G.; Skamene, E.; de Sanctis, J.; Radzioch, D. Dietary n-3 polyunsaturated fatty acids prevent the development of atherosclerotic lesions in mice: modulation of macrophage secretory activities. Arteriosclerosis Thomb. 1993, 13, 1515–1524. [Google Scholar]
- Baumann, K.H.; Hessel, F.; Larass, I.; Muller, T.; Angerer, P.; Kiefl, R.; von Schacky, C. Dietary ω-3, ω-6, and ω-9 unsaturated fatty acids and growth factor and cytokine gene expression in unstimulated and stimulated monocytes. Arteriosclerosis Thromb. Vascular Biol. 1999, 19, 59–66. [Google Scholar] [CrossRef]
- Trebble, T.; Arden, N.K.; Stroud, M.A.; Wootton, S.A.; Burdge, G.C.; Miles, E.A.; Ballinger, A.B.; Thompson, R.L.; Calder, P.C. Inhibition of tumour necrosis factor-α and interleukin-6 production by mononuclear cells following dietary fish-oil supplementation in healthy men and response to antioxidant co-supplementation. Brit. J. Nutr. 2003, 90, 405–412. [Google Scholar]
- Abbate, R.; Gori, A.M.; Martini, F.; Brunelli, T.; Filippini, M.; Francalanci, I.; Paniccia, R.; Prisco, D.; Gensini, G.F.; Serneri, G.G.N. N-3 PUFA supplementation, monocyte PCA expression and interleukin-6 production. Prostagland. Leuk. Essent. Fatty Acids 1996, 54, 439–444. [Google Scholar]
- Thies, F.; Miles, E.A.; Nebe-von-Caron, G.; Powell, J.R.; Hurst, T.L.; Newsholme, E.A.; Calder, P.C. Influence of dietary supplementation with long chain n-3 or n-6 polyunsaturated fatty acids on blood inflammatory cell populations and functions and on plasma soluble adhesion molecules in healthy adults. Lipids 2001, 36, 1183–1193. [Google Scholar]
- Kew, S.; Banerjee, T.; Minihane, A.M.; Finnegan, Y.E.; Muggli, R.; Albers, R.; Williams, C.M.; Calder, P.C. Lack of effect of foods enriched with plant- or marine-derived n-3 fatty acids on human immune function. Am. J. Clin. Nutr. 2003, 77, 1287–1295. [Google Scholar]
- Cooper, A.L.; Gibbins, L.; Horan, M.A.; Little, R.A.; Rothwell, N.J. Effect of dietary fish oil supplementation on fever and cytokine production in human volunteers. Clin. Nutr. 1993, 12, 321–328. [Google Scholar]
- Wallace, F.A.; Miles, E.A.; Calder, P.C. Comparison of the effects of linseed oil and different doses of fish oil on mononuclear cell function in healthy human subjects. Brit. J. Nutr. 2003, 89, 679–689. [Google Scholar]
- Miles, E.A.; Banerjee, T.; Dooper, M.W.B.W.; M'Rabet, L.; Graus, Y.M.F.; Calder, P.C. The influence of different combinations of γ-linolenic acid, stearidonic acid and EPA on immune function in healthy young male subjects. Brit. J. Nutr. 2004, 91, 893–903. [Google Scholar] [CrossRef]
- Blok, W.L.; Deslypere, J-P.; Demacker, P.N.M.; van der Ven-Jonggekrijg, J.; Hectors, M.P.C.; van der Meer, J.M.W.; Katan, M.B. Pro- and anti-inflammatory cytokines in healthy volunteers fed various doses of fish oil for 1 year. Eur. J. Clin. Investig. 1997, 27, 1003–1008. [Google Scholar]
- Cannon, J.G.; Fiatarone, M.A.; Meydani, M.; Gong, J.; Scott, L.; Blumberg, J.B.; Evans, W.J. Aging and dietary modulation of elastase and interleukin-2 beta secretion. Am. J. Physiol. 1995, 268, R208–R213. [Google Scholar]
- Molvig, J.; Pociot, F.; Worsaae, H.; Wogensen, L.D.; Baek, L.; Christensen, P.; Mandruppoulsen, T.; Andersen, K.; Madsen, P.; Dyerberg, J.; Nerup, J. Dietary supplementation with omega 3 polyunsaturated fatty acids decreases mononuclear cell proliferation and interleukin 1 beta content but not monokine secretion in healthy and insulin dependent diabetic individuals. Scand. J. Immunol. 1991, 34, 399–410. [Google Scholar]
- Calder, P.C. n-3 Polyunsaturated fatty acids, inflammation and immunity: pouring oil on troubled waters or another fishy tale? Nutr. Res. 2001, 21, 309–341. [Google Scholar] [CrossRef]
- Grimble, R.F.; Howell, W.M.; O'Reilly, G.; Turner, S.J.; Markovic, O.; Hirrell, S.; East, J.M.; Calder, P.C. The ability of fish oil to suppress tumor necrosis factor-α production by peripheral blood mononuclear cells in healthy men is associated with polymorphisms in genes that influence tumor necrosis factor α production. Am. J. Clin. Nutr. 2002, 76, 454–459. [Google Scholar]
- Calder, P.C. PUFA, inflammatory processes and rheumatoid arthritis. Proc. Nutr. Soc. 2008, 67, 409–418. [Google Scholar]
- Fortin, P.R.; Lew, R.A.; Liang, M.H.; Wright, E.A.; Beckett, L.A.; Chalmers, T.C.; Sperling, R.I. Validation of a meta-analysis: the effects of fish oil in rheumatoid arthritis. J. Clin. Epidemiol. 1995, 48, 1379–1390. [Google Scholar]
- Goldberg, R.J.; Katz, J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain 2007, 129, 210–223. [Google Scholar]
- Calder, P.C. Polyunsaturated fatty acids, inflammatory processes and inflammatory bowel diseases. Mol. Nutr. Food Res. 2008, 52, 885–897. [Google Scholar]
- Calder, P.C. Fatty acids and immune function: relevance to inflammatory bowel diseases. Int. Rev. Immunol. 2009, 28, 506–534. [Google Scholar]
- Kremmyda, L.S.; Vlachava, M.; Noakes, P.S.; Diaper, N.D.; Miles, E.A.; Calder, P.C. Atopy risk in infants and children in relation to early exposure to fish, oily fish, or long-chain omega-3 fatty acids: A systematic review. Clin. Rev. Allergy and Immunol. 2009. [Google Scholar] [CrossRef]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Maternal fish oil supplementation in pregnancy reduces interleukin-13 levels in cord blood of infants at high risk of atopy. Clin. Exp. Allergy 2003, 33, 442–448. [Google Scholar] [PubMed]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Fish oil supplementation in pregnancy modifies neonatal allergen-specific immune responses and clinical outcomes in infants at high risk of atopy: a randomized, controlled trial. J. Allergy Clin. Immunol. 2003, 112, 1178–1184. [Google Scholar]
- Krauss-Etschmann, S.; Hartl, D.; Rzehak, P.; Heinrich, J.; Shadid, R.; Del Carmen Ramírez-Tortosa, M.; Campoy, C.; Pardillo, S.; Schendel, D.J.; Decsi, T.; Demmelmair, H.; Koletzko, B.V. Nutraceuticals for Healthier Life Study Group. Decreased cord blood IL-4, IL-13, and CCR4 and increased TGF-beta levels after fish oil supplementation of pregnant women. J. Allergy Clin. Immunol. 2008, 121, 464–470. [Google Scholar] [PubMed]
- Olsen, S.F.; Østerdal, M.L.; Salvig, J.D.; Mortensen, L.M.; Rytter, D.; Secher, N.J.; Henriksen, T.B. Fish oil intake compared with olive oil intake in late pregnancy and asthma in the offspring: 16 y of registry-based follow-up from a randomized controlled trial. Am. J. Clin. Nutr. 2008, 88, 167–175. [Google Scholar] [PubMed]
- Sala-Vila, A.; Miles, E.A.; Calder, P.C. Fatty acid composition abnormalities in atopic disease: evidence explored and role in the disease process examined. Clin. Exp. Allergy 2008, 38, 1432–1450. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Krauss-Etschmann, S.; de Jong, E.C.; Dupont, C.; Frick, J-S.; Frokiaer, H.; Garn, H.; Koletzko, S.; Lack, G.; Mattelio, G.; Renz, H.; Sangild, P.T.; Schrezenmeir, J.; Stulnig, T.M.; Thymann, T.; Wold, A.E.; Koletzko, B. Workshop Report: Early nutrition and immunity – progress and perspectives. Brit. J. Nutr. 2006, 96, 774–790. [Google Scholar]
- Calder, P.C. N-3 fatty acids and cardiovascular disease: evidence explained and mechanisms explored. Clin. Sci. 2004, 107, 1–11. [Google Scholar]
- Calder, P.C.; Yaqoob, P. Omega-3 (n-3) fatty acids, cardiovascular disease and stability of atherosclerotic plaques. Cell. Mol. Biol. 2010, 56, 28–37. [Google Scholar]
- Ross, R. Mechanisms of disease - Atherosclerosis - An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar]
- Glass, C.K.; Witztum, J.L. Atherosclerosis: The road ahead. Cell 2001, 104, 503–516. [Google Scholar]
- Bucher, H.C.; Hengstler, P.; Schindler, C.; Meier, G. N-3 polyunsaturated fatty acids in coronary heart disease: a meta-analysis of randomized controlled trials. Am. J. Med. 2002, 112, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Studer, M.; Briel, M.; Leimenstoll, B.; Glass, T.R.; Bucher, H.C. Effect of different antilipidemic agents and diets on mortality: a systemic review. Arch. Intern. Med. 2005, 165, 725–730. [Google Scholar]
- Thies, F.; Garry, J.M.C.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: a randomised controlled trial. Lancet 2003, 361, 477–485. [Google Scholar]
- Plutzky, J. Atherosclerotic plaque rupture: emerging insights and opportunities. Am. J. Cardiol. 1999, 84, 15J–20J. [Google Scholar]
- Kremer, J.M.; Lawrence, D.A.; Jubiz, W.; DiGiacomo, R.; Rynes, R.; Bartholomew, L.E.; Sherman, M. Dietary fish oil and olive oil supplementation in patients with rheumatoid arthritis. Arthritis Rheum. 1990, 33, 810–820. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Calder, P.C. Omega-3 Fatty Acids and Inflammatory Processes. Nutrients 2010, 2, 355-374. https://doi.org/10.3390/nu2030355
Calder PC. Omega-3 Fatty Acids and Inflammatory Processes. Nutrients. 2010; 2(3):355-374. https://doi.org/10.3390/nu2030355
Chicago/Turabian StyleCalder, Philip C. 2010. "Omega-3 Fatty Acids and Inflammatory Processes" Nutrients 2, no. 3: 355-374. https://doi.org/10.3390/nu2030355
APA StyleCalder, P. C. (2010). Omega-3 Fatty Acids and Inflammatory Processes. Nutrients, 2(3), 355-374. https://doi.org/10.3390/nu2030355




