1. Introduction
Cardiac glycosides are found in plants worldwide and based on their chemical structure are classified as either bufadienolides or cardenolides. Bufadienolide-containing plants are considered as one of the greatest causes of plant-associated poisoning of livestock in South Africa [
1]. Bufadienolides are C-24 steroids with a double unsaturated six-membered lactone ring attached to C-17β. Bufadienolides can be further divided into non-cumulative and cumulative bufadienolides [
2]. Depending on the type and dose of the bufadienolide-containing plant ingested by the animal, poisoning can manifest as either acute or chronic intoxication.
Non-cumulative bufadienolides cause acute cardiac glycoside poisoning in animals, which affects the cardiovascular, gastrointestinal, respiratory and nervous systems. These non-cumulative bufadienolides can be found in plants belonging to the genera
Moraea and
Drimia, among others.
Moraea pallida (yellow tulip) is well known for causing livestock poisoning and contains the non-cumulative bufadienolide 1α,2α-epoxyscillirosidine as its main toxic principle [
3].
In contrast, cumulative bufadienolides can cause either acute or chronic intoxication depending on the dose and are present in the plants of the family Crassulaceae (‘plakkies’), specifically from the genera
Tylecodon,
Cotyledon and
Kalanchoe [
2]. Although bufadienolide-containing plants have a worldwide distribution, plants containing cumulative bufadienolides have, to date, only been reported in Southern Africa. During chronic intoxication the clinical signs associated with the cardiovascular, gastrointestinal and respiratory systems are less apparent, while the nervous signs predominate. The chronic form of intoxication is a paretic condition known colloquially as ‘krimpsiekte’ and occurs most often in small stock [
1]. Affected animals assume a characteristic posture, from which the name ‘krimpsiekte’ is derived, by standing with their back arched, feet together and head bowed down. Lanceotoxin B is a cumulative bufadienolide, isolated from
Kalanchoe lanceolata, reportedly causing krimpsiekte in sheep and has a subcutaneous LD
50 (half maximal lethal dose) of 0.10 mg/kg in guinea-pigs [
4].
All cardiac glycosides, including bufadienolides, act by inhibiting the Na
+/K
+-ATPase (sodium/potassium-adenosine triphosphatase) causing an increase in the intracellular sodium concentration of the cell and subsequently indirectly inhibiting the Na
+/Ca
2+-exchanger. The ability of the Na
+/K
+-ATPase to act as a scaffold protein that regulates downstream effectors, as well as the increase in intracellular calcium, allow the cardiac glycoside to trigger various cell signaling pathways [
5]. These pathways then proceed to influence cellular functions such as proliferation, cell death mechanisms, protein synthesis and cellular metabolism [
5], and even cellular attachment [
6,
7]. In addition, it was proposed that cumulative bufadienolides can bind to the nicotinic acetylcholine receptors of the neuromuscular junction and that this is what causes the neuromuscular dysfunction [
8]. Based on the cholinergic activity, it was hypothesized that cumulative bufadienolides occupy the neuromuscular receptors, decreasing their functionality. With continuous discharge, the acetylcholine stores of the motor nerve terminal are eventually depleted, and the remaining receptors are occupied with acetylcholine, desensitizing the receptors to further stimulation; as a result, fatigue sets in. This leads to the myasthenia gravis-like weakness seen in affected animals. Chronic intoxication with the cumulative bufadienolide, cotyledoside, has been shown to cause lesions in the white matter of thalamic nuclei, explaining some of the motor function deficiencies seen in affected animals [
9].
The objective of this study was to investigate the in vitro cytotoxicity induced by 1α,2α-epoxyscillirosidine and lanceotoxin B on rat myocardial (H9c2) and mouse neuroblastoma (Neuro-2a) cell lines, to lend credence to the premise that cumulative bufadienolides are neurotoxic. Additionally, the ultrastructural changes caused by the non-cumulative and cumulative bufadienolide respectively, was investigated using electron microscopy.
3. Discussion
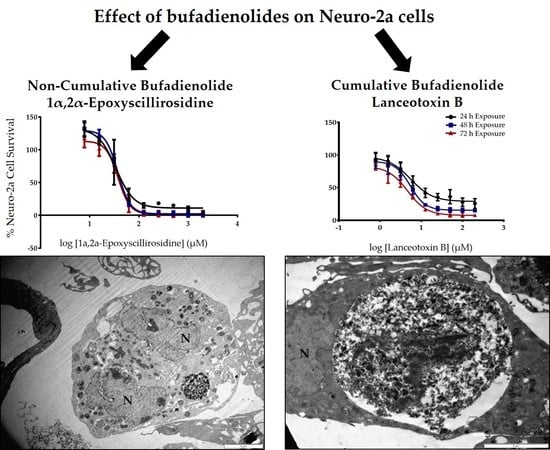
Due to the paretic/paralytic nature of chronic intoxication in small stock, the cumulative bufadienolides are often referred to as neurotoxic. We thus compared the
in vitro cytotoxicity of a cumulative bufadienolide, i.e., lanceotoxin B with a non-cumulative bufadienolide, 1α,2α-epoxyscillirosidine on both myocardial (H9c2) and neuroblastoma (Neuro-2a) cell lines (
Table 1). The H9c2 cells were more susceptible to 1α,2α-epoxyscillirosidine with the EC
50 of lanceotoxin B being more than five times greater than that of 1α,2α-epoxyscillirosidine after 24 h exposure. In contrast, lanceotoxin B had EC
50s below 6 µM at all three exposure times when Neuro-2a cells were assessed, also being much lower than the EC
50s of 1α,2α-epoxyscillirosidine, ranging from 35.7 to 37.6 µM. These results support the theory that lanceotoxin B is neurotoxic, as lanceotoxin B induced greater cytotoxicity towards nerve cells. The neurotoxicity of the cumulative bufadienolides are believed to be due to their stereochemistry that differs from that of non-cumulative bufadienolides and cardenolides [
10]. Unlike the other cardiac glycosides, cumulative bufadienolides have a levorotatory sugar strongly attached to the C3 position of the aglycone that cannot be removed by acid hydrolysis [
10].
Of note, low concentrations of 1α,2α-epoxyscillirosidine had a positive effect on cell survival of both the H9c2 and Neuro-2a cell lines, showing a possible hormetic effect. Similar results have previously been reported when using low concentrations of the cardenolide, ouabain [
11,
12]. The toxicity of the bufadienolides falling within the micromolar range was not unexpected as rodent cells are less susceptible to cardiac glycosides compared to other animals [
13,
14].
The ultrastructural changes observed in both H9c2 and Neuro-2a cells exposed to 1α,2α-epoxyscillirosidine included swollen Golgi complexes, RER and enlarged perinuclear space (
Figure 3 and
Figure 5). These swollen organelles can be ascribed to the disruption of the ionic homeostasis of the cells caused by the bufadienolide. The RER and perinuclear space act as a storage site for intracellular calcium. The increased concentration of intracellular Na
+ and Ca
2+ ions are accompanied by the influx of water, causing the organelles to swell. In addition to mitochondrial damage, 1α,2α-epoxyscillirosidine caused some of the mitochondrial cristae to become slightly ballooned (
Figure 5e), while the mitochondrial matrix remained unchanged. The inner and outer mitochondrial membranes have different permeability capacities thus causing the cristae and matrix to swell separately or subsequently. Exposure to 1α,2α-epoxyscillirosidine also caused large scale vacuolation of the cytoplasm (
Figure 3 and
Figure 5), also possibly due to the disruption of the ion homeostasis of the cell. These vacuoles seem to originate from the Golgi complexes and the RER. The swollen RER, in addition to the dissociated ribosomes clumping within the cytoplasm, suggested that protein synthesis was disrupted.
Lanceotoxin B caused the Golgi complexes of both H9c2 and Neuro-2a cells to swell, also conceivably due to the disruption of the sodium, potassium and calcium homeostasis of the cells. However, in contrast to cells exposed to 1α,2α-epoxyscillirosidine, the RER of the H9c2 cells remained unaffected. The RER and nuclei of the Neuro-2a cells also remained mostly unaffected. The mitochondrial matrix of both H9c2 and Neuro-2a cells became grossly swollen after exposure to lanceotoxin B (
Figure 4 and
Figure 6). The swelling, as mentioned above, is due to the influx of water that accompanies the increased ion concentrations. The cytoskeleton associated with the plasma membrane of the H9c2 cells were also affected. Calcium activates different proteases, and these can subsequently degrade the cytoskeleton [
15]. Lanceotoxin B caused a noticeable increase in the number of autophagic vesicles within the cytoplasm of affected cells. This was especially apparent in Neuro-2a cells, with large parts of the cells sequestered (
Figure 6). Autophagy has historically been classified as a type of cell death, however its role in cell death is often misinterpreted [
16]. Cells induce autophagy as a survival mechanism and often cell death is cell death with autophagy opposed to cell death by autophagy. Autophagy and neuronal cell death have previously been linked [
17,
18], thus corroborating the contention that lanceotoxin B is neurotoxic. The H9c2 cells exposed to lanceotoxin B were apoptotic, with shrunken, rounded cells; many with membrane blebs or what appears to be apoptotic bodies. In contrast, Neuro-2a cells exposed to lanceotoxin B, besides the extensive autophagy, also showed signs of both apoptosis and necrosis. The mixed morphological signs of cell death could be as a result of the various cellular pathways disrupted by the bufadienolides.
In future, molecular studies can be performed to clarify the type of cell death caused by these cumulative bufadienolides which will contribute to our understanding of the mechanism of toxicity underlying ‘krimpsiekte’. In addition, investigating the ultrastructural changes in appropriate cells from poisoned animals and cells believed to not be affected by these bufadienolides could be of interest.
4. Materials and Methods
4.1. Cell Cultures
Rat myocardial (H9c2(2-1) (ATCC® CRL-1446™)) and mouse neuroblastoma (Neuro-2a (ATCC® CRL-131™)) cell lines were obtained from the ATCC (American Type Culture Collection). The cultures were grown in HyClone DMEM (Dulbecco’s Modified Eagle’s Medium)-High Glucose, supplemented with 4 mM glutamine, 1 mM sodium pyruvate, 10% fetal calf serum (Gibco, Life technologies, Carlsbad, CA, USA), 100 U/mL penicillin and 100 U/mL streptomycin (Lonza, Verviers, Belgium). The cells were maintained in an incubator at 37 °C and a humidified atmosphere of 5% CO2.
4.2. Toxins
1α,2α-Epoxyscillirosidine and lanceotoxin B (>95% purity) used in this study were previously isolated from M. pallida and K. lanceolata respectively and were preserved in dried form and stored in a secure place as part of the natural toxin collection of the Department of Paraclinical Sciences, Faculty of Veterinary Science, Onderstepoort. The stock solutions of 1α,2α-epoxyscillirosidine were prepared in a 1:1 organic solvent-complete DMEM media mixture using acetone, while stock solutions for lanceotoxin B were prepared in DMSO (dimethyl sulfoxide).
4.3. Cell Survival Assays
The percentage cell survival of H9c2 and Neuro-2a cells after exposure to either 1α,2α-epoxyscillirosidine or lanceotoxin B was determined using the MTT assay [
19]. The cells were seeded into a 96 well plates at a concentration of 5000 cells/well for the Neuro-2a cells and 10,000 cells/well for the H9c2 cells, respectively in 200 µL complete media per well, 24 h prior to the commencement of the exposure study. The cells were exposed to a 2× serial dilution of 1α,2α-epoxyscillirosidine and lanceotoxin B for 24, 48 and 72 h. The percentage solvent was kept constant for all dilutions at 0.5% solvent/well. After termination of the exposure studies and following rinsing of the wells with 200 µL PBS (phosphate buffered saline), pH 7.4, 20 µL 0.005 g/mL Thiazolyl Blue Tetrazolium Bromide dissolved in PBS (Sigma-Aldrich, St. Louis, MO, USA) and 200 µL complete DMEM media were added to each well. The plates were then incubated in the dark at 37 °C for 2 h, the MTT medium replaced with 100 µL DMSO and shaken for 5 min in the dark to solubilize the MTT formazan crystals. The absorbance of the MTT product was measured at 570 nm and the background at 630 nm using a Synergy HT BioTek microplate reader (BioTek Instruments, Inc., Winooski, VT, USA).
4.4. Statistical Analysis
Data were analyzed using Microsoft Excel (Office 365, Microsoft, Redmond, WA, USA) and GraphPad Prism (Version 6.0, GraphPad Prism Software Inc., La Jolla, CA, USA). All repeats of the assays were grouped together, and the outliers removed. The data was tested for compliance with normality and homogeneous variance using D’Agostino and Pearson omnibus normality tests and Bartlett’s test, respectively. An ANOVA (factorial analysis of variance) was used to see if there were significant differences (p > 0.05) between the EC50s at the different exposure periods; and in the case that there were, Student’s t-test was used. The 95% confidence intervals were calculated by the following equation:
95% CI = Average cell survival at highest concentration of lanceotoxin B ± (1.96× Standard error of the mean).
4.5. Transmission Electron Microscopy
The ultrastructural changes induced by the toxins on H9c2 and Neuro-2a cells were investigated using TEM. H9c2 and Neuro-2a cells were exposed to 5, 25 and 100 µM 1α,2α-epoxyscillirosidine and lanceotoxin B for 24, 48 and 72 h. After termination of the exposure, the cells were fixed with 2.5% glutaraldehyde in 0.0075 M sodium phosphate buffer (pH 7.4) for 1 h. The cells were scraped off from the plate, centrifuged at 950 g and rinsed three times for 10 min with 0.075 M phosphate buffer. The cells were post-fixed with osmium tetroxide and rinsed with the phosphate buffer for 10 min. The cells were serially dehydrated in 30%, 50%, 70%, 90% and three times in 100% ethanol. The samples were then imbedded in TAAB 812 epoxy resin [
20] and sectioned using an ultra-microtome (Leica EM UC7). Each section was contrasted with 2% aqueous uranyl acetate for 10 min and lead citrate for 2 min [
21]. The sections were examined using a Philips CM10 Transmission Electron Microscope (Philips, Amsterdam, The Netherlands).