Artificial Substrates Coupled with qPCR (AS-qPCR) Assay for the Detection of the Toxic Benthopelagic Dinoflagellate Vulcanodinium rugosum
Abstract
:1. Introduction
2. Results
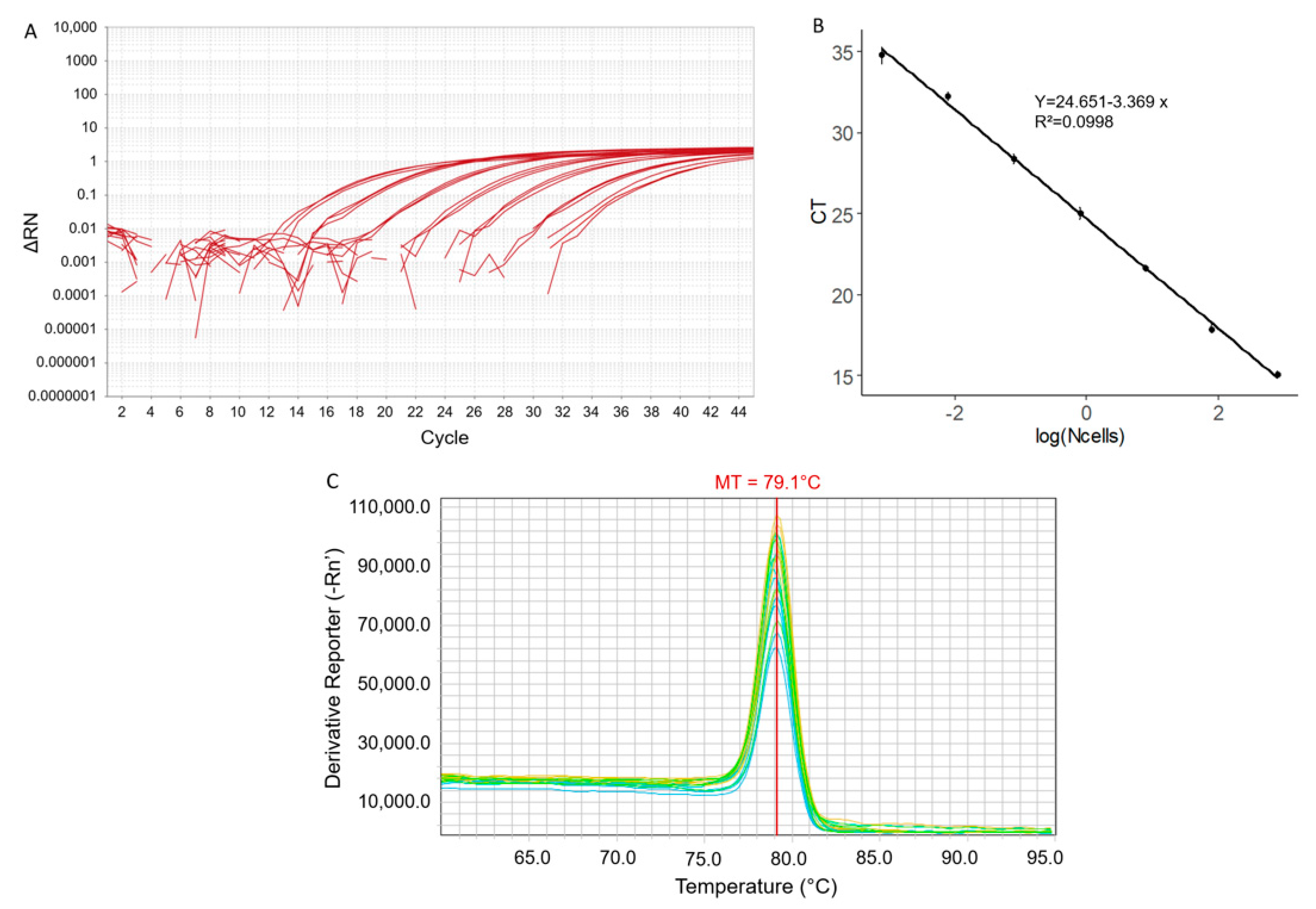
2.1. qPCR Parameters and Sensitivity
2.2. Specificity of the qPCR
2.3. Application of AS-qPCR for Field Survey
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Dinoflagellate Strains Culture
5.2. Artificial Substrates Environmental Sampling
5.2.1. Sampling Area
5.2.2. Deployment and Collection
5.3. DNA Extraction
5.3.1. From Phytoplankton Culture Cells
5.3.2. From Environmental Samples
- Ncells/cm2AS: Equiv. cells per cm2 of artificial substrate (in cells/cm2 substrate)
- Ncells/µL: Equiv. cells per µL in diluted purified DNA extracts (in cells/µL)
- V: Total Volume of diluted purified DNA extracts (in µL)
- F1: Factor associated with the dilution of purified DNA extracts
- F2: Factor associated with the volume of DNA extract used for purification
- F3: Factor associated with the volume of water used for DNA extraction
- AS surface: Artificial Substrates surface (in cm2)
5.4. Primers Design
5.5. qPCR Assay
5.6. Standard Curve and Specificity
5.7. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Moreira, A.R.; Comas, A.; Valle, A.; Seisdedo, M. Bloom of Vulcanodinium rugosum Linked to Skin Lesions in Cienfuegos Bay, Cuba. IOC Newsl. Toxic Algae Algal Bloom. 2016, 55, 10–11. [Google Scholar] [CrossRef]
- Rhodes, L.; Smith, K.; Selwood, A.; McNabb, P.; Munday, R.; Suda, S.; Molenaar, S.; Hallegraeff, G. Dinoflagellate Vulcanodinium rugosum Identified as the Causative Organism of Pinnatoxins in Australia, New Zealand and Japan. Phycologia 2011, 50, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.F.; Rhodes, L.L.; Suda, S.; Selwood, A.I. A Dinoflagellate Producer of Pinnatoxin G, Isolated from Sub-Tropical Japanese Waters. Harmful Algae 2011, 10, 702–705. [Google Scholar] [CrossRef]
- Zeng, N.; Gu, H.; Smith, K.F.; Rhodes, L.L.; Selwood, A.I.; Yang, W. The First Report of Vulcanodinium rugosum (Dinophyceae) from the South China Sea with a Focus on the Life Cycle. N. Z. J. Mar. Freshw. Res. 2012, 46, 511–521. [Google Scholar] [CrossRef] [Green Version]
- Hess, P.; Abadie, E.; Herve, F.; Berteaux, T.; Sechet, V.; Araoz, R.; Molgo, J.; Zakarian, A.; Sibat, M.; Rundberget, T.; et al. Is Responsible for Atypical Toxicity in Mussels (Mytilus galloprovincialis) and Clams (Venerupis decussata) from Ingril, a French Mediterranean Lagoon. Toxicon Off. J. Int. Soc. Toxinology 2013, 75, 16–26. [Google Scholar] [CrossRef] [Green Version]
- Munday, R.; Selwood, A.I.; Rhodes, L. Acute Toxicity of Pinnatoxins, E, F and G to Mice. Toxicon 2012, 60, 995–999. [Google Scholar] [CrossRef]
- Bouquet, A.; Perdrau, M.A.; Laabir, M.; Foucault, E.; Chomérat, N.; Rolland, J.L.; Abadie, E. Liza Ramada Juveniles after Exposure to the Toxic Dinoflagellate Vulcanodinium rugosum: Effects on Fish Viability, Tissue Contamination and Microalgae Survival after Gut Passage. Toxins 2022, 14, 401. [Google Scholar] [CrossRef]
- Abadie, E.; Chiantella, C.; Crottier, A.; Rhodes, L.; Masseret, E.; Berteau, T.; Laabir, M. What are the main environmental factors driving the development of the neurotoxic dinoflagellate Vulcanodinium rugosum in a Mediterranean ecosystem (Ingril lagoon, France)? Harmful Algae 2018, 75, 75–86. [Google Scholar] [CrossRef] [Green Version]
- REPHY—French Observation and Monitoring program for Phytoplankton and Hydrology in coastal waters. In REPHY Dataset—French Observation and Monitoring Program for Phytoplankton and Hydrology in Coastal Waters. Metropolitan Data; SEANOE, Brest, France: 2022. [CrossRef]
- Arnich, N.; Abadie, E.; Delcourt, N.; Fessard, V.; Fremy, J.-M.; Hort, V.; Lagrange, E.; Maignien, T.; Molgó, J.; Peyrat, M.-B.; et al. Health Risk Assessment Related to Pinnatoxins in French Shellfish. Toxicon 2020, 180, 1–10. [Google Scholar] [CrossRef]
- Nézan, E.; Chomérat, N. Vulcanodinium rugosum Gen. Nov., Sp. Nov. (Dinophyceae): A New Marine Dinoflagellate from the French Mediterranean Coast. Cryptogam. Algol. 2011, 32, 3–18. [Google Scholar] [CrossRef]
- Amzil, Z.; Derrien, A.; Terre Terrillon, A.; Duval, A.; Connes, C.; Marco-Miralles, F.; Nézan, E.; Mertens, K.N. Monitoring the Emergence of Algal Toxins in Shellfish: First Report on Detection of Brevetoxins in French Mediterranean Mussels. Mar. Drugs 2021, 19, 393. [Google Scholar] [CrossRef]
- Ben Gharbia, H.; Yahia, O.K.D.; Cecchi, P.; Masseret, E.; Amzil, Z.; Herve, F.; Rovillon, G.; Nouri, H.; M’Rabet, C.; Couet, D.; et al. New Insights on the Species-Specific Allelopathic Interactions between Macrophytes and Marine HAB Dinoflagellates. PLoS ONE 2017, 12, 28. [Google Scholar] [CrossRef] [Green Version]
- Tester, P.A.; Kibler, S.R.; Holland, W.C.; Usup, G.; Vandersea, M.W.; Leaw, C.P.; Teen, L.P.; Larsen, J.; Mohammad-Noor, N.; Faust, M.A.; et al. Sampling Harmful Benthic Dinoflagellates: Comparison of Artificial and Natural Substrate Methods. Harmful Algae 2014, 39, 8–25. [Google Scholar] [CrossRef]
- Jauzein, C.; Fricke, A.; Mangialajo, L.; Lemée, R. Sampling of Ostreopsis Cf. ovata Using Artificial Substrates: Optimization of Methods for the Monitoring of Benthic Harmful Algal Blooms. Mar. Pollut. Bull. 2016, 107, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Tester, P.A.; Litaker, R.W.; Soler-Onís, E.; Fernández-Zabala, J.; Berdalet, E. Using Artificial Substrates to Quantify Gambierdiscus and Other Toxic Benthic Dinoflagellates for Monitoring Purposes. Harmful Algae 2022, 120, 102351. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Zabala, J.; Tuya, F.; Amorim, A.; Soler-Onís, E. Benthic Dinoflagellates: Testing the Reliability of the Artificial Substrate Method in the Macaronesian Region. Harmful Algae 2019, 87, 101634. [Google Scholar] [CrossRef]
- Hernandez-Becerril, D.U.; Rodriguez-Palacio, M.C.; Lozano-Ramirez, C. Morphology and Life Stages of the Potentially Pinnatoxin-Producing Thecate Dinoflagellate Vulcanodinium rugosum from the Tropical Mexican Pacific. Bot. Mar. 2013, 56, 535–540. [Google Scholar] [CrossRef]
- Smith, K.F.; de Salas, M.; Adamson, J.; Rhodes, L.L. Rapid and accurate identification by real-time PCR of biotoxin-producing dinoflagellates from the family gymnodiniaceae. Mar. Drugs 2014, 12, 1361–1376. [Google Scholar] [CrossRef] [Green Version]
- Galluzzi, L.; Penna, A.; Bertozzini, E.; Vila, M.; Garcés, E.; Magnani, M. Development of a Real-Time PCR Assay for Rapid Detection and Quantification of Alexandrium minutum (a Dinoflagellate). Appl. Environ. Microbiol. 2004, 70, 1199–1206. [Google Scholar] [CrossRef] [Green Version]
- Perini, F.; Casabianca, A.; Battocchi, C.; Accoroni, S.; Totti, C.; Penna, A. New Approach Using the Real-Time PCR Method for Estimation of the Toxic Marine Dinoflagellate Ostreopsis Cf. ovata in Marine Environment. PLoS ONE 2011, 6, e17699. [Google Scholar] [CrossRef] [Green Version]
- Yuan, J.; Mi, T.; Zhen, Y.; Yu, Z. Development of a Rapid Detection and Quantification Method of Karenia mikimotoi by Real-Time Quantitative PCR. Harmful Algae 2012, 17, 83–91. [Google Scholar] [CrossRef]
- Bowers, H.A.; Tengs, T.; Glasgow, H.B.; Burkholder, J.M.; Rublee, P.A.; Oldach, D.W. Development of Real-Time PCR Assays for Rapid Detection of Pfiesteria Piscicida and Related Dinoflagellates. Appl. Environ. Microbiol. 2000, 66, 4641–4648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erdner, D.L.; Percy, L.; Keafer, B.; Lewis, J.; Anderson, D.M. A Quantitative Real-Time PCR Assay for the Identification and Enumeration of Alexandrium Cysts in Marine Sediments. Deep Sea Res. Part II Top. Stud. Oceanogr. 2010, 57, 279–287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, P.E.; Counts, R.C.; Clutter, R.I. Changes in Filtering Efficiency of Plankton Nets Due to Clogging Under Tow. ICES J. Mar. Sci. 1968, 32, 232–248. [Google Scholar] [CrossRef]
- Leakey, R.J.G.; Burkill, P.H.; Sleigh, M.A. A Comparison of Fixatives for the Estimation of Abundance and Biovolume of Marine Planktonic Ciliate Populations. J. Plankton Res. 1994, 16, 375–389. [Google Scholar] [CrossRef]
- Menden-Deuer, S.; Lessard, E.J.; Satterberg, J. Effect of Preservation on Dinoflagellate and Diatom Cell Volume and Consequences for Carbon Biomass Predictions. Mar. Ecol. Prog. Ser. 2001, 222, 41–50. [Google Scholar] [CrossRef]
- Faust, M.A. Ciguatera-Causing Dinoflagellates in a Coral-Reef Mangrove Ecosystem, Belize. Atoll Res. Bull. 2009, 569, 1–32. [Google Scholar] [CrossRef]
- Montagnes, D.J.S.; Berges, J.A.; Harrison, P.J.; Taylor, F.J.R. Estimating Carbon, Nitrogen, Protein, and Chlorophyll a from Volume in Marine Phytoplankton. Limnol. Oceanogr. 1994, 39, 1044–1060. [Google Scholar] [CrossRef] [Green Version]
- Bolch, C.J.S.; de Salas, M.F. A Review of the Molecular Evidence for Ballast Water Introduction of the Toxic Dinoflagellates Gymnodinium catenatum and the Alexandrium tamarensis Complex to Australasia. Harmful Algae 2007, 6, 465–485. [Google Scholar] [CrossRef]
- Glibert, P.M. Harmful Algae at the Complex Nexus of Eutrophication and Climate Change. Harmful Algae 2020, 91, 101583. [Google Scholar] [CrossRef]
- Garret, M.J.; Puchulutegui, C.; Selwood, A.I.; Wolny, J.L. Identification of the harmful dinoflagellate Vulcanodinium rugosum recovered from a ballast tank of a globally travelled ship in Port Tampa Bay, USA. Harmful Algae 2014, 39, 202–209. [Google Scholar] [CrossRef]
- Abadie, E. Etude de Vulcanodinium rugosum (Dinoflagellé Producteur de Pinnatoxines) se Développant dans la Lagune Méditerranéenne de l’Ingril. Ph.D. Thesis, Université de Montpellier, Montpellier, France, 2015. Available online: https://archimer.ifremer.fr/doc/00473/58425 (accessed on 10 March 2023).
- Newton, A.; Icely, J.; Cristina, S.; Brito, A.; Cardoso, A.C.; Colijn, F.; Riva, S.D.; Gertz, F.; Hansen, J.W.; Holmer, M.; et al. An overview of ecological status, vulnerability and future perspectives of European large shallow, semi enclosed coastal systems, lagoons and transitional waters. Estuar. Coast. Shelf Sci. 2014, 140, 95–122. [Google Scholar] [CrossRef]
- Harrison, P.J.; Waters, R.E.; Taylor, F.J.R. A Broad-Spectrum Artificial Seawater Medium for Coastal and Open Ocean Phytoplankton. J. Phycol. 1980, 16, 28–35. [Google Scholar] [CrossRef]
- Le Fur, I.; De Wit, R.; Plus, M.; Oheix, J.; Simier, M.; Ouisse, V. Submerged Benthic Macrophytes in Mediterranean Lagoons: Distribution Patterns in Relation to Water Chemistry and Depth. Hydrobiologia 2018, 808, 175–200. [Google Scholar] [CrossRef]
- Corpet, F. Multiple Sequence Alignment with Hierarchical Clustering. Nucleic Acids Res. 1988, 16, 10881–10890. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2020; Available online: https://www.r-project.org/ (accessed on 10 March 2023).



| Species | Strains and/or Origins | Collections | TM (°C) |
|---|---|---|---|
| Fukyuoa paulensis | RCC-6550 | Roscoff Culture Collection (RCC) | Nd |
| Fukyuoa sp. | RCC-6548 | RCC | Nd |
| Gambierdiscus belizeanus | RCC-6344 | RCC | Nd |
| Gambierdiscus carolinianus | RCC-6338 | RCC | Nd |
| Gambierdiscus spp. | RCC-6328 | RCC | Nd |
| Alexandrium minutum | Britany, France | MARBEC, University of Montpellier Collection (UMC) | Nd |
| Alexandrium pseudogonyaulax | Bizerte lagoon, Tunisia | Marbec, UMC | Nd |
| Alexandrium pacificum | ACT03, Thau, France | Marbec, UMC | Nd |
| Alexandrium pacificum | Bizerte lagoon, Tunisia | Marbec, UMC | Nd |
| Amphidinum carterae | SAMS, Oban, UK | Marbec, UMC | Nd |
| Coolia monotis | CMBZT14, Bizerte, Tunisia | Marbec, UMC | Nd |
| Gymnodinium catenatum | M'diq Bay, Morocco | Marbec, UMC | Nd |
| Gyrodinium impudicum | Gulf of Tunis, Tunisia | Marbec, UMC | Nd |
| Karenia selliformis | Gulf of Gabes, Tunisia | Marbec, UMC | Nd |
| Prorocentrum lima | PLBZT14, Bizerte lagoon, Tunisia | Marbec, UMC | Nd |
| Prorocentrum micans | H4-3, Thau lagoon, France | Marbec, UMC | Nd |
| Ostreopsis siamensis. | P550, Gbraltar strait | Marbec, UMC | Nd |
| Scrippsiella trochoidea | ST17, Mellah, Algeria | Marbec, UMC | Nd |
| Scrippsiella accuminata | China Sea | Marbec, UMC | Nd |
| Vulcanodinium rugosum | IFR-VRU-01, Ingril, France | Marbec, UMC | 79.1 |
| Vulcanodinium rugosum | 15-Ing-5.48, Ingril, France | Marbec, UMC | 79.1 |
| Vulcanodinium rugosum | 21-Ing-1.96, Ingril, France | Marbec, UMC | 79.1 |
| Vulcanodinium rugosum | 21-Vic-2.96, Vic, France | Marbec, UMC | 79.1 |
| Collection Date | Lagoons | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Thau | Ingril | Vic | Prévost | |||||||
| Year | Month | Day | LM | qPCR | LM | qPCR | LM | qPCR | LM | qPCR |
| 2021 | May | 4th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd |
| 20th | Nd | Nd | 0.11 | <0.057 | Nd | Nd | Nd | Nd | ||
| Jun. | 3rd | Nd | Nd | Nd | Nd | Nd | <0.057 | Nd | <0.057 | |
| 16th | Nd | Nd | 0.36 | 0.26 | Nd | <0.057 | Nd | Nd | ||
| 29th | Nd | Nd | Nd | <0.057 | Nd | <0.057 | 0.11 | <0.057 | ||
| Jul. | 7th | Nd | Nd | Nd | <0.057 | 0.25 | 0.18 | Nd | Nd | |
| 27th | Nd | Nd | Nd | <0.057 | 0.071 | <0.057 | Nd | Nd | ||
| Aug. | 10th | Nd | <0.057 | 0.036 | <0.057 | 0.036 | <0.057 | Nd | <0.057 | |
| 25th | Nd | Nd | Nd | Nd | 0.036 | <0.057 | Nd | Nd | ||
| Sep. | 8th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | <0.057 | |
| 21th | Nd | <0.057 | Nd | Nd | Nd | Nd | Nd | ND | ||
| Oct. | 4th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | |
| 19th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | ||
| Nov. | 3rd | Nd | Nd | Nd | Nd | Nd | <0.057 | Nd | Nd | |
| 19th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | ||
| Dec. | 2nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | |
| 16th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | ||
| 2022 | Jan. | 4th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd |
| 20th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | ||
| Feb. | 3rd | Nd | Nd | Nd | Nd | Nd | <0.057 | Nd | Nd | |
| 15th | Nd | <0.057 | Nd | Nd | Nd | Nd | Nd | Nd | ||
| Mar. | 1st | Nd | <0.057 | Nd | Nd | Nd | Nd | Nd | Nd | |
| 17th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | ||
| 29th | Nd | Nd | Nd | Nd | Nd | Nd | Nd | Nd | ||
| Apr. | 12th | Nd | Nd | Nd | Nd | Nd | <0.057 | Nd | Nd | |
| LSRU Genes | Sequences | Sequences Lengths |
|---|---|---|
| Isolate G | MG826107.1 | 1351 |
| Clone c650 | MK236574.1 | 620 |
| Clone c13139 | MK236578.1 | 620 |
| Clone c5437 | MK236579.1 | 619 |
| Clone c12163 | MK236577.1 | 622 |
| Clone c30140 | MK236582.1 | 619 |
| Clone c10134 | MK236580.1 | 617 |
| Clone c3918 | MK236576.1 | 619 |
| Clone c13589 | MK236581.1 | 620 |
| Clone c31575 | MK236575.1 | 618 |
| Isolate C | MG826106.1 | 928 |
| Isolate IFR10-017 | HQ622103.1 | 1384 |
| Isolate CAWD166 | JF267773.1 | 805 |
| Isolate CAWD180 | JF683380.1 | 788 |
| Strain G65 | JX457352.1 | 596 |
| Isolate QQCCCM92 | KX853181.1 | 856 |
| Isolate QQCCCM93 | KX853179.1 | 846 |
| Isolate CAWD188 | JF683382.1 | 788 |
| Strain OPMS30976 | LC228963.1 | 599 |
| Isolate CAFWC516 | KM252944.1 | 845 |
| Isolate QUCCCM91 | KX853180.1 | 847 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouquet, A.; Felix, C.; Masseret, E.; Reymond, C.; Abadie, E.; Laabir, M.; Rolland, J.L. Artificial Substrates Coupled with qPCR (AS-qPCR) Assay for the Detection of the Toxic Benthopelagic Dinoflagellate Vulcanodinium rugosum. Toxins 2023, 15, 217. https://doi.org/10.3390/toxins15030217
Bouquet A, Felix C, Masseret E, Reymond C, Abadie E, Laabir M, Rolland JL. Artificial Substrates Coupled with qPCR (AS-qPCR) Assay for the Detection of the Toxic Benthopelagic Dinoflagellate Vulcanodinium rugosum. Toxins. 2023; 15(3):217. https://doi.org/10.3390/toxins15030217
Chicago/Turabian StyleBouquet, Aurélien, Christine Felix, Estelle Masseret, Coralie Reymond, Eric Abadie, Mohamed Laabir, and Jean Luc Rolland. 2023. "Artificial Substrates Coupled with qPCR (AS-qPCR) Assay for the Detection of the Toxic Benthopelagic Dinoflagellate Vulcanodinium rugosum" Toxins 15, no. 3: 217. https://doi.org/10.3390/toxins15030217
APA StyleBouquet, A., Felix, C., Masseret, E., Reymond, C., Abadie, E., Laabir, M., & Rolland, J. L. (2023). Artificial Substrates Coupled with qPCR (AS-qPCR) Assay for the Detection of the Toxic Benthopelagic Dinoflagellate Vulcanodinium rugosum. Toxins, 15(3), 217. https://doi.org/10.3390/toxins15030217





