Programmed Cell Death-Like and Accompanying Release of Microcystin in Freshwater Bloom-Forming Cyanobacterium Microcystis: From Identification to Ecological Relevance
Abstract
1. Introduction
2. Detection of Programmed Death in Microcystis
2.1. Transmission Electronic Microscopy (TEM)
2.2. Gel Electrophoresis
2.3. Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling (TUNEL) Assay
2.4. Hoechst Staining Assay
2.5. Caspase Assay
2.6. Annexin V Staining Assay
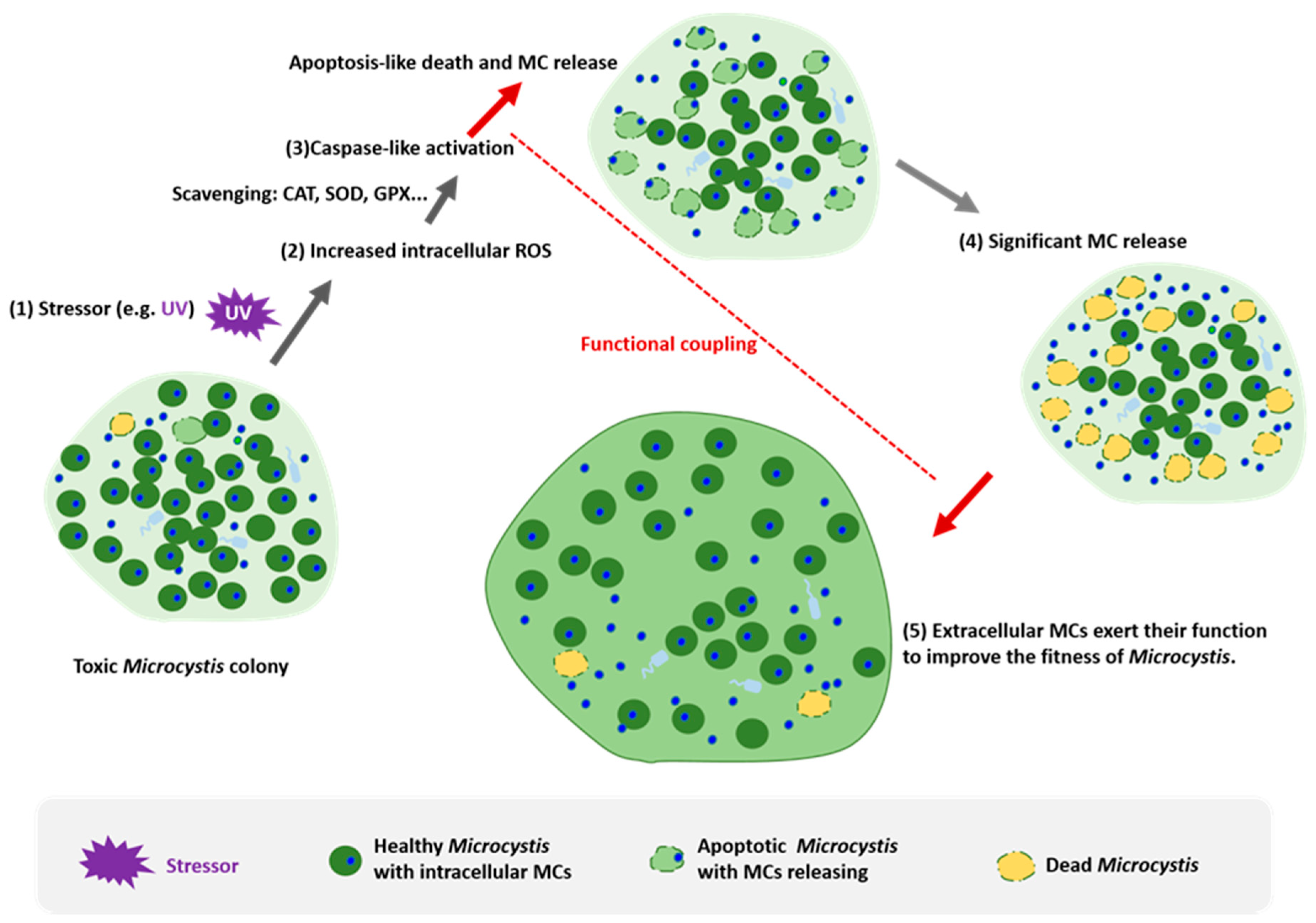
3. The Mechanisms of Programmed Death in Microcystis
4. The Functional Coupling between Apoptosis and Microcystin in Microcystis
5. Future Research Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Harke, M.J.; Steffen, M.M.; Gobler, C.J.; Otten, T.G.; Wilhelm, S.W.; Wood, S.A.; Paerl, H.W. A review of the global ecology, genomics, and biogeography of the toxic cyanobacterium, Microcystis spp. Harmful Algae 2016, 54, 4–20. [Google Scholar] [CrossRef] [PubMed]
- Brunberg, A.K.; Blomqvist, P. Benthic overwintering of Microcystis colonies under different environmental conditions. J. Plankton Res. 2002, 24, 1247–1252. [Google Scholar] [CrossRef]
- Bridgeman, T.B.; Chaffin, J.D.; Filbrun, J.E. A novel method for tracking western Lake Erie Microcystis blooms, 2002–2011. J. Great Lakes Res. 2013, 39, 83–89. [Google Scholar] [CrossRef]
- Hisbergues, M.; Christiansen, G.; Rouhiainen, L.; Sivonen, K.; Borner, T. PCR-based identification of microcystin-producing genotypes of different cyanobacterial genera. Arch. Microbiol. 2003, 180, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Mackintosh, C.; Beattie, K.A.; Klumpp, S.; Cohen, P.; Codd, G.A. Cyanobacterial Microcystin-Lr is a Potent and Specific Inhibitor of Protein Phosphatase-1 and Phosphatase-2a from Both Mammals and Higher-Plants. FEBS Lett. 1990, 264, 187–192. [Google Scholar] [CrossRef]
- Massey, I.Y.; Yang, F.; Ding, Z.; Yang, S.; Guo, J.; Tezi, C.; Al-Osman, M.; Kamegni, R.B.; Zeng, W.M. Exposure routes and health effects of microcystins on animals and humans: A mini-review. Toxicon 2018, 151, 156–162. [Google Scholar] [CrossRef]
- Nishiwakimatsushima, R.; Nishiwaki, S.; Ohta, T.; Yoshizawa, S.; Suganuma, M.; Harada, K.; Watanabe, M.F.; Fujiki, H. Structure-Function-Relationships of Microcystins, Liver-Tumor Promoters, in Interaction with Protein Phosphatase. Jpn. J. Cancer Res. 1991, 82, 993–996. [Google Scholar] [CrossRef]
- Panou, M.; Zervou, S.K.; Kaloudis, T.; Hiskia, A.; Gkelis, S. A Greek Cylindrospermopsis raciborskii strain: Missing link in tropic invader’s phylogeography tale. Harmful Algae 2018, 80, 96–106. [Google Scholar] [CrossRef]
- Spoof, L.; Catherine, A. Appendix 3: Tables of Microcystins and Nodularins. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Codd, G.A., Eds.; Wiley Online Library: Hoboken, NJ, USA, 2016; pp. 526–537. [Google Scholar] [CrossRef]
- Hu, C.L.; Gan, N.Q.; Chen, Y.Y.; Bi, L.J.; Zhang, X.E.; Song, L.R. Detection of microcystins in environmental samples using surface plasmon resonance biosensor. Talanta 2009, 80, 407–410. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality. Addendum to Volume 2, Health Criteria and Other Supporting Information, 2nd ed.; World Health Organization: Geneva, Switzerland, 1998. [Google Scholar]
- Nishizawa, T.; Ueda, A.; Asayama, M.; Fujii, K.; Harada, K.; Ochi, K.; Shirai, M. Polyketide synthase gene coupled to the peptide synthetase module involved in the biosynthesis of the cyclic heptapeptide microcystin. J. Biochem. 2000, 127, 779–789. [Google Scholar] [CrossRef]
- Tillett, D.; Dittmann, E.; Erhard, M.; von Dohren, H.; Borner, T.; Neilan, B.A. Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: An integrated peptide-polyketide synthetase system. Chem. Biol. 2000, 7, 753–764. [Google Scholar] [CrossRef]
- Pearson, L.A.; Neilan, B.A. The molecular genetics of cyanobacterial toxicity as a basis for monitoring water quality and public health risk. Curr. Opin. Biotech. 2008, 19, 281–288. [Google Scholar] [CrossRef] [PubMed]
- McLellan, N.L.; Manderville, R.A. Toxic mechanisms of microcystins in mammals. Toxicol. Res. 2017, 6, 391–405. [Google Scholar] [CrossRef] [PubMed]
- Dittmann, E.; Wiegand, C. Cyanobacterial toxins-occurrence, biosynthesis and impact on human affairs. Mol. Nutr. Food Res. 2006, 50, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Omidi, A.; Esterhuizen-Londt, M.; Pflugmacher, S. Still challenging: The ecological function of the cyanobacterial toxin microcystin-What we know so far. Toxin Rev. 2018, 37, 87–105. [Google Scholar] [CrossRef]
- Kaplan, A.; Harel, M.; Kaplan-Levy, R.N.; Hadas, O.; Sukenik, A.; Dittmann, E. The languages spoken in the water body (or the biological role of cyanobacterial toxins). Front. Microbiol. 2012, 3. [Google Scholar] [CrossRef]
- Makower, A.K.; Schuurmans, J.M.; Groth, D.; Zilliges, Y.; Matthijs, H.C.P.; Dittmann, E. Transcriptomics-Aided Dissection of the Intracellular and Extracellular Roles of Microcystin in Microcystis aeruginosa PCC 7806. Appl. Environ. Microb. 2015, 81, 544–554. [Google Scholar] [CrossRef]
- Gan, N.; Wei, N.; Song, L. Recent progress in research of the biological function of microcystins. J. Lake Sci. 2017, 29, 8. [Google Scholar]
- Utkilen, H.; Gjolme, N. Iron-Stimulated Toxin Production in Microcystis-Aeruginosa. Appl. Environ. Microb. 1995, 61, 797–800. [Google Scholar]
- Van de Waal, D.B.; Verspagen, J.M.H.; Finke, J.F.; Vournazou, V.; Immers, A.K.; Kardinaal, W.E.A.; Tonk, L.; Becker, S.; Van Donk, E.; Visser, P.M.; et al. Reversal in competitive dominance of a toxic versus non-toxic cyanobacterium in response to rising CO2. ISME J. 2011, 5, 1438–1450. [Google Scholar] [CrossRef]
- Rohrlack, T.; Dittmann, E.; Borner, T.; Christoffersen, K. Effects of cell-bound microcystins on survival and feeding of Daphnia spp. Appl. Environ. Microb. 2001, 67, 3523–3529. [Google Scholar] [CrossRef] [PubMed]
- Zilliges, Y.; Kehr, J.C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Borner, T.; Dittmann, E. The Cyanobacterial Hepatotoxin Microcystin Binds to Proteins and Increases the Fitness of Microcystis under Oxidative Stress Conditions. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Dittmann, E.; Erhard, M.; Kaebernick, M.; Scheler, C.; Neilan, B.A.; von Dohren, H.; Borner, T. Altered expression of two light-dependent genes in a microcystin-lacking mutant of Microcystis aeruginosa PCC 7806. Microbiology 2001, 147, 3113–3119. [Google Scholar] [CrossRef] [PubMed]
- Schatz, D.; Keren, Y.; Vardi, A.; Sukenik, A.; Carmeli, S.; Borner, T.; Dittmann, E.; Kaplan, A. Towards clarification of the biological role of microcystins, a family of cyanobacterial toxins. Environ. Microbiol. 2007, 9, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Downing, T.G.; Phelan, R.R.; Downing, S. A potential physiological role for cyanotoxins in cyanobacteria of arid environments. J. Arid. Environ. 2015, 112, 147–151. [Google Scholar] [CrossRef]
- Pflugmacher, S. Possible allelopathic effects of cyanotoxins, with reference to microcystin-LR, in aquatic ecosystems. Environ. Toxicol. 2002, 17, 407–413. [Google Scholar] [CrossRef]
- Gan, N.Q.; Xiao, Y.; Zhu, L.; Wu, Z.X.; Liu, J.; Hu, C.L.; Song, L.R. The role of microcystins in maintaining colonies of bloom-forming Microcystis spp. Environ. Microbiol. 2012, 14, 730–742. [Google Scholar] [CrossRef]
- Hu, C.; Rea, C.; Yu, Z.; Lee, J. Relative importance of Microcystis abundance and diversity in determining microcystin dynamics in Lake Erie coastal wetland and downstream beach water. J. Appl. Microbiol. 2016, 120, 138–151. [Google Scholar] [CrossRef]
- Dziallas, C.; Grossart, H.P. Increasing Oxygen Radicals and Water Temperature Select for Toxic Microcystis sp. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Blooms Bite the Hand That Feeds Them. Science 2013, 342, 433–434. [Google Scholar] [CrossRef]
- Jones, G.J.; Orr, P.T. Release and Degradation of Microcystin Following Algicide Treatment of a Microcystis-Aeruginosa Bloom in a Recreational Lake, as Determined by Hplc and Protein Phosphatase Inhibition Assay. Water Res. 1994, 28, 871–876. [Google Scholar] [CrossRef]
- Papadimitriou, T.; Kormas, K.; Dionysiou, D.D.; Laspidou, C. Using H2O2 treatments for the degradation of cyanobacteria and microcystins in a shallow hypertrophic reservoir. Environ. Sci. Pollut. Res. 2016, 23, 21523–21535. [Google Scholar] [CrossRef] [PubMed]
- Broker, L.E.; Kruyt, F.A.; Giaccone, G. Cell death independent of caspases: A review. Clin. Cancer Res. 2005, 11, 3155–3162. [Google Scholar] [CrossRef] [PubMed]
- Skulachev, V.P. The programmed death phenomena, aging, and the Samurai law of biology. Exp. Gerontol. 2001, 36, 995–1024. [Google Scholar] [CrossRef]
- Bidle, K.D.; Falkowski, P.G. Cell death in planktonic, photosynthetic microorganisms. Nat. Rev. Microbiol. 2004, 2, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Bidle, K.D. Programmed Cell Death in Unicellular Phytoplankton. Curr. Biol. 2016, 26, R594–R607. [Google Scholar] [CrossRef]
- Franklin, D.J.; Brussaard, C.P.D.; Berges, J.A. What is the role and nature of programmed cell death in phytoplankton ecology? Eur. J. Phycol. 2006, 41, 1–14. [Google Scholar] [CrossRef]
- Franklin, D.J. Explaining the causes of cell death in cyanobacteria: What role for asymmetric division? J. Plankton Res. 2014, 36, 11–17. [Google Scholar] [CrossRef]
- Li, J.; Ding, Y.; Xiang, R.; Song, L. Programmed cell death in phytoplankton. Ecol. Environ. 2010, 19, 6. [Google Scholar]
- Hu, C.L.; Ludsin, S.A.; Martin, J.F.; Dittrnann, E.; Lee, J. Mycosporine-like amino acids (MAAs)-producing Microcystis in Lake Erie: Development of a qPCR assay and insight into its ecology. Harmful Algae 2018, 77, 1–10. [Google Scholar] [CrossRef]
- Visser, P.M.; Ibelings, B.W.; Mur, L.R.; Walsby, A.E. The Ecophysiology of the Harmful Cyanobacterium Microcystis. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C., Visser, P.M., Eds.; Springer: Berlin, Germany, 2005; Volume 3. [Google Scholar]
- Sigee, D.C.; Selwyn, A.; Gallois, P.; Dean, A.P. Patterns of cell death in freshwater colonial cyanobacteria during the late summer bloom. Phycologia 2007, 46, 284–292. [Google Scholar] [CrossRef]
- Yang, K.; Chen, Q.L.; Zhang, D.Y.; Zhang, H.J.; Lei, X.Q.; Chen, Z.R.; Li, Y.; Hong, Y.L.; Ma, X.H.; Zheng, W.; et al. The algicidal mechanism of prodigiosin from Hahella sp KA22 against Microcystis aeruginosa. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Song, L.R.; Sedmak, B. UVB Radiation as a Potential Selective Factor Favoring Microcystin Producing Bloom Forming Cyanobacteria. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.R.; Zheng, J.; Cao, H.S.; Wang, X.J.; Lou, K.; Zhang, X.H.; Tao, Y. Growth suppression and apoptosis-like cell death in Microcystis aeruginosa by H2O2: A new insight into extracellular and intracellular damage pathways. Chemosphere 2018, 211, 1098–1108. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Gan, N.Q.; Li, J.; Sedmak, B.; Song, L.R. Hydrogen peroxide induces apoptotic-like cell death in Microcystis aeruginosa (Chroococcales, Cyanobacteria) in a dose-dependent manner. Phycologia 2012, 51, 567–575. [Google Scholar] [CrossRef]
- Du, Y.P.; Ye, J.; Wu, L.; Yang, C.Y.; Wang, L.M.; Hu, X.J. Physiological effects and toxin release in Microcystis aeruginosa and Microcystis viridis exposed to herbicide fenoxaprop-p-ethyl. Environ. Sci. Pollut. Res. 2017, 24, 7752–7763. [Google Scholar] [CrossRef]
- Wu, L.; Qiu, Z.H.; Zhou, Y.; Du, Y.P.; Liu, C.N.; Ye, J.; Hu, X.J. Physiological effects of the herbicide glyphosate on the cyanobacterium Microcystis aeruginosa. Aquat. Toxicol. 2016, 178, 72–79. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Sha, J.; Tian, Y.; Zhang, X.Z.; Liu, B.Y.; Wu, Z.B. Polyphenolic allelochemical pyrogallic acid induces caspase-3(like)-dependent programmed cell death in the cyanobacterium Microcystis aeruginosa. Algal Res. 2017, 21, 148–155. [Google Scholar] [CrossRef]
- He, Y.; Zhou, Q.H.; Liu, B.Y.; Cheng, L.; Tian, Y.; Zhang, Y.Y.; Wu, Z.B. Programmed cell death in the cyanobacterium Microcystis aeruginosa induced by allelopathic effect of submerged macrophyte Myriophyllum spicatum in co-culture system. J. Appl. Phycol. 2016, 28, 2805–2814. [Google Scholar] [CrossRef]
- Ross, C.; Santiago-Vazquez, L.; Paul, V. Toxin release in response to oxidative stress and programmed cell death in the cyanobacterium Microcystis aeruginosa. Aquat. Toxicol. 2006, 78, 66–73. [Google Scholar] [CrossRef]
- Hu, L.B.; Zhou, W.; Yang, J.D.; Chen, J.A.; Yin, Y.F.; Shi, Z.Q. Cinnamaldehyde Induces PCD-Like Death of Microcystis aeruginosa via Reactive Oxygen Species. Water Air Soil Pollut. 2011, 217, 105–113. [Google Scholar] [CrossRef]
- Frangeul, L.; Quillardet, P.; Castets, A.M.; Humbert, J.F.; Matthijs, H.C.P.; Cortez, D.; Tolonen, A.; Zhang, C.C.; Gribaldo, S.; Kehr, J.C.; et al. Highly plastic genome of Microcystis aeruginosa PCC 7806, a ubiquitous toxic freshwater cyanobacterium. BMC Genom. 2008, 9. [Google Scholar] [CrossRef] [PubMed]
- Klemencic, M.; Novinec, M.; Dolinar, M. Orthocaspases are proteolytically active prokaryotic caspase homologues: The case of Microcystis aeruginosa. Mol. Microbiol. 2015, 98, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Saraste, A.; Pulkki, K. Morphologic and biochemical hallmarks of apoptosis. Cardiovasc. Res. 2000, 45, 528–537. [Google Scholar] [CrossRef]
- Martinez, M.M.; Reif, R.D.; Pappas, D. Detection of apoptosis: A review of conventional and novel techniques. Anal. Methods 2010, 2, 996–1004. [Google Scholar] [CrossRef]
- Li, J.; Song, L.R. Applicability of the MTT assay for measuring viability of cyanobacteria and algae, specifically for Microcystis aeruginosa (Chroococcales, Cyanobacteria). Phycologia 2007, 46, 593–599. [Google Scholar] [CrossRef]
- Li, J.; Ou, D.Y.; Zheng, L.L.; Gan, N.Q.; Song, L.R. Applicability of the fluorescein diacetate assay for metabolic activity measurement of Microcystis aeruginosa (Chroococcales, Cyanobacteria). Phycol. Res. 2011, 59, 200–207. [Google Scholar] [CrossRef]
- Bouchard, J.N.; Purdie, D.A. Effect of Elevated Temperature, Darkness, and Hydrogen Peroxide Treatment on Oxidative Stress and Cell Death in the Bloom-Forming Toxic Cyanobacterium Microcystis Aeruginosa. J. Phycol. 2011, 47, 1316–1325. [Google Scholar] [CrossRef]
- Crowley, L.C.; Marfell, B.J.; Waterhouse, N.J. Analyzing Cell Death by Nuclear Staining with Hoechst 33342. Cold Spring Harb. Protoc. 2016, 2016. [Google Scholar] [CrossRef]
- Ross, C.; Warhurst, B.C.; Brown, A.; Huff, C.; Ochrietor, J.D. Mesohaline conditions represent the threshold for oxidative stress, cell death and toxin release in the cyanobacterium Microcystis aeruginosa. Aquat. Toxicol. 2019, 206, 203–211. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Li, J.; Wei, J.; Kawan, A.; Wang, L.; Zhang, X.Z. Vitamin C modulates Microcystis aeruginosa death and toxin release by induced Fenton reaction. J. Hazard. Mater. 2017, 321, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Van Engeland, M.; Nieland, L.J.W.; Ramaekers, F.C.S.; Schutte, B.; Reutelingsperger, C.P.M. Annexin V-affinity assay: A review on an apoptosis detection system based on phosphatidylserine exposure. Cytometry 1998, 31, 1–9. [Google Scholar] [CrossRef]
- Vermes, I.; Haanen, C.; Steffensnakken, H.; Reutelingsperger, C. A Novel Assay for Apoptosis-Flow Cytometric Detection of Phosphatidylserine Expression on Early Apoptotic Cells Using Fluorescein-Labeled Annexin-V. J. Immunol. Methods 1995, 184, 39–51. [Google Scholar] [CrossRef]
- Ye, J.; Huang, C.; Qiu, Z.H.; Wu, L.; Xu, C. The Growth, Apoptosis and Oxidative Stress in Microcystis viridis Exposed to Glyphosate. Bull. Environ. Contam. Toxicol. 2019, 103, 585–589. [Google Scholar] [CrossRef]
- Segovia, M.; Berges, J.A. Inhibition of Caspase-Like Activities Prevents the Appearance of Reactive Oxygen Species and Dark-Induced Apoptosis in the Unicellular Chlorophyte Dunaliella Tertiolecta. J. Phycol. 2009, 45, 1116–1126. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.M.; Lu, Z.Y.; Liu, B.Y.; Zhou, Q.H.; Zhang, Y.Y.; Wu, Z.B. Allelopathic effects of pyrogallic acid secreted by submerged macrophytes on Microcystis aeruginosa: Role of ROS generation. Allelopath. J. 2014, 33, 121–129. [Google Scholar]
- Sharon, A.; Finkelstein, A.; Shlezinger, N.; Hatam, I. Fungal apoptosis: Function, genes and gene function. FEMS Microbiol. Rev. 2009, 33, 833–854. [Google Scholar] [CrossRef]
- Chandra, J.; Samali, A.; Orrenius, S. Triggering and modulation of apoptosis by oxidative stress. Free Radic. Biol. Med. 2000, 29, 323–333. [Google Scholar] [CrossRef]
- Bayles, K.W. Bacterial programmed cell death: Making sense of a paradox. Nat. Rev. Microbiol. 2014, 12, 63–69. [Google Scholar] [CrossRef]
- Kasuba, K.C.; Vavilala, S.L.; D’Souza, J.S. Apoptosis-like cell death in unicellular photosynthetic organisms—A review. Algal Res. 2015, 12, 126–133. [Google Scholar] [CrossRef]
- Engelberg-Kulka, H.; Sat, B.; Reches, M.; Amitai, S.; Hazan, R. Bacterial programmed cell death systems as targets for antibiotics. Trends Microbiol. 2004, 12, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, D.G. Programmed Cell Death in Bacterial Community: Mechanisms of Action, Causes and Consequences. J. Microbiol. Biotechnol. 2019, 29, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Peeters, S.H.; de Jonge, M.I. For the greater good: Programmed cell death in bacterial communities. Microbiol. Res. 2018, 207, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Berman-Frank, I.; Bidle, K.D.; Haramaty, L.; Falkowski, P.G. The demise of the marine cyanobacterium, Trichodesmium spp., via an autocatalyzed cell death pathway. Limnol. Oceanogr. 2004, 49, 997–1005. [Google Scholar] [CrossRef]
- Green, D.R.; Llambi, F. Cell Death Signaling. CSH Perspect. Biol. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, K.; Christensen, S.K.; Gerdes, K. Rapid induction and reversal of a bacteriostatic condition by controlled expression of toxins and antitoxins. Mol. Microbiol. 2002, 45, 501–510. [Google Scholar] [CrossRef]
- Erental, A.; Sharon, I.; Engelberg-Kulka, H. Two Programmed Cell Death Systems in Escherichia coli: An Apoptotic-Like Death Is Inhibited by the mazEF-Mediated Death Pathway. PLoS Biol. 2012, 10. [Google Scholar] [CrossRef]
- Klemencic, M.; Dolinar, M. Orthocaspase and toxin-antitoxin loci rubbing shoulders in the genome of Microcystis aeruginosa PCC 7806. Curr. Genet. 2016, 62, 669–675. [Google Scholar] [CrossRef]
- Erental, A.; Kalderon, Z.; Saada, A.; Smith, Y.; Engelberg-Kulka, H. Apoptosis-Like Death, an Extreme SOS Response in Escherichia coli. MBio 2014, 5. [Google Scholar] [CrossRef]
- Little, J.W. Mechanism of Specific Lexa Cleavage-Autodigestion and the Role of Reca Coprotease. Biochimie 1991, 73, 411–422. [Google Scholar] [CrossRef]
- Li, S.; Xu, M.L.; Su, Z.C. Computational analysis of LexA regulons in Cyanobacteria. BMC Genom. 2010, 11. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Morimoto, D.; Sako, Y.; Yoshida, T. LexA Binds to Transcription Regulatory Site of Cell Division Gene ftsZ in Toxic Cyanobacterium Microcystis aeruginosa. Mar. Biotechnol. 2018, 20, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Tsiatsiani, L.; Van Breusegem, F.; Gallois, P.; Zavialov, A.; Lam, E.; Bozhkov, P.V. Metacaspases. Cell Death Differ. 2011, 18, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Klemencic, M.; Funk, C. Structural and functional diversity of caspase homologues in non-metazoan organisms. Protoplasma 2018, 255, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Asplund-Samuelsson, J. The art of destruction: Revealing the proteolytic capacity of bacterial caspase homologs. Mol. Microbiol. 2015, 98, 1–6. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Drury, G.E.; Rotari, V.I.; Gordon, A.; Willer, M.; Farzaneh, T.; Woltering, E.J.; Gallois, P. Metacaspase-8 modulates programmed cell death induced by ultraviolet light and H2O2 in Arabidopsis. J. Biol. Chem. 2008, 283, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Herker, E.; Maldener, C.; Wissing, S.; Lachelt, S.; Herian, M.; Fehr, M.; Lauber, K.; Sigrist, S.J.; Wesselborg, S.; et al. A caspase-related protease regulates apoptosis in yeast. Mol. Cell 2002, 9, 911–917. [Google Scholar] [CrossRef]
- Van Creveld, S.G.; Ben-Dor, S.; Mizrachi, A.; Alcolombri, U.; Hopes, A.; Mock, T.; Rosenwasser, S.; Vardi, A. A redox-regulated type III metacaspase controls cell death in a marine diatom. BioRxiv 2018. [Google Scholar] [CrossRef]
- Gautam, S.; Sharma, A. Involvement of caspase-3-like protein in rapid cell death of Xanthomonas. Mol. Microbiol. 2002, 44, 393–401. [Google Scholar] [CrossRef]
- Lee, R.E.C.; Puente, L.G.; Kaern, M.; Megeney, L.A. A Non-Death Role of the Yeast Metacaspase: Yca1p Alters Cell Cycle Dynamics. PLoS ONE 2008, 3. [Google Scholar] [CrossRef]
- Asplund-Samuelsson, J.; Bergman, B.; Larsson, J. Prokaryotic Caspase Homologs: Phylogenetic Patterns and Functional Characteristics Reveal Considerable Diversity. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Qin, S.; Wu, Q.Y. Genome-wide comparative analysis of metacaspases in unicellular and filamentous cyanobacteria. BMC Genom. 2010, 11. [Google Scholar] [CrossRef] [PubMed]
- Klemencic, M.; Asplund-Samuelsson, J.; Dolinar, M.; Funk, C. Phylogenetic Distribution and Diversity of Bacterial Pseudo-Orthocaspases Underline Their Putative Role in Photosynthesis. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Asplund-Samuelsson, J.; Sundh, J.; Dupont, C.L.; Allen, A.E.; McCrow, J.P.; Celepli, N.A.; Bergman, B.; Ininbergs, K.; Ekman, M. Diversity and Expression of Bacterial Metacaspases in an Aquatic Ecosystem. Front. Microbiol. 2016, 7, 1043. [Google Scholar] [CrossRef] [PubMed]
- Spungin, D.; Bidle, K.D.; Berman-Frank, I. Metacaspase involvement in programmed cell death of the marine cyanobacterium Trichodesmium. Environ. Microbiol. 2019, 21, 667–681. [Google Scholar] [CrossRef] [PubMed]
- Bar-Zeev, E.; Avishay, I.; Bidle, K.D.; Berman-Frank, I. Programmed cell death in the marine cyanobacterium Trichodesmium mediates carbon and nitrogen export. ISME J. 2013, 7, 2340–2348. [Google Scholar] [CrossRef]
- Suarez, M.F.; Filonova, L.H.; Smertenko, A.; Savenkov, E.I.; Clapham, D.H.; von Arnold, S.; Zhivotovsky, B.; Bozhkov, P.V. Metacaspase-dependent programmed cell death is essential for plant embryogenesis. Curr. Biol. 2004, 14, R339–R340. [Google Scholar] [CrossRef]
- Lewis, K. Programmed death in bacteria. Microbiol. Mol. Biol. Res. 2000, 64, 503–514. [Google Scholar] [CrossRef]
- Michod, R.E. The Theory of Kin Selection. Annu. Rev. Ecol. Syst. 1982, 13, 23–55. [Google Scholar] [CrossRef]
- Allocati, N.; Masulli, M.; Di Ilio, C.; De Laurenzi, V. Die for the community: An overview of programmed cell death in bacteria. Cell Death Dis. 2015, 6. [Google Scholar] [CrossRef]
- Rantala, A.; Fewer, D.P.; Hisbergues, M.; Rouhiainen, L.; Vaitomaa, J.; Borner, T.; Sivonen, K. Phylogenetic evidence for the early evolution of microcystin synthesis. Proc. Natl. Acad. Sci. USA 2004, 101, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Takashima, Y.; Tomaru, Y.; Shirai, Y.; Takao, Y.; Hiroishi, S.; Nagasaki, K. Isolation and characterization of a cyanophage infecting the toxic cyanobacterium Microcystis aeruginosa. Appl. Environ. Microb. 2006, 72, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.W.; Ha, C.; Lee, S.; Kwon, J.; Cho, H.; Gorham, T.; Lee, J. Characterization of Cyanophages in Lake Erie: Interaction Mechanisms and Structural Damage of Toxic Cyanobacteria. Toxins 2019, 11, 444. [Google Scholar] [CrossRef] [PubMed]
- Cerbin, S.; Wejnerowski, L.; Dziuba, M. Aphanizomenon gracile increases in width in the presence of Daphnia. A defence mechanism against grazing? J. Limnol. 2013, 72, 505–511. [Google Scholar] [CrossRef]
| Microcystis Strain | Stimulus | Approaches for Analyzing PCD | ROS * Production | Reference |
|---|---|---|---|---|
| M. aeruginosa | H2O2 (100 µM) | Caspase-3 Assay | Yes | [53] |
| Field Microcystis samples | Natural environmental condition (e.g., pH > 9.2 and depletion of CO2) | Evans blue staining, Hoechst 33342 staining, and TUNEL assay | Yes | [44] |
| M. aeruginosa FACHB905 | Cinnamaldehyde (0.15–1.5 mM) | No available | Yes | [54] |
| M. aeruginosa CCAP1450 ⁄ 16 | Dark and oxidative stress (0.5 mM) | Caspase 3-like assay and TUNEL assay | Yes | [61] |
| M. aeruginosa FACHB905 | H2O2 (150–325 µM) | TEM, Hoechst 33342 staining assay, TUNEL assay, gel electrophoresis, and Caspase-3 assay | Yes | [48] |
| M. aeruginosa PCC7005, PCC7806, and FACHB905 | UV-B irradiation (0.5 and 0.99 W/m2) | TUNEL assay | Yes | [46] |
| M. aeruginosa FACH905 | Allelopathic submerged macrophyte, Myriophyllum spicatum, | Caspase assay and gel electrophoresis | Yes | [52] |
| M. aeruginosa | Glyphosate (1–10 mg/L) | Phosphatidylserine apoptosis assay based flow cytometry | Yes | [50] |
| M. aeruginosa FACHB905 and M. viridis (FACHB1337) | fenoxaprop-p-ethyl (1–10 mg/L) | Phosphatidylserine apoptosis assay | Yes | [49] |
| M. aeruginosa FACHB905 | Vitamin C (0.6 mM) | TEM, Caspase-3 assay, and Hoechst 33342/PI staining assay | Yes | [64] |
| M. aeruginosa TAIHU98 | Prodigiosin (20–50 µg/mL) from Hahella sp. KA22 | TEM, gel electrophoresis, Annexin V assay based on flow cytometry | Yes | [45] |
| M. aeruginosa DIANCHI 905 | Polyphenolic allelochemical pyrogallic acid (14 mg/L) | TEM, gel electrophoresis, and Annexin V apoptosis assay | Yes | [51] |
| M. aeruginosa FACHB905, | H2O2 (0.1–1.5 mM) | SEM, Caspase-3 assay, and Hoechst 33342 staining assay | Yes | [47] |
| M. aeruginosa LB-2385 | Mesohaline conditions (<7 ppt) | Caspase-3 assay | Yes | [63] |
| M.viridis FACHB979 | 0.2 to 10 mg/L in glyphosate | Phosphatidylserine Apoptosis Assay Kit | Yes | [67] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, C.; Rzymski, P. Programmed Cell Death-Like and Accompanying Release of Microcystin in Freshwater Bloom-Forming Cyanobacterium Microcystis: From Identification to Ecological Relevance. Toxins 2019, 11, 706. https://doi.org/10.3390/toxins11120706
Hu C, Rzymski P. Programmed Cell Death-Like and Accompanying Release of Microcystin in Freshwater Bloom-Forming Cyanobacterium Microcystis: From Identification to Ecological Relevance. Toxins. 2019; 11(12):706. https://doi.org/10.3390/toxins11120706
Chicago/Turabian StyleHu, Chenlin, and Piotr Rzymski. 2019. "Programmed Cell Death-Like and Accompanying Release of Microcystin in Freshwater Bloom-Forming Cyanobacterium Microcystis: From Identification to Ecological Relevance" Toxins 11, no. 12: 706. https://doi.org/10.3390/toxins11120706
APA StyleHu, C., & Rzymski, P. (2019). Programmed Cell Death-Like and Accompanying Release of Microcystin in Freshwater Bloom-Forming Cyanobacterium Microcystis: From Identification to Ecological Relevance. Toxins, 11(12), 706. https://doi.org/10.3390/toxins11120706






