The Activity of Isoquinoline Alkaloids and Extracts from Chelidonium majus against Pathogenic Bacteria and Candida sp.
Abstract
:1. Introduction
2. Results
2.1. Comparison of Extraction Effectiveness
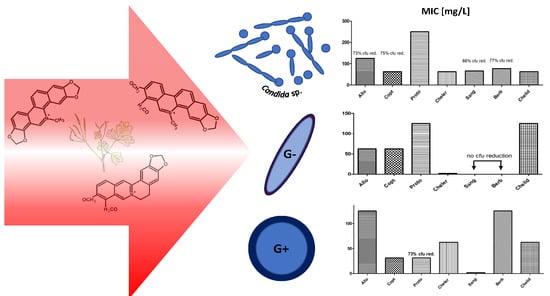
2.2. MIC Evaluation
2.3. Cytotoxicity
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plant Material
5.1.1. Wild Growing Plants
5.1.2. In Vitro Plant Material
5.2. Phytochemistry
5.2.1. Reagents and Standards
5.2.2. Sample Preparation
5.2.3. HPLC Analysis
5.3. Statistical Analysis
5.4. Experimental Design for Bioactivity Assays
5.4.1. Strains
5.4.2. MIC Evaluation
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zielinska, S.; Jezierska-Domaradzka, A.; Wójciak-Kosior, M.; Sowa, I.; Junka, A.; Matkowski, A.M. Greater Celandine’s ups and downs—21 centuries of medicinal uses of Chelidonium majus from the viewpoint of today’s pharmacology. Front. Pharmacol. 2018, 9, 1–29. [Google Scholar] [CrossRef]
- Miao, F.; Yanga, X.-J.; Zhou, L.; Hu, H.-J.; Zheng, F.; Ding, X.-D.; Sun, D.-M.; Zhou, C.-D.; Sun, W. Structural modification of sanguinarine and chelerythrine and their antibacterial activity. Nat. Prod. Res. 2011, 25, 863–875. [Google Scholar] [CrossRef]
- Hossain, M.; Khan, A.Y.; Kumar, G.S. Interaction of the anticancer plant alkaloid sanguinarine with bovine serum albumin. PLoS ONE 2011, 6, e18333. [Google Scholar] [CrossRef]
- Giri, P.; Kumar, G.S. Specific binding and self-structure induction to poly(A) by the cytotoxic plant alkaloid sanguinarine. Biochim. Biophys. Acta 2007, 1770, 1419–1426. [Google Scholar] [CrossRef]
- Bai, L.-P.; Zhao, Z.-Z.; Cai, Z.; Jiang, Z.-H. DNA-binding affinities and sequence selectivity of quaternary benzophenanthridine alkaloids sanguinarine, chelerythrine, and nitidine. Bioorgan. Med. Chem. 2006, 14, 5439–5445. [Google Scholar] [CrossRef]
- Barreto, M.C.; Pinto, R.E.; Arrabaça, J.D.; Pavão, M.L. Inhibition of mouse liver respiration by Chelidonium majus isoquinoline alkaloids. Toxicol. Lett. 2003, 146, 37–47. [Google Scholar] [CrossRef] [Green Version]
- Sedo, A.; Vlasicova, K.; Bartak, P.; Vespalec, R.; Vicar, J.; Simanek, V.; Ulrichova, J. Quaternary Benzo[c]phenanthridine Alkaloids as Inhibitors of Aminopeptidase N and Dipeptidyl Peptidase IV. Phytother. Res. 2002, 16, 84–87. [Google Scholar] [CrossRef]
- Ulrichova, J.; Dvorak, Z.; Vicar, J.; Lata, J.; Smrzova, J.; Sedo, A.; Simanek, V. Cytotoxicity of natural compounds in hepatocyte cell culture models. The case of quaternary benzo[c]phenanthridine alkaloids. Toxicol. Lett. 2001, 125, 125–132. [Google Scholar] [CrossRef]
- Zielinska, S.; Wójciak-Kosior, M.; Płachno, B.J.; Sowa, I.; Włodarczyk, M.; Matkowski, A. Quaternary alkaloids in Chelidonium majus in vitro cultures. Ind. Crop. Prod. 2018, 123, 17–24. [Google Scholar] [CrossRef]
- Sowa, I.; Zielinska, S.; Sawicki, J.; Bogucka-Kocka, A.; Staniak, M.; Bartusiak-Szczesniak, E.; Podolska-Fajks, M.; Kocjan, R.; Wojciak-Kosior, M. Systematic evaluation of chromatographic parameters for isoquinoline alkaloids on XB-C18 core-shell column using different mobile phase compositions. J. Anal. Methods Chem. 2018, 3, 1–8. [Google Scholar] [CrossRef]
- Borghini, A.; Pietra, D.; di Trapani, C.; Madau, P.; Lubinu, G.; Bianucci, A.M. Data mining as a predictive model for Chelidonium majus extracts production. Ind. Crop Prod. 2015, 64, 25–32. [Google Scholar] [CrossRef]
- Gilca, M.; Gaman, L.; Panait, E.; Stoian, I.; Atanasiu, V. Chelidonium majus—An integrative review: Traditional knowledge versus modern findings. Complement. Med. Res. 2010, 17, 241–248. [Google Scholar] [CrossRef]
- Sarközi, A.; Janicsak, G.; Kursinszki, L.; Kery, A. Alkaloid composition of Chelidonium majus L. studied by different chromatographic techniques. Chromatographia 2006, 63, S81–S86. [Google Scholar] [CrossRef]
- Osbaldeston, T.A.; Wood, R.P.A. Dioscorides de Materia Medica, Being a Herbal with Many other Materials Written in Greek in the First Century of the Common Era; An Indexed Version in Modern English; Ibidis Press: Johannesburg, South Africa, 2000; pp. 352–355. [Google Scholar]
- Jones, W.H.S. Pliny Natural History with an English Translation in Ten Volumes; Harvard University Press: London, UK, 1966. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Gamborg, O.L.; Miller, R.A.; Ojima, K. Nutrient requirements of suspension culture of soybean root cells. Exp. Cell Res. 1968, 50, 151–158. [Google Scholar] [CrossRef]
- Kędzia, B.; Hołderna-Kędzia, E.; Goździcka-Józefiak, A.; Buchwald, W. The antimicrobial activity of Chelidonium majus L. Postępy Fitoterapii 2013, 4, 236–243. [Google Scholar]
- Artini, M.; Papa, R.; Barbato, G.; Scoarughi, G.L.; Cellini, A.; Morazzoni, P.; Bombardelli, E.; Selan, L. Bacterial biofilm formation inhibitory activity revealed for plant derived natural compounds. Bioorg. Med. Chem. 2012, 20, 920–926. [Google Scholar] [CrossRef]
- Mitscher, L.A.; Leu, R.P.; Bathala, M.S.; Wu, W.N.; Beal, J.L. Antimicrobial agents from higher plants. I. Introduction, rationale, and methodology. Lloydia 1972, 35, 157–166. [Google Scholar]
- Mitscher, L.A.; Park, Y.H.; Clark, D.; Clark, G.W.; Hammesfahr, P.D.; Wu, W.N.; Beal, J.L. Antimicrobial agents from higher plants. An investigation of Hunnemannia fumariaefolia pseudoalcoholates of sanguinarine and chelerythrine. Lloydia 1978, 41, 145–149. [Google Scholar]
- Kokoška, L.; Polesny, Z.; Rada, V.; Nepovim, A.; Vanek, T. Screening of some Siberian medicinal plants for antimicrobial activity. J. Ethnopharmacol. 2002, 82, 51–53. [Google Scholar] [CrossRef]
- Ciric, A.; Vinterhalter, B.; Šavikin-Fodulovi’c, K.; Sokovic, M.; Vinterhalter, D. Chemical analysis and antimicrobial activity of methanol extracts of celandine (Chelidonium majus L.) plants growing in nature and cultured in vitro. Arch. Biol. Sci. 2008, 60, 7–8. [Google Scholar] [CrossRef]
- Wongbutdee, J. Physiological effects of berberine—Review article. Thai Pharm. Health Sci. J. 2009, 4, 78–83. [Google Scholar]
- Imanshahidi, M.; Hosseinzadeh, H. Pharmacological and therapeutic effects of Berberis vulgaris and its active constituent, Berberine—Review article. Phytother. Res. 2008, 22, 999–1012. [Google Scholar] [CrossRef]
- Kedzia, B.; Hołderna-Kedzia, E. The effect of alkaloids and other groups of plant compounds on bacteria and fungi. Postęp. Fitoter. 2013, 1, 8–16. [Google Scholar]
- Ma, W.G.; Fukushi, Y.; Tahava, S.; Osawa, T. Fungitoxic alkaloids from Hokkaido Papaveraceae. Fitoterapia 2000, 71, 527–534. [Google Scholar] [CrossRef]
- Meng, F.; Zuo, G.; Hao, X.; Wang, G.; Xiao, H.; Zhang, J.; Xu, G. Antifungal activity of the benzo [c] phenanthridine alkaloids from Chelidonium majus Linn against resistant clinical yeast isolates. J. Ethnopharmacol. 2009, 125, 494–496. [Google Scholar] [CrossRef]
- Young, I.M.; Crawford, J.W. Interactions and Self-Organization in the Soil-Microbe Complex. Science 2004, 304, 1634–1637. [Google Scholar] [CrossRef]
- Djenane, Z.; Nateche, F.; Amziane, M.; Gomis-Cebolla, J.; El-Aichar, F.; Khorf, H.; Ferre, J. Assessment of the Antimicrobial Activity and the Entomocidal Potential of Bacillus thuringiensis Isolates from Algeria. Toxins 2017, 9, 139. [Google Scholar] [CrossRef]
- Schirawski, J.; Perlin, H. Plant-Microbe Interaction 2017—The Good, the Bad and the Diverse. Int. J. Mol. Sci. 2018, 19, 1374. [Google Scholar] [CrossRef]
- Wand, M.E.; Bock, L.J.; Bonney, L.C.; Sutton, J.M. Mechanisms of Increased Resistance to Chlorhexidine and Cross-Resistance to Colistin following Exposure of Klebsiella pneumoniae Clinical Isolates to Chlorhexidine. Antimicrob. Agents Chemother. 2017, 61, 01162-16. [Google Scholar] [CrossRef]
- Kulp, M.; Bragina, O.; Kogerman, P.; Kaljurand, M. Capillary electrophoresis with LED-induced native fluorescence detection for determination of isoquinoline alkaloids and their cytotoxicity in extracts of Chelidonium majus L. J. Chromatogr. A 2011, 1218, 5298–5304. [Google Scholar] [CrossRef]







| Solvent/Plant Material | Candida albicans | Staphylococcus aureus |
|---|---|---|
| MeOH aerial | − | − |
| MeOH roots | − | 62.5 * |
| Culture medium/plant material | Candida albicans | Staphylococcus aureus |
| MS roots | 125 | 500 * |
| MS shoots | 500 * | 500 * |
| MS+N roots | 3.9 | 500 * |
| MS+N shoots | 3.9 | 500 * |
| ½ MS roots | − | − |
| ½ MS shoots | − | − |
| 1.5%suc. + N roots | − | − |
| 1.5%suc.+ N shoots | 500 | 250 |
| 1.5%suc. roots | 250 | 125 |
| 1.5%suc. shoots | 500 | 500 * |
| B5 roots | − | 250 * |
| B5 shoots | − | 500 * |
| B5 + N roots | − | − |
| B5 + N shoots | − | 250 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zielińska, S.; Wójciak-Kosior, M.; Dziągwa-Becker, M.; Gleńsk, M.; Sowa, I.; Fijałkowski, K.; Rurańska-Smutnicka, D.; Matkowski, A.; Junka, A. The Activity of Isoquinoline Alkaloids and Extracts from Chelidonium majus against Pathogenic Bacteria and Candida sp. Toxins 2019, 11, 406. https://doi.org/10.3390/toxins11070406
Zielińska S, Wójciak-Kosior M, Dziągwa-Becker M, Gleńsk M, Sowa I, Fijałkowski K, Rurańska-Smutnicka D, Matkowski A, Junka A. The Activity of Isoquinoline Alkaloids and Extracts from Chelidonium majus against Pathogenic Bacteria and Candida sp. Toxins. 2019; 11(7):406. https://doi.org/10.3390/toxins11070406
Chicago/Turabian StyleZielińska, Sylwia, Magdalena Wójciak-Kosior, Magdalena Dziągwa-Becker, Michał Gleńsk, Ireneusz Sowa, Karol Fijałkowski, Danuta Rurańska-Smutnicka, Adam Matkowski, and Adam Junka. 2019. "The Activity of Isoquinoline Alkaloids and Extracts from Chelidonium majus against Pathogenic Bacteria and Candida sp." Toxins 11, no. 7: 406. https://doi.org/10.3390/toxins11070406